Salidroside Protects Caenorhabditis elegans Neurons from Polyglutamine-Mediated Toxicity by Reducing Oxidative Stress
Abstract
:1. Introduction
2. Results and Discussion
2.1. Salidroside Prevents PolyQ-Mediated Neuronal Death in C. elegans
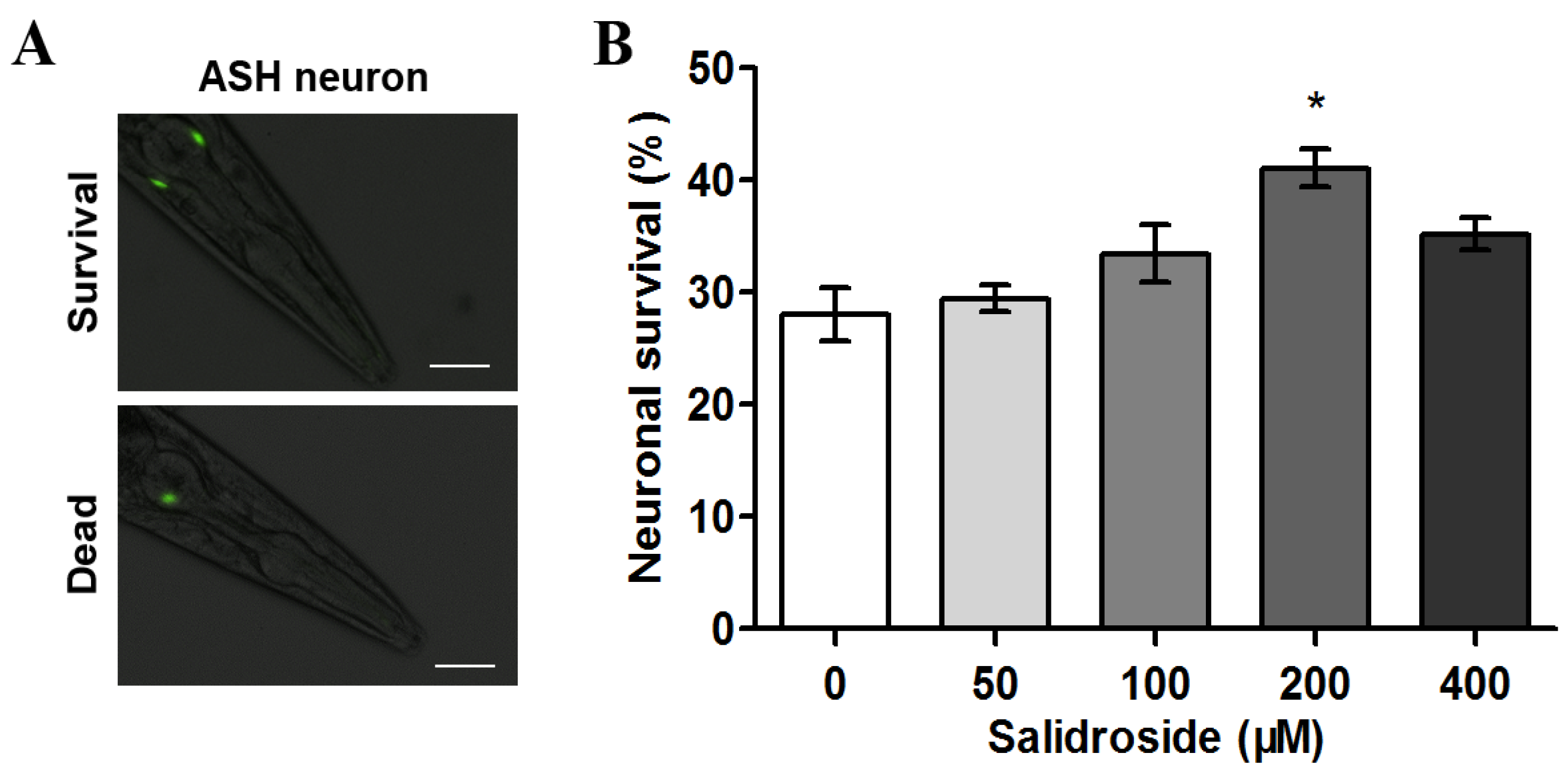
2.2. Salidroside Reduces PolyQ-Mediated Behavioral Dysfunction in C. elegans
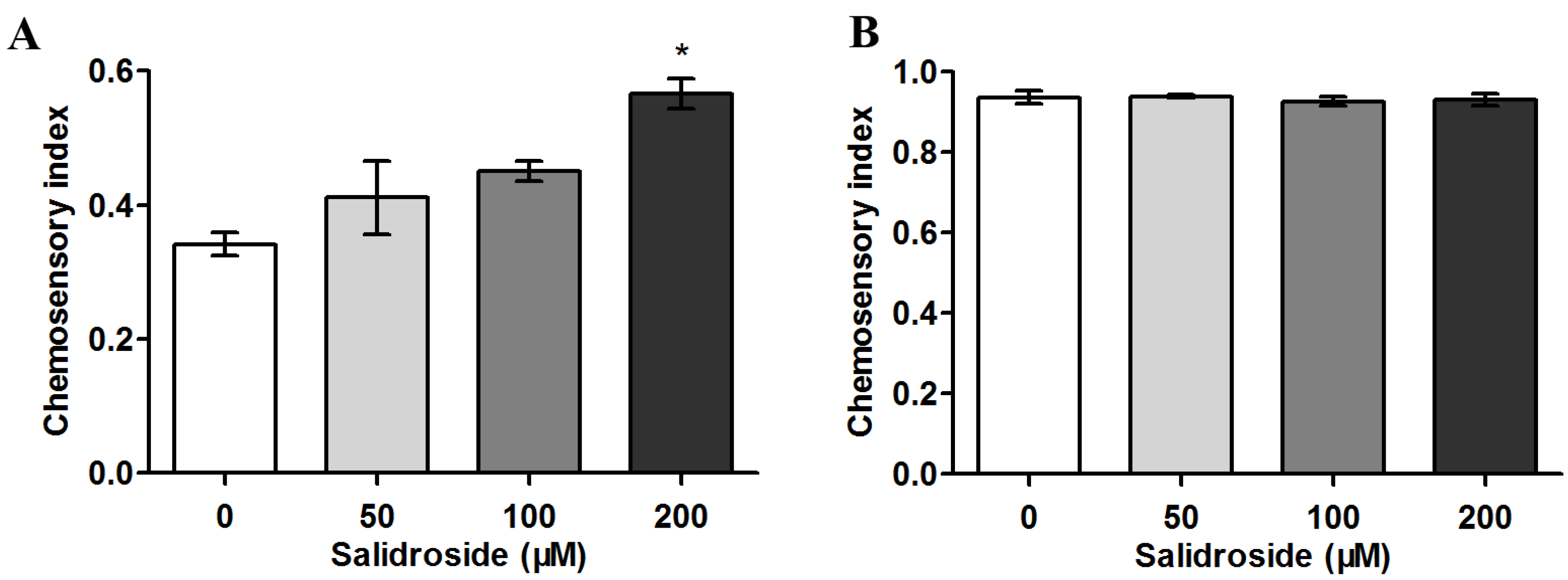
2.3. Salidroside Does not Inhibit PolyQ Aggregation
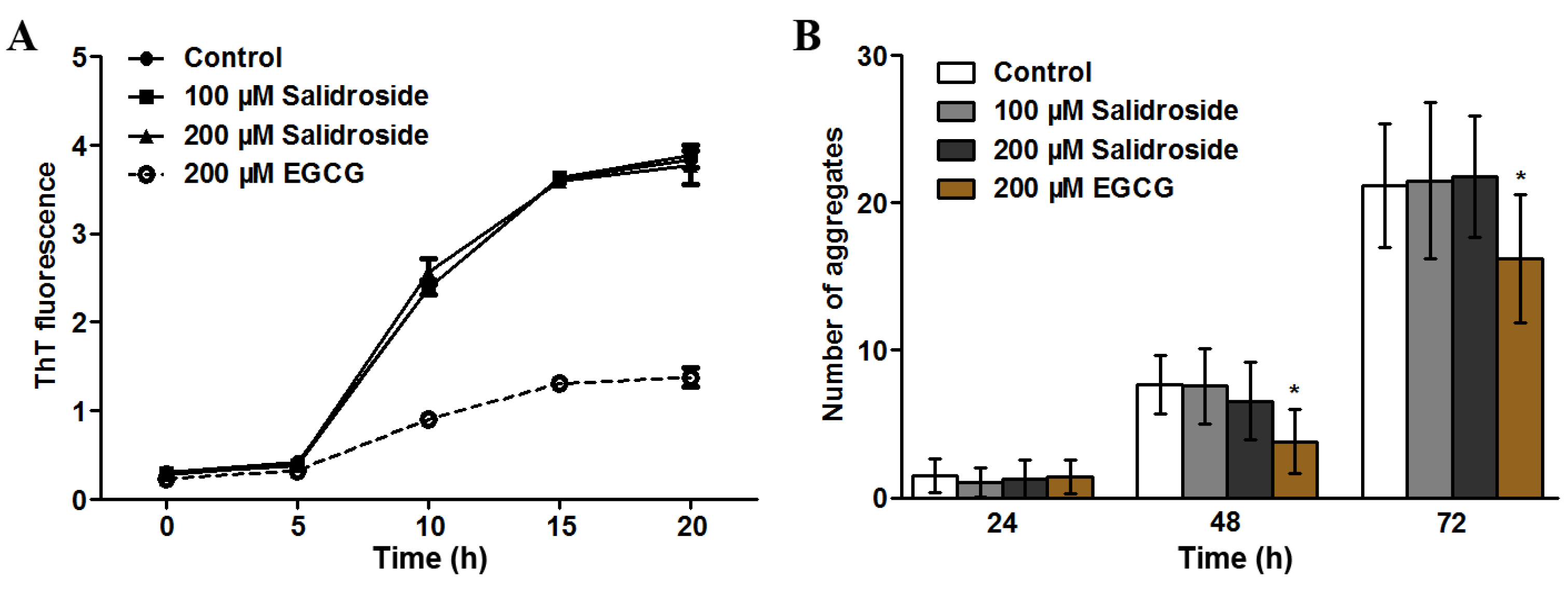
2.4. Salidroside Decreases ROS Level and Increases Oxidative Survival in C. elegans
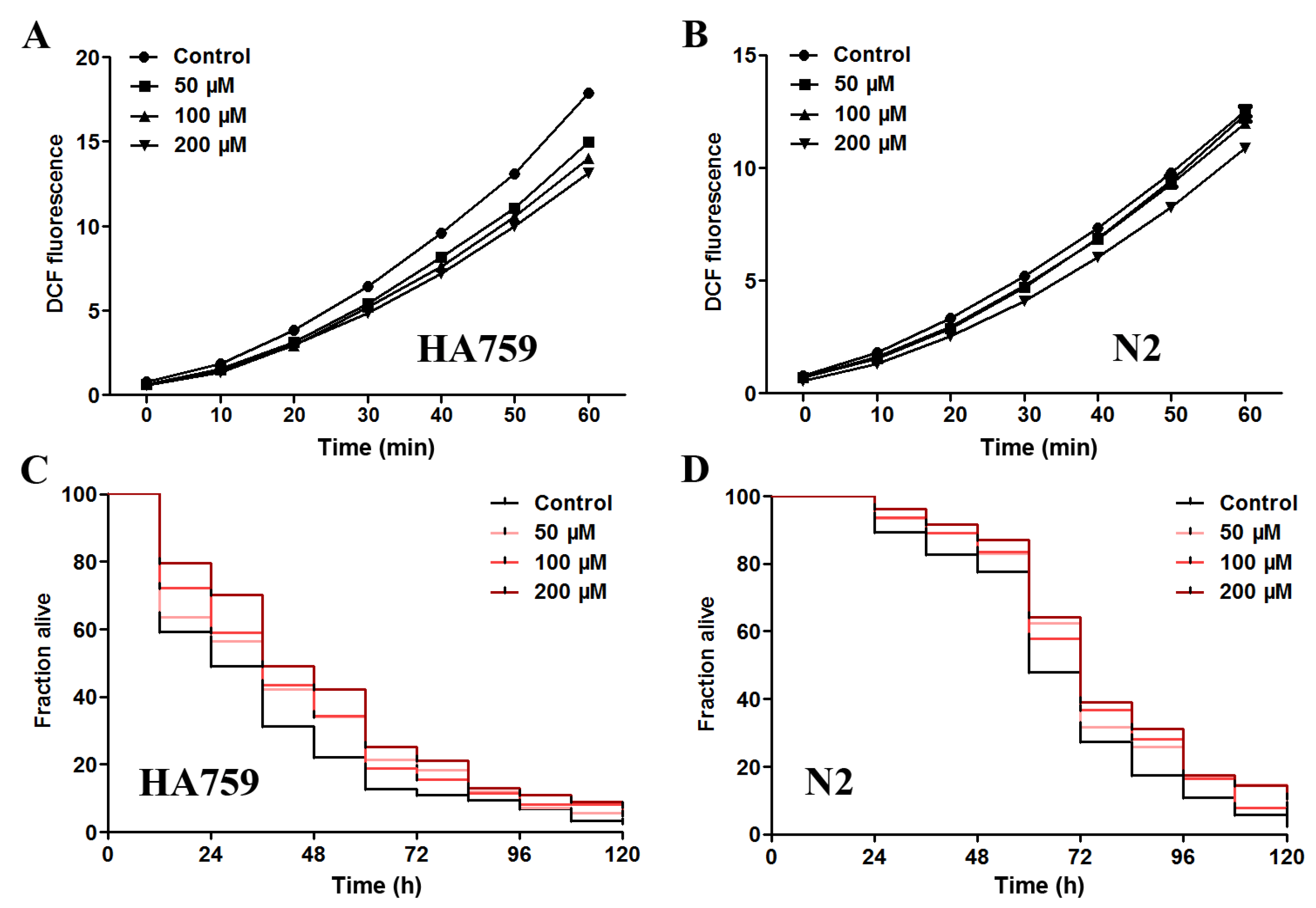
2.5. Salidroside Increases Antioxidant Enzyme Activities and Inhibits Lipid Peroxidation in PolyQ Nematodes
| Strain | Salidroside (μM) | Antioxidant Enzyme Activity a | MDA Content b | |
|---|---|---|---|---|
| SOD | CAT | |||
| HA759 | 0 | 16.2 ± 0.35 | 870 ± 78 | 0.66 ± 0.037 |
| 50 | 20.6 ± 1.37 | 1104 ± 74 | 0.51 ± 0.084 | |
| 100 | 21.5 ± 1.78 c | 1427 ± 188 c | 0.45 ± 0.14 | |
| 200 | 21.3 ± 0.63 c | 1418 ± 37 c | 0.24 ± 0.031 c | |
| N2 | 0 | 19.01 ± 0.24 | 1147 ± 26 | 0.59 ± 0.060 |
| 50 | 19.42 ± 0.78 | 1063 ± 80 | 0.45 ± 0.15 | |
| 100 | 18.30 ± 0.62 | 1066 ± 90 | 0.43 ±0.061 | |
| 200 | 20.06 ± 0.90 | 1176 ± 11 | 0.30 ± 0.059 c | |
3. Experimental Section
3.1. Chemicals and Materials
3.2. Strains and Maintenance
3.3. Neuronal Survival Assay
3.4. Chemosensory Assay
3.5. PolyQ Aggregation Assay
3.6. ROS Level Measurement
3.7. Paraquat Assay
3.8. Determination of Antioxidant Enzyme Activity and MDA Content
3.9. Statistic Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vonsattel, J.P.G.; DiFiglia, M. Huntington disease. J. Neuropathol. Exp. Neurol. 1998, 57, 369–384. [Google Scholar] [CrossRef]
- Penney, J.B., Jr.; Vonsattel, J.P.; MacDonald, M.E.; Gusella, J.F.; Myers, R.H. CAG repeat number governs the development rate of pathology in Huntington’s disease. Ann. Neurol. 1997, 41, 689–692. [Google Scholar] [CrossRef]
- Williams, A.J.; Paulson, H.L. Polyglutamine neurodegeneration: Protein misfolding revisited. Trends Neurosci. 2008, 31, 521–528. [Google Scholar] [CrossRef]
- Bates, G. Huntingtin aggregation and toxicity in Huntington’s disease. Lancet 2003, 361, 1642–1644. [Google Scholar] [CrossRef]
- Duennwald, M.L.; Jagadish, S.; Giorgini, F.; Muchowski, P.J.; Lindquist, S. A network of protein interactions determines polyglutamine toxicity. Proc. Natl. Acad. Sci. USA 2006, 103, 11051–11056. [Google Scholar] [CrossRef]
- Ross, C.A.; Poirier, M.A. Protein aggregation and neurodegenerative disease. Nat. Med. 2004, 10, S10–S17. [Google Scholar] [CrossRef]
- Ehrnhoefer, D.E.; Duennwald, M.; Markovic, P.; Wacker, J.L.; Engemann, S.; Roark, M.; Legleiter, J.; Marsh, J.L.; Thompson, L.M.; Lindquist, S.; et al. Green tea (−)-epigallocatechin-gallate modulates early events in huntingtin misfolding and reduces toxicity in Huntington’s disease models. Hum. Mol. Genet. 2006, 15, 2743–2751. [Google Scholar] [CrossRef]
- Holmes, C.; Boche, D.; Wilkinson, D.; Yadegarfar, G.; Hopkins, V.; Bayer, A.; Jones, R.W.; Bullock, R.; Love, S.; Neal, J.W.; et al. Long-term effects of Abeta42 immunisation in Alzheimer’s disease: Follow-up of a randomised, placebo-controlled phase I trial. Lancet 2008, 372, 216–223. [Google Scholar] [CrossRef]
- Browne, S.E.; Beal, M.F. Oxidative damage in Huntington’s disease pathogenesis. Antioxid. Redox Sign. 2006, 8, 2061–2073. [Google Scholar] [CrossRef]
- Bogdanov, M.B.; Andreassen, O.A.; Dedeoglu, A.; Ferrante, R.J.; Beal, M.F. Increased oxidative damage to DNA in a transgenic mouse model of Huntington’s disease. J. Neurochem. 2001, 79, 1246–1249. [Google Scholar]
- Wang, J.; Pfleger, C.M.; Friedman, L.; Vittorino, R.; Zhao, W.; Qian, X.; Conley, L.; Ho, L.; Pasinetti, G.M. Potential Application of Grape Derived Polyphenols in Huntington’s Disease. Transl. Neurosci. 2010, 1, 95–100. [Google Scholar] [CrossRef]
- Ferrante, R.J.; Andreassen, O.A.; Dedeoglu, A.; Ferrante, K.L.; Jenkins, B.G.; Hersch, S.M.; Beal, M.F. Therapeutic effects of coenzyme Q10 and remacemide in transgenic mouse models of Huntington’s disease. J. Neurosci. 2002, 22, 1592–1599. [Google Scholar]
- Sluder, A.E.; Baumeister, R. From genes to drugs: Target validation in Caenorhabditis elegans. Drug Discov. Today Technol. 2004, 1, 171–177. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, F.; Guo, W.; Zhang, J.; Xiao, L.; Li, H.; Jia, W.; Huang, Z. Caenorhabditis elegans in Chinese Medicinal Studies: Making the Case for Aging and Neurodegeneration. Rejuvnation Res. 2014, 17, 205–208. [Google Scholar] [CrossRef]
- Wu, Y.; Wu, Z.; Butko, P.; Christen, Y.; Lambert, M.P.; Klein, W.L.; Link, C.D.; Luo, Y. Amyloid-beta-induced pathological behaviors are suppressed by Ginkgo biloba extract EGb 761 and ginkgolides in transgenic Caenorhabditis elegans. J. Neurosci. 2006, 26, 13102–13113. [Google Scholar]
- Lakso, M.; Vartiainen, S.; Moilanen, A.M.; Sirvio, J.; Thomas, J.H.; Nass, R.; Blakely, R.D.; Wong, G. Dopaminergic neuronal loss and motor deficits in Caenorhabditis elegans overexpressing human alpha-synuclein. J. Neurochem. 2003, 86, 165–172. [Google Scholar]
- Parker, A.J.; Arango, M.; Abderrahmane, S.; Lambert, E.; Tourette, C.; Catoire, H.; Neri, C. Resveratrol rescues mutant polyglutamine cytotoxicity in nematode and mammalian neurons. Med. Sci. 2005, 21, 556–557. [Google Scholar]
- Zhang, H.; Pan, N.; Xiong, S.; Zou, S.; Li, H.; Xiao, L.; Cao, Z.; Tunnacliffe, A.; Huang, Z. Inhibition of polyglutamine-mediated proteotoxicity by Astragalus membranaceus polysaccharide through the DAF-16/FOXO transcription factor in Caenorhabditis elegans. Biochem. J. 2012, 441, 417–424. [Google Scholar] [CrossRef]
- Yu, S.; Liu, M.; Gu, X.; Ding, F. Neuroprotective effects of salidroside in the PC12 cell model exposed to hypoglycemia and serum limitation. Cell. Mol. Neurobiol. 2008, 28, 1067–1078. [Google Scholar] [CrossRef]
- Mao, G.; Wang, Y.; Qiu, Q.; Deng, H.; Yuan, L.; Li, R.; Song, D.; Li, Y.; Li, D.; Wang, Z. Salidroside protects human fibroblast cells from premature senescence induced by H2O2 partly through modulating oxidative status. Mech. Ageing Dev. 2010, 131, 723–731. [Google Scholar] [CrossRef]
- Guan, S.; Feng, H.; Song, B.; Guo, W.; Xiong, Y.; Huang, G.; Zhong, W.; Huo, M.; Chen, N.; Lu, J.; et al. Salidroside attenuates LPS-induced pro-inflammatory cytokine responses and improves survival in murine endotoxemia. Int. Immunopharmacol. 2011, 11, 2194–2199. [Google Scholar] [CrossRef]
- Liu, Z.; Li, X.; Simoneau, A.R.; Jafari, M.; Zi, X. Rhodiola rosea extracts and salidroside decrease the growth of bladder cancer cell lines via inhibition of the mTOR pathway and induction of autophagy. Mol. Carcinog. 2012, 51, 257–267. [Google Scholar] [CrossRef]
- Chen, X.; Liu, J.; Gu, X.; Ding, F. Salidroside attenuates glutamate-induced apoptotic cell death in primary cultured hippocampal neurons of rats. Brain Res. 2008, 1238, 189–198. [Google Scholar]
- Voisine, C.; Varma, H.; Walker, N.; Bates, E.A.; Stockwell, B.R.; Hart, A.C. Identification of potential therapeutic drugs for huntington’s disease using Caenorhabditis elegans. PLoS One 2007, 2, e504. [Google Scholar]
- Hilliard, M.A.; Apicella, A.J.; Kerr, R.; Suzuki, H.; Bazzicalupo, P.; Schafer, W.R. In vivo imaging of C. elegans ASH neurons: Cellular response and adaptation to chemical repellents. EMBO J. 2005, 24, 63–72. [Google Scholar]
- Yang, X.; Zhang, P.; Wu, J.; Xiong, S.; Jin, N.; Huang, Z. The neuroprotective and lifespan-extension activities of Damnacanthus officinarum extracts in Caenorhabditis elegans. J. Ethnopharmacol. 2012, 141, 41–47. [Google Scholar] [CrossRef]
- LeVine, H., 3rd. Thioflavine T interaction with synthetic Alzheimer’s disease beta-amyloid peptides: Detection of amyloid aggregation in solution. Protein Sci. 1993, 2, 404–410. [Google Scholar] [CrossRef]
- Hamuro, L.; Zhang, G.; Tucker, T.J.; Self, C.; Strittmatter, W.J.; Burke, J.R. Optimization of a polyglutamine aggregation inhibitor peptide (QBP1) using a thioflavin T fluorescence assay. Assay Drug Dev. Technol. 2007, 5, 629–636. [Google Scholar] [CrossRef]
- Roberts, B.E.; Shorter, J. Escaping amyloid fate. Nat. Struct. Mol. Biol. 2008, 15, 544–546. [Google Scholar] [CrossRef]
- Nollen, E.A.; Garcia, S.M.; van Haaften, G.; Kim, S.; Chavez, A.; Morimoto, R.I.; Plasterk, R.H. Genome-wide RNA interference screen identifies previously undescribed regulators of polyglutamine aggregation. Proc. Natl. Acad. Sci. USA 2004, 101, 6403–6408. [Google Scholar]
- Chini, A. Application of yeast-two hybrid assay to chemical genomic screens: A high-throughput system to identify novel molecules modulating plant hormone receptor complexes. Methods Mol. Biol. 2014, 1056, 35–43. [Google Scholar]
- Gutierrez-Zepeda, A.; Santell, R.; Wu, Z.; Brown, M.; Wu, Y.; Khan, I.; Link, C.D.; Zhao, B.; Luo, Y. Soy isoflavone glycitein protects against beta amyloid-induced toxicity and oxidative stress in transgenic Caenorhabditis elegans. BMC Neurosci. 2005, 6, 54. [Google Scholar] [CrossRef]
- Firdaus, W.J.; Wyttenbach, A.; Diaz-Latoud, C.; Currie, R.W.; Arrigo, A.P. Analysis of oxidative events induced by expanded polyglutamine huntingtin exon 1 that are differentially restored by expression of heat shock proteins or treatment with an antioxidant. FEBS J. 2006, 273, 3076–3093. [Google Scholar] [CrossRef]
- Yuan, Y.; Wu, S.-J.; Liu, X.; Zhang, L.-L. Antioxidant effect of salidroside and its protective effect against furan-induced hepatocyte damage in mice. Food Funct. 2013, 4, 763–769. [Google Scholar] [CrossRef]
- Smith, J.V.; Luo, Y. Ginkgo biloba extract EGb761 extends life span and attenuates H2O2 levels in a Caenorhabditis elegans model of Alzheimer’s Disease. In Oxidative Stress and Age-Related Neurodegeneration, 1st ed.; Luo, Y., Ed.; CRC Press: Boca Raton, FL, USA, 2005; pp. 300–326. [Google Scholar]
- Chen, X.; Wu, J.; Luo, Y.; Liang, X.; Supnet, C.; Kim, M.W.; Lotz, G.P.; Yang, G.; Muchowski, P.J.; Kodadek, T.; et al. Expanded polyglutamine-binding peptoid as a novel therapeutic agent for treatment of Huntington’s disease. Chem. Biol. 2011, 18, 1113–1125. [Google Scholar] [CrossRef]
- Li, H.; Xu, J.; Liu, Y.; Ai, S.; Qin, F.; Li, Z.; Zhang, H.; Huang, Z. Antioxidant and moisture-retention activities of the polysaccharide from Nostoc commune. Carbohydr. Polym. 2011, 83, 1821–1827. [Google Scholar]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant. Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Method Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef]
- Mihara, M.; Uchiyama, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem. 1978, 86, 271–278. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds salidroside are available from the authors.
© 2014 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xiao, L.; Li, H.; Zhang, J.; Yang, F.; Huang, A.; Deng, J.; Liang, M.; Ma, F.; Hu, M.; Huang, Z. Salidroside Protects Caenorhabditis elegans Neurons from Polyglutamine-Mediated Toxicity by Reducing Oxidative Stress. Molecules 2014, 19, 7757-7769. https://doi.org/10.3390/molecules19067757
Xiao L, Li H, Zhang J, Yang F, Huang A, Deng J, Liang M, Ma F, Hu M, Huang Z. Salidroside Protects Caenorhabditis elegans Neurons from Polyglutamine-Mediated Toxicity by Reducing Oxidative Stress. Molecules. 2014; 19(6):7757-7769. https://doi.org/10.3390/molecules19067757
Chicago/Turabian StyleXiao, Lingyun, Haifeng Li, Ju Zhang, Fan Yang, Aizhen Huang, Jingjing Deng, Ming Liang, Fangli Ma, Minghua Hu, and Zebo Huang. 2014. "Salidroside Protects Caenorhabditis elegans Neurons from Polyglutamine-Mediated Toxicity by Reducing Oxidative Stress" Molecules 19, no. 6: 7757-7769. https://doi.org/10.3390/molecules19067757
APA StyleXiao, L., Li, H., Zhang, J., Yang, F., Huang, A., Deng, J., Liang, M., Ma, F., Hu, M., & Huang, Z. (2014). Salidroside Protects Caenorhabditis elegans Neurons from Polyglutamine-Mediated Toxicity by Reducing Oxidative Stress. Molecules, 19(6), 7757-7769. https://doi.org/10.3390/molecules19067757





