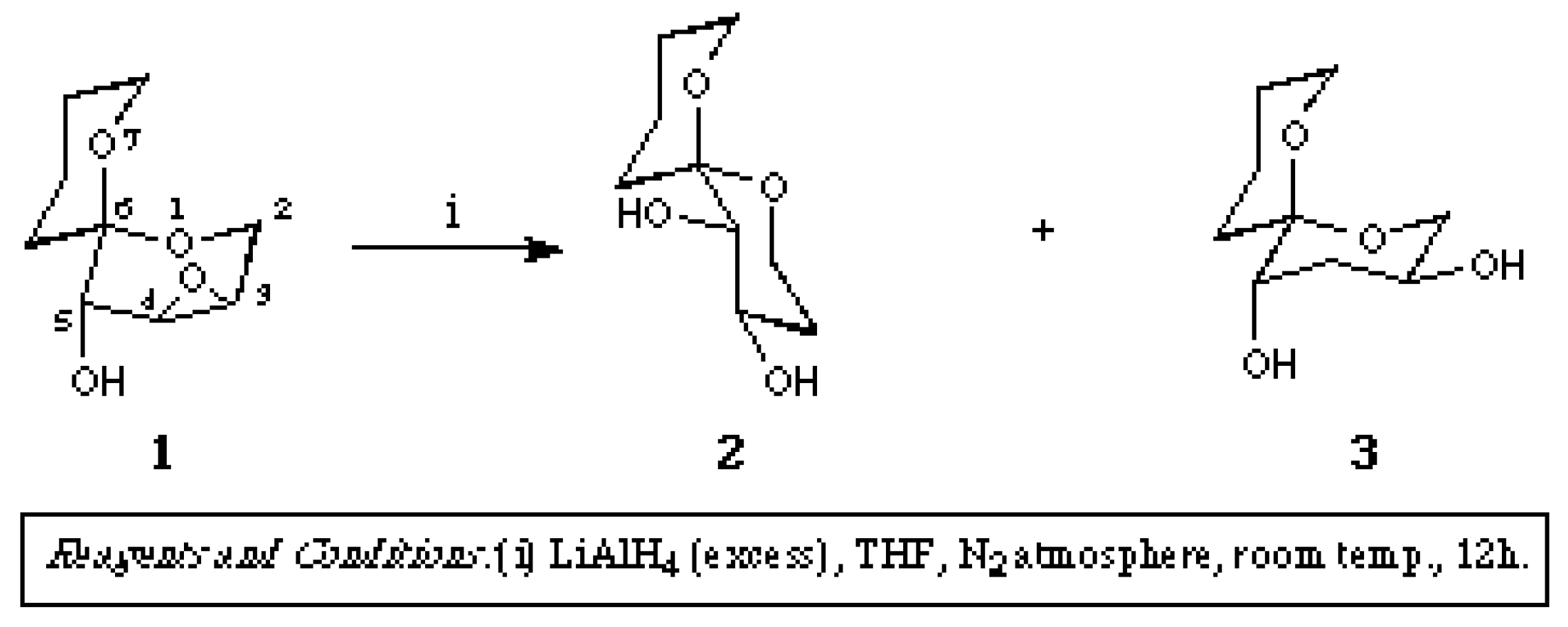
[4R*,5R*,6S*]-1,7-Dioxaspiro[5.5]undecane-4,5-diol
Supplementary materials
Supplementary File 1Supplementary File 2Acknowledgment:
References and Notes
- Another product (3 in the Scheme, a colourless glass, 33mg, 6%) will be reported in the following short note. Brimble, M. A.; Johnston, A. D. Molecules 1997, 2, M20.
- Sample Availability: No sample available.
© 1997 MDPI. All rights reserved
Share and Cite
Brimble, M.A.; Johnston, A.D. [4R*,5R*,6S*]-1,7-Dioxaspiro[5.5]undecane-4,5-diol. Molecules 1997, 2, M19. https://doi.org/10.3390/M19
Brimble MA, Johnston AD. [4R*,5R*,6S*]-1,7-Dioxaspiro[5.5]undecane-4,5-diol. Molecules. 1997; 2(6):M19. https://doi.org/10.3390/M19
Chicago/Turabian StyleBrimble, Margaret A, and Andrew D Johnston. 1997. "[4R*,5R*,6S*]-1,7-Dioxaspiro[5.5]undecane-4,5-diol" Molecules 2, no. 6: M19. https://doi.org/10.3390/M19
APA StyleBrimble, M. A., & Johnston, A. D. (1997). [4R*,5R*,6S*]-1,7-Dioxaspiro[5.5]undecane-4,5-diol. Molecules, 2(6), M19. https://doi.org/10.3390/M19



