Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus)
Abstract
:1. Introduction
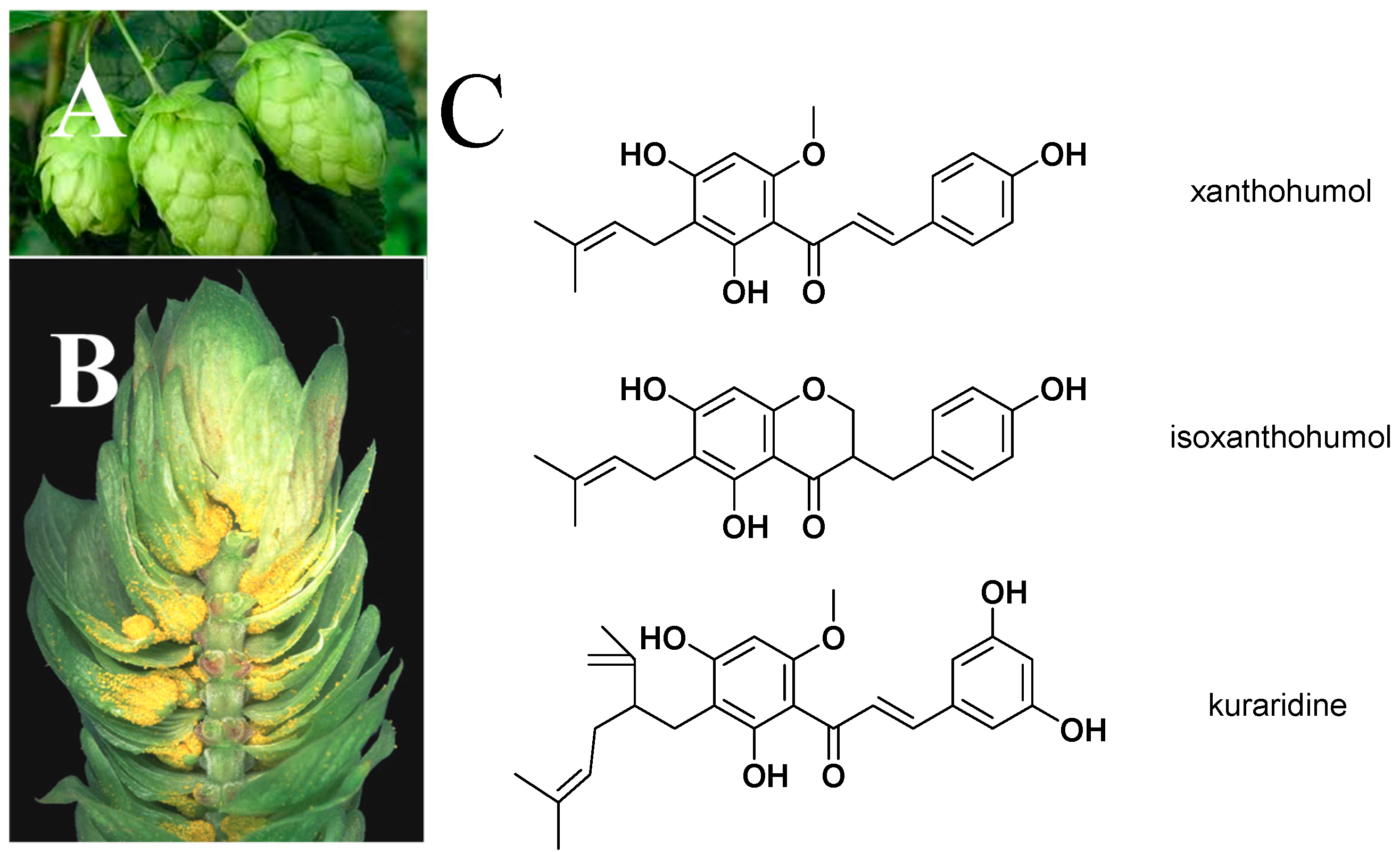
2. Bioactivity, Pharmacokinetics, and Safety of XN
2.1. Effect on Metabolic Syndrome and Related Disorders
2.1.1. Anti-Obesity Activities
2.1.2. Hypoglycemic Activities
2.1.3. Anti-Hyperlipidemia Activities
2.2. Cancer Related Bioactivities
2.2.1. Cancer Chemo-Preventive Effect
2.2.2. Anti-Angiogenic Activity
2.2.3. Proapoptosis Activity and Modulation of Autophagy
2.2.4. Anti-Invasion Activities
2.3. Anti-Inflammatory Activity
2.4. Central Nervous Systems Modulation Properties
2.5. Antimicrobial Activity
2.6. Anti-Parasite Effects
2.7. Effect on Bone Disease
2.8. Hepatic Protection
2.8.1. Protection in Chemical Hepatic Injury
2.8.2. Protection in Liver Ischemia/Reperfusion Injur
2.8.3. Benefits in Liver Diseases Associated with Virus Infection
2.9. Effects on Skin Disease
2.10. Thyroid Diseases
2.11. Benefits in the Thromboembolic Disease
2.12. Pharmacokinetics and Biotransformation of XN
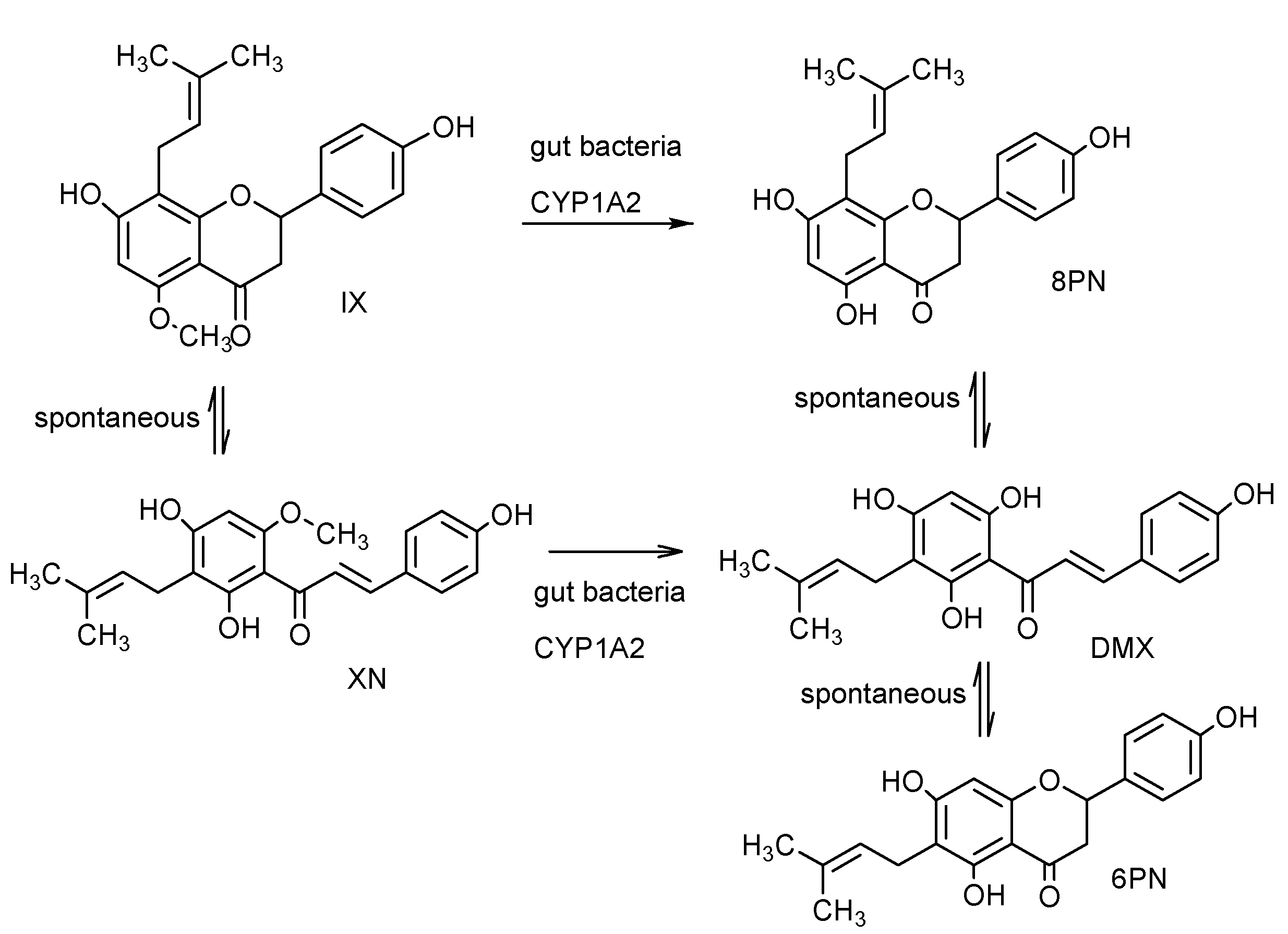
2.13. Safety of XN
3. Conclusions
Acknowledgments
Author Contributions
Abbreviation
| 6PN | 6-Prenylnaringenin |
| 8PN | 8-Prenylnaringenin |
| ALPL | Alkaline Phosphatase |
| ApoB | Apolipoprotein B |
| BACE1 | β-Site APP Cleaving Enzyme 1 |
| BVDV | Bovine Viral Diarrhea Virus |
| CXC | Cysteine X Cysteine |
| CXCR4 | Cysteine X Cysteine Chemokine Receptor 4 |
| CYP | Cytochrome P450 Protein |
| DCs | Dendritic Cells |
| DMX | Desmethylxanthohumol |
| DR | Death Receptor |
| HCV | Hepatitis C Virus |
| HIV | Human Immunodeficiency Virus |
| ICAM-1 | Intercellular Adhesion Molecule 1 |
| IFN-γ | Interferon-γ |
| IL | Interleukin |
| iNOS | Inducible NO Synthase |
| IX | Isoxanthohumol |
| Keap1 | Kelch-Like ECH-Associated Protein 1 |
| LPS | Lipopolysaccharide |
| MD-2 | Myeloid Differentiation Protein 2 |
| MIC | Minimum Inhibitory Concentration |
| MMP | Matrix Metalloproteinase |
| MRP5 | Multidrug Resistance Associated Protein 5 |
| NFκB | Nuclear Factor κB |
| NO | Nitric Oxide |
| NQO1 | NAD(P)H-Quinone Oxidoreductase |
| Nrf2 | Nuclear Factor E2-Related Factor 2 |
| PMA | Phorbol 12-Myristate 13-Acetate |
| Raf-1 | Rapidly Accelerated Fibrosarcoma-1 |
| RANKL | Receptor Activator NFκB Ligand |
| ROS | Reactive Oxygen Species |
| STAT | Signal Transducer And Activator Of Transcription |
| TLR4 | Toll-Likereceptor-4 |
| TNF-α | Tumor Necrosis Factor-α |
| VCP | Valosin-Containing Protein |
| XN | Xanthohumol |
Conflicts of Interest
References
- Zanoli, P.; Zavatti, M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008, 116, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, X.; Yang, L.; Yang, X. Advance on isolation and purification of xanthohumol from hops (Humulus lupulus L.). Food Ferment. Ind. 2013, 39, 143–148. [Google Scholar]
- Chen, Q.H.; Fu, M.L.; Chen, M.M.; Liu, J.; Liu, X.J.; He, G.Q.; Pu, S.C. Preparative isolation and purification of xanthohumol from hops (Humulus lupulus L.) by high-speed counter-current chromatography. Food Chem. 2012, 132, 619–623. [Google Scholar] [CrossRef]
- Khupse, R.S.; Erhardt, P.W. Total Synthesis of Xanthohumol. J. Nat. Prod. 2007, 70, 1507–1509. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Taylor, A.W.; Clawson, J.E.; Deinzer, M.L. Fate of Xanthohumol and Related Prenylflavonoids from Hops to Beer. J. Agric. Food Chem. 1999, 47, 2421–2428. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, S.; Zurcher, A.; Back, W. Enrichment of xanthohumol in the brewing process. Mol. Nutr. Food Res. 2005, 49, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Karabín, M.; Jelínek, L.; Kinčl, T.; Hudcová, T.; Kotlíková, B.; Dostálek, P. New approach to the production of xanthohumol-enriched beers. J. Inst. Brew. 2013, 119, 98–102. [Google Scholar]
- Vinson, J.A.; Mandarano, M.; Hirst, M.; Trevithick, J.R.; Bose, P. Phenol antioxidant quantity and quality in foods: Beers and the effect of two types of beer on an animal model of atherosclerosis. J. Agric. Food Chem. 2003, 51, 5528–5533. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Miranda, C.L.; Frei, B.; Buhler, D.R. Inhibition of peroxynitrite-mediated LDL oxidation by prenylated flavonoids: The α, β-unsaturated keto functionality of 2'-hydroxychalcones as a novel antioxidant pharmacophore. Chem. Res. Toxicol. 2003, 16, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Verzele, M.; Stockx, J.; Fontijn, F.; Anteunis, M. Xanthohumol, a New Natural Chalkone. Bull. Soc. Chim. Belg. 1957, 66, 452–475. [Google Scholar] [CrossRef]
- Stevens, J.F.; Page, J.E. Xanthohumol and related prenylflavonoids from hops and beer: To your good health! Phytochemistry 2004, 65, 1317–1330. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, P.J.; Carvalho, D.O.; Cruz, J.M.; Guido, L.F.; Barros, A.A. Fundamentals and health benefits of xanthohumol, a natural product derived from hops and beer. Nat. Prod. Commun. 2009, 4, 591–610. [Google Scholar] [PubMed]
- Turner-McGrievy, G.; Harris, M. Key elements of plant-based diets associated with reduced risk of metabolic syndrome. Curr. Diabete Rep. 2014, 14, 524. [Google Scholar] [CrossRef]
- Yang, J.-Y.; Della-Fera, M.; Rayalam, S.; Baile, C. Effect of xanthohumol and isoxanthohumol on 3T3-L1 cell apoptosis and adipogenesis. Apoptosis 2007, 12, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Mendes, V.; Monteiro, R.R.; Pestana, D.; Teixeira, D.; Calhau, C.A.O.; Azevedo, I. Xanthohumol Influences Preadipocyte Differentiation: Implication of Antiproliferative and Apoptotic Effects. J. Agric. Food Chem. 2008, 56, 11631–11637. [Google Scholar] [CrossRef] [PubMed]
- Kiyofuji, A.; Yui, K.; Takahashi, K.; Osada, K. Effects of xanthohumol-rich hop extract on the differentiation of preadipocytes. J. Oleo Sci. 2014, 63, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Rayalam, S.; Yang, J.Y.; Della-Fera, M.A.; Park, H.J.; Ambati, S.; Baile, C.A. Anti-obesity effects of xanthohumol plus guggulsterone in 3T3-L1 adipocytes. J. Med. Food 2009, 12, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Della-Fera, M.A.; Rayalam, S.; Baile, C.A. Enhanced effects of xanthohumol plus honokiol on apoptosis in 3T3-L1 adipocytes. Obesity 2008, 16, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, J.S.; Legette, L.L.; Miranda, C.L.; Jiang, Y.; Stevens, J.F. A metabolomics driven elucidation of the anti-obesity mechanisms of xanthohumol. J. Biol. Chem. 2013, 288, 19000–19013. [Google Scholar] [CrossRef] [PubMed]
- Yui, K.; Kiyofuji, A.; Osada, K. Effects of xanthohumol-rich extract from the hop on fatty acid metabolism in rats fed a high-fat diet. J. Oleo Sci. 2014, 63, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Quan, X.; Wang, Y.; Ma, X.; Liang, Y.; Tian, W.; Ma, Q.; Jiang, H.; Zhao, Y. α-Mangostin induces apoptosis and suppresses differentiation of 3T3-L1 cells via inhibiting fatty acid synthase. PLoS One 2012, 7, e33376. [Google Scholar] [CrossRef] [PubMed]
- Legette, L.L.; Moreno Luna, A.Y.; Reed, R.L.; Miranda, C.L.; Bobe, G.; Proteau, R.R.; Stevens, J.F. Xanthohumol lowers body weight and fasting plasma glucose in obese male Zucker fa/fa rats. Phytochemistry 2013, 91, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Nozawa, H. Xanthohumol, the chalcone from beer hops (Humulus lupulus L.), is the ligand for farnesoid X receptor and ameliorates lipid and glucose metabolism in KK-Ay mice. Biochem. Biophys. Res. Commun. 2005, 336, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.; Negrao, R.; Valente, I.; Castela, A.; Duarte, D.; Guardao, L.; Magalhaes, P.J.; Rodrigues, J.A.; Guimaraes, J.T.; Gomes, P.; et al. Xanthohumol modulates inflammation, oxidative stress, and angiogenesis in type 1 diabetic rat skin wound healing. J. Nat. Prod. 2013, 76, 2047–2053. [Google Scholar] [CrossRef] [PubMed]
- Araújo, J.R.; Gonçalves, P.; Martel, F. Modulation of glucose uptake in a human choriocarcinoma cell line (BeWo) by dietary bioactive compounds and drugs of abuse. J. Biochem. 2008, 144, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yin, H.; Liu, G.; Dong, J.; Qian, Z.; Miao, J. Xanthohumol, a prenylated chalcone from beer hops, acts as an α-glucosidase inhibitor in vitro. J. Agric. Food Chem. 2014, 62, 5548–5554. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Ryu, Y.B.; Kang, N.S.; Lee, B.W.; Heo, J.S.; Jeong, I.Y.; Park, K.H. Glycosidase inhibitory flavonoids from Sophora flavescens. Biol. Pharm. Bull. 2006, 29, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Casaschi, A.; Maiyoh, G.K.; Rubio, B.K.; Li, R.W.; Adeli, K.; Theriault, A.G. The chalcone xanthohumol inhibits triglyceride and apolipoprotein B secretion in HepG2 cells. J. Nutr. 2004, 134, 1340–1346. [Google Scholar] [PubMed]
- Tabata, N.; Ito, M.; Tomoda, H.; Ōmura, S. Xanthohumols, diacylglycerol acyltransferase inhibitors, from Humulus lupulus. Phytochemistry 1997, 46, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Kamisaka, Y.; Goto, R.; Shibakami, M.; Yoshioka, K.; Sato, Y. Surface plasmon resonance analysis of interactions between diacylglycerol acyltransferase and its interacting molecules. Biosci. Biotechnol. Biochem. 2011, 75, 1135–1139. [Google Scholar] [CrossRef] [PubMed]
- Inokoshi, J.; Kawamoto, K.; Takagi, Y.; Matsuhama, M.; Omura, S.; Tomoda, H. Expression of two human acyl-CoA: Diacylglycerol acyltransferase isozymes in yeast and selectivity of microbial inhibitors toward the isozymes. J. Antibiot. 2009, 62, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Santos-Gallego, C.G.; Badimon, J.J.; Rosenson, R.S. Beginning to understand high-density lipoproteins. Endocrinol. Metab. Clin. N. Am. 2014, 43, 913–947. [Google Scholar] [CrossRef]
- Hirata, H.; Takazumi, K.; Segawa, S.; Okada, Y.; Kobayashi, N.; Shigyo, T.; Chiba, H. Xanthohumol, a prenylated chalcone from Humulus lupulus L., inhibits cholesteryl ester transfer protein. Food Chem. 2012, 134, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Hirata, H.; Yimin; Segawa, S.; Ozaki, M.; Kobayashi, N.; Shigyo, T.; Chiba, H. Xanthohumol prevents atherosclerosis by reducing arterial cholesterol content via CETP and apolipoprotein E in CETP-transgenic mice. PLoS One 2012, 7, e49415. [Google Scholar] [CrossRef] [PubMed]
- Doddapattar, P.; Radović, B.; Patankar, J.V.; Obrowsky, S.; Jandl, K.; Nusshold, C.; Kolb, D.; Vujić, N.; Doshi, L.; Chandak, P.G.; et al. Xanthohumol ameliorates atherosclerotic plaque formation, hypercholesterolemia, and hepatic steatosis in ApoE-deficient mice. Mol. Nutr. Food Res. 2013, 57, 1718–1728. [Google Scholar] [PubMed]
- Heinecke, J.W. Oxidants and antioxidants in the pathogenesis of atherosclerosis: Implications for the oxidized low density lipoprotein hypothesis. Atherosclerosis 1998, 141, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Schempp, H.; Vogel, S.; Hückelhoven, R.; Heilmann, J. Re-evaluation of superoxide scavenging capacity of xanthohumol. Free Radic. Res. 2010, 44, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.L.; Stevens, J.F.; Ivanov, V.; McCall, M.; Frei, B.; Deinzer, M.L.; Buhler, D.R. Antioxidant and prooxidant actions of prenylated and nonprenylated chalcones and flavanones in vitro. J. Agric. Food Chem. 2000, 48, 3876–3884. [Google Scholar] [CrossRef] [PubMed]
- Plazar, J.; Žegura, B.; Lah, T.T.; Filipič, M. Protective effects of xanthohumol against the genotoxicity of benzo(a)pyrene (BaP), 2-amino-3-methylimidazo[4,5-f]quinoline (IQ) and tert-butyl hydroperoxide (t-BOOH) in HepG2 human hepatoma cells. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2007, 632, 1–8. [Google Scholar] [CrossRef]
- Dorn, C.; Massinger, S.; Wuzik, A.; Heilmann, J.; Hellerbrand, C. Xanthohumol suppresses inflammatory response to warm ischemia–reperfusion induced liver injury. Exp. Mol. Pathol. 2013, 94, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Strathmann, J.; Gerhauser, C. Natural Compounds as Inducers of Cell Death; Springer: Dordrecht, The Netherland, 2012; Volume 1, pp. 69–93. [Google Scholar]
- Kac, J.; Plazar, J.; Mlinarič, A.; Žegura, B.; Lah, T.T.; Filipič, M. Antimutagenicity of hops (Humulus lupulus L.): Bioassay-Directed fractionation and isolation of xanthohumol. Phytomedicine 2008, 15, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.L.; Yang, Y.-H.; Henderson, M.C.; Stevens, J.F.; Santana-Rios, G.; Deinzer, M.L.; Buhler, D.R. Prenylflavonoids from Hops Inhibit the Metabolic Activation of the Carcinogenic Heterocyclic Amine 2-Amino-3-methylimidazo[4,5-f]quinoline, Mediated by cDNA-Expressed Human CYP1A2. Drug Metab. Dispos. 2000, 28, 1297–1302. [Google Scholar] [PubMed]
- Plazar, J.; Filipič, M.; Groothuis, G.M.M. Antigenotoxic effect of xanthohumol in rat liver slices. Toxicol. in Vitro 2008, 22, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.L.; Aponso, G.L.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. Prenylated chalcones and flavanones as inducers of quinone reductase in mouse Hepa 1c1c7 cells. Cancer Lett. 2000, 149, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Dietz, B.M.; Kang, Y.-H.; Liu, G.; Eggler, A.L.; Yao, P.; Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R.; Mesecar, A.D.; van Breemen, R.B.; et al. Xanthohumol isolated from Humulus lupulus inhibits menadione-induced DNA damage through induction of quinone reductase. Chem. Res. Toxicol. 2005, 18, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Gerhauser, C.; Alt, A.; Heiss, E.; Gamal-Eldeen, A.; Klimo, K.; Knauft, J.; Neumann, I.; Scherf, H.R.; Frank, N.; Bartsch, H.; et al. Cancer chemopreventive activity of Xanthohumol, a natural product derived from hop. Mol. Cancer Ther. 2002, 1, 959–969. [Google Scholar] [PubMed]
- Albini, A.; Dell’Eva, R.; Vene, R.; Ferrari, N.; Buhler, D.R.; Noonan, D.M.; Fassina, G. Mechanisms of the antiangiogenic activity by the hop flavonoid xanthohumol: NF-κB and Akt as targets. FASEB J. 2006, 20, 527–529. [Google Scholar] [PubMed]
- Negrao, R.; Incio, J.; Lopes, R.; Azevedo, I.; Soares, R. Evidence for the effects of Xanthohumol in disrupting angiogenic, but not stable vessels. Int. J. Biomed. Sci. 2007, 3, 279–286. [Google Scholar] [PubMed]
- Gerhauser, C.; Hussong, R.; Bertl, E.; Klimo, K.; Frank, N.; Bartsch, H.; Becker, H. Cancer chemopreventive and anti-angiogenic activities of xanthohumol from hop (Humulus lupulus L.). Eur. J. Cancer Suppl. 2006, 4, 56. [Google Scholar] [CrossRef]
- Negrão, R.; Costa, R.; Duarte, D.; Taveira Gomes, T.; Mendanha, M.; Moura, L.; Vasques, L.; Azevedo, I.; Soares, R. Angiogenesis and inflammation signaling are targets of beer polyphenols on vascular cells. J. Cell. Biochem. 2010, 111, 1270–1279. [Google Scholar] [CrossRef] [PubMed]
- Shamoto, T.; Matsuo, Y.; Shibata, T.; Tsuboi, K.; Takahashi, H.; Funahashi, H.; Okada, Y.; Takeyama, H. Xanthohumol inhibits angiogenesis through VEGF and IL-8 in pancreatic cancer. Pancreatology 2013, 13, S52–S53. [Google Scholar] [CrossRef]
- Rudzitis-Auth, J.; Körbel, C.; Scheuer, C.; Menger, M.D.; Laschke, M.W. Xanthohumol inhibits growth and vascularization of developing endometriotic lesions. Hum. Reprod. 2012, 27, 1735–1744. [Google Scholar] [CrossRef] [PubMed]
- Negrão, R.; Costa, R.; Duarte, D.; Gomes, T.T.; Coelho, P.; Guimarães, J.T.; Guardão, L.; Azevedo, I.; Soares, R. Xanthohumol-supplemented beer modulates angiogenesis and inflammation in a skin wound healing model. Involvement of local adipocytes. J. Cell. Biochem. 2012, 113, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; Mei, Y.; Sinha, S. Role of the crosstalk between autophagy and apoptosis in cancer. J. Oncol. 2013, 2013. [Google Scholar] [CrossRef]
- Liu, F. Xanthohumol Inhibits the Proliferation of Prostate Cancer Cells by Reducing the Activity of Notch Signaling Pathway. Master’s Thesis, Lanzhou University, Lanzhou, China, 2014. [Google Scholar]
- Delmulle, L.; Bellahcène, A.; Dhooge, W.; Comhaire, F.; Roelens, F.; Huvaere, K.; Heyerick, A.; Castronovo, V.; de Keukeleire, D. Anti-proliferative properties of prenylated flavonoids from hops (Humulus lupulus L.) in human prostate cancer cell lines. Phytomedicine 2006, 13, 732–734. [Google Scholar] [CrossRef] [PubMed]
- Monteghirfo, S.; Tosetti, F.; Ambrosini, C.; Stigliani, S.; Pozzi, S.; Frassoni, F.; Fassina, G.; Soverini, S.; Albini, A.; Ferrari, N. Antileukemia effects of xanthohumol in Bcr/Abl-transformed cells involve nuclear factor-κB and p53 modulation. Mol. Cancer Ther. 2008, 7, 2692–2702. [Google Scholar] [CrossRef] [PubMed]
- Harikumar, K.B.; Kunnumakkara, A.B.; Ahn, K.S.; Anand, P.; Krishnan, S.; Guha, S.; Aggarwal, B.B. Modification of the cysteine residues in IκBα kinase and NF-κB (p65) by xanthohumol leads to suppression of NF-κB–regulated gene products and potentiation of apoptosis in leukemia cells. Blood 2009, 113, 2003–2013. [Google Scholar] [CrossRef] [PubMed]
- Drenzek, J.G.; Seiler, N.L.; Jaskula-Sztul, R.; Rausch, M.M.; Rose, S.L. Xanthohumol decreases Notch1 expression and cell growth by cell cycle arrest and induction of apoptosis in epithelial ovarian cancer cell lines. Gynecol. Oncol. 2011, 122, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Krajka-Kuźniak, V.; Paluszczak, J.; Baer-Dubowska, W. Xanthohumol induces phase II enzymes via Nrf2 in human hepatocytes in vitro. Toxicol. in Vitro 2013, 27, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.C.; Liu, C.H.; Chen, C.N.; Duan, K.J.; Lin, M.T. Inhibitory effects of xanthohumol from hops (Humulus lupulus L.) on human hepatocellular carcinoma cell lines. Phytother. Res. 2008, 22, 1465–1468. [Google Scholar] [CrossRef] [PubMed]
- Tronina, T.; Bartmańska, A.; Filip-Psurska, B.; Wietrzyk, J.; Popłoński, J.; Huszcza, E. Fungal metabolites of xanthohumol with potent antiproliferative activity on human cancer cell lines in vitro. Bioorg. Med. Chem. 2013, 21, 2001–2006. [Google Scholar] [CrossRef] [PubMed]
- Festa, M.; Capasso, A.; D’Acunto, C.W.; Masullo, M.; Rossi, A.G.; Pizza, C.; Piacente, S. xanthohumol induces apoptosis in human malignant glioblastoma cells by increasing reactive oxygen species and activating MAPK pathways. J. Nat. Prod. 2011, 74, 2505–2513. [Google Scholar] [CrossRef] [PubMed]
- Zajc, I.; Filipic, M.; Lah, T.T. Xanthohumol induces different cytotoxicity and apoptotic pathways in malignant and normal astrocytes. Phytother. Res. 2012, 26, 1709–1713. [Google Scholar] [CrossRef] [PubMed]
- Bartmanska, A.; Tronina, T.; Poplonski, J.; Huszcza, E. Biotransformations of prenylated hop flavonoids for drug discovery and production. Curr. Drug Metab. 2013, 14, 1083–1097. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Lee, I.-S.; Moon, A. 2-Hydroxychalcone and xanthohumol inhibit invasion of triple negative breast cancer cells. Chem. Biol. Interact. 2013, 203, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Deeb, D.; Gao, X.; Jiang, H.; Arbab, A.S.; Dulchavsky, S.A.; Gautam, S.C. Growth Inhibitory and Apoptosis-inducing Effects of Xanthohumol, a Prenylated Chalone Present in Hops, in Human Prostate Cancer Cells. Anticancer Res. 2010, 30, 3333–3339. [Google Scholar] [PubMed]
- Pan, L.; Becker, H.; Gerhäuser, C. Xanthohumol induces apoptosis in cultured 40–16 human colon cancer cells by activation of the death receptor- and mitochondrial pathway. Mol. Nutr. Food Res. 2005, 49, 837–843. [Google Scholar] [CrossRef] [PubMed]
- Strathmann, J.; Klimo, K.; Sauer, S.W.; Okun, J.G.; Prehn, J.H.M.; Gerhäuser, C. Xanthohumol-induced transient superoxide anion radical formation triggers cancer cells into apoptosis via a mitochondria-mediated mechanism. FASEB J. 2010, 24, 2938–2950. [Google Scholar] [CrossRef] [PubMed]
- Blanquer-Rosselló, M.M.; Oliver, J.; Valle, A.; Roca, P. Effect of xanthohumol and 8-prenylnaringenin on MCF-7 breast cancer cells oxidative stress and mitochondrial complexes expression. J. Cell. Biochem. 2013, 114, 2785–2794. [Google Scholar] [CrossRef] [PubMed]
- Lust, S.; Vanhoecke, B.; van Gele, M.; Boelens, J.; van Melckebeke, H.; Kaileh, M.; vanden Berghe, W.; Haegeman, G.; Philephus, J.; Bracke, M.; et al. Xanthohumol activates the proapoptotic arm of the unfolded protein response in chronic lymphocytic leukemia. Anticancer Res. 2009, 29, 3797–3805. [Google Scholar] [PubMed]
- Cook, M.R.; Luo, J.; Ndiaye, M.; Chen, H.; Kunnimalaiyaan, M. Xanthohumol inhibits the neuroendocrine transcription factor achaete-scute complex-like 1, suppresses proliferation, and induces phosphorylated ERK1/2 in medullary thyroid cancer. Am. J. Surg. 2010, 199, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Colgate, E.C.; Miranda, C.L.; Stevens, J.F.; Bray, T.M.; Ho, E. Xanthohumol, a prenylflavonoid derived from hops induces apoptosis and inhibits NF-kappaB activation in prostate epithelial cells. Cancer Lett. 2007, 246, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kim, H.J.; Lee, J.S.; Lee, I.S.; Kang, B.Y. Inhibition of topoisomerase I activity and efflux drug transporters’ expression by xanthohumol. from hops. Arch. Pharm. Res. 2007, 30, 1435–1439. [Google Scholar] [CrossRef]
- Monteiro, R.; Faria, A.; Azevedo, I.; Calhau, C. Modulation of breast cancer cell survival by aromatase inhibiting hop (Humulus lupulus L.) flavonoids. J. Steroid Biochem. Mol. Biol. 2007, 105, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Nozawa, H.; Daikonnya, A.; Kondo, K.; Kitanaka, S. Inhibitors of nitric oxide production from hops (Humulus lupulus L.). Biol. Pharm. Bull. 2003, 26, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Park, M.A.; Heo, S.W.; Park, S.Y.; Kang, K.W.; Park, P.H.; Kim, J.A. The radio-sensitizing effect of xanthohumol is mediated by STAT3 and EGFR suppression in doxorubicin-resistant MCF-7 human breast cancer cells. BBA–Gen. Subj. 2013, 1830, 2638–2648. [Google Scholar] [CrossRef]
- Szliszka, E.; Jaworska, D.; Ksek, M.; Czuba, Z.P.; Krol, W. Targeting death receptor TRAIL-R2 by chalcones for TRAIL-induced apoptosis in cancer cells. Int. J. Mol. Sci. 2012, 13, 15343–15359. [Google Scholar] [CrossRef] [PubMed]
- Szliszka, E.; Czuba, Z.; Mazur, B.; Sedek, L.; Paradysz, A.; Krol, W. Chalcones enhance TRAIL-induced apoptosis in prostate cancer cells. Int. J. Mol. Sci. 2009, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.W.; Cooney, J.; Jensen, D.; Li, Y.; Paxton, J.W.; Birch, N.P.; Scheepens, A. Hop-derived prenylflavonoids are substrates and inhibitors of the efflux transporter breast cancer resistance protein (BCRP/ABCG2). Mol. Nutr. Food Res. 2014, 14, 2099–2110. [Google Scholar] [CrossRef]
- Lust, S.; Vanhoecke, B.; Janssens, A.; Philippe, J.; Bracke, M.; Offner, F. Xanthohumol kills B-chronic lymphocytic leukemia cells by an apoptotic mechanism. Mol. Nutr. Food Res. 2005, 49, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Benelli, R.; Vene, R.; Ciarlo, M.; Carlone, S.; Barbieri, O.; Ferrari, N. The AKT/NF-κB inhibitor xanthohumol is a potent anti-lymphocytic leukemia drug overcoming chemoresistance and cell infiltration. Biochem. Pharmacol. 2012, 83, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Sasazawa, Y.; Kanagaki, S.; Tashiro, E.; Nogawa, T.; Muroi, M.; Kondoh, Y.; Osada, H.; Imoto, M. Xanthohumol impairs autophagosome maturation through direct inhibition of valosin-containing protein. ACS Chem. Biol. 2012, 7, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Vanhoecke, B.; Derycke, L.; van Marck, V.; Depypere, H.; de Keukeleire, D.; Bracke, M. Antiinvasive effect of xanthohumol, a prenylated chalcone present in hops (Humulus lupulus L.) and beer. Int. J. Cancer 2005, 117, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Jongthawin, J.; Techasen, A.; Loilome, W.; Yongvanit, P.; Namwat, N. Anti-inflammatory agents suppress the prostaglandin E2 production and migration ability of cholangiocarcinoma cell lines. Asian Pac. J. Cancer Prev. 2012, 13, 47–51. [Google Scholar] [PubMed]
- Wang, Y.; Chen, Y.; Wang, J.; Chen, J.; Aggarwal, B.B.; Pang, X.; Liu, M. Xanthohumol, a prenylated chalcone derived from hops, suppresses cancer cell invasion through inhibiting the expression of CXCR4 chemokine receptor. Curr. Mol. Med. 2012, 12, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Viola, K.; Kopf, S.; Rarova, L.; Jarukamjorn, K.; Kretschy, N.; Teichmann, M.; Vonach, C.; Atanasov, A.G.; Giessrigl, B.; Huttary, N.; et al. Xanthohumol attenuates tumour cell-mediated breaching of the lymphendothelial barrier and prevents intravasation and metastasis. Arch. Toxicol. 2013, 87, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-C.; Kim, H.J.; Kim, Y.-J.; Lee, K.Y.; Choi, H.J.; Lee, I.-S.; Kang, B.Y. Differential anti-inflammatory pathway by xanthohumol in IFN-γ and LPS-activated macrophages. Int. Immunopharmacol. 2008, 8, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.R.; Miranda, C.L.; Hobbs, D.J.; Proteau, R.R.; Stevens, J.F. Xanthohumol and related prenylated flavonoids inhibit inflammatory cytokine production in LPS-activated THP-1 monocytes: Structure-Activity relationships and in silico binding to myeloid differentiation protein-2 (MD-2). Planta Med. 2010, 76, 1536–1543. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-C.; You, S.-K.; Kim, H.J.; Cho, C.-W.; Lee, I.-S.; Kang, B.Y. Xanthohumol inhibits IL-12 production and reduces chronic allergic contact dermatitis. Int. Immunopharmacol. 2010, 10, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.M.; Kim, H.J.; Lee, K.Y.; Choi, H.J.; Lee, I.S.; Kang, B.Y. Increased IL-2 production in T cells by xanthohumol through enhanced NF-AT and AP-1 activity. Int. Immunopharmacol. 2009, 9, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Deeb, D.; Liu, Y.; Gautam, S.; Dulchavsky, S.A.; Gautam, S.C. Immunomodulatory activity of xanthohumol: Inhibition of T cell proliferation, cell-mediated cytotoxicity and Th1 cytokine production through suppression of NF-κB. Immunopharmacol. Immunotoxicol. 2009, 31, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Lupinacci, E.; Meijerink, J.; Vincken, J.-P.; Gabriele, B.; Gruppen, H.; Witkamp, R.F. Xanthohumol from hop (Humulus lupulus L.) is an efficient inhibitor of monocyte chemoattractant protein-1 and tumor necrosis factor-α release in LPS-stimulated RAW 264.7 mouse macrophages and U937 human monocytes. J. Agric. Food Chem. 2009, 57, 7274–7281. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Tyagi, A.K.; Deshmukh-Taskar, P.; Hinojosa, M.; Prasad, S.; Aggarwal, B.B. Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch. Biochem. Biophys. 2014, 559C:91–559C:99. [Google Scholar] [CrossRef]
- Lee, I.-S.; Lim, J.; Gal, J.; Kang, J.C.; Kim, H.J.; Kang, B.Y.; Choi, H.J. Anti-inflammatory activity of xanthohumol involves heme oxygenase-1 induction via NRF2-ARE signaling in microglial BV2 cells. Neurochem. Int. 2011, 58, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Xuan, N.T.; Shumilina, E.; Gulbins, E.; Gu, S.; Götz, F.; Lang, F. Triggering of dendritic cell apoptosis by xanthohumol. Mol. Nutr. Food Res. 2010, 54, S214–S224. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Kim, B.-W.; Jung, J.H.; Yokozawa, T.; Jung, H.A. Selective inhibition of prenylated flavonoids from Sophora flavescens against BACE1 and cholinesterases. Am. J. Chin. Med. 2010, 38, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Yen, T.L.; Hsu, C.K.; Lu, W.J.; Hsieh, C.Y.; Hsiao, G.; Chou, D.S.; Wu, G.J.; Sheu, J.R. Neuroprotective effects of xanthohumol, a prenylated flavonoid from hops (Humulus lupulus), in ischemic stroke of rats. J. Agric. Food Chem. 2012, 60, 1937–1944. [Google Scholar] [CrossRef] [PubMed]
- Oberbauer, E.; Urmann, C.; Steffenhagen, C.; Bieler, L.; Brunner, D.; Furtner, T.; Humpel, C.; Bäumer, B.; Bandtlow, C.; Couillard-Despres, S.; et al. Chroman-like cyclic prenylflavonoids promote neuronal differentiation and neurite outgrowth and are neuroprotective. J. Nutr. Biochem. 2013, 24, 1953–1962. [Google Scholar] [CrossRef] [PubMed]
- Meissner, O.; Haberlein, H. Influence of xanthohumol on the binding behavior of GABAA receptors and their lateral mobility at hippocampal neurons. Planta Med. 2006, 72, 656–658. [Google Scholar] [CrossRef] [PubMed]
- Ceremuga, T.E.; Johnson, L.A.; Adams-Henderson, J.M.; McCall, S.; Johnson, D. Investigation of the anxiolytic effects of xanthohumol, a component of humulus lupulus (Hops), in the male Sprague-Dawley rat. AANA J. 2013, 81, 193–198. [Google Scholar] [PubMed]
- Kireev, R.; Salinas, G.; Bermudo, B.; Vara, E.; Tresguerres, J. Xanthohumol activated pro-survival pathways in the brain of femal senescence accelerated mouse. Clin. Nutr. 2013, 32 (Suppl. 1), S221–S222. [Google Scholar]
- Zamzow, D.R.; Elias, V.; Legette, L.L.; Choi, J.; Stevens, J.F.; Magnusson, K.R. Xanthohumol improved cognitive flexibility in young mice. Behav. Brain Res. 2014, 275, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ding, Z.-H.; Liu, J.-K.; Zheng, Y.-T. Xanthohumol, a novel anti-HIV-1 agent purified from Hops Humulus lupulus. Antivir. Res. 2004, 64, 189–194. [Google Scholar] [PubMed]
- Buckwold, V.E.; Wilson, R.J.H.; Nalca, A.; Beer, B.B.; Voss, T.G.; Turpin, J.A.; Buckheit Iii, R.W.; Wei, J.; Wenzel-Mathers, M.; Walton, E.M.; et al. Antiviral activity of hop constituents against a series of DNA and RNA viruses. Antivir. Res. 2004, 61, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Lou, S.; Zheng, Y.M.; Liu, S.L.; Qiu, J.; Han, Q.; Li, N.; Zhu, Q.; Zhang, P.; Yang, C.; Liu, Z. Inhibition of hepatitis C virus replication in vitro by xanthohumol, a natural product present in hops. Planta Med. 2014, 80, 171–176. [Google Scholar] [PubMed]
- Yamaguchi, N.; Satoh-Yamaguchi, K.; Ono, M. In vitro evaluation of antibacterial, anticollagenase, and antioxidant activities of hop components (Humulus lupulus) addressing acne vulgaris. Phytomedicine 2009, 16, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Virani, S.; Zavro, M.; Haas, G. Inhibition ofStreptococcus mutans and Other Oral streptococci by hop (Humulus lupulus L.) constituents. Econ. Bot. 2003, 57, 118–125. [Google Scholar] [CrossRef]
- Rozalski, M.; Micota, B.; Sadowska, B.; Stochmal, A.; Jedrejek, D.; Wieckowska-Szakiel, M.; Rozalska, B. Antiadherent and Antibiofilm Activity of Humulus lupulus L. Derived Products: New Pharmacological Properties. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Gerhauser, C. Broad spectrum anti-infective potential of xanthohumol from hop (Humulus lupulus L.) in comparison with activities of other hop constituents and xanthohumol metabolites. Mol. Nutr. Food Res. 2005, 49, 827–831. [Google Scholar] [CrossRef] [PubMed]
- Hanske, L.; Hussong, R.; Frank, N.; Gerhäuser, C.; Blaut, M.; Braune, A. Xanthohumol does not affect the composition of rat intestinal microbiota. Mol. Nutr. Food Res. 2005, 49, 868–873. [Google Scholar] [CrossRef] [PubMed]
- Allen, P.C. Anticoccidial effects of xanthohumol. Avian Dis. 2007, 51, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Frölich, S.; Schubert, C.; Bienzle, U.; Jenett-Siems, K. In vitro antiplasmodial activity of prenylated chalcone derivatives of hops (Humulus lupulus) and their interaction with haemin. J. Antimicrob. Chemother. 2005, 55, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Tobe, H.; Muraki, Y.; Kitamura, K.; Komiyama, O.; Sato, Y.; Sugioka, T.; Maruyama, H.B.; Matsuda, E.; Nagai, M. Bone resorption inhibitors from hop extract. Biosci. Biotechnol. Biochem. 1997, 61, 158–159. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.M.; Han, E.H.; Jin, Y.H.; Choi, Y.H.; Lee, K.Y.; Jeong, H.G. Xanthohumol from the hop plant stimulates osteoblast differentiation by RUNX2 activation. Biochem. Biophys. Res. Commun. 2011, 409, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Milligan, S.R.; Kalita, J.C.; Pocock, V.; Van De Kauter, V.; Stevens, J.F.; Deinzer, M.L.; Rong, H.; de Keukeleire, D. The endocrine activities of 8-prenylnaringenin and related hop (Humulus lupulus L.) flavonoids. J. Clin. Endocrinol. Metab. 2000, 85, 4912–4915. [Google Scholar] [CrossRef] [PubMed]
- Suh, K.S.; Rhee, S.Y.; Kim, Y.S.; Lee, Y.S.; Choi, E.M. Xanthohumol modulates the expression of osteoclast-specific genes during osteoclastogenesis in RAW264.7 cells. Food Chem. Toxicol. 2013, 62, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Stracke, D.; Schulz, T.; Prehm, P. Inhibitors of hyaluronan export from hops prevent osteoarthritic reactions. Mol. Nutr. Food Res. 2011, 55, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Kathagen, N.; Prehm, P. Regulation of intracellular pH by glycosaminoglycans. J. Cell. Physiol. 2013, 228, 2071–2075. [Google Scholar] [CrossRef] [PubMed]
- Prehm, P. Curcumin analogue identified as hyaluronan export inhibitor by virtual docking to the ABC transporter MRP5. Food Chem. Toxicol. 2013, 62, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Dorn, C.; Kraus, B.; Motyl, M.; Weiss, T.S.; Gehrig, M.; Schölmerich, J.; Heilmann, J.; Hellerbrand, C. Xanthohumol, a chalcon derived from hops, inhibits hepatic inflammation and fibrosis. Mol. Nutr. Food Res. 2010, 54, S205–S213. [Google Scholar] [CrossRef] [PubMed]
- Dorn, C.; Heilmann, J.; Hellerbrand, C. Protective effect of xanthohumol on toxin-induced liver inflammation and fibrosis. Int. J. Clin. Exp. Pathol. 2012, 5, 29–36. [Google Scholar] [PubMed]
- Pinto, C.; Duque, A.L.; Rodriguez-Galdon, B.; Cestero, J.J.; Macias, P. Xanthohumol prevents carbon tetrachloride-induced acute liver injury in rats. Food Chem. Toxicol. 2012, 50, 3405–3412. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.J.; Miranda, C.L.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. Influence of prenylated and non-prenylated flavonoids on liver microsomal lipid peroxidation and oxidative injury in rat hepatocytes. Food Chem. Toxicol. 2001, 39, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Dietz, B.M.; Hagos, G.K.; Eskra, J.N.; Wijewickrama, G.T.; Anderson, J.R.; Nikolic, D.; Guo, J.; Wright, B.; Chen, S.-N.; Pauli, G.F.; et al. Differential regulation of detoxification enzymes in hepatic and mammary tissue by hops (Humulus lupulus) in vitro and in vivo. Mol. Nutr. Food Res. 2013, 57, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Cestero, J.J.; Rodríguez-Galdón, B.; Macías, P. Xanthohumol, a prenylated flavonoid from hops (Humulus lupulus L.), protects rat tissues against oxidative damage after acute ethanol administration. Toxicol. Rep. 2014, 1, 726–733. [Google Scholar] [CrossRef]
- Hartkorn, A.; Hoffmann, F.; Ajamieh, H.; Vogel, S.; Heilmann, J.; Gerbes, A.L.; Vollmar, A.M.; Zahler, S. Antioxidant effects of xanthohumol and functional impact on hepatic ischemia-reperfusion injury. J. Nat. Prod. 2009, 72, 1741–1747. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Liu, Z.; Han, Q.; Chen, J.; Lou, S.; Qiu, J.; Zhang, G. Inhibition of bovine viral diarrhea virus in vitro by xanthohumol: Comparisons with ribavirin and interferon-α and implications for the development of anti-hepatitis C virus agents. Eur. J. Pharm. Sci. 2009, 38, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Liu, Z.; Han, Q.; Chen, J.; Lv, Y. Xanthohumol enhances antiviral effect of interferon α-2b against bovine viral diarrhea virus, a surrogate of hepatitis C virus. Phytomedicine 2010, 17, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Li, N.; Li, F.; Zhu, Q.; Liu, X.; Han, Q.; Wang, Y.; Chen, Y.; Zeng, X.; Lv, Y.; et al. Xanthohumol, a main prenylated chalcone from hops, reduces liver damage and modulates oxidative reaction and apoptosis in hepatitis C virus infected Tupaia belangeri. Int. Immunopharmacol. 2013, 16, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Koo, J.H.; Kim, H.T.; Yoon, H.Y.; Kwon, K.B.; Choi, I.W.; Jung, S.H.; Kim, H.U.; Park, B.H.; Park, J.W. Effect of xanthohumol on melanogenesis in B16 melanoma cells. Exp. Mol. Med. 2008, 40, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Philips, N.; Samuel, M.; Arena, R.; Chen, Y.J.; Conte, J.; Natarajan, P.; Haas, G.; Gonzalez, S. Direct inhibition of elastase and matrixmetalloproteinases and stimulation of biosynthesis of fibrillar collagens, elastin, and fibrillins by xanthohumol. J. Cosmet. Sci. 2010, 61, 125–132. [Google Scholar] [PubMed]
- Chen, W.; Becker, T.; Qian, F.; Ring, J. Beer and beer compounds: Physiological effects on skin health. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 142–150. [Google Scholar] [CrossRef]
- Radović, B.; Schmutzler, C.; Köhrle, J. Xanthohumol stimulates iodide uptake in rat thyroid-derived FRTL-5 cells. Mol. Nutr. Food Res. 2005, 49, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Proteau, R.; Mata, J.E.; Miranda, C.L.; Fan, Y.; Brown, J.J.; Buhler, D.R. Plant polyphenols and multidrug resistance: Effects of dietary flavonoids on drug transporters in Caco-2 and MDCKII-MDR1 cell transport models. Xenobiotica 2006, 36, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Taur, J.S.; Rodriguez-Proteau, R. Effects of dietary flavonoids on the transport of cimetidine via P-glycoprotein and cationic transporters in Caco-2 and LLC-PK1 cell models. Xenobiotica 2008, 38, 1536–1550. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Rodriguez-Proteau, R. Ketoconazole and the modulation of multidrug resistance-mediated transport in Caco-2 and MDCKII-MDR1 drug transport models. Xenobiotica 2008, 38, 107–129. [Google Scholar] [CrossRef] [PubMed]
- Radović, B.; Hussong, R.; Gerhäuser, C.; Meinl, W.; Frank, N.; Becker, H.; Köhrle, J. Xanthohumol, a prenylated chalcone from hops, modulates hepatic expression of genes involved in thyroid hormone distribution and metabolism. Mol. Nutr. Food Res. 2010, 54, S225–S235. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Hsieh, K.H.; Lu, W.J.; Chou, H.C.; Chou, D.S.; Lien, L.M.; Sheu, J.R.; Lin, K.H. Xanthohumol, a prenylated flavonoid from hops (Humulus lupulus), prevents platelet activation in human platelets. Evid. Based Complement. Altern. Med. 2012, 2012, 1–10. [Google Scholar]
- Qadri, S.M.; Mahmud, H.; Föller, M.; Lang, F. Inhibition of suicidal erythrocyte death by xanthohumol. J. Agric. Food Chem. 2009, 57, 7591–7595. [Google Scholar] [CrossRef] [PubMed]
- Yilmazer, M.; Stevens, J.F.; Buhler, D.R. In vitro glucuronidation of xanthohumol, a flavonoid in hop and beer, by rat and human liver microsomes. FEBS Lett. 2001, 491, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Yilmazer, M.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. In vitro biotransformation of xanthohumol, a flavonoid from hops (Humulus lupulus), by rat liver microsomes. Drug Metab. Dispos. 2001, 29, 223–231. [Google Scholar] [PubMed]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Pauli, G.F.; van Breemen, R.B. Metabolism of xanthohumol and isoxanthohumol, prenylated flavonoids from hops (Humulus lupulus L.), by human liver microsomes. J. Mass Spectrom. 2005, 40, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Possemiers, S.; Heyerick, A.; Robbens, V.; de Keukeleire, D.; Verstraete, W. Activation of proestrogens from hops (Humulus lupulus L.) by intestinal microbiota; conversion of isoxanthohumol into 8-prenylnaringenin. J. Agric. Food Chem. 2005, 53, 6281–6288. [Google Scholar] [CrossRef] [PubMed]
- Legette, L.; Karnpracha, C.; Reed, R.L.; Choi, J.; Bobe, G.; Christensen, J.M.; Rodriguez-Proteau, R.; Purnell, J.Q.; Stevens, J.F. Human pharmacokinetics of xanthohumol, an antihyperglycemic flavonoid from hops. Mol. Nutr. Food Res. 2014, 58, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Van Breemen, R.B.; Yuan, Y.; Banuvar, S.; Shulman, L.P.; Qiu, X.; Ramos Alvarenga, R.F.; Chen, S.N.; Dietz, B.M.; Bolton, J.L.; Pauli, G.F.; et al. Pharmacokinetics of prenylated hop phenols in women following oral administration of a standardized extract of hops. Mol. Nutr. Food Res. 2014, 17. [Google Scholar] [CrossRef]
- Hanske, L.; Loh, G.; Sczesny, S.; Blaut, M.; Braune, A. Recovery and metabolism of xanthohumol in germ-free and human microbiota-associated rats. Mol. Nutr. Food Res. 2010, 54, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Avula, B.; Ganzera, M.; Warnick, J.E.; Feltenstein, M.W.; Sufka, K.J.; Khan, I.A. High-performance liquid chromatographic determination of xanthohumol in rat plasma, urine, and fecal samples. J. Chromatogr. Sci. 2004, 42, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Nookandeh, A.; Frank, N.; Steiner, F.; Ellinger, R.; Schneider, B.; Gerhäuser, C.; Becker, H. Xanthohumol metabolites in faeces of rats. Phytochemistry 2004, 65, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Jirásko, R.; Holčapek, M.; Vrublová, E.; Ulrichová, J.; Šimánek, V. Identification of new phase II metabolites of xanthohumol in rat in vivo biotransformation of hop extracts using high-performance liquid chromatography electrospray ionization tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 4100–4108. [Google Scholar] [CrossRef] [PubMed]
- Legette, L.; Ma, L.; Reed, R.L.; Miranda, C.L.; Christensen, J.M.; Rodriguez-Proteau, R.; Stevens, J.F. Pharmacokinetics of xanthohumol and metabolites in rats after oral and intravenous administration. Mol. Nutr. Food Res. 2012, 56, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Wolff, H.; Motyl, M.; Hellerbrand, C.; Heilmann, J.; Kraus, B. Xanthohumol uptake and intracellular kinetics in hepatocytes, hepatic stellate cells, and intestinal cells. J. Agric. Food Chem. 2011, 59, 12893–12901. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Nikolic, D.; Zhu, D.; Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R.; van Breemen, R.B. Binding of the hop (Humulus lupulus L.) chalcone xanthohumol to cytosolic proteins in Caco-2 intestinal epithelial cells. Mol. Nutr. Food Res. 2007, 51, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Arczewska, M.; Kamiński, D.M.; Górecka, E.; Pociecha, D.; Rój, E.; Sławińska-Brych, A.; Gagoś, M. The molecular organization of prenylated flavonoid xanthohumol in DPPC multibilayers: X-ray diffraction and FTIR spectroscopic studies. Biochim. Biophys. Acta–Biomembr. 2013, 1828, 213–222. [Google Scholar] [CrossRef]
- Wesolowska, O.; Gasiorowska, J.; Petrus, J.; Czarnik-Matusewicz, B.; Michalak, K. Interaction of prenylated chalcones and flavanones from common hop with phosphatidylcholine model membranes. Biochim. Biophys. Acta 2014, 1, 173–184. [Google Scholar] [CrossRef]
- Vanhoecke, B.W.; Delporte, F.; van Braeckel, E.; Heyerick, A.; Depypere, H.T.; Nuytinck, M.; de Keukeleire, D.; Bracke, M.E. A safety study of oral tangeretin and xanthohumol administration to laboratory mice. In Vivo 2005, 19, 103–107. [Google Scholar] [PubMed]
- Dorn, C.; Bataille, F.; Gaebele, E.; Heilmann, J.; Hellerbrand, C. Xanthohumol feeding does not impair organ function and homoeostasis in mice. Food Chem. Toxicol. 2010, 48, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- Hussong, R.; Frank, N.; Knauft, J.; Ittrich, C.; Owen, R.; Becker, H.; Gerhäuser, C. A safety study of oral xanthohumol administration and its influence on fertility in Sprague Dawley rats. Mol. Nutr. Food Res. 2005, 49, 861–867. [Google Scholar] [CrossRef] [PubMed]
- XAN—An elixier for life. Available online: http://www.ta-xanag.com/en/xanthohumol/grundlagen (accessed on 28 August 2014).
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Hansen, P.E.; Wang, G.; Qiu, L.; Dong, J.; Yin, H.; Qian, Z.; Yang, M.; Miao, J. Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus). Molecules 2015, 20, 754-779. https://doi.org/10.3390/molecules20010754
Liu M, Hansen PE, Wang G, Qiu L, Dong J, Yin H, Qian Z, Yang M, Miao J. Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus). Molecules. 2015; 20(1):754-779. https://doi.org/10.3390/molecules20010754
Chicago/Turabian StyleLiu, Ming, Poul Erik Hansen, Genzhu Wang, Lin Qiu, Jianjun Dong, Hua Yin, Zhonghua Qian, Mei Yang, and Jinlai Miao. 2015. "Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus)" Molecules 20, no. 1: 754-779. https://doi.org/10.3390/molecules20010754
APA StyleLiu, M., Hansen, P. E., Wang, G., Qiu, L., Dong, J., Yin, H., Qian, Z., Yang, M., & Miao, J. (2015). Pharmacological Profile of Xanthohumol, a Prenylated Flavonoid from Hops (Humulus lupulus). Molecules, 20(1), 754-779. https://doi.org/10.3390/molecules20010754






