Enhanced Antioxidant Capacity and Anti-Ageing Biomarkers after Diet Micronutrient Supplementation
Abstract
:1. Introduction
2. Results and Discussion
| Parameter | Week 0 | Week 12 |
|---|---|---|
| TG [mg/dL] | 80.05 ± 30.46 | 83.27 ± 37.77 |
| TC [mg/dL] | 211.41 ± 34.87 | 208.09 ± 36.01 |
| HDL [mg/dL] | 72.72 ± 16.51 | 72.48 ± 17.31 |
| LDL [mg/dL] | 122.73 ± 29.49 | 118.99 ± 29.93 |
| ASPAT [U/L] | 19.62 ± 5.05 | 19.20 ± 6.32 |
| ALAT [U/L] | 18.15 ± 8.65 | 17.98 ± 13.06 |
| Creatinine [mg/dL] | 0.65 ± 0.10 | 0.71 ± 0.10 |
| Vit D [ng/mL] # | 15.61 ± 6.66 | 29.16 ± 8.59 |
2.1. Redox Homeostasis of Blood after NucleVital®Q10 Complex Intake
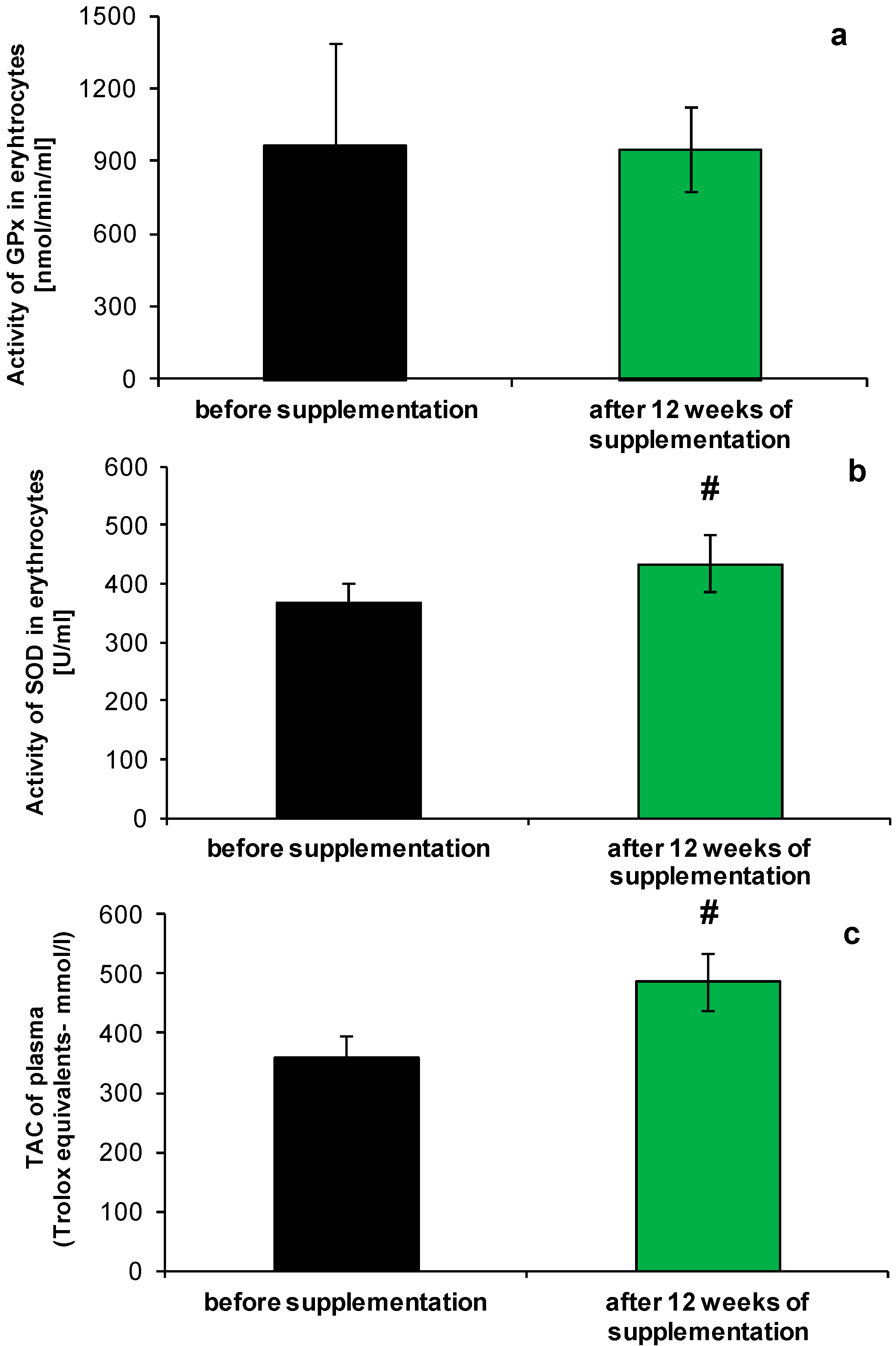
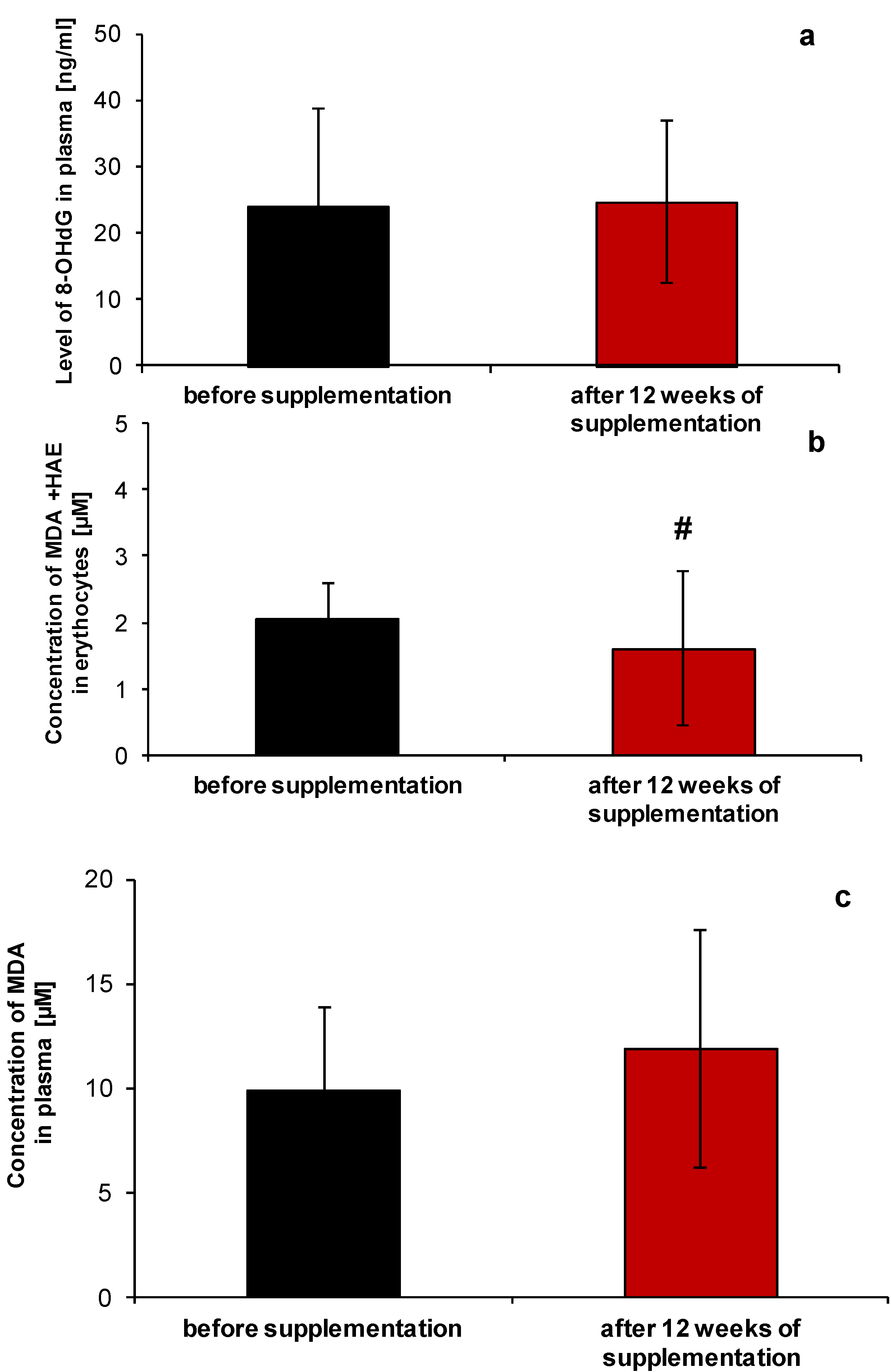
2.2. Anti-Ageing Abilities of NucleVital®Q10 Complex
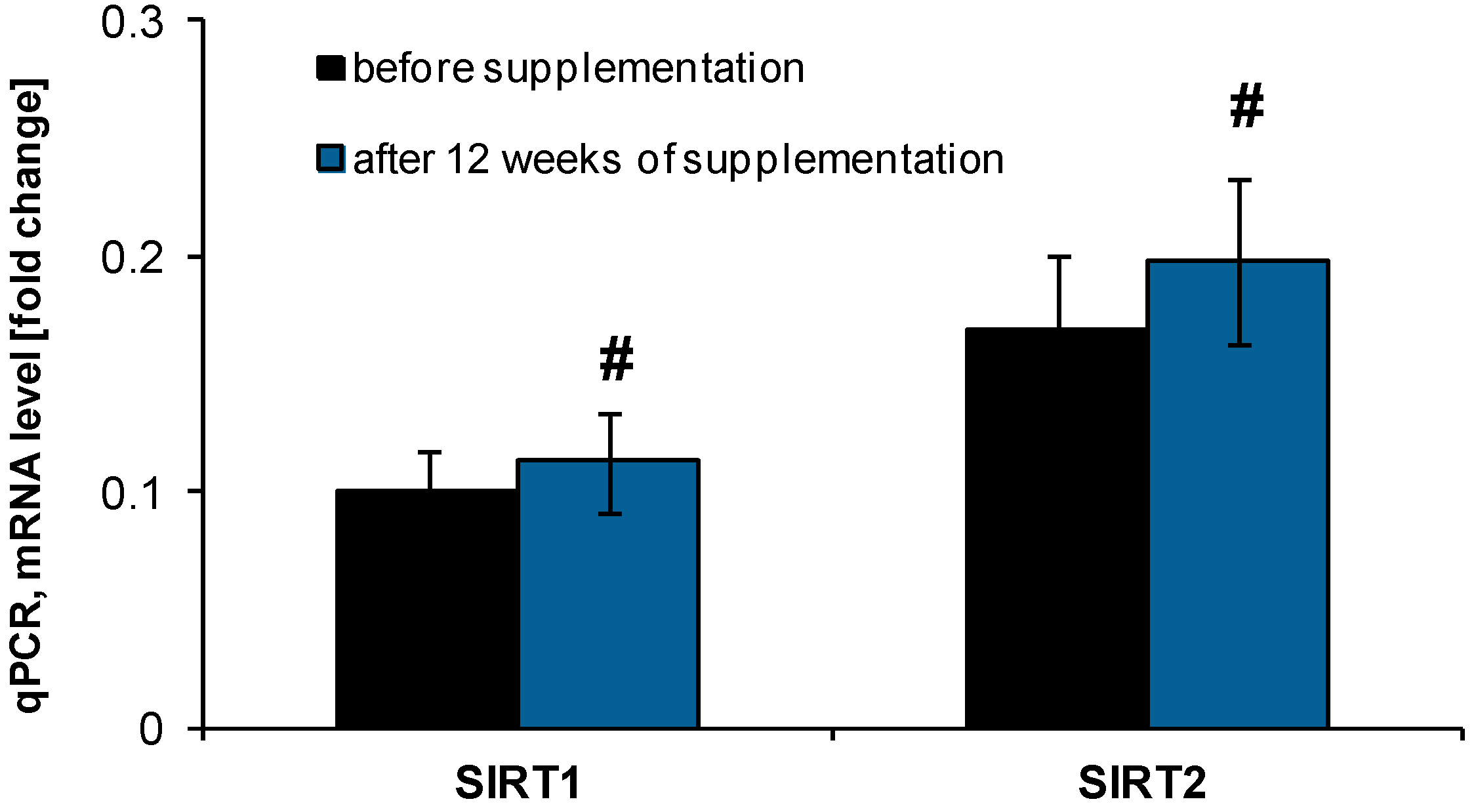
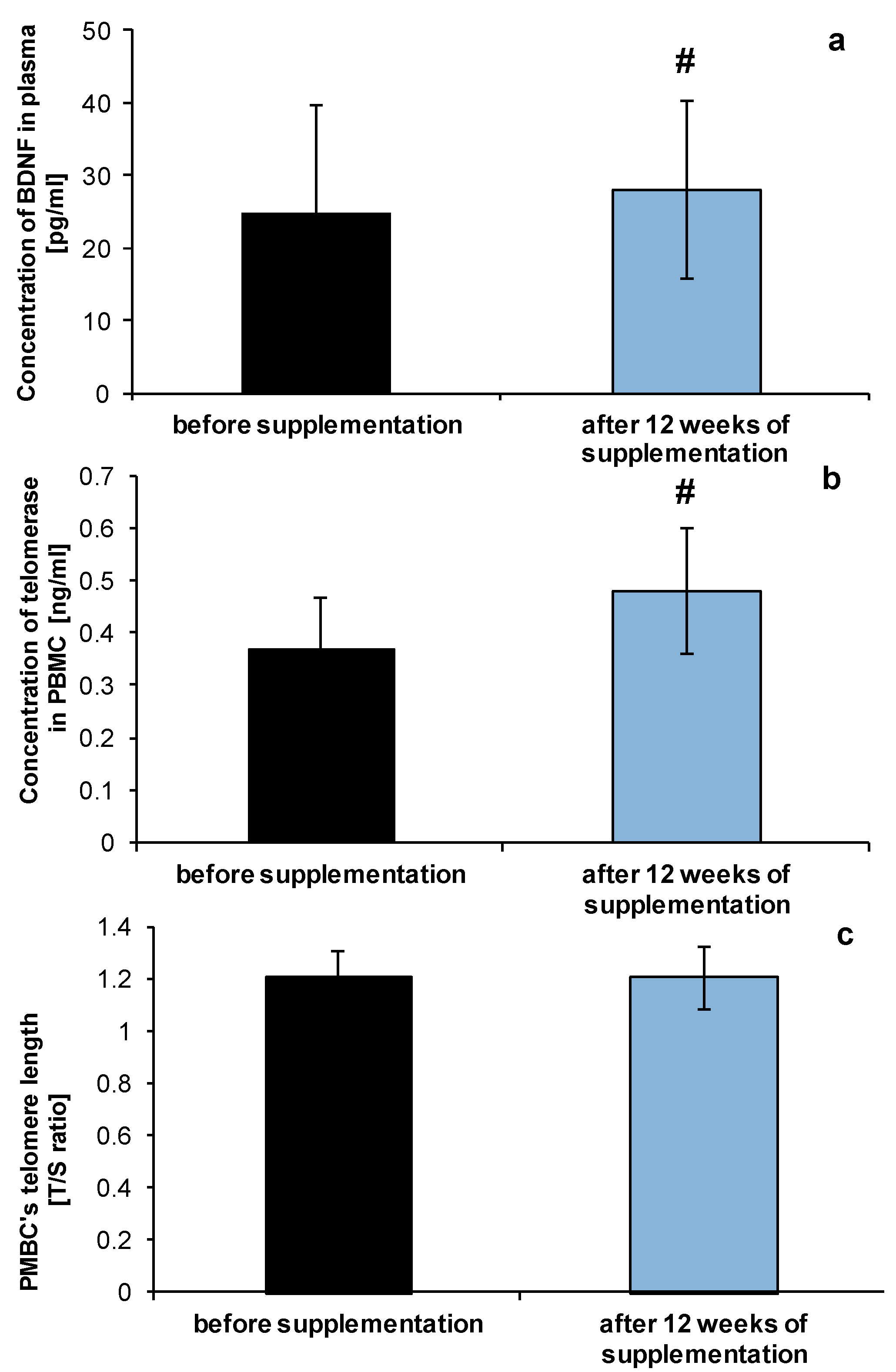
2.3. Effect of NucleVital®Q10 Complex on Vitamin D Deficiencies
3. Experimental Section
3.1. Material
3.1.1. Study Design
3.1.2. Inclusion/Exclusion Criteria
3.2. Analysis of Selected Parameters of Redox Homeostasis and Oxidative Stress
3.2.1. Measurement of Superoxide Dismutase Activity
3.2.2. Measurement of Glutathione Peroxidase (GPx) Activity
3.2.3. Total Antioxidant Capacity
3.2.4. Lipid Peroxidation Assay
3.2.5. Measurement of 8-OHdG Level
3.3. Analysis of Ageing-Related Parameters
3.3.1. RNA Isolation and cDNA Synthesis
3.3.2. Estimation of SIRT1 and SIRT2 Expression
3.3.3. Measurement of BDNF Level
3.3.4. Measurement of Telomerase Level
3.3.5. Measurement of PBMC’s Telomere Length
3.4. Measurement of Vitamin D Level
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef]
- Speakman, J.R.; Selman, C. The free-radical damage theory: Accumulating evidence against a simple link of oxidative stress to ageing and lifespan. Bioessays 2011, 33, 255–259. [Google Scholar] [CrossRef]
- Kirkwood, T.B.; Kowald, A. The free-radical theory of ageing—Older, wiser and still alive: Modelling positional effects of the primary targets of ROS reveals new support. Bioessays 2012, 34, 692–700. [Google Scholar] [CrossRef]
- Zimniak, P. Relationship of electrophilic stress to aging. Free Radic. Biol. Med. 2011, 51, 1087–1105. [Google Scholar] [CrossRef]
- Pallauf, K.; Bendall, J.K.; Scheiermann, C.; Watschinger, K.; Hoffmann, J.; Roeder, T.; Rimbach, G. Vitamin C and lifespan in model organisms. Food Chem. Toxicol. 2013, 58, 255–263. [Google Scholar] [CrossRef]
- Banks, R.; Speakman, J.R.; Selman, C. Vitamin E supplementation and mammalian lifespan. Mol. Nutr. Food Res. 2010, 54, 719–725. [Google Scholar] [CrossRef]
- Ernst, I.M.; Pallauf, K.; Bendall, J.K.; Paulsen, L.; Nikolai, S.; Huebbe, P.; Roeder, T.; Rimbach, G. Vitamin E supplementation and lifespan in model organisms. Ageing Res. Rev. 2013, 12, 365–375. [Google Scholar] [CrossRef]
- Carney, J.M.; Starke-Reed, P.E.; Oliver, C.N.; Landum, R.W.; Cheng, M.S.; Wu, J.F.; Floyd, R.A. Reversal of age-related increase in brain protein oxidation, decrease in enzyme activity, and loss in temporal and spatial memory by chronic administration of the spin-trapping compound N-tert-butyl-alpha-phenylnitrone. Proc. Natl. Acad. Sci. USA. 1991, 88, 3633–3636. [Google Scholar] [CrossRef]
- Shetty, R.A.; Forster, M.J.; Sumien, N. Coenzyme Q(10) supplementation reverses age-related impairments in spatial learning and lowers protein oxidation. Age (Dordr) 2013, 35, 1821–1834. [Google Scholar] [CrossRef]
- Bai, H.; Liu, R.; Chen, H.L.; Zhang, W.; Wang, X.; Zhang, X.D.; Li, W.L.; Hai, C.X. Enhanced antioxidant effect of caffeic acid phenethyl ester and Trolox in combination against radiation induced-oxidative stress. Chem. Biol. Interact. 2014, 207, 7–15. [Google Scholar] [CrossRef]
- Stefanska, B.; Salamé, P.; Bednarek, A.; Fabianowska-Majewska, K. Comparative effects of retinoic acid, vitamin D and resveratrol alone and in combination with adenosine analogues on methylation and expression of phosphatase and tensin homologue tumour suppressor gene in breast cancer cells. Br. J. Nutr. 2012, 107, 781–790. [Google Scholar] [CrossRef]
- Bouayed, J.; Bohn, J. Exogenous antioxidants—Double-edged swords in cellular redox state. Oxidative Med. Cell. Longev. 2010, 3, 228–237. [Google Scholar] [CrossRef]
- Nogala-Kałucka, M.; Dwiecki, K.; Siger, A.; Górnaś, P.; Polewski, K.; Ciosek, S. Antioxidant synergism and antagonism between tocotrienols, quercetin and rutin in model system. Acta Aliment. 2013, 42, 360–370. [Google Scholar] [CrossRef]
- Podmore, I.D.; Griffiths, H.R.; Herbert, K.E.; Mistry, N.; Mistry, P.; Lunec, J. Vitamin C exhibits pro-oxidant properties. Nature 1998, 392, 559. [Google Scholar] [CrossRef]
- Halliwell, B. Phagocyte-derived reactive species: Salvation or suicide? Trends Biochem. Sci. 2006, 31, 509–515. [Google Scholar]
- Rice-Evans, C. Flavonoid antioxidants. Curr. Med. Chem. 2001, 8, 797–807. [Google Scholar] [CrossRef]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517S–520S. [Google Scholar]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Priemé, H.; Loft, S.; Nyyssönen, K.; Salonen, J.T.; Poulsen, H.E. No effect of supplementation with vitamin E, ascorbic acid, or coenzyme Q10 on oxidative DNA damage estimated by 8-oxo-7,8-dihydro-2'-deoxyguanosine excretion in smokers. Am. J. Clin. Nutr. 1997, 65, 503–507. [Google Scholar]
- Bartosz, G. Non-enzymatic antioxidant capacity assays: Limitations of use in biomedicine. Free Radic. Res. 2010, 44, 711–720. [Google Scholar]
- Nagyova, A.; Krajcovivova-Kudlackova, M.; Horska, A.; Smolkowa, B.; Blazicek, P.; Raslova, K.; Collins, A.; Dusinska, M. Lipid peroxidation in men after dietary supplementation with a mixure of antioxidant nutrients. Bratisl. Lek. Listy 2004, 105, 277–280. [Google Scholar]
- Heinrich, U.; Tronnier, H.; Stahl, W.; Bejot, M.; Maurette, J.M. Antioxidant supplements improve parameters related to skin structure in humans. Skin Pharmacol. Physiol. 2006, 19, 224–231. [Google Scholar] [CrossRef]
- Goraca, A. Assessment of total antioxidant capacity in human plasma. Folia Med. (Plovdiv.) 2004, 46, 16–21. [Google Scholar]
- Zhan, Z.J.; Zhou, Z.G.; Shan, W.G. Preparation and characterization of Cu,Zn-superoxide dismutase covalently modified by polyunsaturated fatty acids. Biochemistry (Mosc.) 2009, 74, 1266–1269. [Google Scholar]
- Mali, P.Y. Beneficial effect of extracts of Premna integrifolia root on human leucocytes and erythrocytes against hydrogen peroxide induced oxidative damage. Chron. Young Sci. 2014, 5, 53–58. [Google Scholar] [CrossRef]
- Scott, M.D.; Eaton, J.W.; Kuypers, F.A.; Chiu, D.T.; Lubin, B.H. Enhancement of erythrocyte superoxide dismutase activity: Effects on cellular oxidant defense. Blood 1989, 74, 2542–2549. [Google Scholar]
- Guerra-Araiza, C.; Álvarez-Mejía, A.L.; Sánchez-Torres, S.; Farfan-García, E.; Mondragón-Lozano, R.; Pinto-Almazán, R.; Salgado-Ceballos, H. Effect of natural exogenous antioxidants on aging and on neurodegenerative diseases. Free Radic. Res. 2013, 47, 451–462. [Google Scholar]
- Pan, M.H.; Lai, C.S.; Tsai, M.L.; Wu, J.C.; Ho, C.T. Molecular mechanisms for anti-aging by natural dietary compounds. Mol. Nutr. Food Res. 2012, 56, 88–115. [Google Scholar] [CrossRef]
- Hwang, J.W.; Yao, H.; Caito, S.; Sundar, I.K.; Rahman, I. Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic. Biol. Med. 2013, 61C, 95–110. [Google Scholar] [CrossRef]
- Ha, Ch.W.; Huh, W-K. The implication of Sir2 in replicative aging and senescence in Saccharomyces cerevisiae. Aging 2011, 3, 319–324. [Google Scholar]
- Komulainen, P.; Pedersen, M.; Hänninen, T.; Bruunsgaard, H.; Lakka, T.A.; Kivipelto, M.; Hassinen, M.; Rauramaa, T.H.; Pedersen, B.K.; Rauramaa, R. BDNF is a novel marker of cognitive function in ageing women: The DR’s EXTRA Study. Neurobiol. Learn. Mem. 2008, 90, 596–603. [Google Scholar] [CrossRef]
- Laske, C.; Stransky, E.; Leyhe, T.; Eschweiler, G.W.; Wittorf, A.; Richartz, E.; Bartels, M.; Buchkremer, G.; Schott, K. Stage-dependent BDNF serum concentrations in Alzheimer’s disease. J. Neural Transm. 2006, 113, 1217–1224. [Google Scholar] [CrossRef]
- Yasutake, C.; Kuroda, K.; Yanagawa, T.; Okamura, T.; Yoneda, H. Serum BDNF, TNF-alpha and IL-1beta levels in dementia patients: Comparison between Alzheimer’s disease and vascular dementia. Eur. Arch. Psychiatry Clin. Neurosci. 2006, 256, 402–406. [Google Scholar] [CrossRef]
- Takubo, K.; Aida, J.; Izumiyama-Shimomura, N.; Ishikawa, N.; Sawabe, M.; Kurabayashi, R.; Shiraishi, H.; Arai, T.; Nakamura, K.-I. Changes of telomere length with aging. Geriatr. Gerontol. Int. 2010, 10, 197–206. [Google Scholar] [CrossRef]
- Cassidy, A.; de Vivo, I.; Liu, Y.; Han, J.; Prescott, J.; Hunter, D.J.; Rimm, E.B. Associations between diet, lifestyle factors, and telomere length in women. Am. J. Clin. Nutr. 2010, 91, 1273–1280. [Google Scholar] [CrossRef]
- Celec, P.; Hodosy, J.; Palffy, R.; Gardlik, R.; Halcak, L.; Ostatnikova, D. The short-term effects of soybean intake on oxidative and carbonyl stress in mena and women. Molecules 2013, 18, 5190–5200. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, F.R.; Brincat, M.; Erel, C.T.; Tremollieres, F.; Gambacciani, M.; Lambrinoudaki, I.; Moen, M.H.; Schenck-Gustafsson, K.; Vujovic, S.; Rozenberg, S.; et al. EMAS position statement: Vitamin D and postmenopausal health. Maturitas 2012, 71, 83–88. [Google Scholar] [CrossRef]
- Komoroski, M.; Azad, N.; Camacho, P. Disorders of bone and bone mineral metabolism. Handb. Clin. Neurol. 2014, 120, 865–887. [Google Scholar]
- Haussler, M.R.; Whitfield, G.K.; Kaneko, I.; Haussler, C.A.; Hsieh, D.; Hsieh, J.C.; Jurutka, P.W. Molecular mechanisms of vitamin D action. Calcif. Tissue Int. 2013, 92, 77–98. [Google Scholar] [CrossRef]
- Dong, J.; Lau, C.W.; Wong, S.L.; Huang, Y. Cardiovascular benefits of vitamin D. Sheng Li Xue Bao 2014, 25, 30–36. [Google Scholar]
- Norman, P.E.; Powell, J.T. Vitamin D and cardiovascular disease. Circ. Res. 2014, 114, 379–393. [Google Scholar] [CrossRef]
- Rauchová, H.; Battino, M.; Fato, R.; Lenaz, G.; Drahota, Z. Coenzyme Q-pool function in glycerol-3-phosphate oxidation in hamster brown adipose tissue mitochondria. J. Bioenerg. Biomembr. 1992, 24, 235–241. [Google Scholar] [CrossRef]
- Lenaz, G.; Battino, M.; Castelluccio, C.; Fato, R.; Cavazzoni, M.; Rauchova, H.; Bovina, C.; Formiggini, G.; Parenti Castelli, G. Studies on the role of ubiquinone in the control of the mitochondrial respiratory chain. Free Radic. Res. Commun. 1990, 8, 317–27. [Google Scholar]
- Malmsten, C.L.; Lignell, A. Dietary Supplementation with Astaxanthin-Rich Algal Meal Improves Strength Endurance—A Double Blind Placebo Controlled Study on Male Students. Carotenoid Sci. 2008, 13, 20–22. [Google Scholar]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and Health: Evolution, Biologic Functions, and Recommended Dietary Intakes for Vitamin D. Clin. Rev. Bone Miner. Metab. 2009, 7, 2–19. [Google Scholar] [CrossRef]
- Etminan, M.; Gill, S.S.; Samii, A. Intake of vitamin E, vitamin C, and carotenoids and the risk of Parkinson’s disease: A meta-analysis. Lancet Neurol. 2005, 4, 362–365. [Google Scholar] [CrossRef]
- Engin, K.N.; Engin, G.; Kucuksahin, H.; Oncu, M.; Engin, G.; Guvener, B. Clinical evaluation of the neuroprotective effect of alpha-tocopherol against glaucomatous damage. Eur. J. Ophthalmol. 2007, 17, 528–533. [Google Scholar]
- Benzie, F.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antoxidant activity of biological fluids and modified version for simultaneous measurements of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–23. [Google Scholar]
- Cawthon, R.M. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucl. Acids Res. 2009, 37, e21. [Google Scholar] [CrossRef]
- Sample Availability: The NucleVital®Q10 Complex preparation is already a commercially available product.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Balcerczyk, A.; Gajewska, A.; Macierzyńska-Piotrowska, E.; Pawelczyk, T.; Bartosz, G.; Szemraj, J. Enhanced Antioxidant Capacity and Anti-Ageing Biomarkers after Diet Micronutrient Supplementation. Molecules 2014, 19, 14794-14808. https://doi.org/10.3390/molecules190914794
Balcerczyk A, Gajewska A, Macierzyńska-Piotrowska E, Pawelczyk T, Bartosz G, Szemraj J. Enhanced Antioxidant Capacity and Anti-Ageing Biomarkers after Diet Micronutrient Supplementation. Molecules. 2014; 19(9):14794-14808. https://doi.org/10.3390/molecules190914794
Chicago/Turabian StyleBalcerczyk, Aneta, Agnieszka Gajewska, Ewa Macierzyńska-Piotrowska, Tomasz Pawelczyk, Grzegorz Bartosz, and Janusz Szemraj. 2014. "Enhanced Antioxidant Capacity and Anti-Ageing Biomarkers after Diet Micronutrient Supplementation" Molecules 19, no. 9: 14794-14808. https://doi.org/10.3390/molecules190914794
APA StyleBalcerczyk, A., Gajewska, A., Macierzyńska-Piotrowska, E., Pawelczyk, T., Bartosz, G., & Szemraj, J. (2014). Enhanced Antioxidant Capacity and Anti-Ageing Biomarkers after Diet Micronutrient Supplementation. Molecules, 19(9), 14794-14808. https://doi.org/10.3390/molecules190914794




