Nopal Cactus (Opuntia ficus-indica) as a Source of Bioactive Compounds for Nutrition, Health and Disease
Abstract
:1. Introduction
2. General Compound Content of Cactus
3. Individual Classes of Cactus Compounds and Related Biological Activities
3.1. Phenolic Compounds
| Plant tissue | Main Component Identified | Content in mg/100 g | References |
|---|---|---|---|
| Flower | Gallic acid | 1630–4900 | [20,25,26,27] |
| Quercetin 3-O-Rutinoside | 709 | ||
| 4 Kaempferol 3-O-Rutinoside | 400 | ||
| 5 Quercetin 3-O-Glucoside | 447 | ||
| 6 Isorhamnetin 3-O-Robinobioside | 4269 | ||
| 7 Isorhamnetin 3-O-Galactoside | 979 | ||
| 8 Isorhamnetin 3-O-Glucoside | 724 | ||
| 9 Kaempferol 3-O-Arabinoside | 324 | ||
| Pulp | Total phenolic acid | 218.8 | [13,28,29,31,32,38] |
| Quercetin | 9 | ||
| Isorhamnetin | 4.94 | ||
| Kaempferol | 0.78 | ||
| Luteolin | 0.84 | ||
| isorhamnetin glycosides | 50.6 | ||
| Kaempferol | 2.7 | ||
| Seed | Total phenolic acid | 48–89 | [33] |
| Feruloyl-sucrose isomer 1 | 7.36–17.62 | ||
| Feruloyl-sucrose isomer 2 | 2.9–17.1 | ||
| Sinapoyl-diglucoside | 12.6–23.4 | ||
| Total Flavonoids | 1.5–2.6 | ||
| Total Tannins | 4.1–6.6 | ||
| Skin fruits | Total phenolic acid | 45,700 | [5,30,34] |
| Total Flavonoid | 6.95 | ||
| Kaempferol | 0.22 | ||
| Quercetin | 4.32 | ||
| Isorhamnetin | 2.41–91 | ||
| Cladode | Gallic acid | 0.64–2.37 | [4,29,35,36,37] |
| Coumaric | 14.08–16.18 | ||
| 3,4-dihydroxybenzoic | 0.06–5.02 | ||
| 4-hydroxybenzoic | 0.5–4.72 | ||
| Ferulicacid | 0.56–34.77 | ||
| Salicylicacid | 0.58–3.54 | ||
| Isoquercetin | 2.29–39.67 | ||
| Isorhamnetin-3-O-glucoside | 4.59–32.21 | ||
| Nicotiflorin | 2.89–146.5 | ||
| Rutin | 2.36–26.17 | ||
| Narcissin | 14.69–137.1 |
3.2. Fatty Acids
| Fatty Acid | C12:0 | C14:0 | C16:0 | C16:1 | C18:0 | C18:1 | C18:2 | C18:3 | C20:0 | C22:0 | C22:1 | C24:0 | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cladode | 1.33 | 1.96 | 13.87 | 0.24 | 3.33 | 11.16 | 34.87 | 33.23 | - | - | - | - | [50] |
| Cactus seed oil | - | - | 20.1 | 1.80 | 2.72 | 18.3 | 53.5 | 2.58 | - | - | - | - | [52] |
| Cactus seed oil | - | - | 9.32 | 1.42 | 3.11 | 16.77 | 70.29 | nd | - | - | - | - | [53] |
| Fruits pulp oil | - | 1.13 | 34.4 | 1.62 | 2.37 | 10.8 | 37 | 12.68 | - | - | - | - | [52] |
| Prickly pear peel | 0.71 | 1.95 | 23.1 | 2.48 | 2.67 | 24.1 | 32.3 | 9.27 | nd | 0.5 | - | 0.41 | [21] |
| Argan oil | - | 0.10 | 11.7 | 0.14 | 4.9 | 36.6 | 31.3 | 0.09 | 0.33 | 0.12 | - | 0.06 | [54] |
| Olive oil | - | 11.5 | 0.9 | 1.4 | 61.9 | 3.8 | 1.1 | 0.23 | - | - | - | - | [55] |
| Grape seed oil | - | 0.06 | 8.3 | 0.1 | 3 | 12 | 67.6 | 0.3 | 0.2 | 0.1 | 0.02 | 0.01 | [56] |
| Soybean oil | - | - | 6 | 0.4 | 2.2 | 26.1 | 50.1 | 14.5 | - | - | - | - | [57] |
| Corn oil | - | - | 13.4 | Traces | 1.5 | 27.4 | 56 | 0.9 | 0.2 | - | - | - | [58] |
| Sunflower oil | - | 0.08 | 7.4 | 0.09 | 4.56 | 25.17 | 60.15 | 0.3 | - | - | - | 0.34 | [59] |
3.3. Vitamins
| Pulp | Seeds | Skin | Cladode | Source | |
|---|---|---|---|---|---|
| Vitamin K1 | 53.2 | 52.5 | 109 | ---- | [5,21,28,31,32,60,66] |
| Vitamin C, | 34–40 | ---- | ---- | 7–22 | |
| Vitamin B1 | ---- | ---- | ---- | 0.14 | |
| Vitamin B2 | ---- | ---- | ---- | 0.60 | |
| Vitamin B3 | ---- | ---- | ---- | 0.46 | |
| α-Tocopherol, | 84.9 | 56 | 1760 | ---- | |
| β-Tocopherol, | 12.6 | 12 | 222 | ---- | |
| γ-Tocopherol, | 7.9 | 33 | 174 | ---- | |
| σ-Tocopherol | 422 | 5 | 26 | ---- | |
| Total vitamin E | 527.4 | 106 | 2182 | ---- |
3.4. Sterols
| Main Component Identified | Pulp | Seed | Skin | References |
|---|---|---|---|---|
| Campesterol | 8.74 | 1.66 | 8.76 | [21,60] |
| Stigmasterol | 0.73 | 0.30 | 2.12 | |
| Lanosterol | 0.76 | 0.28 | 1.66 | |
| β-Sitosterol | 11.2 | 6.75 | 21.1 | |
| Δ5-Avenasterol, Δ7-Avenasterol | 1.43 | 0.29 | 2.71 | |
| Δ7-Avenasterol | ---- | 0.05 | ---- | |
| Ergosterol | ---- | ---- | 0.68 |
3.5. Mineral Compounds
| Main component identified | Pulp | Seed | Cladode | References |
|---|---|---|---|---|
| Calcium | 27.6 | 16.2 | 5.64–17.95 | [65,66,70,71,72,74] |
| Calcium oxalate | ---- | ---- | 11.5 to 4.3 | |
| Magnesium | 27.7 | 74.8 | 8.80 | |
| Sodium | 0.8 | 67.6 | 0.3–0.4 | |
| Potassium | 161 | 163 | 2.35–55.20 | |
| Iron | 1.5 | 9.45 | 0.09 | |
| Phosphorus | ---- | 152 | 0.15–2.59 | |
| Zinc | ---- | 1.45 | 0.08 | |
| Copper | ---- | 0.32 | ---- | |
| Manganese | ---- | Trace | 0.19–0.29 |
3.6. Amino Acids
| Amino Acid | Cladode | Fruit | Seeds | References |
|---|---|---|---|---|
| Alanine | 1.25 | 3.17 | 4.75 | [65,73] |
| Arginine | 5.01 | 1.11 | 6.63 | |
| Asparagine | 3.13 | 1.51 | Trace | |
| Asparaginic acid | 4.38 | Trace | 10.42 | |
| Glutamic acid | 5.43 | 2.40 | 21.68 | |
| Glutamine | 36.12 | 12.59 | Trace | |
| Cystine | 1.04 | 0.41 | 0.37 | |
| Histidine | 4.18 | 1.64 | 3.11 | |
| Isoleucine | 3.97 | 1.13 | 6.20 | |
| Leucine | 2.71 | 0.75 | 9.94 | |
| Lysine | 5.22 | 0.63 | 6.79 | |
| Methionine | 2.92 | 2.01 | 0.70 | |
| Phenylalanine | 3.55 | 0.85 | 5.25 | |
| Serine | 6.68 | 6.34 | 8.46 | |
| Threonine | 4.18 | 0.48 | 1.53 | |
| Tyrosine | 1.46 | 0.45 | 3.09 | |
| Tryptophane | 1.04 | 0.46 | Trace | |
| Valine | 7.72 | 1.43 | 6.02 | |
| α-Aminobutyric acid | Trace | 0.04 | Trace | |
| Carnosine | Trace | 0.21 | Trace | |
| Citrulline | Trace | 0.59 | Trace | |
| Ornithine | Trace | Trace | Trace | |
| Proline | Trace | 46.00 | Trace | |
| Taurine | Trace | 15.79 | Trace | |
| Glycine | Trace | Trace | 5.06 |
4. Cactus and Compounds in Nutritional and Medical Practice
4.1. Cactus in Nutrition and Prevention of Disease
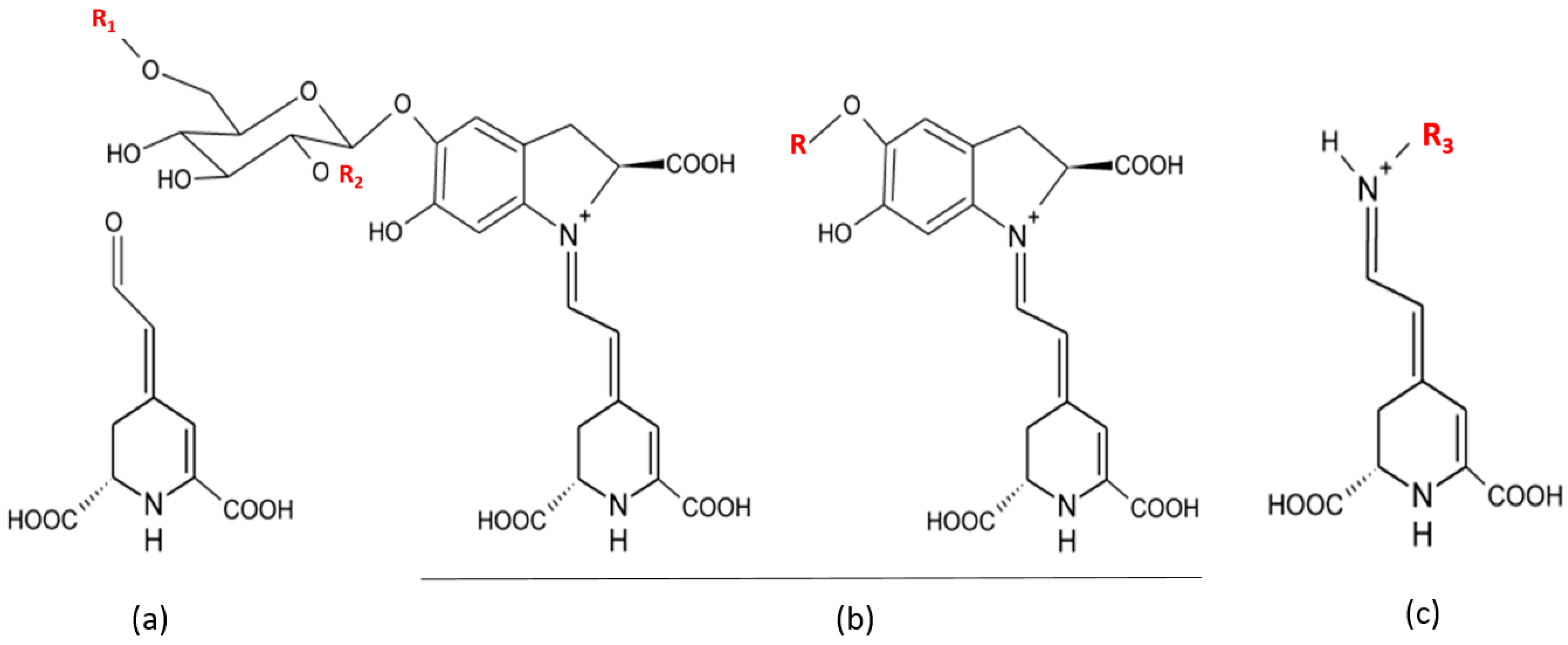
| Compounds | Name | Radical | Opuntia Specie | References |
|---|---|---|---|---|
| Betaxanthins | Portulacaxanthin I | R3 = hydroxyproline | O. ficus-indica | [78] |
| Portulacaxanthin III | R3 = glycine | O. ficus-indica | [78,81] | |
| Muscaaurin | R3 = histidine | O. robusta, O. ficus-indica, O. megacantha | [78,82] | |
| Indicaxanthin | R3 = proline | O. robusta Wendl, O. robusta, O. streptacantha Lemaire, O. ficus-indica, O. megacantha, O. albi-carpa | [12,78] | |
| (S)-serine-betaxanthin | R3 = serine | O. ficus-indica | [78,81] | |
| (S)-valine-betaxanthin | R3 = valine | O. ficus-indica | [81] | |
| (S)-isoleucine-betaxanthin | R3 = isoleucine | O. ficus-indica | [81] | |
| γ-Aminobutyric acid-Bx | R = butyric acid | O. spp | [82] | |
| Methionine-betaxanthin | R3 = methionine | O. spp | [82] | |
| (S)- Phenylalaine-betaxanthin | R3 = phenylalaine | O. ficus-indica | [81] | |
| Vulgaxanthin I | R3 = glutamine | O. robusta Wendl, O. ficus-indica | [81] | |
| Vulgaxantin Ii | R = glutamic acid | O. streptacantha; O. beta vulgaris L.spp. V. Pablo; O. bergeriana; O. ficus indica; O. alba-carba; O. robusta and O. Spp | [82] | |
| Vulgaxantin III | R = asparagine | O. streptacantha; O. beta vulgaris L.spp. V. Pablo; O. alba-carba; O. robusta and O. Spp | [82] | |
| Vulgaxanthin IV | R3 = leucine | O. streptacantha; O. beta vulgaris; O. alba-carba; O. robusta Wendl, O. ficus-indica | [78,81] | |
| Miraxanthin II | R3 = aspartic acid | O. bergeriana; O. ficus indica | [12] | |
| Betacyanins | Betanin | R1 = R2 = H | O. robusta Wendl, O. robusta, O. streptacantha Lemaire, O. ficus-indica, O. megacantha, O. albi-carpa, O. xoconostle | [8,12,78,82] |
| iso-Betanin | R1 = R2 = H | O. robusta Wendl, O. robusta, O. streptacantha Lemaire, O. ficus-indica, O. xoconostle | [8,78,82] | |
| Betanidin | R = H | O. robusta Wendl, O. robusta, O. streptacantha Lemaire, O. ficus-indica, O. megacantha, O. xoconostle | [8,12,78,82] | |
| Gomphrenin i | R1 = R2 = H | O. robusta Wendl, O. robusta, O. ficus-indica | [78,82] | |
| Phyllocactin | R1 = malonyl R2 = H | O. xoconostle | [8,12,81,82] |
4.2. Cactus in Health and Disease
4.3. Cactus Use in Traditional Medicine
5. Medical Relevance of Cactus Compounds: The State of the Art
5.1. Experimental Models and Randomized Trials
5.2. Pharmacological Potentials of Antioxidant and Antiinflammatory Effects of Cactus
5.2.1. In Vitro Studies (on Intact Cells)
5.2.2. In Vivo Studies (on the Whole Animals)
5.3. Pharmacological Potentialities of Cactus Effects on Non-Alcoholic Fatty Liver Disease
5.4. Pharmacological Potentials of Antimicrobial Activities of Cactus
5.5. Pharmacological Potentials in Targeting Alcoholism with Cactus Extracts
5.6. Side Effects Caused by Cactus Compounds
6. Conclusions
| Biological Activity | Source of Cactus Products | In Vivo and in Vitro Models | References |
|---|---|---|---|
| Hypolipidemic and Hypocholesterolemic | Cladodes powder | Rats | [14] |
| Cladodes (Glycoproteine) | Mice | [100] | |
| Seeds powder and seeds oil | Rats | [53] | |
| Anti-diabetic | Capsule: cladode and fruit skin extract | Human | [101] |
| Cactus powder in capsule | Human (Man and women) | [102] | |
| Aqueous extract of the cladode and fruit and mixture | Rats | [103] | |
| Cladode and fruit skin extract capsule | Man | [104] | |
| Hypoglycemic | Polysaccharide extract from the cladode | Rats | [105] |
| Extract powder racket after drying | Rats | [106] | |
| Anti-Inflammatory | Indicaxanthin, from fruit | Human intestinal epithelial cell line (Caco-2 cells) stimulated by cytokine IL-1b | [88] |
| Lyophilized extracts of cladodes | Human chondrocyte cultures stimulated with IL-1β | [89] | |
| Indicaxanthin from Cactus Pear Fruit | Rat Pleurisy obtained by injection of 0.2 ml of λ-carrageenin into the pleural cavity | [93] | |
| Methanol extract of cactus stems (active substance: β-sitosterol) | Mice (male) | [107] | |
| Methanolic extracts of prickly pear fruits (Betalain Indicaxanthin) | In vitro study of the interaction between purified Betalains and HOCL and human myeloperoxidase | [93,108] | |
| Anti-Inflammatory and Antioxidant | Butanol and methanol fruit extract | In vivo studies in gerbils and In vitro studies in cultured mouse cortical cells | [92] |
| Antioxidant | Betalain a pigment purified from fresh pulp of cactus pear | Endothelial cells human umbilical vein (HUVEC) | [90] |
| Betanin prickly pear fruit Extracts | Chemical and biological (human RBC, LDL) systems | [1] | |
| Ethanol extract of the stem | Chemical and biological systems (mouse splenocytes) | [22] | |
| Flavonoid fraction of juice of whole fruits | Rats | [18] | |
| Glycoprotein (90 kDa) isolated from Opuntia ficus-indica var. saboten MAKINO | Mice induced by Triton WR-1339 | [100] | |
| Cactus pear fruit | Healthy humans (10 women and 8 men) supplemented with cactus pear or Vit C | [109] | |
| Quercetine ether 3-O-méthyl isolated from Opuntia ficus-indica var. saboten | Primary cultured rat cortical cells | [17] | |
| Antimicrobial | Methanol extract of cladode | Bacteria: Campylobacter jejuni and Campylobacter coli | [95] |
| Methanolic, ethanolic, and aqueous extracts of cladode | Bacteria: Vibrio cholerae | [96] | |
| Hexane extracts from flowers | Bacteria: Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa and Bacillus subtilis | [110] | |
| Aqueous and alcoholic extracts of cladode | Bacteria: Proteus mirabilis | [111] |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Butera, D.; Tesoriere, L.; di Gaudio, F.; Bongiorno, A.; Allegra, M.; Pintaudi, A.M.; Kohen, R.; Livrea, M.A. Antioxidant activities of sicilian prickly pear ( Opuntia ficus indica ) fruit extracts and reducing properties of its betalains: Betanin and indicaxanthin. J. Agric. Food Chem. 2002, 50, 6895–6901. [Google Scholar]
- Alimi, H.; Hfaiedh, N.; Bouoni, Z.; Hfaiedh, M.; Sakly, M.; Zourgui, L.; Rhouma, K.B. Antioxidant and antiulcerogenic activities of Opuntia ficus indica f. inermis root extract in rats. Phytomedicine 2010, 17, 1120–1126. [Google Scholar]
- Morales, P.; Ramírez-Moreno, E.; de Cortes Sanchez-Mata, M.; Carvalho, A.M.; Ferreira, I.C.F.R. Nutritional and antioxidant properties of pulp and seeds of two xoconostle cultivars (Opuntia joconostle F.A.C. Weber ex Diguet and Opuntia matudae Scheinvar) of high consumption in Mexico. Food Res. Int. 2012, 46, 279–285. [Google Scholar]
- Valente, L.M.M.; da Paixão, D.; do Nascimento, A.C.; dos Santos, P.F.P.; Scheinvar, L.A.; Moura, M.R.L.; Tinoco, L.W.; Gomes, L.N.F.; da Silva, J.F.M. Antiradical activity, nutritional potential and flavonoids of the cladodes of Opuntia monacantha (Cactaceae). Food Chem. 2010, 123, 1127–1131. [Google Scholar]
- Kuti, J.O. Antioxidant compounds from four Opuntia cactus pear fruit varieties. Food Chem. 2004, 85, 527–533. [Google Scholar]
- Valente, L.; Scheinvar, L.; da Silva, G.; Antunes, A.; dos Santos, F.; Oliveira, T.; Tappin, M.; Aquino Neto, F.; Pereira, A.; Carvalhaes, S.; et al. Evaluation of the antitumor and trypanocidal activities and alkaloid profile in species of Brazilian Cactaceae. Pharmacogn. Mag. 2007, 3, 167–172. [Google Scholar]
- Yang, N.; Zhao, M.; Zhu, B.; Yang, B.; Chen, C.; Cui, C.; Jiang, Y. Anti-diabetic effects of polysaccharides from Opuntia monacantha cladode in normal and streptozotocin-induced diabetic rats. Innov. Food Sci. Emerg. Technol. 2008, 9, 570–574. [Google Scholar]
- Osorio-Esquivel, O.; Alicia-Ortiz-Moreno; Álvarez, V.B.; Dorantes-Álvarez, L.; Giusti, M.M. Phenolics, betacyanins and antioxidant activity in Opuntia joconostle fruits. Food Res. Int. 2011, 44, 2160–2168. [Google Scholar]
- Paiz, R.C.; Juárez-Flores, B.I.; Aguirre, R.J.R.; Cárdenas, O.C.; Reyes, A.J.A.; García, C.E.; Álvarez, F.G. Glucose-lowering effect of xoconostle (Opuntia joconostle A. Web. Cactaceae) in diabetic rats. J. Med. Plants Res. 2010, 4, 2326–2333. [Google Scholar]
- Schaffer, S.; Schmitt-Schillig, S.; Müller, W.E.; Eckert, G.P. Antioxidant properties of Mediterranean food plant extracts: Geographical differences. J. Physiol. Pharmacol. 2005, 56 (Suppl. S1), 115–124. [Google Scholar]
- Stintzing, F.C.; Schieber, A.; Carle, R. Evaluation of colour properties and chemical quality parameters of cactus juices. Eur. Food Res. Technol. 2003, 216, 303–311. [Google Scholar]
- Stintzing, F.C.; Schieber, A.; Carle, R. Phytochemical and nutritional significance of cactus pear. Eur. Food Res. Technol. 2001, 212, 396–407. [Google Scholar]
- Galati, E.M.; Mondello, M.R.; Giuffrida, D.; Dugo, G.; Miceli, N.; Pergolizzi, S.; Taviano, M.F. Chemical characterization and biological effects of Sicilian Opuntia ficus indica (L.) mill. Fruit juice: Antioxidant and antiulcerogenic activity. J. Agric. Food Chem. 2003, 51, 4903–4908. [Google Scholar]
- Galati, E.M.; Mondello, M.R.; Monforte, M.T.; Galluzzo, M.; Miceli, N.; Tripodo, M.M. Effect of Opuntia ficus-indica (L.) Mill. cladodes in the wound-healing process. J. Prof. Assoc. Cactus Dev. 2003, 5, 1–16. [Google Scholar]
- Tesoriere, L.; Allegra, M.; Butera, D.; Livrea, M.A. Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: Potential health effects of betalains in humans. Am. J. Clin. Nutr. 2004, 80, 941–945. [Google Scholar]
- Zou, D.-M.; Brewer, M.; Garcia, F.; Feugang, J.M.; Wang, J.; Zang, R.; Liu, H.; Zou, C. Cactus pear: a natural product in cancer chemoprevention. Nutr. J. 2005, 4, 25. [Google Scholar]
- Dok-Go, H.; Lee, K.H.; Kim, H.J.; Lee, E.H.; Lee, J.; Song, Y.S.; Lee, Y.-H.; Jin, C.; Lee, Y.S.; Cho, J. Neuroprotective effects of antioxidative flavonoids, quercetin, (+)-dihydroquercetin and quercetin 3-Methyl ether, isolated from Opuntia ficus-indica var. saboten. Brain Res. 2003, 965, 130–136. [Google Scholar]
- Galati, E.M.; Mondello, M.R.; Lauriano, E.R.; Taviano, M.F.; Galluzzo, M.; Miceli, N. Opuntia ficus indica (L.) Mill. fruit juice protects liver from carbon tetrachloride-induced injury. Phytother. Res. 2005, 19, 796–800. [Google Scholar]
- Sreekanth, D.; Arunasree, M.K.; Roy, K.R.; Chandramohan Reddy, T.; Reddy, G.V.; Reddanna, P. Betanin a betacyanin pigment purified from fruits of Opuntia ficus-indica induces apoptosis in human chronic myeloid leukemia Cell line-K562. Phytomedicine 2007, 14, 739–746. [Google Scholar]
- De Leo, M.; Abreu, M.B.D.; Pawlowska, A.M.; Cioni, P.L.; Braca, A. Profiling the chemical content of Opuntia ficus-indica flowers by HPLC–PDA-ESI-MS and GC/EIMS analyses. Phytochem. Lett. 2010, 3, 48–52. [Google Scholar]
- Ramadan, M.F.; Mörsel, J.-T. Oil cactus pear (Opuntia ficus-indica L.). Food Chem. 2003, 82, 339–345. [Google Scholar]
- Lee, J.-C.; Kim, H.-R.; Kim, J.; Jang, Y.-S. Antioxidant property of an ethanol extract of the stem of Opuntia ficus-indica var. saboten. J. Agric. Food Chem. 2002, 50, 6490–6496. [Google Scholar]
- Stintzing, F.C.; Carle, R. Cactus stems (Opuntia spp.): A review on their chemistry, technology, and uses. Mol. Nutr. Food Res. 2005, 49, 175–194. [Google Scholar]
- Laughton, M.J.; Evans, P.J.; Moroney, M.A.; Hoult, J.R.; Halliwell, B. Inhibition of mammalian 5-lipoxygenase and cyclo-oxygenase by flavonoids and phenolic dietary additives. Relationship to antioxidant activity and to iron ion-reducing ability. Biochem. Pharmacol. 1991, 42, 1673–1681. [Google Scholar]
- Ahmed, M.S.; Tanbouly, N.D.E.; Islam, W.T.; Sleem, A.A.; Senousy, A.S.E. Antiinflammatory flavonoids from Opuntia dillenii (Ker-Gawl) Haw. flowers growing in Egypt. Phytother. Res. 2005, 19, 807–809. [Google Scholar]
- Ammar, I.; Ennouri, M.; Khemakhem, B.; Yangui, T.; Attia, H. Variation in chemical composition and biological activities of two species of Opuntia flowers at four stages of flowering. Ind. Crop. Prod. 2012, 37, 34–40. [Google Scholar]
- Clark, W.D.; Brown, G.K.; Mays, R.L. Flower flavonoids of Opuntia subgenus Cylindropuntia. Phytochemistry 1980, 19, 2042–2043. [Google Scholar]
- Fernández-López, J.A.; Almela, L.; Obón, J.M.; Castellar, R. Determination of Antioxidant Constituents in Cactus Pear Fruits. Plant Food Hum. Nutr. 2010, 65, 253–259. [Google Scholar]
- Bensadón, S.; Hervert-Hernández, D.; Sáyago-Ayerdi, S.G.; Goñi, I. By-Products of Opuntia ficus-indica as a Source of Antioxidant Dietary Fiber. Plant Food Hum. Nutr. 2010, 65, 210–216. [Google Scholar]
- Moussa-Ayoub, T.E.; El-Samahy, S.K.; Kroh, L.W.; Rohn, S. Identification and quantification of flavonol aglycons in cactus pear (Opuntia ficus indica) fruit using a commercial pectinase and cellulase preparation. Food Chem. 2011, 124, 1177–1184. [Google Scholar]
- Salim, N.; Abdelwaheb, C.; Rabah, C.; Ahcene, B. Chemical composition of Opuntia ficus-indica (L.) fruit. Afr. J. Biotechnol. 2009, 8, 1623–1624. [Google Scholar]
- Tesoriere, L.; Fazzari, M.; Allegra, M.; Livrea, M.A. Biothiols, Taurine, and Lipid-Soluble Antioxidants in the Edible Pulp of Sicilian Cactus Pear (Opuntia ficus-indica) Fruits and Changes of Bioactive Juice Components upon Industrial Processing. J. Agric. Food Chem. 2005, 53, 7851–7855. [Google Scholar]
- Chougui, N.; Tamendjari, A.; Hamidj, W.; Hallal, S.; Barras, A.; Richard, T.; Larbat, R. Oil composition and characterisation of phenolic compounds of Opuntia ficus-indica seeds. Food Chem. 2013, 139, 796–803. [Google Scholar]
- Jorge, A.J.; de La Garza, T.H.; Alejandro, Z.C.; Ruth, B.C.; Noé, A.C. The optimization of phenolic compounds extraction from cactus pear (Opuntia ficus-indica) skin in a reflux system using response surface methodology. Asian Pac. J. Trop. Biomed. 2013, 3, 436–442. [Google Scholar]
- Gallegos-Infante, J.-A.; Rocha-Guzman, N.-E.; González-Laredo, R.-F.; Reynoso-Camacho, R.; Medina-Torres, L.; Cervantes-Cardozo, V. Effect of air flow rate on the polyphenols content and antioxidant capacity of convective dried cactus pear cladodes (Opuntia ficus indica ). Int. J. Food Sci. Nutr. 2009, 60, 80–87. [Google Scholar]
- Ginestra, G.; Parker, M.L.; Bennett, R.N.; Robertson, J.; Mandalari, G.; Narbad, A.; Lo Curto, R.B.; Bisignano, G.; Faulds, C.B.; Waldron, K.W. Anatomical, Chemical, and Biochemical Characterization of Cladodes from Prickly Pear [Opuntia ficus-indica (L.) Mill.]. J. Agric. Food Chem. 2009, 57, 10323–10330. [Google Scholar]
- Guevara-Figueroa, T.; Jiménez-Islas, H.; Reyes-Escogido, M.L.; Mortensen, A.G.; Laursen, B.B.; Lin, L.-W.; de León-Rodríguez, A.; Fomsgaard, I.S.; Barba de la Rosa, A.P. Proximate composition, phenolic acids, and flavonoids characterization of commercial and wild nopal (Opuntia spp.). J. Food Compos. Anal. 2010, 23, 525–532. [Google Scholar]
- Khatabi, O.; Hanine, H.; Elothmani, D.; Hasib, A. Extraction and determination of polyphenols and betalain pigments in the Moroccan Prickly pear fruits (Opuntia ficus indica). Arab. J. Chem. 2013. [Google Scholar] [CrossRef]
- Khan, N.S.; Ahmad, A.; Hadi, S.M. Anti-oxidant, pro-oxidant properties of tannic acid and its binding to DNA. Chem. Biol. Interact. 2000, 125, 177–189. [Google Scholar]
- Yen, G.-C.; Duh, P.-D.; Tsai, H.-L. Antioxidant and pro-oxidant properties of ascorbic acid and gallic acid. Food Chem. 2002, 79, 307–313. [Google Scholar]
- You, B.R.; Park, W.H. Gallic acid-induced lung cancer cell death is related to glutathione depletion as well as reactive oxygen species increase. Toxicol. In Vitro 2010, 24, 1356–1362. [Google Scholar]
- Li, R.; Guo, M.; Zhang, G.; Xu, X.; Li, Q. Nicotiflorin reduces cerebral ischemic damage and upregulates endothelial nitric oxide synthase in primarily cultured rat cerebral blood vessel endothelial cells. J. Ethnopharmacol. 2006, 107, 143–150. [Google Scholar]
- Nakayama, M.; Aihara, M.; Chen, Y.-N.; Araie, M.; Tomita-Yokotani, K.; Iwashina, T. Neuroprotective effects of flavonoids on hypoxia-, glutamate-, and oxidative stress–induced retinal ganglion cell death. Mol. Vis. 2011, 17, 1784. [Google Scholar]
- Huang, J.-L.; Fu, S.-T.; Jiang, Y.-Y.; Cao, Y.-B.; Guo, M.-L.; Wang, Y.; Xu, Z. Protective effects of Nicotiflorin on reducing memory dysfunction, energy metabolism failure and oxidative stress in multi-infarct dementia model rats. Pharmacol. Biochem. Behav. 2007, 86, 741–748. [Google Scholar]
- Kim, J.-E.; Lee, D.-E.; Lee, K.W.; Son, J.E.; Seo, S.K.; Li, J.; Jung, S.K.; Heo, Y.-S.; Mottamal, M.; Bode, A.M.; et al. Isorhamnetin Suppresses Skin Cancer through Direct Inhibition of MEK1 and PI3-K. Cancer Prev. Res. 2011, 4, 582–591. [Google Scholar]
- Zhang, N.; Pei, F.; Wei, H.; Zhang, T.; Yang, C.; Ma, G.; Yang, C. Isorhamnetin protects rat ventricular myocytes from ischemia and reperfusion injury. Exp. Toxicol. Pathol. 2011, 63, 33–38. [Google Scholar]
- Kim, B.; Choi, Y.-E.; Kim, H.-S. Eruca sativa and its Flavonoid Components, Quercetin and Isorhamnetin, Improve Skin Barrier Function by Activation of Peroxisome Proliferator-Activated Receptor (PPAR)-α and Suppression of Inflammatory Cytokines. Phytother. Res. 2014. [Google Scholar] [CrossRef]
- Lee, J.; Jung, E.; Lee, J.; Kim, S.; Huh, S.; Kim, Y.; Kim, Y.; Byun, S.Y.; Kim, Y.-S.; Park, D. Isorhamnetin represses adipogenesis in 3T3-L1 cells. Obesity 2009, 17, 226–232. [Google Scholar]
- Ramachandran, L.; Manu, K.A.; Shanmugam, M.K.; Li, F.; Siveen, K.S.; Vali, S.; Kapoor, S.; Abbasi, T.; Surana, R.; Smoot, D.T.; et al. Isorhamnetin inhibits proliferation and invasion and induces apoptosis through the modulation of peroxisome proliferator-activated receptor γ activation pathway in gastric cancer. J. Biol. Chem. 2012, 287, 38028–38040. [Google Scholar]
- Abidi, S.; Ben Salem, H.; Vasta, V.; Priolo, A. Supplementation with barley or spineless cactus (Opuntia ficus indica f. inermis) cladodes on digestion, growth and intramuscular fatty acid composition in sheep and goats receiving oaten hay. Small Rumin. Res. 2009, 87, 9–16. [Google Scholar]
- Charouf, Z.; Guillaume, D. Phenols and Polyphenols from Argania spinosa. Am. J. Food Technol. 2007, 2, 679–683. [Google Scholar]
- Ramadan, M.F.; Moersel, J.-T. Lipid profile of prickly pear pulp fractions. J. Food Agric. Environ. 2003, 1, 66–70. [Google Scholar]
- Ennouri, M.; Evelyne, B.; Laurence, M.; Hamadi, A. Fatty acid composition and rheological behaviour of prickly pear seed oils. Food Chem. 2005, 93, 431–437. [Google Scholar]
- Marfil, R.; Cabrera-Vique, C.; Gimenez, R.; Bouzas, P.R.; Martinez, O.; Sanchez, J.A. Metal content and physicochemical parameters used as quality criteria in virgin argan oil: Influence of the extraction method. J. Agric. Food Chem. 2008, 56, 7279–7284. [Google Scholar]
- Ollivier, D.; Artaud, J.; Pinatel, C.; Durbec, J.P.; Guerere, M. Triacylglycerol and fatty acid compositions of French virgin olive oils. Characterization by chemometrics. J. Agric. Food Chem. 2003, 51, 5723–5731. [Google Scholar]
- Rubio, M.; Alvarez-Ortí, M.; Alvarruiz, A.S.; Fernández, E.; Pardo, J.E. Characterization of Oil Obtained from Grape Seeds Collected during Berry Development. J. Agric. Food Chem. 2009, 57, 2812–2815. [Google Scholar]
- Ayorinde, F.O.; Garvin, K.; Saeed, K. Determination of the fatty acid composition of saponified vegetable oils using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 608–615. [Google Scholar]
- Karoui, I.J.; Wannes, W.A.; Marzouk, B. Refined corn oil aromatization by Citrus aurantium peel essential oil. Ind. Crop. Prod. 2010, 32, 202–207. [Google Scholar]
- Filip, S.; Hribar, J.; Vidrih, R. Influence of natural antioxidants on the formation of trans-fatty-acid isomers during heat treatment of sunflower oil. Eur. J. Lipid Sci. Technol. 2011, 113, 224–230. [Google Scholar]
- Ramadan, M.F.; Mörsel, J.-T. Recovered lipids from prickly pear [Opuntia ficus-indica (L.) Mill] peel: A good source of polyunsaturated fatty acids, natural antioxidant vitamins and sterols. Food Chem. 2003, 83, 447–456. [Google Scholar]
- Soel, S.M.; Choi, O.S.; Bang, M.H.; Yoon Park, J.H.; Kim, W.K. Influence of conjugated linoleic acid isomers on the metastasis of colon cancer cells in vitro and in vivo. J. Nutr. Biochem. 2007, 18, 650–657. [Google Scholar]
- Cayuela, J.A.; Rada, M.; del Carmen Pérez-Camino, M.; Benaissa, M.; Abdelaziz, E.; Guinda, Á. Characterization of artisanally and semiautomatically extracted argan oils from Morocco. Eur. J. Lipid Sci. Technol. 2008, 110, 1159–1166. [Google Scholar]
- Khallouki, F.; Younos, C.; Soulimani, R.; Oster, T.; Charrouf, Z.; Spiegelhalder, B.; Bartsch, H.; Owen, R.W. Consumption of argan oil (Morocco) with its unique profile of fatty acids, tocopherols, squalene, sterols and phenolic compounds should confer valuable cancer chemopreventive effects. Eur. J. Cancer Prev. 2003, 12, 67–75. [Google Scholar]
- Piga, A. Cactus pear: A fruit of nutraceutical and functional importance. J. Prof. Assoc. Cactus Dev. 2004, 6, 9–22. [Google Scholar]
- Feugang, J.M.; Konarski, P.; Zou, D.; Stintzing, F.C.; Zou, C. Nutritional and medicinal use of Cactus pear (Opuntia spp.) cladodes and fruits. Front. Biosci. 2006, 11, 2574–2589. [Google Scholar]
- Sawaya, W.N.; Khan, P. Chemical Characterization of Prickly Pear Seed Oil, Opuntia ficus-indica. J. Food Sci. 1982, 47, 2060–2061. [Google Scholar]
- Gharby, S.; Harhar, H.; Guillaume, D.; Haddad, A.; Matthäus, B.; Charrouf, Z. Oxidative stability of edible argan oil: A two-year study. LWT-Food Sci. Technol. 2011, 44, 1–8. [Google Scholar]
- El Kossori, R.L.; Villaume, C.; El Boustani, E.; Sauvaire, Y.; Méjean, L. Composition of pulp, skin and seeds of prickly pears fruit (Opuntia ficus indica sp.). Plant Food Hum. Nutr. 1998, 52, 263–270. [Google Scholar]
- Sawaya, W.N.; Khatchadourian, H.A.; Safi, W.M.; Al-Muhammad, H. Chemical characterization of prickly pear pulp, Opuntia ficus-indica, and the manufacturing of prickly pear jam. Int. J. Food Sci. Technol. 1983, 18, 183–193. [Google Scholar]
- Medina, E.M.D.; Rodríguez, E.M.R.; Romero, C.D. Chemical characterization of Opuntia dillenii and Opuntia ficus indica fruits. Food Chem. 2007, 103, 38–45. [Google Scholar]
- Ayadi, M.A.; Abdelmaksoud, W.; Ennouri, M.; Attia, H. Cladodes from Opuntia ficus indica as a source of dietary fiber: Effect on dough characteristics and cake making. Ind. Crop. Prod. 2009, 30, 40–47. [Google Scholar]
- Trachtenberg, S.; Mayer, A.M. Mucilage Cells, Calcium Oxalate Crystals and Soluble Calcium in Opuntia ficus-indica. Ann. Bot. 1982, 50, 549–557. [Google Scholar]
- Sawaya, W.N.; Khalil, J.K.; Al-Mohammad, M.M. Nutritive value of prickly pear seeds, Opuntia ficus-indica. Plant Food Hum. Nutr. 1983, 33, 91–97. [Google Scholar]
- Contreras-Padilla, M.; Pérez-Torrero, E.; Hernández-Urbiola, M.I.; Hernández-Quevedo, G.; del Real, A.; Rivera-Muñoz, E.M.; Rodríguez-García, M.E. Evaluation of oxalates and calcium in nopal pads (Opuntia ficus-indica var. redonda) at different maturity stages. J. Food Compos. Anal. 2011, 24, 38–43. [Google Scholar]
- Nassar, A.G. Chemical composition and functional properties of prickly pear (Opuntia ficus indica) seeds flour and protein concentrate. World J. Dairy Food Sci. 2008, 3, 11–16. [Google Scholar]
- Uchoa, A.F.; Souza, P.A.S.; Zarate, R.M.L.; Gomes-Filho, E.; Campos, F.A.P. Isolation and characterization of a reserve protein from the seeds of Opuntia ficus-indica (Cactaceae). Braz. J. Med. Biol. Res. 1998, 31, 757–761. [Google Scholar]
- Zito, P.; Sajeva, M.; Bruno, M.; Rosselli, S.; Maggio, A.; Senatore, F. Essential oils composition of two Sicilian cultivars of Opuntia ficus-indica (L.) Mill. (Cactaceae) fruits (prickly pear). Nat. Prod. Res. 2013, 27, 1305–1314. [Google Scholar]
- Castellanos-Santiago, E.; Yahia, E.M. Identification and Quantification of Betalains from the Fruits of 10 Mexican Prickly Pear Cultivars by High-Performance Liquid Chromatography and Electrospray Ionization Mass Spectrometry. J. Agric. Food Chem. 2008, 56, 5758–5764. [Google Scholar]
- Fernández-López, J.A.; Castellar, R.; Obón, J.M.; Almela, L. Screening and mass-spectral confirmation of betalains in cactus pears. Chromatographia 2002, 56, 591–595. [Google Scholar]
- Fernández-López, J.A.; Giménez, P.J.; Angosto, J.M.; Moreno, J.I. A process of recovery of a natural yellow colourant from opuntia fruits. Food Technol. Biotechnol. 2012, 50, 246–251. [Google Scholar]
- Strack, D.; Vogt, T.; Schliemann, W. Recent advances in betalain research. Phytochemistry 2003, 62, 247–269. [Google Scholar]
- Stintzing, F.C.; Herbach, K.M.; Mosshammer, M.R.; Carle, R.; Yi, W.; Sellappan, S.; Akoh, C.C.; Bunch, R.; Felker, P. Color, betalain pattern, and antioxidant properties of cactus pear (Opuntia spp.) clones. J. Agric. Food Chem. 2005, 53, 442–451. [Google Scholar]
- Ahmad, A.; Davies, J.; Randall, S.; Skinner, G.R.B. Antiviral properties of extract of Opuntia streptacantha. Antivir. Res. 1996, 30, 75–85. [Google Scholar]
- Kaur, M.; Kaur, A.; Sharma, R. Pharmacological actions of Opuntia ficus indica: A Review. J. Appl. Pharm. Sci. 2012, 2, 1. [Google Scholar]
- Lee, J.-A.; Jung, B.-G.; Kim, T.-H.; Lee, S.-G.; Park, Y.-S.; Lee, B.-J. Dietary feeding of Opuntia humifusa inhibits UVB radiation-induced carcinogenesis by reducing inflammation and proliferation in hairless mouse model. Photochem. Photobiol. 2013, 89, 1208–1215. [Google Scholar]
- Lee, J.-A.; Jung, B.-G.; Lee, B.-J. Inhibitory effects of Opuntia humifusa on 7, 12-dimethyl- benz[a]anthracene and 12-O-tetradecanoylphorbol-13- acetate induced two-stage skin carcinogenesis. Asian Pac. J. Cancer Prev. 2012, 13, 4655–4660. [Google Scholar]
- Tomczyk, M.; Zovko-Koncić, M.; Chrostek, L. Phytotherapy of alcoholism. Nat. Prod. Commun. 2012, 7, 273–280. [Google Scholar]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Gentile, C.; Livrea, M.A. Indicaxanthin inhibits NADPH oxidase (NOX)-1 activation and NF-κB-dependent release of inflammatory mediators and prevents the increase of epithelial permeability in IL-1β-exposed Caco-2 cells. Br. J. Nutr. 2014, 111, 415–423. [Google Scholar]
- Panico, A.M.; Cardile, V.; Garufi, F.; Puglia, C.; Bonina, F.; Ronsisvalle, S. Effect of hyaluronic acid and polysaccharides from Opuntia ficus indica (L.) cladodes on the metabolism of human chondrocyte cultures. J. Ethnopharmacol. 2007, 111, 315–321. [Google Scholar]
- Gentile, C.; Tesoriere, L.; Allegra, M.; Livrea, M.A.; D’Alessio, P. Antioxidant betalains from cactus pear (Opuntia ficus-indica) inhibit endothelial ICAM-1 expression. Ann. N. Y. Acad. Sci. 2004, 1028, 481–486. [Google Scholar]
- Lee, M.H.; Kim, J.Y.; Yoon, J.H.; Lim, H.J.; Kim, T.H.; Jin, C.; Kwak, W.-J.; Han, C.-K.; Ryu, J.-H. Inhibition of nitric oxide synthase expression in activated microglia and peroxynitrite scavenging activity by Opuntia ficus indica var. saboten. Phytother. Res. 2006, 20, 742–747. [Google Scholar]
- Kim, J.-H.; Park, S.-M.; Ha, H.-J.; Moon, C.-J.; Shin, T.-K.; Kim, J.-M.; Lee, N.-H.; Kim, H.-C.; Jang, K.-J.; Wie, M.-B. Opuntia ficus-indica attenuates neuronal injury in in vitro and in vivo models of cerebral ischemia. J. Ethnopharmacol. 2006, 104, 257–262. [Google Scholar]
- Allegra, M.; Ianaro, A.; Tersigni, M.; Panza, E.; Tesoriere, L.; Livrea, M.A. Indicaxanthin from cactus pear fruit exerts anti-inflammatory effects in carrageenin-induced rat pleurisy. J. Nutr. 2014, 144, 185–192. [Google Scholar]
- Morán-Ramos, S.; Avila-Nava, A.; Tovar, A.R.; Pedraza-Chaverri, J.; López-Romero, P.; Torres, N. Opuntia ficus indica (nopal) attenuates hepatic steatosis and oxidative stress in obese Zucker (fa/fa) rats. J. Nutr. 2012, 142, 1956–1963. [Google Scholar]
- Castillo, S.L.; Heredia, N.; Contreras, J.F.; García, S. Extracts of edible and medicinal plants in inhibition of growth, adherence, and cytotoxin production of Campylobacter jejuni and Campylobacter coli. J. Food Sci. 2011, 76, M421–M426. [Google Scholar]
- Sánchez, E.; García, S.; Heredia, N. Extracts of Edible and Medicinal Plants Damage Membranes of Vibrio cholerae. Appl. Environ. Microbiol. 2010, 76, 6888–6894. [Google Scholar]
- Vázquez-Ramírez, R.; Olguín-Martínez, M.; Kubli-Garfias, C.; Hernández-Muñoz, R. Reversing gastric mucosal alterations during ethanol-induced chronic gastritis in rats by oral administration of Opuntia ficus-indica mucilage. World J. Gastroenterol. 2006, 12, 4318–4324. [Google Scholar]
- Wiese, J.; McPherson, S.; Odden, M.C.; Shlipak, M.G. Effect of Opuntia ficus indica on symptoms of the alcohol hangover. Arch. Intern. Med. 2004, 164, 1334–1340. [Google Scholar]
- Kleiner, O.; Cohen, Z.; Mares, A.J. Low colonic obstruction due to Opuntia ficus indica seeds: The aftermath of enjoying delicious cactus fruits. Acta Paediatr. 2002, 91, 606–607. [Google Scholar]
- Oh, P.-S.; Lim, K.-T. Glycoprotein (90 kDa) isolated from Opuntia ficus-indica var. saboten MAKINO lowers plasma lipid level through scavenging of intracellular radicals in Triton WR-1339-induced mice. Biol. Pharm. Bull. 2006, 29, 1391–1396. [Google Scholar]
- Deldicque, L.; van Proeyen, K.; Ramaekers, M.; Pischel, I.; Sievers, H.; Hespel, P. Additive insulinogenic action of Opuntia ficus-indica cladode and fruit skin extract and leucine after exercise in healthy males. J. Int. Soc. Sports Nutr. 2013, 10, 45. [Google Scholar]
- Godard, M.P.; Ewing, B.A.; Pischel, I.; Ziegler, A.; Benedek, B.; Feistel, B. Acute blood glucose lowering effects and long-term safety of OpunDia supplementation in pre-diabetic males and females. J. Ethnopharmacol. 2010, 130, 631–634. [Google Scholar]
- Butterweck, V.; Semlin, L.; Feistel, B.; Pischel, I.; Bauer, K.; Verspohl, E.J. Comparative evaluation of two different Opuntia ficus-indica extracts for blood sugar lowering effects in rats. Phytother. Res. 2011, 25, 370–375. [Google Scholar]
- Van Proeyen, K.; Ramaekers, M.; Pischel, I.; Hespel, P. Opuntia ficus-indica ingestion stimulates peripheral disposal of oral glucose before and after exercise in healthy men. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 284–291. [Google Scholar]
- Alarcon-Aguilar, F.J.; Valdes-Arzate, A.; Xolalpa-Molina, S.; Banderas-Dorantes, T.; Jimenez-Estrada, M.; Hernandez-Galicia, E.; Roman-Ramos, R. Hypoglycemic activity of two polysaccharides isolated from Opuntia ficus-indica and O. streptacantha. Proc. West. Pharmacol. Soc. 2003, 46, 139–142. [Google Scholar]
- Nuñez-López, M.A.; Paredes-López, O.; Reynoso-Camacho, R. Functional and Hypoglycemic Properties of Nopal Cladodes (O. ficus-indica) at Different Maturity Stages Using in Vitro and in Vivo Tests. J. Agric. Food Chem. 2013, 61, 10981–10986. [Google Scholar]
- Park, E.H.; Kahng, J.H.; Lee, S.H.; Shin, K.H. An anti-inflammatory principle from cactus. Fitoterapia 2001, 72, 288–290. [Google Scholar]
- Allegra, M.; Furtmuller, P.G.; Jantschko, W.; Zederbauer, M.; Tesoriere, L.; Livrea, M.A.; Obinger, C. Mechanism of interaction of betanin and indicaxanthin with human myeloperoxidase and hypochlorous acid. Biochem. Biophys. Res. Commun. 2005, 332, 837–844. [Google Scholar]
- Tesoriere, L.; Butera, D.; Pintaudi, A.M.; Allegra, M.; Livrea, M.A. Supplementation with cactus pear (Opuntia ficus-indica) fruit decreases oxidative stress in healthy humans: A comparative study with vitamin C. Am. J. Clin. Nutr. 2004, 80, 391–395. [Google Scholar]
- Ennouri, M.; Ammar, I.; Khemakhem, B.; Attia, H. Chemical Composition and Antibacterial Activity of Opuntia Ficus-Indica F. Inermis (Cactus Pear) Flowers. J. Med. Food 2014, 17, 908–914. [Google Scholar]
- Yasmeen, R.; Hashmi, A.S.; Anjum, A.A.; Saeed, S.; Muhammad, K. Antibacterial activity of indigenous herbal extracts against urease producing bacteria. J. Anim. Plant. Sci. 2012, 22, 416–419. [Google Scholar]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Mostafa, K.; El Kharrassi, Y.; Badreddine, A.; Andreoletti, P.; Vamecq, J.; El Kebbaj, M.S.; Latruffe, N.; Lizard, G.; Nasser, B.; Cherkaoui-Malki, M. Nopal Cactus (Opuntia ficus-indica) as a Source of Bioactive Compounds for Nutrition, Health and Disease. Molecules 2014, 19, 14879-14901. https://doi.org/10.3390/molecules190914879
El-Mostafa K, El Kharrassi Y, Badreddine A, Andreoletti P, Vamecq J, El Kebbaj MS, Latruffe N, Lizard G, Nasser B, Cherkaoui-Malki M. Nopal Cactus (Opuntia ficus-indica) as a Source of Bioactive Compounds for Nutrition, Health and Disease. Molecules. 2014; 19(9):14879-14901. https://doi.org/10.3390/molecules190914879
Chicago/Turabian StyleEl-Mostafa, Karym, Youssef El Kharrassi, Asmaa Badreddine, Pierre Andreoletti, Joseph Vamecq, M'Hammed Saïd El Kebbaj, Norbert Latruffe, Gérard Lizard, Boubker Nasser, and Mustapha Cherkaoui-Malki. 2014. "Nopal Cactus (Opuntia ficus-indica) as a Source of Bioactive Compounds for Nutrition, Health and Disease" Molecules 19, no. 9: 14879-14901. https://doi.org/10.3390/molecules190914879
APA StyleEl-Mostafa, K., El Kharrassi, Y., Badreddine, A., Andreoletti, P., Vamecq, J., El Kebbaj, M. S., Latruffe, N., Lizard, G., Nasser, B., & Cherkaoui-Malki, M. (2014). Nopal Cactus (Opuntia ficus-indica) as a Source of Bioactive Compounds for Nutrition, Health and Disease. Molecules, 19(9), 14879-14901. https://doi.org/10.3390/molecules190914879








