1. Introduction
The successful delivery of cargo-molecules into cells can be based on constitutive endocytic processes by exploiting vectors that can ensure: (
i) high-yield, non-toxic cellular uptake of the cargo moiety, and (
ii) efficient endosomal escape of the cargo in order to let it reach its specific target. In recent reports by some of us [
1,
2,
3], these properties were conferred to a novel chimeric peptide in which Tat
11 arginine-rich motif (YGRKKRRQRRR, residues 47–57 of HIV-1 Tat protein, a classical cell-penetrating peptide, CPP) [
4] is fused to CM
18, a membrane-perturbing amphipathic α-helical sequence derived from the cecropin-A/melittin hybrid antimicrobial peptides (AMPs) series (KWKLFKKIGAVLKVLTTG, residues 1–7 of cecropin-A and 2–12 of melittin) [
5,
6]. CM
18-Tat
11 was exploited to promote the cytosolic delivery of a vast range of co-administered cargoes (calcein, GFP, dextrans) [
1], and to afford efficient transfection of complexed DNA plasmids [
2,
3]. CM
18-Tat
11 was shown to retain both Tat
11 ability to enter eukaryotic cells by endocytosis [
7] and CM
18 membrane-perturbing properties. Yet, the mechanism through which this chimera actually alters endosomal-membrane integrity in eukaryotic cells is still unknown. Since CM
18-Tat
11 does contain a functional antimicrobial peptide (CM
18), it is conceivable that it may act by one of the three “canonical” mechanisms of membrane permeabilization proposed for this class of sequences,
i.e., the “barrel-stave”, the “toroidal”, and the “carpet” model (reviewed in [
8,
9]). Briefly, in the barrel-stave mechanism the peptide monomers aggregate (as staves) around a central lumen, forming a pore (the barrel): the peptide hydrophobic segments align with the lipid core region of the bilayer, while their hydrophilic segments face the pore lumen. In the toroidal-pore mechanism, the polar segments of the peptides associate instead with the polar head groups of the lipids, so that the lipids are forced to tilt up to form a continuous bend from one side to the other of the membrane. Finally, in the carpet mechanism, the strong electrostatic interaction between the peptides and the phospholipid head groups lead to a peptide-induced membrane ‘carpeting’ effect. Eventually, the peptides can form micelles, thus leading to bilayer disintegration in a detergent-like manner. The possibility to probe these mechanisms directly on eukaryotic-cell membranes would be paramount to understand the mode of action of CM
18-Tat
11. To this end, an experimental platform has been recently proposed by some of us, consisting in the whole-cell voltage-clamp analysis of currents on the plasma membrane of rod outer segments isolated from frog retinae, where endogenous conductance can be fully blocked by bright light [
10,
11,
12,
13]. By this approach the membrane permeabilization properties of various synthetic or naturally occurring peptides were investigated, and a rationale was proposed to distinguish between the “barrel-stave”, the “toroidal-pore”, and the “carpet” mechanisms, as follows. The barrel-stave model requires that a certain number of monomers bind together once in the plasma membrane to form an ion conductive pore. If the number of monomers inserted in the membrane is small, as it occurs at low concentrations of a peptide like alamethicin, it is expected that the pores form and disaggregate frequently, producing sustained single channel events. The application of larger peptide concentrations is expected to produce macroscopic currents that recover to
zero upon extracellular peptide removal, because the interaction between the peptide monomers and the membrane is not strong enough to keep the peptides stably inserted once the extracellular supply is ceased. Consequently, the kinetics of current activation and deactivation and its amplitude are expected to be constant at each peptide application. All these features were observed with alamethicin F50/5 [
9,
10], and are therefore considered in the following as a “signature” of the barrel-stave permeabilization process. In the case of the toroidal model, the molecular attractive forces between the polar head groups of the lipids and peptides are such that the lipids are forced to tilt up and form pores whose walls are constituted by both lipids and peptide monomers. Such a strong interaction, however, is not expected to produce rapid pore formation and disaggregation at low peptide concentrations (
i.e., sustained single channel events), while larger peptide concentrations are expected to produce macroscopic currents with a
‘delay’ (see Experimental Section), and time constants of current activation (
τa) and recovery (deactivation;
τd) larger than in the case of the barrel-stave process. This is because of the longer time needed to form pores whose walls are not composed just by peptide monomers but by tilted lipids and peptides, and because of the longer time to disassemble this stable structure when extracellular peptide supply is ceased. If the peptide application is short and at low concentration, the monomers left on the membrane after peptide removal do not have a concentration large enough to give rise to conductive pores: these peptides may yet contribute to form additional pores once the peptide is applied again extracellularly. Therefore, repeated peptide applications are expected to elicit currents that only initially recover to the
zero level upon peptide removal, and then produce a progressive acceleration of the current-activation kinetics (
i.e., decrease of
delay and
τa), and a progressive increase of the steady-state current amplitude. This would eventually lead to the formation of stable conductive channels, which would produce progressively larger background currents upon peptide removal. All these features were observed with CM
15 [
11], a peptide well-known to form pores according to the toroidal model [
13], and are therefore considered in the following as a “signature” of this particular permeabilization process. Finally, the “signature” of the carpet mechanism is given by two main observables: (
i) a larger
delay with respect to the one characterizing current activation in the toroidal and barrel-stave mechanisms (due to the expected slow process of micelle formation); and (
ii) the substantial irreversibility of the permeabilization process, due to the membrane disruption in a detergent-like fashion, as observed in the case of viroporins [
12].
In the following, we have applied the above approach to investigate the permeabilization mechanism of CM18-Tat11 (and of isolated modules) inserted in the plasma membrane of single CHO-K1 cells. We find that the addition of the Tat11 module to CM18 is able to trigger a shift in the mechanism of membrane destabilization from “toroidal” to “carpet”, promoting a detergent-like membrane disruption. Collectively, these results rationalize our previous observations on CM18-Tat11 delivery properties.
2. Results and Discussion
Our goal here is to employ the patch-clamp technique to study the mechanism of membrane permeabilization induced by the pore-forming peptides, under strict physiological conditions. This goal is achieved by recording the ion current through the channels formed by these peptides, once inserted in a cell plasma membrane. To avoid contamination by the cell membrane currents, all the endogenous current sources must be blocked, possibly without using any drug (such as TTX, TEA, dihydropyridines,
etc.) that could obstruct the peptide pores or interfere with the pore formation. We previously have found that the photoreceptor rod outer segment, mechanically isolated from the frog retina, is a suitable preparation because its sole membrane conductance can be fully closed by bright light [
9]. In line with our previous reports on CM
18-Tat
11 [
1,
2], we used the CHO-K1 cell line as case study. Nicely, we found that, in symmetric ionic conditions, CHO-K1 cells have no voltage- and/or calcium- and/or time-dependent conductances, but only a very small background one.
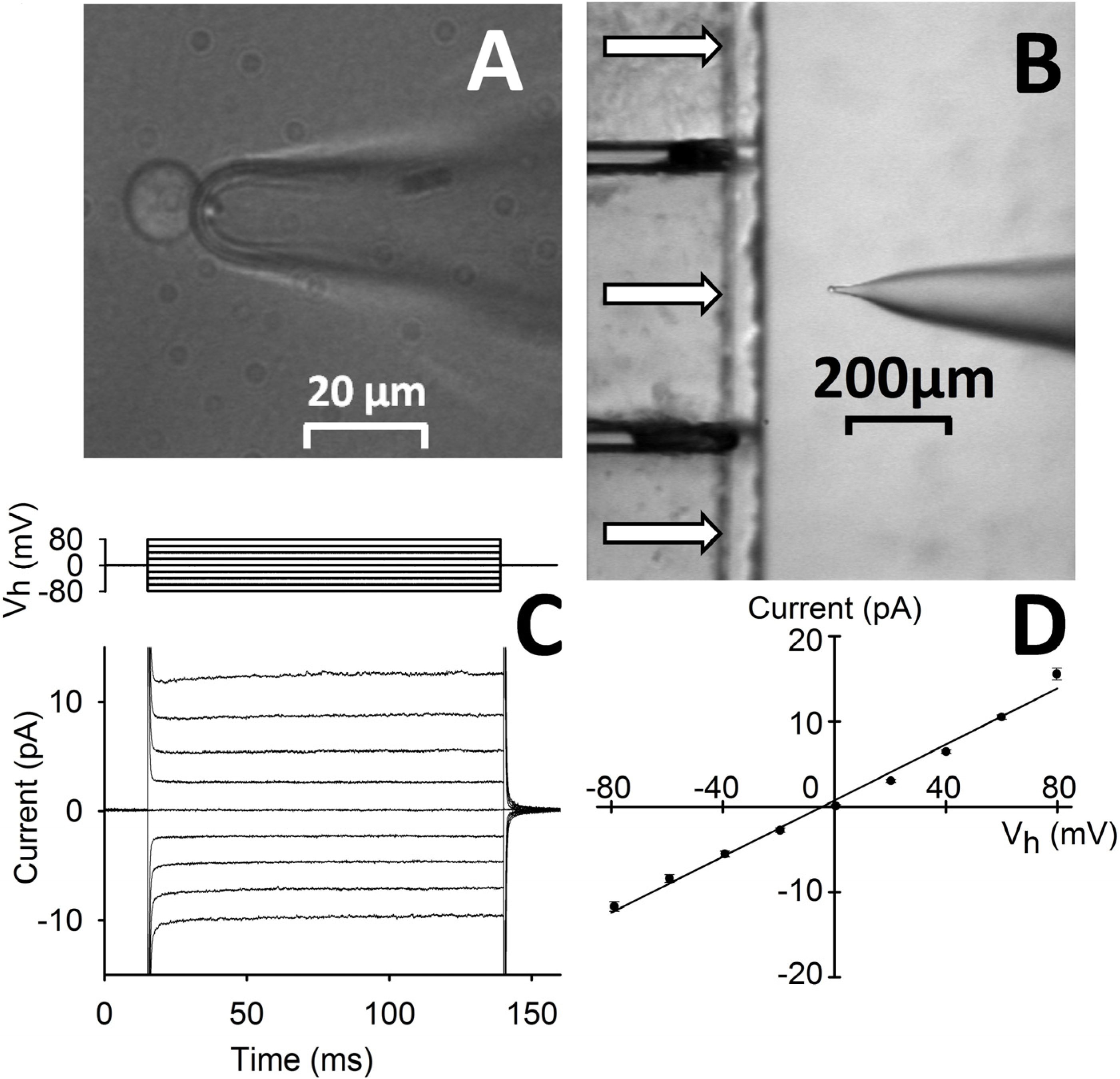
Indeed, the current amplitude is time-independent and very small for any physiological voltage value (
Figure 1C). We observe a linear (ohmic) current-to-voltage behavior (
Figure 1D) and a membrane resistance (
Rm) > 5 GΩ. Such high
Rm values make it possible to measure current amplitudes as small as 1 pA in a bandwidth of at least 1 kHz (
i.e., any exogenous peptide-induced current can be detected down to the single-channel level). Thanks to this high recording resolution, we set
Vh = −20 mV in order to limit the current amplitude induced by the strongly permeabilizing peptides while ensuring a detectable current through the weaker ones. In a characteristics measurement, isolated CHO-K1 are held at
Vh and
Rm is checked before peptide delivery by means of a brief −10 mV step (
Figure 2,
Figure 3 and
Figure 4); various concentrations of CM
18-Tat
11 (or CM
18, or Tat
11) are then delivered using the fast-perfusion system. Once current stabilizes, the cell is returned to the control solution (without peptide) to assess the possible current level recovery and
Rm is again measured. In the control solution, repetitive 10 mV pulses are routinely applied to check access resistance (
Ra) stability (
Figure 3), otherwise the recording is terminated.
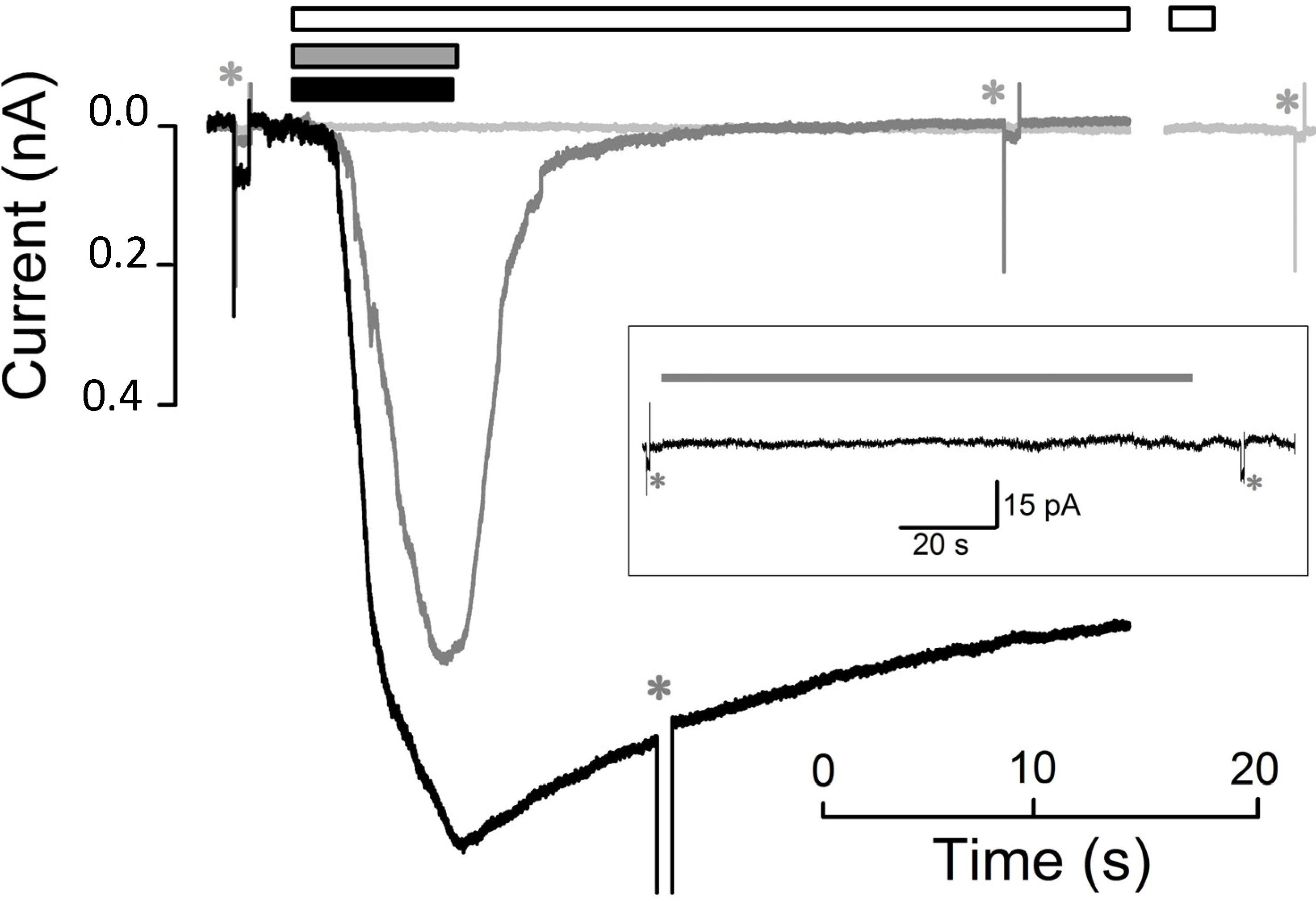
CM
18-Tat
11 continuously applied for more than 1 min at various concentrations (0.5 µM,
n = 2; 1 µM,
n = 3; 2 µM,
n = 4; 3 µM,
n = 5) fails to elicit detectable currents down to single-channel events. A typical example is shown in
Figure 2,
inset, in which the current does not significantly deviate from the
zero level following a ~100 s application of 3 µM CM
18-Tat
11. On the contrary, application of 4 µM CM
18-Tat
11 elicits a current that develops in a roughly exponential trend (
τa = 1.5 ± 0.4 s) up to a steady-state amplitude of 1.0 ± 0.2 nA (
n = 15 cells) with a
delay of 2.6 ± 0.5 s (see Experimental Section). As shown by the representative curve in
Figure 2 (
black trace), the induced membrane permeabilization is extensive, as the evoked current does not fully recover to the baseline even after several seconds from peptide removal (see
Figure 4 for more details): these recordings are very similar to the ones obtained with a viroporin-derived peptide [
13], acting in a carpet-like fashion. Given the structural/functional modularity of CM
18-Tat
11, we used the two isolated CM
18 and Tat
11 peptides as controls for the observed behavior. CHO-K1 cells exposed to CM
18 concentrations from 0.5 to 4 µM invariably show detectable membrane currents,
i.e., membrane destabilization (4 µM: steady state amplitude = 1.0 ± 0.1 nA,
delay = 0.9 ± 0.2 s,
τa = 2.1 ± 0.7 s,
τd =3. 7 ± 1.0 s,
n = 7 cells; 2 µM: steady state amplitude = 0.36 ± 0.07 nA,
delay = 3.6 ± 1.1 s,
τa = 4.0 ± 1.1 s,
τd = 3.2 ± 1.0 s,
n = 6 cells; concentrations < 2 µM yield activation and deactivation currents that cannot be simply fitted with a single exponential: statistics are therefore not reported). Contrary to what was found for CM
18-Tat
11, however, in this case permeabilization is fully reversible for all tested concentrations upon peptide removal,
i.e., current returns roughly exponentially to 0, and
Rm fully recovers to its former level (representative curve in
Figure 2,
dark grey trace).
Figure 1.
Outline of the technique employed. (A) CHO-K1 cell recorded in whole-cell with a pressure-polished pipette; cell is aligned in front of the perfusion pipette (at low magnification in (B) formed by square glass capillaries (500 µm of side) glued together; horizontal arrows denote perfusion flows. (C) Average whole-cell current recorded from representative cells (lower panel; pipette and external solution: 130 mM K+ + 1 mM Ca2+), subjected to 125 ms voltage steps from −80 mV to +80 mV in 20 mV increments (top panel) starting from Vh = 0 mV (traces are the average of n = 6 cells); (D) the average current evoked by each voltage step of C is plotted against the voltage step amplitude; the points are well fitted by a straight line (correlation coefficient ~0.99), whose angular coefficient gave Rm ~6.1 GΩ.
Figure 1.
Outline of the technique employed. (A) CHO-K1 cell recorded in whole-cell with a pressure-polished pipette; cell is aligned in front of the perfusion pipette (at low magnification in (B) formed by square glass capillaries (500 µm of side) glued together; horizontal arrows denote perfusion flows. (C) Average whole-cell current recorded from representative cells (lower panel; pipette and external solution: 130 mM K+ + 1 mM Ca2+), subjected to 125 ms voltage steps from −80 mV to +80 mV in 20 mV increments (top panel) starting from Vh = 0 mV (traces are the average of n = 6 cells); (D) the average current evoked by each voltage step of C is plotted against the voltage step amplitude; the points are well fitted by a straight line (correlation coefficient ~0.99), whose angular coefficient gave Rm ~6.1 GΩ.
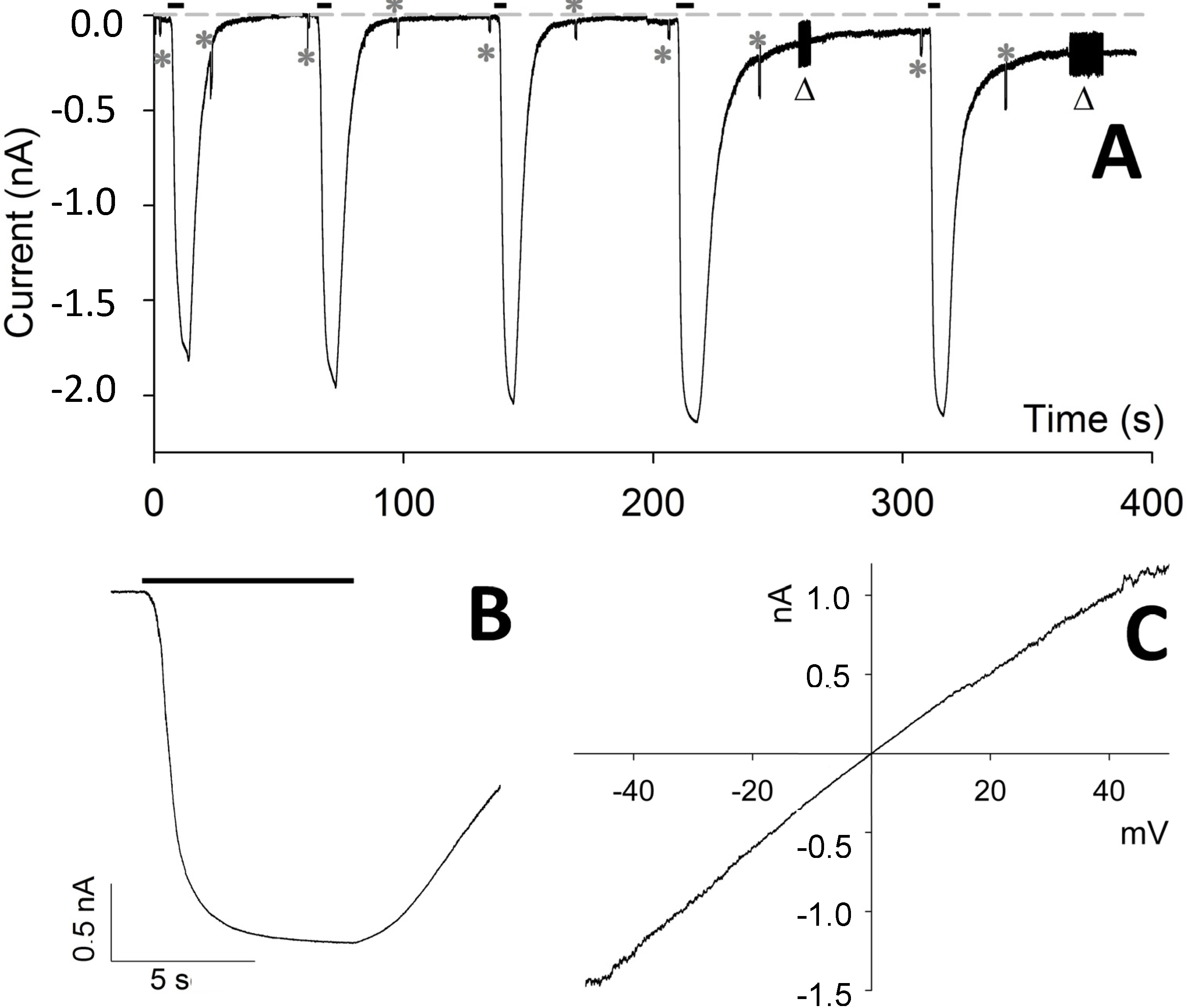
However, for repetitive CM
18 applications at 4 µM (
Figure 3A), the recovery of current and of
Rm was progressively more incomplete; moreover, the current-to-voltage relationship was found to be linear (a representative recording is illustrated in
Figure 3C), showing that the pore formation is voltage-insensitive (differently from other pore-forming peptides, as alamethicin). This relationship was obtained by applying voltage ramps (from −50 to +50 mV, slope: 0.25 mV/ms) during CM
18 perfusion at V
h = −20 mV. To avoid the loss of voltage control due to
Ra at extreme voltages (−50 and +50 mV, where currents may become very large), CM
18 was applied at 2 µM concentration, corresponding to a current not exceeding 2 nA at extreme voltages. The responses to three consecutive voltage ramps during CM
18 perfusion were averaged and corrected by subtracting the average response recorded in control conditions (
Figure 1D and
Figure 3C). The obtained relationship is almost perfectly ohmic at physiological voltages, and in all the cells examined (
n = 4). Notably, all the recordings obtained with CM
18 were very similar to the ones obtained with the analogous cecropin-A/melittin CM
15 hybrid peptide, inserted in the plasma membrane of isolated photoreceptor rod outer segments [
12] and in CHO-K1 cells (data not shown). Since CM
15 forms transient toroidal pores in the membrane, as previously demonstrated by patch-clamp analysis [
12] and site-directed spin-labeling electron-paramagnetic-resonance studies [
14], it is concluded that these two variants share the same membrane-destabilization mechanism. Finally, continuous application of Tat
11 (up to 5 min) at concentrations from 0.5 to 8 µM failed to elicit any current for all voltage values tested (from −80 mV to +80 mV), thus demonstrating that this module alone is not able to significantly permeabilize the membrane (
Figure 2,
light grey trace). In order to provide a direct comparison between membrane-destabilization properties of CM
18-Tat
11 and CM
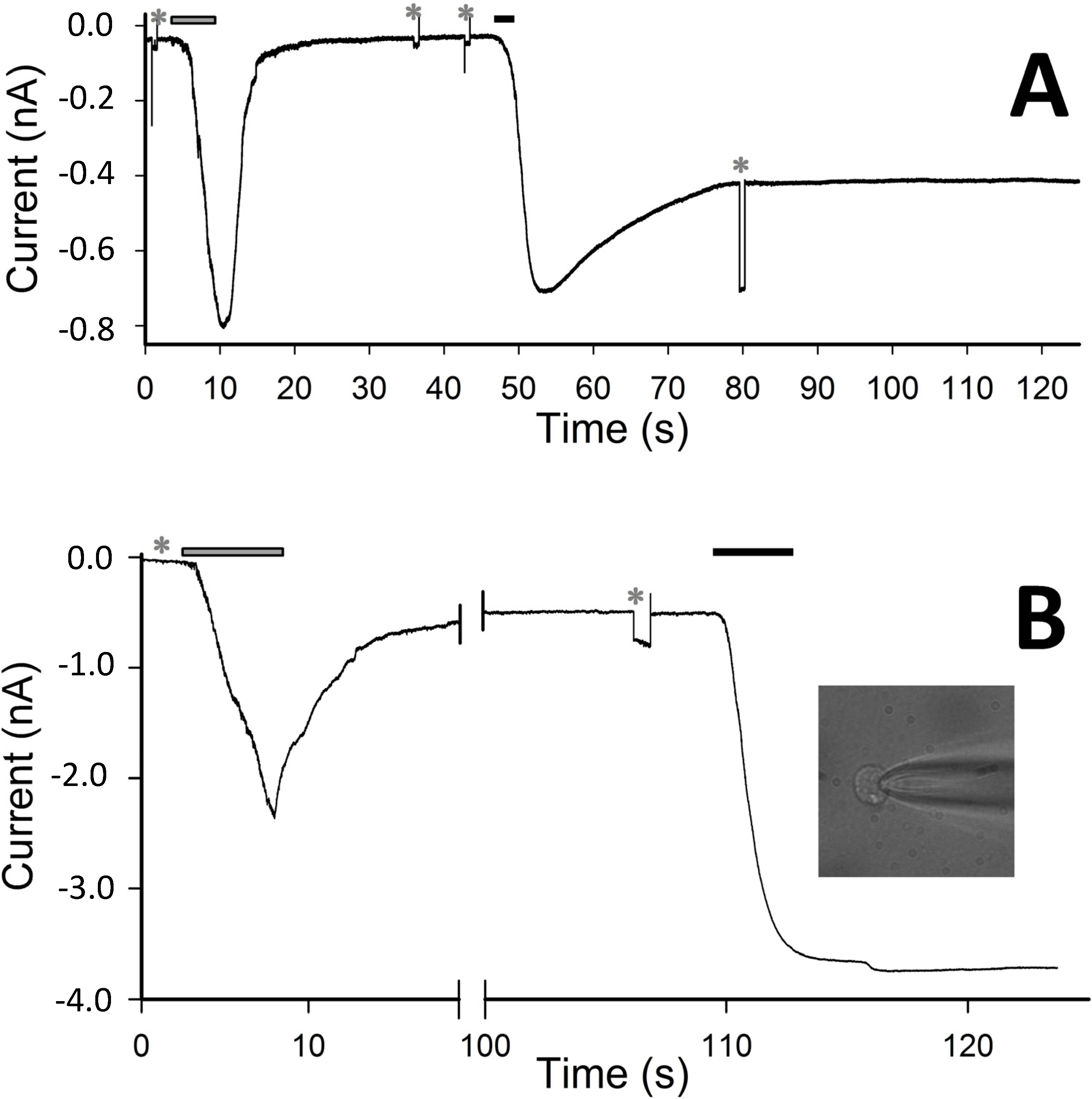
18, we recorded data during sequential administration of these peptides to the same cell at 4 µM (
Figure 4A). As expected, CM
18 produces a reversible current, while CM
18-Tat
11 leads to irreversible membrane destabilization of the same cell. Increasing the peptide concentration to 8 µM still produces a reversible effect in the case of CM
18 (although with an incomplete recovery,
Figure 4B) while cell lysis and death are the outcomes in the case of CM
18-Tat
11 (
Figure 4B, and 4B inset, experiment performed on the same cell).
Figure 2.
Kinetics of peptide-induced membrane permeabilization of CHO-K1 cells. Whole-cell currents recording elicited by the application (in three different cells), of CM18-Tat11 (6.7 s, 4 µM; black bar and black trace), CM18 (7.0 s, grey bar and grey trace), and Tat11 (71 s, 8 µM; white bar and light grey trace; trace break corresponds to 33 s of uninterrupted Tat11 perfusion); traces were aligned with peptide timing application; Vh = −20 mV. Inset, 113 s application of 3 µM of CM18-Tat11; Rm was 1.3 GΩ before and 1.0 GΩ after CM18-Tat11 application. Asterisks indicate −10 mV pulse delivery, used to measure Rm.
Figure 2.
Kinetics of peptide-induced membrane permeabilization of CHO-K1 cells. Whole-cell currents recording elicited by the application (in three different cells), of CM18-Tat11 (6.7 s, 4 µM; black bar and black trace), CM18 (7.0 s, grey bar and grey trace), and Tat11 (71 s, 8 µM; white bar and light grey trace; trace break corresponds to 33 s of uninterrupted Tat11 perfusion); traces were aligned with peptide timing application; Vh = −20 mV. Inset, 113 s application of 3 µM of CM18-Tat11; Rm was 1.3 GΩ before and 1.0 GΩ after CM18-Tat11 application. Asterisks indicate −10 mV pulse delivery, used to measure Rm.
Figure 3.
Repeated applications of CM18 on CHO-K1 cells. (A) Whole-cell current recording elicited by five consecutive applications and withdrawals of 4 µM of CM18 lasting, in sequence, 6.1, 5.2, 4.7, 6.2, and 4.1 s; the parameters characterizing the current following each one of the five peptide applications were respectively: delay: 1.7, 1.0, 0.9, 0.9, 0.9 s; τa: 1.5, 1.2, 0.8, 0.7, 0.7 s; current amplitude: 1.8, 1.9, 2.0, 2.1, 2.0 nA; τd: 4.3, 4.4, 4.3, 6.0, 5.2 s. (B) A detailed view of the fourth peptide application. (C) Voltage dependence of the current elicited by 2 µM CM18 corrected for the leakage (in a cell different than A). In A and B: peptide application is indicated by the black bar; Rm, Ra, and membrane capacitance were checked before and after each peptide application by means of a single −10 mV pulse, indicated by an asterisk, and by repeated 10 mV pulses, indicated by a triangle; the grey dotted line indicates zero current; Vh = −20 mV.
Figure 3.
Repeated applications of CM18 on CHO-K1 cells. (A) Whole-cell current recording elicited by five consecutive applications and withdrawals of 4 µM of CM18 lasting, in sequence, 6.1, 5.2, 4.7, 6.2, and 4.1 s; the parameters characterizing the current following each one of the five peptide applications were respectively: delay: 1.7, 1.0, 0.9, 0.9, 0.9 s; τa: 1.5, 1.2, 0.8, 0.7, 0.7 s; current amplitude: 1.8, 1.9, 2.0, 2.1, 2.0 nA; τd: 4.3, 4.4, 4.3, 6.0, 5.2 s. (B) A detailed view of the fourth peptide application. (C) Voltage dependence of the current elicited by 2 µM CM18 corrected for the leakage (in a cell different than A). In A and B: peptide application is indicated by the black bar; Rm, Ra, and membrane capacitance were checked before and after each peptide application by means of a single −10 mV pulse, indicated by an asterisk, and by repeated 10 mV pulses, indicated by a triangle; the grey dotted line indicates zero current; Vh = −20 mV.
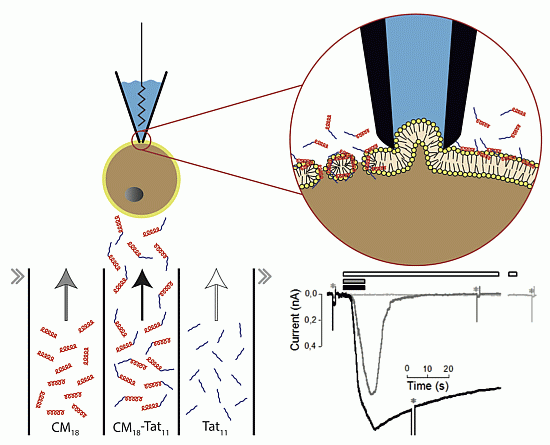
Based on these results, we conclude that the addition of the Tat
11 sequence to the pore-forming CM
18 module is determinant to trigger a switch in the membrane destabilization mechanism (schematic representation in
Figure 5). The appearance of irreversible destabilization following an increase in CM
18-Tat
11 concentration from 3 to 4 µM seems to indicate a threshold-effect. This is somewhat surprising in light of the well-known ability of the Tat
11 peptide to accumulate on the plasma membrane [
1]. We believe this observation can be linked to the fact that the main constituents of the CHO-K1 extracellular matrix (e.g., heparan sulfates and membrane-associated proteoglycans, which are the electrostatic counterparts of Tat
11 responsible for its accumulation on the plasma membrane [
1]) are altered by the trypsinization procedure used here to detach the cells before the patch-clamp analysis. We wish to stress, however, the important result reported here,
i.e., the demonstration of Tat
11 ability to favor irreversible carpet-like membrane destabilization.
Figure 4.
(A) Current elicited by the application (for 7 s) and withdrawal of 4 µM CM18 (grey bar) and of 4 µM CM18-Tat11 (3 s, black bar) on the same cell. (B) Whole-cell current recording elicited by the application and withdrawal of CM18 (9 s, 8 µM; grey bar) and of CM18-Tat11 (7 s, 8 µM, black bar) on the same cell; inset, lysis and death of cell following >10 min application of CM18-Tat11. Rm in A and B was checked by means of −10 mV pulses, indicated by the asterisks, i.e., before CM18 application, after CM18 withdrawal (and before CM18-Tat11 application) and after CM18-Tat11 application, respectively, and found to be: 0.6 GΩ, 0.6 GΩ, 35 MΩ in A and 0.6 GΩ, 110 MΩ, and not measurable (too small) in B; Vh = −20 mV.
Figure 4.
(A) Current elicited by the application (for 7 s) and withdrawal of 4 µM CM18 (grey bar) and of 4 µM CM18-Tat11 (3 s, black bar) on the same cell. (B) Whole-cell current recording elicited by the application and withdrawal of CM18 (9 s, 8 µM; grey bar) and of CM18-Tat11 (7 s, 8 µM, black bar) on the same cell; inset, lysis and death of cell following >10 min application of CM18-Tat11. Rm in A and B was checked by means of −10 mV pulses, indicated by the asterisks, i.e., before CM18 application, after CM18 withdrawal (and before CM18-Tat11 application) and after CM18-Tat11 application, respectively, and found to be: 0.6 GΩ, 0.6 GΩ, 35 MΩ in A and 0.6 GΩ, 110 MΩ, and not measurable (too small) in B; Vh = −20 mV.
We believe this stems from the high positive charge density of Tat
11, which leads to a stronger interaction of the peptide with the phospholipid head groups and to consequent membrane carpeting effect. However, it cannot be excluded that Tat
11 may strongly promote membrane partitioning of CM
18 (
Figure 5B,a) up to a point that some of the resulting toroidal pores (characteristic of CM
18 action) group together to delimit the contour of a micelle (
Figure 5B,b), that may eventually separate from the membrane and pass in solution (
Figure 5B,c).
Figure 5.
(A) Schematic representation of the membrane destabilization mechanism proposed for CM18-Tat11 (left) and CM18 (right) peptides. CM18 moiety is pictured as a red helix, while Tat11 moiety is represented as an unstructured blue segment. The upper part describes for each peptide its hypothetical membrane distribution when it is administered in the range between 0.5–3 µM. Instead the lower section represents the membrane distribution at concentrations higher than 4 µM. (B) Alternative mechanism of permeabilization operated by the CM18-Tat11 monomers, illustrated as red cylinders (CM18) plus blue segments (Tat11).
Figure 5.
(A) Schematic representation of the membrane destabilization mechanism proposed for CM18-Tat11 (left) and CM18 (right) peptides. CM18 moiety is pictured as a red helix, while Tat11 moiety is represented as an unstructured blue segment. The upper part describes for each peptide its hypothetical membrane distribution when it is administered in the range between 0.5–3 µM. Instead the lower section represents the membrane distribution at concentrations higher than 4 µM. (B) Alternative mechanism of permeabilization operated by the CM18-Tat11 monomers, illustrated as red cylinders (CM18) plus blue segments (Tat11).