Two New Lanostane Triterpenoids from the Branches and Leaves of Polyalthia oblique
Abstract
:1. Introduction
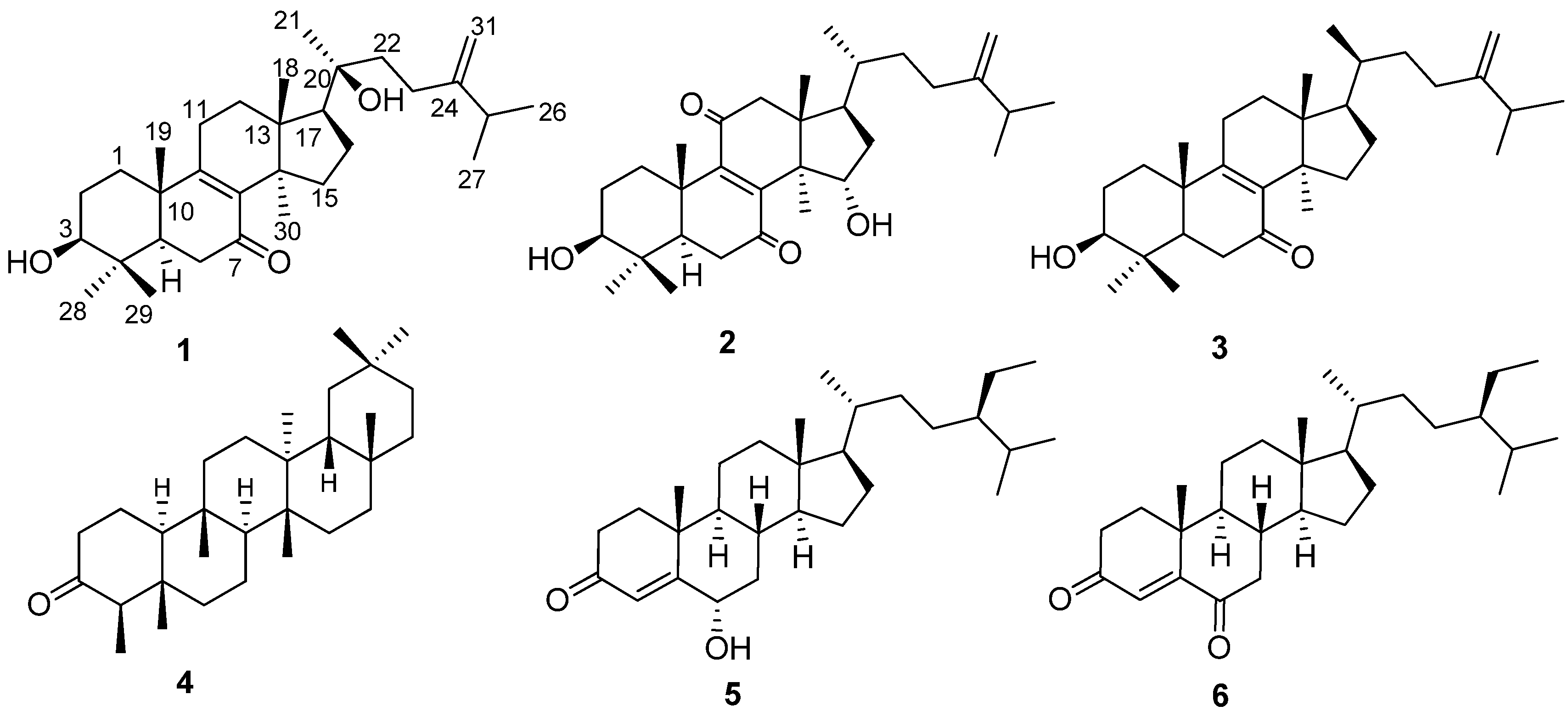
2. Results and Discussion
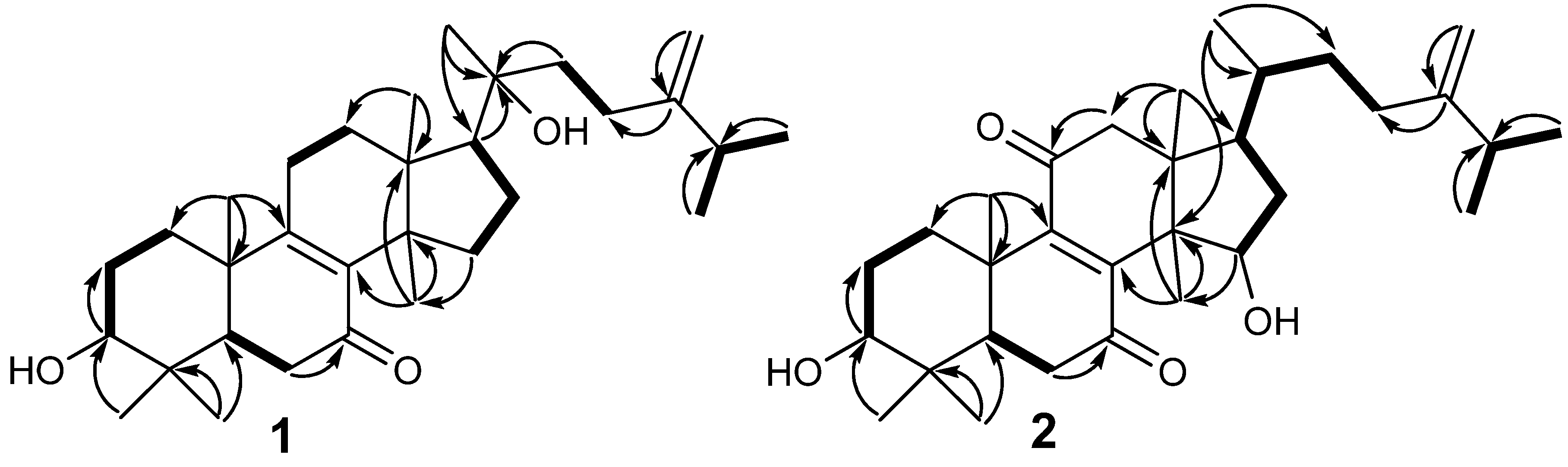
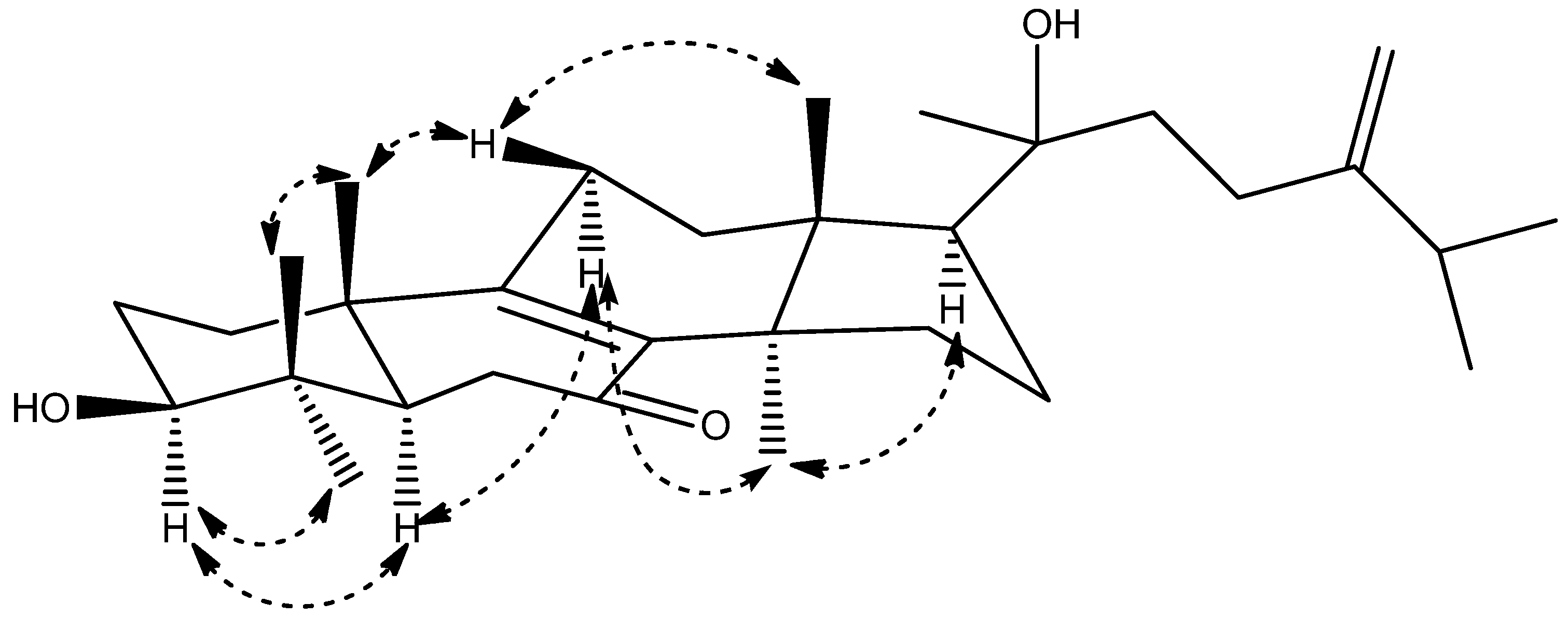
| No. | 1 | 2 | ||
|---|---|---|---|---|
| δC | δH | δC | δH | |
| 1 | 34.9, CH2 | 1.46 (1H, m, H-α) | 34.3, CH2 | 1.26 (1H, m, H-α) |
| 1.81 (1H, m, H-β) | 2.84 (1H, m, H-β) | |||
| 2 | 27.6, CH2 | 1.65 (1H, m, H-α) | 27.7, CH2 | 1.74 (2H, m) |
| 1.75 (1H, m, H-β) | ||||
| 3 | 78.1, CH | 3.26 (1H, dd, 11.6, 3.2) | 77.6, CH | 3.28 (1H, dd, 11.2, 4.4) |
| 4 | 39.1, C | 38.9, C | ||
| 5 | 49.9, CH | 1.61 (1H, overlapped) | 49.9, CH | 1.55 (1H, overlapped) |
| 6 | 36.8, CH2 | 2.39 (1H, m, H-α) | 35.6, CH2 | 2.55 (2H, m) |
| 2.44 (1H, m, H-β) | ||||
| 7 | 199.2, C | 205.6, C | ||
| 8 | 138.5, C | 150.1, C | ||
| 9 | 164.8, C | 154.8, C | ||
| 10 | 40.0, C | 40.1, C | ||
| 11 | 22.9, CH2 | 1.80 (1H, m, H-α) | 201.9, C | |
| 2.29 (1H, m, H-β) | ||||
| 12 | 30.7, CH2 | 1.79 (1H, m, H-α) | 52.2, CH2 | 2.82 (1H, d, 15.6, H-α) |
| 1.86 (1H, m, H-β) | 2.86 (1H, d, 15.6, H-β) | |||
| 13 | 45.5, C | 48.2, C | ||
| 14 | 48.1, C | 48.0, C | ||
| 15 | 31.6, CH2 | 1.77 (1H, m, H-α) | 72.3, CH | 4.34 (1H, dd, 14.4, 7.2) |
| 2.12 (1H, m, H-β) | ||||
| 16 | 23.8, CH2 | 1.26 (1H, m, H-α) | 33.7, CH2 | 1.55 (1H, m, H-α) |
| 2.25 (1H, m, H-β) | 2.21 (1H, m, H-β) | |||
| 17 | 51.0, CH | 1.80 (1H, m) | 48.4, CH | 1.82 (1H, m) |
| 18 | 17.4, CH3 | 0.83 (3H, s) | 17.3, CH3 | 0.86 (3H, s) |
| 19 | 18.6, CH3 | 1.16 (3H, s) | 17.5, CH3 | 1.29 (3H, s) |
| 20 | 75.3, C | 36.3, CH | 1.93 (1H, m) | |
| 21 | 26.3, CH3 | 1.28 (3H, s) | 18.4, CH3 | 0.89 (3H, s) |
| 22 | 42.8, CH2 | 1.56 (2H, m) | 34.5, CH2 | 1.17 (1H, m, H-α) |
| 2.56 (1H, m, H-β) | ||||
| 23 | 29.1, CH2 | 1.26 (1H, m, H-α) | 31.0, CH2 | 1.86 (1H, m, H-α) |
| 1.33 (1H, m, H-β) | 2.07 (1H, m, H-β) | |||
| 24 | 156.3, C | 156.3, C | ||
| 25 | 34.0, CH | 2.25 (1H, m) | 33.9, CH | 2.24 (1H, m) |
| 26 | 22.1, CH3 | 1.01 (3H, d, 6.6) | 22.0, CH3 | 1.01 (3H, d, 7.2) |
| 27 | 22.0, CH3 | 1.03 (3H, d, 6.6) | 21.9, CH3 | 1.03 (3H, d, 7.2) |
| 28 | 27.6, CH3 | 1.00 (3H, s) | 27.7, CH3 | 1.03 (3H, s) |
| 29 | 15.4, CH3 | 0.88 (3H, s) | 15.4, CH3 | 0.89 (3H, s) |
| 30 | 25.4, CH3 | 0.94 (3H, s) | 20.4, CH3 | 1.15 (3H, s) |
| 31 | 106.5, CH2 | 4.67 (1H, br s) | 106.6, CH2 | 4.65 (1H, br s) |
| 4.73 (1H, br s) | 4.72 (1H, br s) | |||

3. Experimental
3.1. General Information
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Characterization of Compounds 1–2
3.5. Antimicrobial Activity Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jiang, Y.; Li, B.T. Flora of China; Science Press: Beijing, China, 1979; Volume 30, pp. 93–95. [Google Scholar]
- Lu, Z.M.; Zhang, Q.J.; Chen, R.Y.; Yu, D.Q. Aporphine alkaloids from branches and leaves of Polyalthia nemoralis. China J. Chin. Mater. Med. 2009, 34, 2343–2345. [Google Scholar]
- Zhang, Y.; Wang, J.; Liu, B.J.; Yuan, Y.; Chen, G.Y.; Zhang, X.W. Study the proliferation inhibition in vitro of four plants of Polyalthia to three cancer cells. Shandong Med. J. 2011, 51, 62–64. [Google Scholar]
- Hui, Y.L.; Nan, J.S.; Yoshikkia, K.; Li, S. Anti-AIDS agents, 9. Suberosol, a new C31 lanostane-type triterpene and anti-HIV principle from Polyalthia Suberosa. J. Nat. Prod. 1993, 56, 1130–1133. [Google Scholar] [CrossRef]
- Wang, J.H.; Ji, M.H.; Chen, G.Y.; Song, X.P.; Wang, J. Chemical constituents from the roots of Polyalthia oblique. China J. Nat. Med. 2012, 4, 303–306. [Google Scholar]
- Lue, Y.P.; Mu, Q.; Zheng, H.L.; Li, C.M. 24-methylene tetracyclic triterpenes from Polyalthia lancilimba. Phytochemistry 1998, 49, 2053–2056. [Google Scholar]
- Apollinaire, T.; Kamnaing, P. Terpenoids constituents of Euphorbia sapinii. Phytochem. Lett. 2011, 4, 218–221. [Google Scholar] [CrossRef]
- Lu, Z.Q.; Chen, G.T.; Zhang, J.Q.; Huang, H.L.; Guan, S.H.; Guo, D.A. Four new lanostane triterpenoids from Euphorbia humifusa. Helv. Chim. Acta 2007, 90, 2245–2250. [Google Scholar] [CrossRef]
- Joy, K.; Winston, F.T.; Stewart, M.; William, F.R. Friedelane triterpenoids from Perztassa compta: Complete 1H and 13C assignments by 2D NMR spectroscopy. J. Nat. Prod. 1992, 55, 1626–1630. [Google Scholar] [CrossRef]
- Fan, M.S.; Ye, G.; Huang, C.G. The advances of chemistry and pharmacological study of Ficus genus. Nat. Prod. Res. Dev. 2005, 17, 497–504. [Google Scholar]
- Wang, X.H.; Han, G.Q. The chemical constituents of aristolochia KUNMINGENSIS. Acta Botanica. Yunnanica 1993, 15, 306–308. [Google Scholar]
- Solis, P.N.; Wright, C.W.; Anderson, M.M.; Gupta, M.P.; Phillipson, J.D. Antimicrobial activity and brine shrimp lethality bioassay of the leaves extract of Dillenia indica Linn. Planta Med. 1993, 59, 250–252. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–6 are available from the authors.
© 2014 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, L.-K.; Zheng, C.-J.; Li, X.-B.; Chen, G.-Y.; Han, C.-R.; Chen, W.-H.; Song, X.-P. Two New Lanostane Triterpenoids from the Branches and Leaves of Polyalthia oblique. Molecules 2014, 19, 7621-7628. https://doi.org/10.3390/molecules19067621
Wang L-K, Zheng C-J, Li X-B, Chen G-Y, Han C-R, Chen W-H, Song X-P. Two New Lanostane Triterpenoids from the Branches and Leaves of Polyalthia oblique. Molecules. 2014; 19(6):7621-7628. https://doi.org/10.3390/molecules19067621
Chicago/Turabian StyleWang, Liu-Kai, Cai-Juan Zheng, Xiao-Bao Li, Guang-Ying Chen, Chang-Ri Han, Wen-Hao Chen, and Xiao-Ping Song. 2014. "Two New Lanostane Triterpenoids from the Branches and Leaves of Polyalthia oblique" Molecules 19, no. 6: 7621-7628. https://doi.org/10.3390/molecules19067621
APA StyleWang, L.-K., Zheng, C.-J., Li, X.-B., Chen, G.-Y., Han, C.-R., Chen, W.-H., & Song, X.-P. (2014). Two New Lanostane Triterpenoids from the Branches and Leaves of Polyalthia oblique. Molecules, 19(6), 7621-7628. https://doi.org/10.3390/molecules19067621





