Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isothermal Crystallization Study
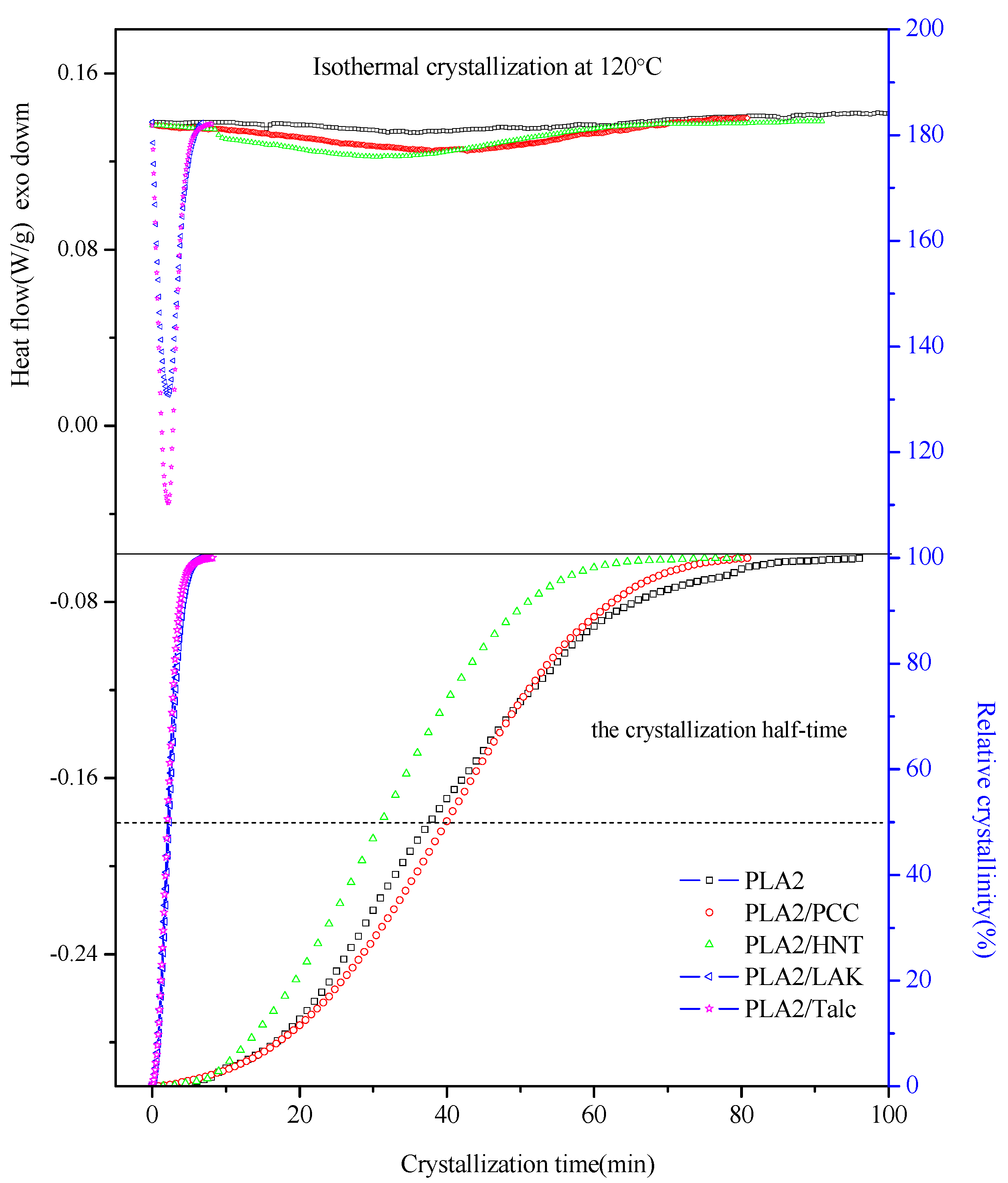
| Sample Code | ∆Hc (J/g) | Xc (%) | n | log(k) | t0.5 (min) |
|---|---|---|---|---|---|
| PLA2002D | / | / | 2.45 | −4.03 | 38 |
| PLA2/PCC | 29.14 | 31.3 | 2.56 | −4.23 | 40 |
| PLA2/HNT | 28.58 | 30.7 | 2.44 | −3.79 | 31 |
| PLA2/talc | 29.50 | 31.7 | 2.05 | −0.81 | 2.1 |
| PLA2/LAK | 25.43 | 27.3 | 2.04 | −0.89 | 2.2 |
2.2. Non-Isothermal Crystallization Study
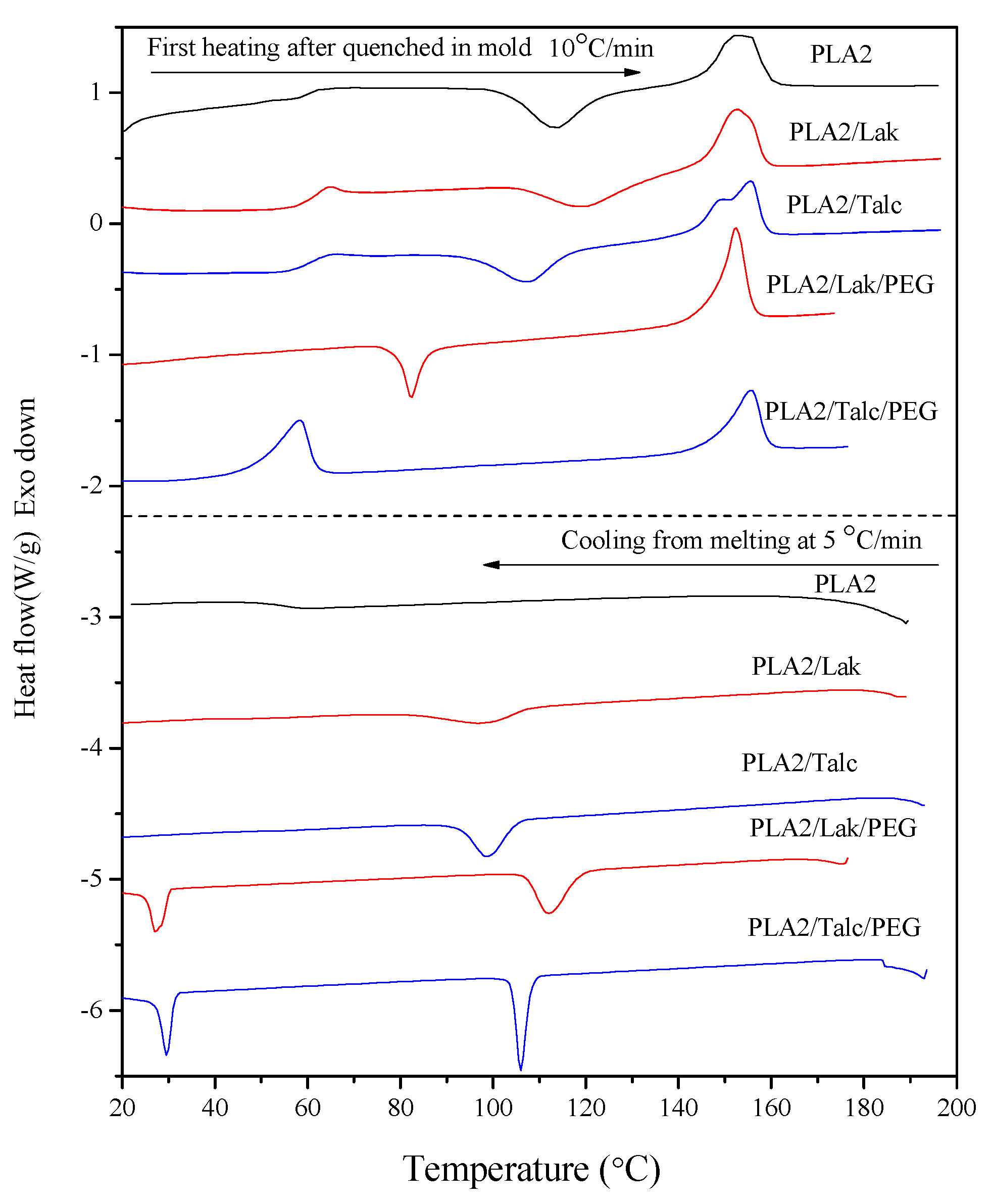
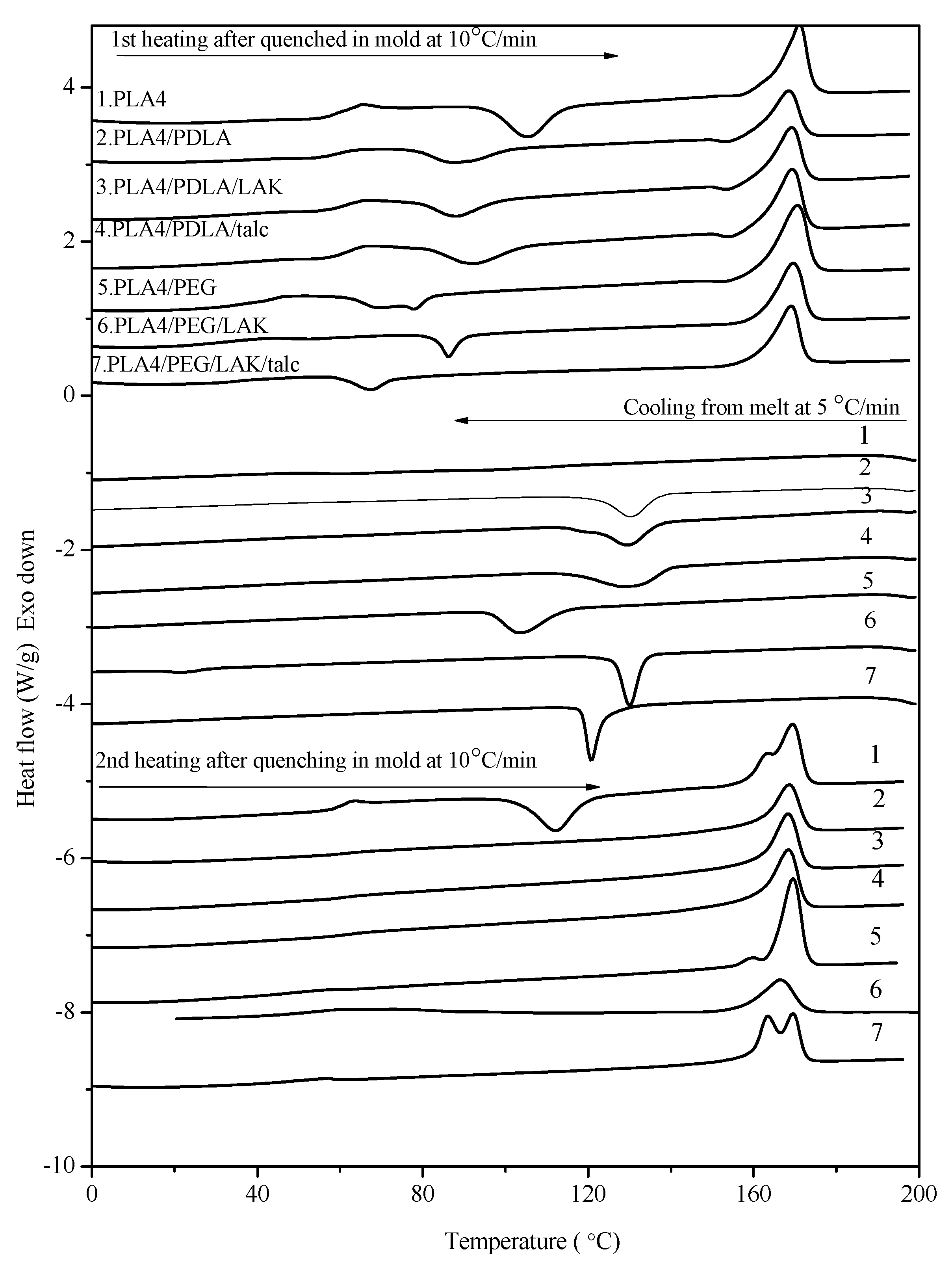
2.3. DMTA Tests
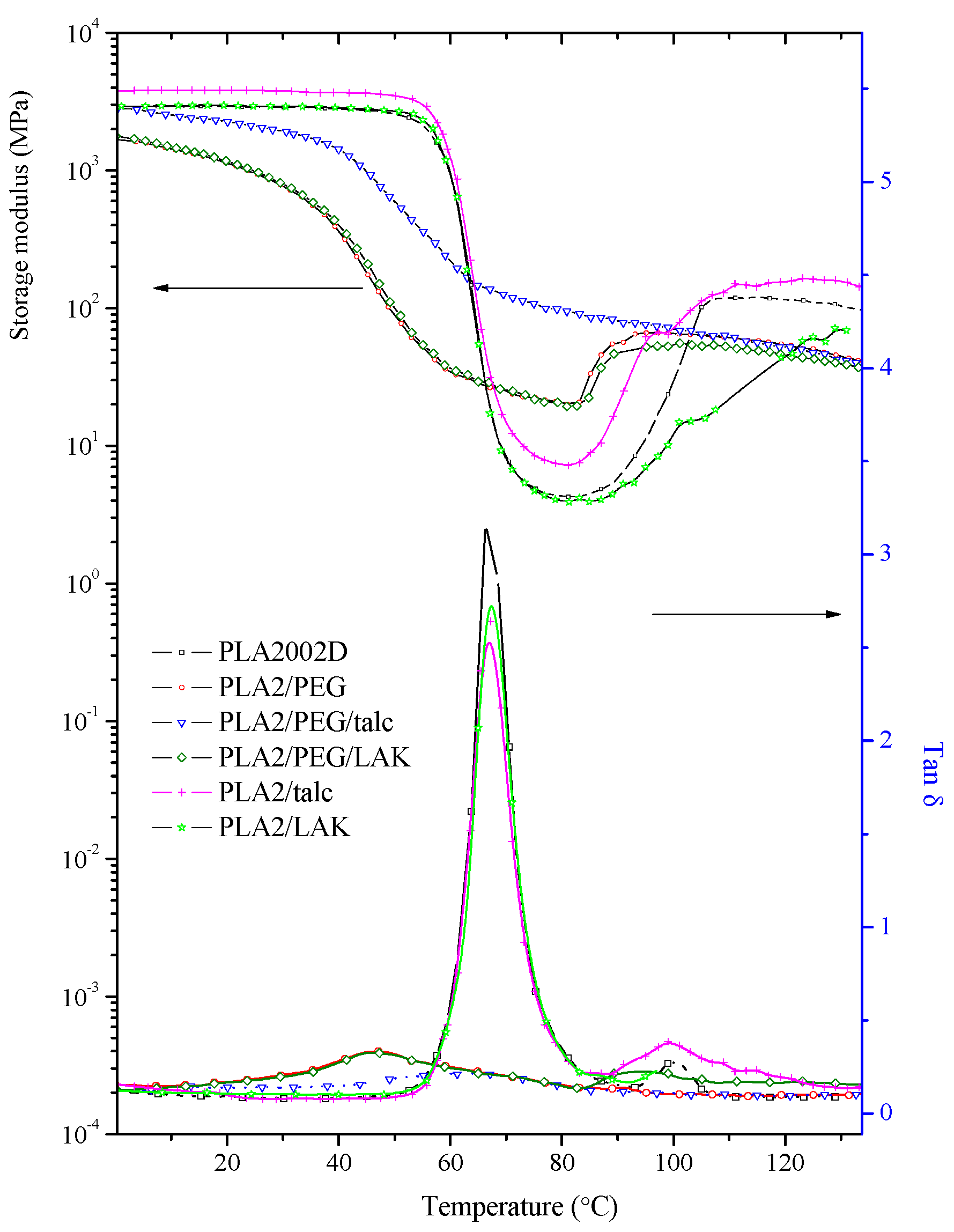
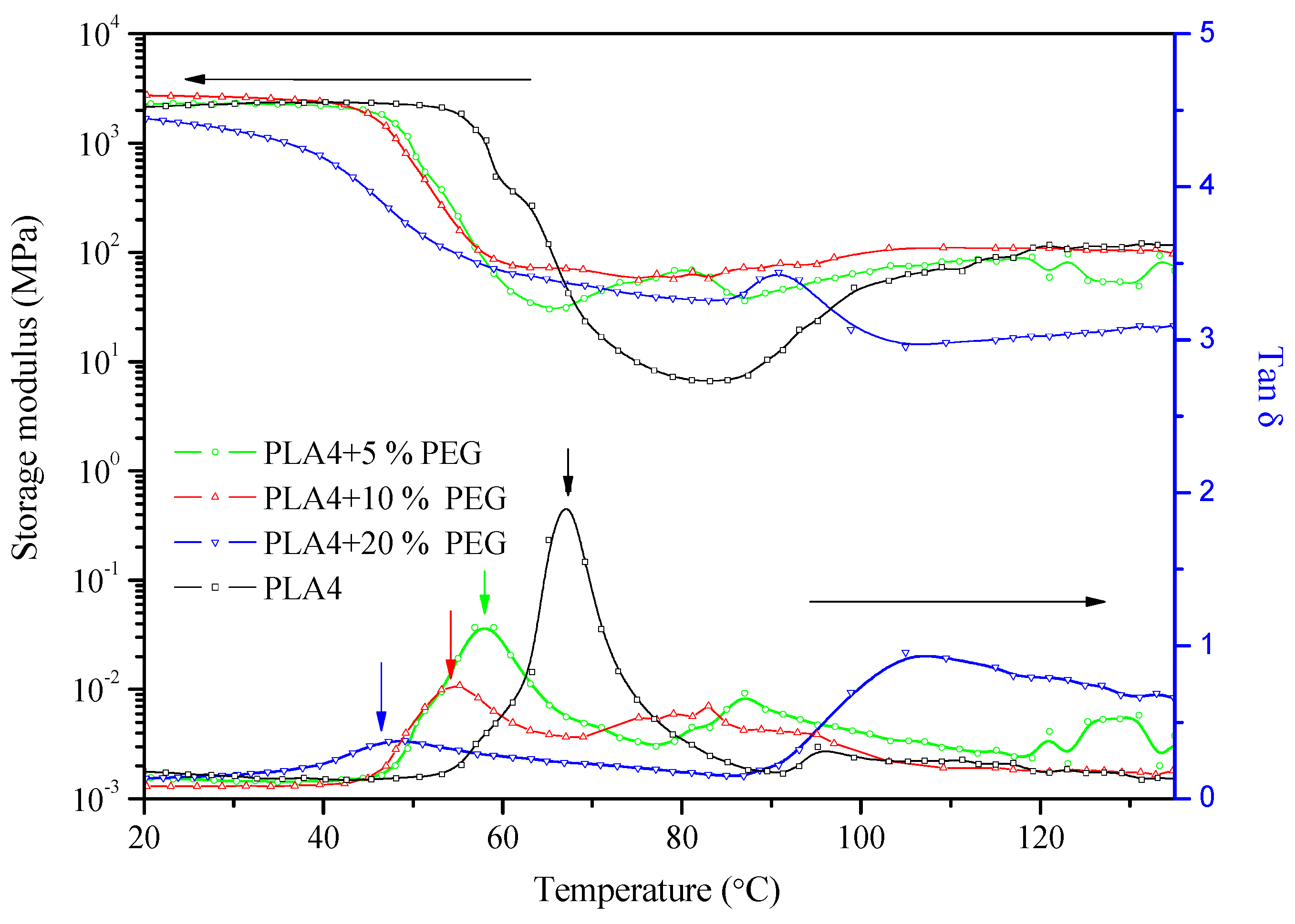
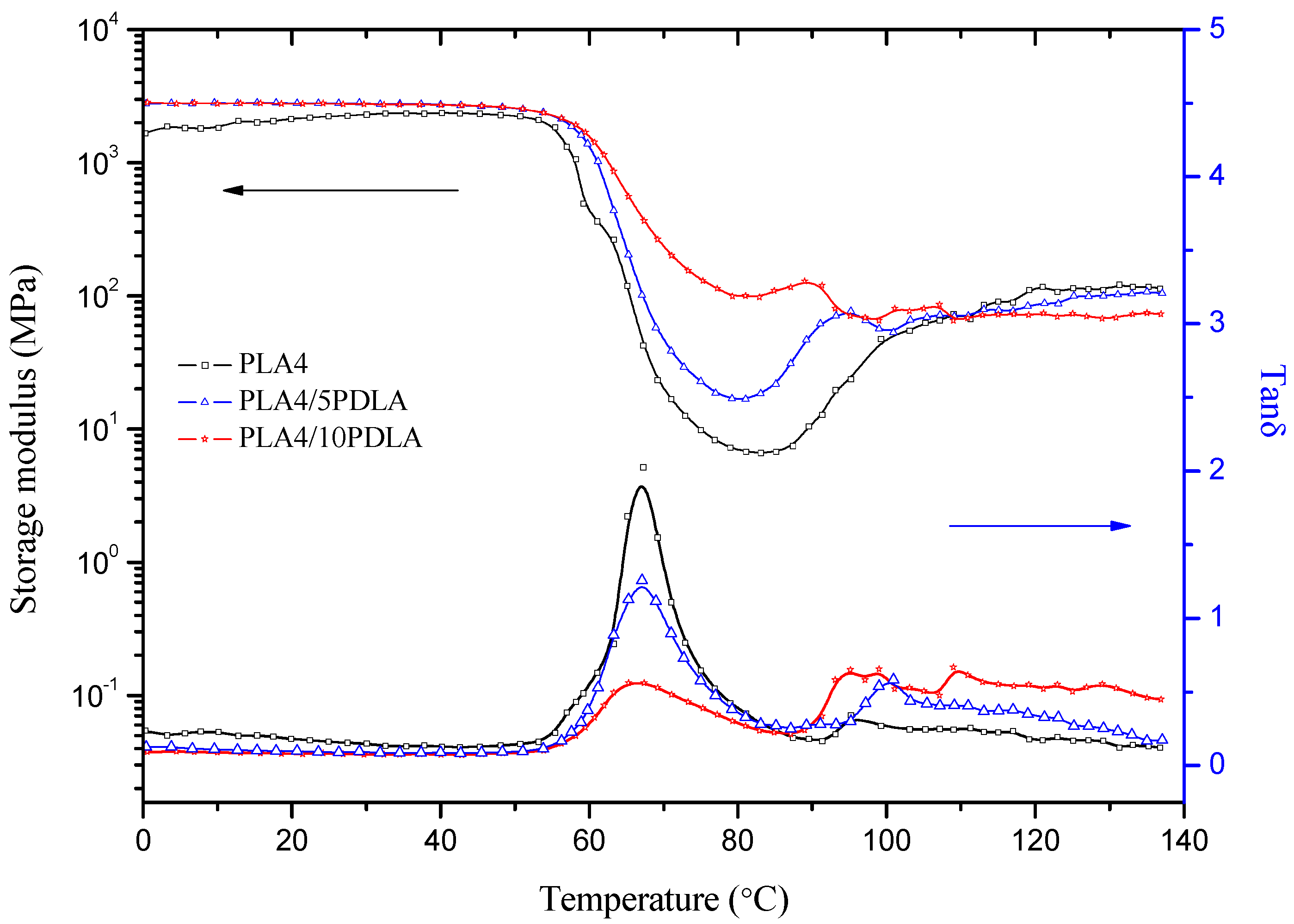
2.4. Mechanical Properties
| PLA Sample Code | Composition | Young’s Modulus (GPa) | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|---|
| PLA2 | 100 | 3.48 | 65.15 | 3.29 |
| PLA2/LAK | 99/1 | 3.98 | 64.37 | 5.30 |
| PLA2/talc | 95/5 | / | 59.87 | 2.04 |
| PLA2/PEG | 80/20 | / | 29.59 | 202.7 |
| PLA2/PEG/LAK | 79/20/1 | 1.42 | 29.44 | 177.0 |
| PLA2/PEG/talc | 75/20/5 | / | 35.64 | 132.6 |
| PLA4 | 100 | 3.83 | 69.06 | 2.48 |
| PLA4/PDLA | 99/1 | 3.65 | 71.57 | 4.30 |
| PLA4/PDLA | 95/5 | 3.61 | 67.40 | 2.98 |
| PLA4/PDLA | 90/10 | 4.19 | 68.79 | 3.32 |
| PLA4/PEG | 95/5 | 3.55 | 58.45 | 2.83 |
| PLA4/PEG | 90/10 | 3.16 | 53.83 | 10.25 |
| PLA4/PEG | 80/20 | 1.12 | 20.08 | 174.0 |
| PLA4/PDLA/LAK | 94/5/1 | 3.18 | 64.45 | 5.34 |
| PLA4/PDLA/talc | 90/5/5 | 3.86 | 64.22 | 2.28 |
| PLA/PEG/PDLA | 79/20/1 | 1.08 | 30.40 | 210.30 |
| PLA/PEG/LAK | 79/20/1 | 1.02 | 31.90 | 195.20 |
| PLA/PEG/LAK/talc | 79/10/1/10 | 4.24 | 46.10 | 174.88 |
3. Experimental Section
3.1. Materials
3.2. Sample Preparation
3.3. Characterization
3.3.1. Thermal Properties
3.3.2. Mechanical Properties
3.3.3. Dynamic Mechanical Properties
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly(lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Gandini, A. Polymers from Renewable Resources: A Challenge for the Future of Macromolecular Materials. Macromolecules 2008, 41, 9491–9504. [Google Scholar] [CrossRef]
- Lim, L.T.; Auras, R.; Rubino, M. Processing technologies for poly(lactic acid). Prog. Polym. Sci. 2008, 33, 820–852. [Google Scholar] [CrossRef]
- Inkinen, S.; Hakkarainen, M.; Albertsson, A.C.; Södergard, A. From Lactic Acid to Poly(lactic acid) (PLA): Characterization and Analysis of PLA and Its Precursors. Biomacromolecules 2011, 12, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.P.; Kumar, V. New emerging trends in synthetic biodegradable polymers—Polylactide: A critique. Eur. Polym. J. 2007, 43, 4053–4074. [Google Scholar] [CrossRef]
- Pan, P.J.; Inoue, Y. Polymorphism and isomorphism in biodegradable polyesters. Prog. Polym. Sci. 2009, 34, 605–640. [Google Scholar] [CrossRef]
- Pan, P.J.; Zhu, B.; Kai, W.H.; Dong, T.; Inoue, Y. Polymorphic Transition in Disordered Poly(l-lactide) Crystals Induced by Annealing at Elevated Temperatures. Macromolecules 2008, 41, 4296–4304. [Google Scholar] [CrossRef]
- Li, H.B.; Huneault, M.A. Effect of nucleation and plasticization on the crystallization of poly(lactic acid). Polymer 2007, 48, 6855–6866. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Achilias, D.S.; Nanaki, S.; Beslikas, T.; Bikiaris, D. PLA nanocomposites: Effect of filler type on non-isothermal crystallization. Thermochim. Acta 2010, 511, 129–139. [Google Scholar] [CrossRef]
- Li, Y.L.; Wu, H.Y.; Wang, Y.; Liu, L.; Han, L.; Wu, J.; Xiang, F.M. Synergistic Effects of PEG and MWCNTs on Crystallization Behavior of PLLA. J. Polym. Sci. Part B: Polym. Phys. 2010, 48, 520–528. [Google Scholar] [CrossRef]
- Yamane, H.; SaSai, K. Effect of the addition of poly(d-lactic acid) on the thermal property of poly(l-lactic acid). Polymer 2003, 44, 2569–2575. [Google Scholar] [CrossRef]
- Nampoothiri, K.M.; Nair, N.R.; Pappy John, R. An overview of the recent developments in polylactide (PLA) research. Bioresour. Technol. 2010, 101, 8493–8501. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Z.; Chen, F.; Liu, B.; Estep, G.; Zhang, J.W. Super Toughened Poly(lactic acid) Ternary Blends by Simultaneous Dynamic Vulcanization and Interfacial Compatibilization. Macromolecules 2010, 43, 6058–6066. [Google Scholar] [CrossRef]
- Piorkowska, E.; Kulinski, Z.; Galeski, A.; Masirek, R. Plasticization of semicrystalline poly(l-lactide) with poly(propylene glycol). Polymer 2006, 47, 7178–7188. [Google Scholar] [CrossRef]
- Kulinski, Z.; Piorkowska, E.; Gadzinowska, K.; Stasiak, M. Plasticization of poly(l-lactide) with Poly(propylene glycol). Biomacromolecules 2006, 7, 2128–2135. [Google Scholar] [CrossRef] [PubMed]
- Ljungberg, N.; Wesslén, B. Preparation and Properties of Plasticized Poly(lactic acid) Films. Biomacromolecules 2005, 6, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, H.; Takai, H.; Saha, K.S. Isothermal and non-isothermal crystallization behavior of poly(l-lactic acid): Effects of stereocomplex as nucleating agent. Polymer 2006, 47, 3826–3837. [Google Scholar] [CrossRef]
- Nijenhuis, A.J.; Colstee, E.; Grijpma, D.W.; Pennings, A.J. High molecular weight poly(l-lactide) and poly(ethylene oxide) blends: Thermal characterization and physical properties. Polymer 1996, 34, 5849–5857. [Google Scholar] [CrossRef]
- Fischer, E.W.; Sterzel, H.J.; Wegner, G. Investigation of the structure of solution grown crystals of lactide copolymers by means of chemical reactions. Colloid Polym. Sci. 1973, 251, 980–990. [Google Scholar]
- Kulinski, Z.; Piorkowska, E. Crystallization, structure and properties of plasticized poly(l-lactide). Polymer 2005, 46, 10290–10300. [Google Scholar] [CrossRef]
- Lee, S.H.; Wang, S.Q.; Teramoto, Y. Isothermal crystallization behavior of hybrid biocomposite consisting of regenerated cellulose fiber, clay, and poly(lactic acid). J. Appl. Polym. Sci. 2008, 108, 870–875. [Google Scholar] [CrossRef]
- Schmidt, S.C.; Hillmyer, M.A. Polylactide stereocomplex crystallites as nucleating agents for isotactic polylactide. J. Polym. Sci. Part B: Polym. Phys. 2001, 39, 300–313. [Google Scholar] [CrossRef]
- Chan, C.M.; Wu, J.S.; Li, J.X.; Cheung, Y.K. Isothermal crystallization behavior of hybrid biocomposite consisting of regenerated cellulose fiber, clay, and poly(lactic acid). Polymer 2002, 43, 2981–2992. [Google Scholar] [CrossRef]
- Shi, X.T.; Rosa, R.; Lazzeri, A. On the Coating of Precipitated Calcium Carbonate with Stearic Acid in Aqueous Medium. Langmuir 2010, 26, 8474–8482. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the PLA composites are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, X.; Zhang, G.; Phuong, T.V.; Lazzeri, A. Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid). Molecules 2015, 20, 1579-1593. https://doi.org/10.3390/molecules20011579
Shi X, Zhang G, Phuong TV, Lazzeri A. Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid). Molecules. 2015; 20(1):1579-1593. https://doi.org/10.3390/molecules20011579
Chicago/Turabian StyleShi, Xuetao, Guangcheng Zhang, Thanh Vu Phuong, and Andrea Lazzeri. 2015. "Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid)" Molecules 20, no. 1: 1579-1593. https://doi.org/10.3390/molecules20011579
APA StyleShi, X., Zhang, G., Phuong, T. V., & Lazzeri, A. (2015). Synergistic Effects of Nucleating Agents and Plasticizers on the Crystallization Behavior of Poly(lactic acid). Molecules, 20(1), 1579-1593. https://doi.org/10.3390/molecules20011579





