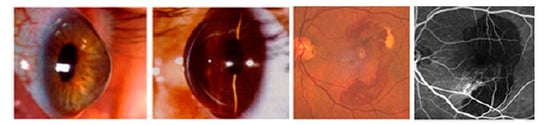
Role of Protease-Inhibitors in Ocular Diseases
Abstract
:1. Introduction
| Ocular Disorders and Site | Proteases and Protease-Inhibitors Mainly Involved in Ocular Diseases | ||
|---|---|---|---|
| Degenerative Disorder: keratoconus and keratoglobus | α2-macroglobulin↓ α1-proteinase inhinitor (α1-PI)↓ | Cathepsin B and G↑ Matrix Metalloproteinase-1,2,3 (MMP-1,2,3)↑ | Acid phosphatase↑ Catalase↑ |
| Keratitis: angiogenesis, tumor, inflammation, oxidative stress, and fibrosis activity | Serpins↑: ovalbumin, α1-antitrypsin, plasminogen activator inhibitor, maspin, pigment epithelial derived factor (PEDF), SERPINA3K | Cathepsin↑ MMP-8↑ | Calpain inhibitor↑ Catalase↑ |
| Iris and ciliary body | Secretory leukocyte protease inhibitor (SLPI)↑ | MMPs↑ | Calpain inhibitor↑ |
| Endophthalmitis, keratitis, vitreous and retina disorders | SLPI↑ | MMPs↑ | Calpain inhibitor↑ |
| Retina: angiogenesis, tumor, inflammation, oxidative stress, and fibrosis activity | SERPINA3K↑ | Metalloproteinase inhibitor-3 (TIMP-3)↓ [age-related macular degeneration, aging, Sorsby dystrophy] | Calpain inhibitor↑ |
| Retina/optic nerve: damage to the retinal ganglion cells (RGC) in glaucoma, multiple sclerosis (MS), retinitis pigmentosa, diabetes, etc. | Caspase 3 inhibitor↑ [apoptosis of RGC and damage to optic nerve fibers] | MMPs↑ | Calpain inhibitor↑ [apoptosis of RGC, damage to optic nerve fibers and photoreceptors] |
2. Protease-Inhibitors and Eye Diseases
2.1. Keratoconus
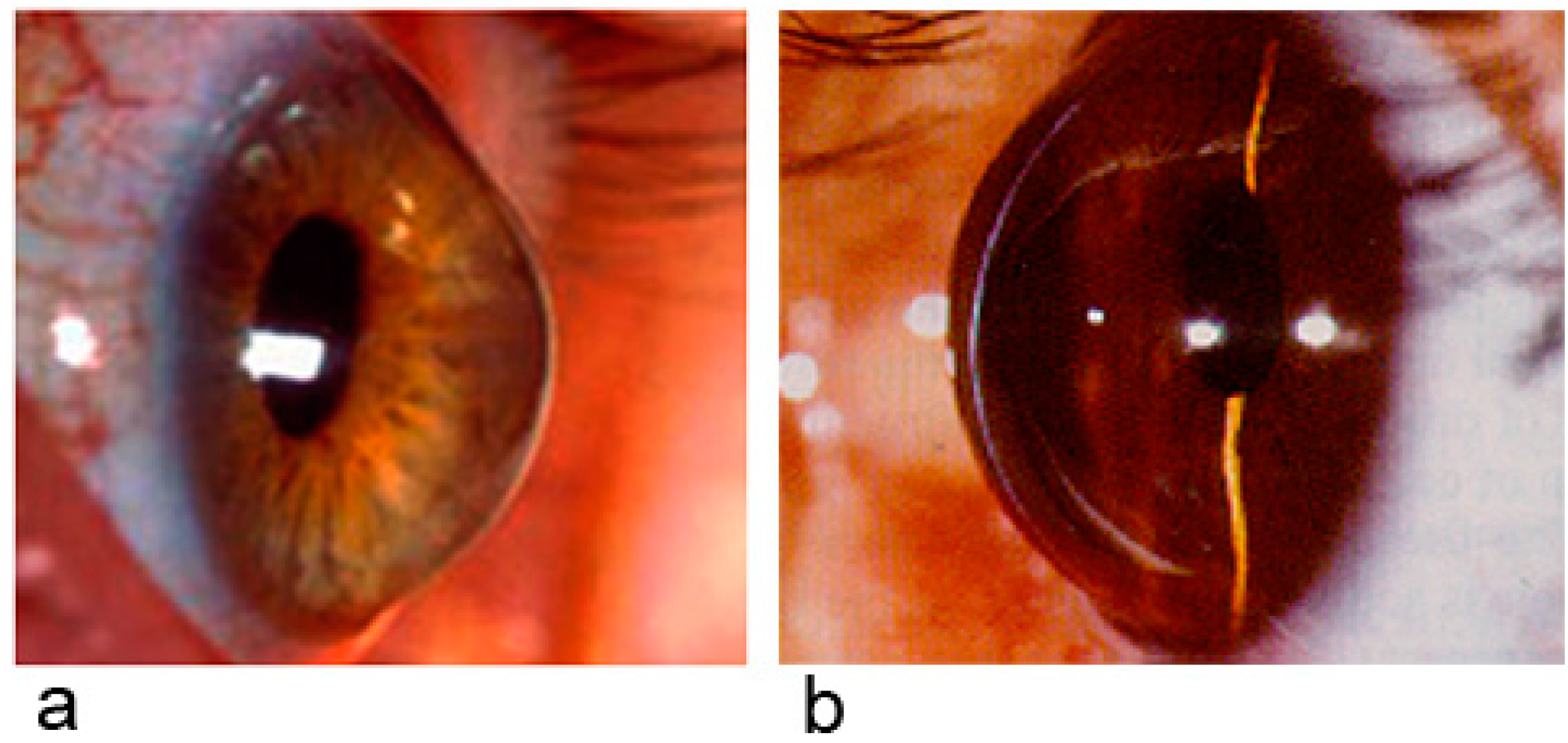
2.2. Keratoconus and α2-Macroglobulin
2.3. Keratoconus and Dysregulation
2.4. Oxidative Stress in Corneal Degenerative Disorder
2.5. Keratoglobus and Keratoconus
2.6. Cornea and Serine Protease Inhibitors
2.7. Angiogenesis, Inflammation, Oxidative Stress, and Serine Protease Inhibitors
2.8. Neurodegenerative Disorder in Retina and Optic Nerve
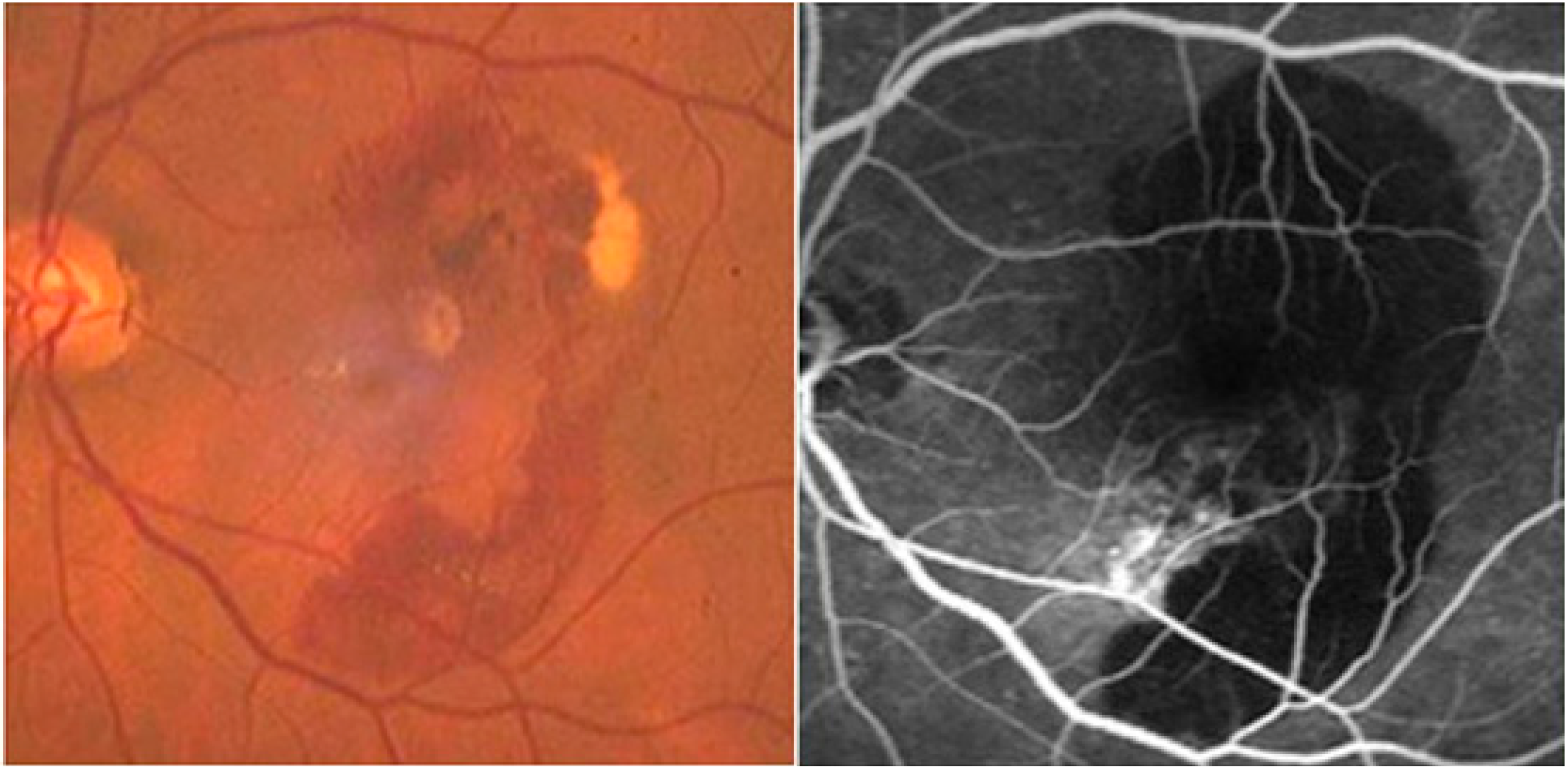
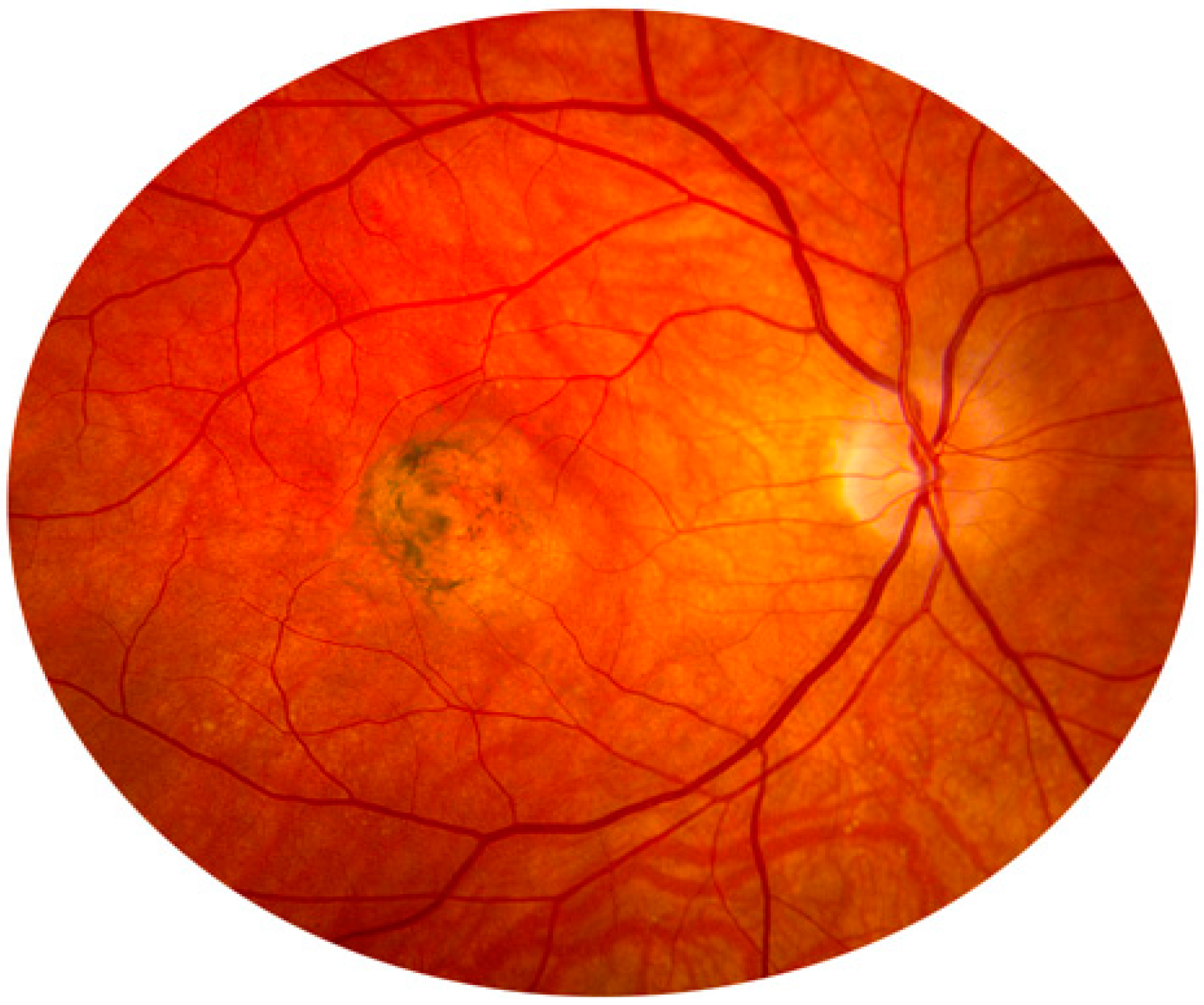
3. Eye Infections and Inflammations by Protease Inhibitors
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- López-Otín, C.; Bond, J.S. Proteases: Multifunctional enzymes in life and disease. J. Biol. Chem. 2008, 283, 30433–30437. [Google Scholar] [CrossRef]
- Habib, H.; Fazili, K.M. Plant protease inhibitors: A defense strategy in plants. Biotechnol. Mol. Biol. Rev. 2007, 2, 068–085. [Google Scholar]
- Qi, R.F.; Song, Z.W.; Chi, C.W. Structural features and molecular evolution of Bowman-Birk protease inhibitors and their potential application. Acta Biochim. Biophys. Sin. (Shanghai) 2005, 37, 283–292. [Google Scholar] [CrossRef]
- Liu, Q.; Yu, L.; Ga, J.; Fu, Q.; Zhang, J.; Zhang, P.; Chen, J.; Zhao, S. Cloning, tissue expression pattern and genomic organization of latexin, a human homologue of rat carboxypeptidase A inhibitor. Mol. Biol. Rep. 2000, 27, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Coyle, J.T. Keratoconus and eye rubbing. Am. J. Ophthalmol. 1984, 97, 527–528. [Google Scholar] [CrossRef] [PubMed]
- Macsai, M.S.; Varley, G.A.; Krachmer, J.H. Development of keratoconus after contact lens wear: Patient characteristics. Arch. Ophthalmol. 1990, 108, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Krachmer, J.H.; Feder, R.S.; Belin, M.W. Keratoconus and related non inflammatory corneal thinning disorders. Surv. Ophthalmol. 1984, 28, 293–322. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, Y.S. Keratoconus. Surv. Ophthalmol. 1998, 42, 297–319. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, M.B.; France, T.D. The ocular features of Down’s syndrome. Am. J. Ophthalmol. 1985, 99, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.Y.J.T.; Sugar, J.; Beneviniste, K. Heterogeneity in keratoconus: Possible biochemical basis. Proc. Soc. Exp. Biol. Med. 1985, 175, 336–341. [Google Scholar] [CrossRef]
- Sawaguchi, S.; Twining, S.; Yue, B.; Chang, S.; Zhou, X.; Loushin, G.; Sugar, J.; Feder, R.S. α2-macroglobulin levels in normal human and keratoconus corneas. Investig. Ophthalmol. Vis. Sci. 1994, 35, 4008–4014. [Google Scholar]
- Prause, J.U. Cellular and biochemical mechanisms involved in the degradation and healing of the cornea: The polymorphonuclear leukocyte and tear fluid serum antiproteases in human melting central corneal ulcers. Acta Ophthalmol. 1984, 168, l–40. [Google Scholar]
- Ando, H.; Twining, S.S.; Yue, B.Y.J.T.; Zhou, X.; Fini, M.E.; Kaiya, T.; Higginbotham, E.J.; Sugar, J. Matrix metalloproteinases and protease inhibitors in the human aqueous humor. Investig. Ophthalmol. Vis. Sci. 1993, 34, 3541–3548. [Google Scholar]
- Twining, S.S.; Takeo Fukuchi, T.; Wilson, P.M.; Boskovic, G. Corneal synthesis of α1-proteinase inhibitor (αl-Antitrypsin). Investig. Ophthalmol. Vis. Sci. 1994, 35, 458–462. [Google Scholar]
- Whitelock, R.B.; Fukuchi, T.; Zhou, L.; Twining, S.S.; Sugar, J.; Feder, R.S.; Yue, B.Y. Cathepsin G, acid phosphatase, and αl-proteinase inhibitor messenger RNA levels in keratoconus corneas. Investig. Ophthalmol. Vis. Sci. 1997, 38, 529–534. [Google Scholar]
- Zhou, L.; Sawaguchi, S.; Twining, S.S.; Sugar, J.; Feder, R.S.; Yue, B.Y. Expression of degradative enzymes and protease inhibitors in corneas with keratoconus. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1117–1124. [Google Scholar]
- Li, Y.; Zhou, L.; Twining, S.S.; Sugar, J.; Yue, B.Y. Involvement of Sp1 elements in the promoter activity of the α1-proteinase inhibitor gene. J. Biol. Chem. 1998, 273, 9959–9965. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, Y.; Wang, X.; Li, Y.; Yue, B.Y. Involvement of Sp1 elements in the promoter activity of genes affected in keratoconus. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1980–1985. [Google Scholar]
- Shen, X.; Park, J.S.; Qiu, Y.; Sugar, J.; Yue, B.Y. Effects of Sp1 overexpression on cultured human corneal stromal Cells. Genes Cells 2009, 14, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Chiambaretta, F.; Nakamura, H.; de Graeve, F.; Sakai, H.; Marceau, G.; Maruyama, Y.; Rigal, D.; Dastugue, B.; Sugar, J.; Yue, B.Y.; et al. Krüppel-like factor 6 (KLF6) affects the promoter activity of the α1-proteinase inhibitor gene. Investig. Ophthalmol. Vis. Sci. 2006, 47, 582–590. [Google Scholar] [CrossRef]
- Maruyama, I.; Zhou, L.; Sugar, J.; Yue, B.Y. Normal expression levels of cathepsins, protease inhibitors, and Sp1 in conjunctival tissues from patients with keratoconus. Curr. Eye Res. 2000, 21, 886–890. [Google Scholar] [CrossRef] [PubMed]
- Buddi, R.; Lin, B.; Atilano, S.R.; Zorapapel, N.C.; Kenney, M.C.; Brown, D.J. Evidence of oxidative stress in human corneal diseases. J. Histochem. Cytochem. 2002, 50, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Gondhowiardjo, T.D.; van Haeringen, N.J. Corneal aldehyde dehydrogenase, glutathione reductase, and glutathione S-transferase in pathologic corneas. Cornea 1993, 12, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Behndig, A.; Karlsson, K.; Johansson, B.O.; Brännström, T.; Marklund, S.L. Superoxide dismutase isoenzymes in the normal and diseased human cornea. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2293–2296. [Google Scholar]
- Gondhowiardjo, T.D.; van Haeringen, N.J.; Volker-Dieben, H.J.; Beekhuis, H.W.; Kok, J.H.; van Rij, G.; Pels, L.; Kijlstra, A. Analysis of corneal aldehyde dehydrogenase patterns in pathologic corneas. Cornea 1993, 12, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Sibata, T.; Wasada, H.; Toyokuni, S.; Uchida, K. Structural basis of proteinbound endogenous aldehydes: Chemical and immunochemical characterizations of configurational isomers of a 4-hydroxy-2-nonenal-histidine adduct. J. Biol. Chem. 2003, 278, 5044–5051. [Google Scholar] [CrossRef] [PubMed]
- Kenney, M.C.; Chwa, M.; Atilano, S.R.; Tran, A.; Carballo, M.; Saghizadeh, M.; Vasiliou, V.; Adachi, W.; Brown, D.J. Increased levels of catalase and cathepsin V/L2 but decreased TIMP-1 in keratoconus corneas: Evidence that oxidative stress plays a role in this disorder. Investig. Ophthalmol. Vis. Sci. 2005, 46, 823–832. [Google Scholar] [CrossRef]
- Meghpara, B.; Nakamura, H.; Vemuganti, G.K.; Murthy, S.I.; Sugar, J.; Yue, B.Y.; Edward, D.P. Histopathologic and immunohistochemical studies of keratoglobus. Arch. Ophthalmol. 2009, 127, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Zicari, A.M.; Nebbioso, M.; Lollobrigida, V.; Bardanzellu, F.; Celani, C.; Occasi, F.; Cesoni Marcelli, A.; Duse, M. Vernal keratoconjunctivitis: Atopy and autoimmunity. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1419–1423. [Google Scholar] [PubMed]
- Narayan, M.; Twining, S. Focus on molecules: Maspin. Exp. Eye Res. 2010, 90, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Anisowicz, A.; Hendrix, M.J.; Thor, A.; Neveu, M.; Sheng, S.; Rafidi, K.; Seftor, E.; Sager, R. Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells. Science 1994, 263, 526–529. [Google Scholar] [CrossRef]
- Ngamkitidechakul, C.; Burke, M.J.; O’Brien, W.J.; Twining, S.S. Maspin: Synthesis by human cornea and regulation of in vitro stromal cell adhesion to extracellular matrix. Investig. Ophthalmol. Vis. Sci. 2001, 42, 3135–3141. [Google Scholar]
- Horswill, M.A.; Narayan, M.; Warejcka, D.J.; Cirillo, L.A.; Twining, S.S. Epigenetic silencing of maspin expression occurs early in the conversion of keratocytes to fibroblasts. Exp. Eye Res. 2008, 86, 586–600. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Volpert, O.; Shi, Y.H.; Bouck, N. Maspin is an angiogenesis inhibitor. Nat. Med. 2000, 6, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Narayan, M.; Mirza, S.P.; Twining, S. Identification of phosphorylation sites on extracellular corneal epithelial cell maspin. Proteinomics 2011, 11, 1382–1390. [Google Scholar] [CrossRef]
- Shellenberger, T.D.; Mazumdar, A.; Henderson, Y.; Briggs, K.; Wang, M.; Chattopadhyay, C.; Jayakumar, A.; Frederick, M.; Clayman, G.L. Headpin: A serpin with endogenous and exogenous suppression of angiogenesis. Cancer Res. 2005, 65, 11501–11509. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Hu, Y.; Ma, J.X. Anti-inflammatory and antioxidant effects of SERPINA3K in the retina. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3943–3952. [Google Scholar] [CrossRef]
- Liu, X.; Lin, Z.; Zhou, T.; Zong, R.; He, H.; Liu, Z.; Ma, J.X.; Liu, Z.; Zhou, Y. Anti-angiogenic and anti-inflammatory effects of SERPINA3K on corneal injury. PLoS One 2011, 6, e16712. [Google Scholar] [CrossRef] [PubMed]
- Nebbioso, M.; Federici, M.; Rusciano, D.; Evangelista, M.; Pescosolido, N. Oxidative stress in pre-retinopathic diabetic subjects and antioxidants. Diabetes Technol. Ther. 2012, 14, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Nebbioso, M.; Pranno, F.; Pescosolido, N. Lipoic acid in animal models and clinical use in diabetic retinopathy. Expert Opin. Pharmacother. 2013, 14, 1829–1838. [Google Scholar] [CrossRef] [PubMed]
- Macgregor, A.M.; Eberhart, C.G.; Fraig, M.; Lu, J.; Halushka, M.K. Tissue inhibitor of matrix metalloproteinase-3 levels in the extracellular matrix of lung, kidney, and eye increase with age. J. Histochem. Cytochem. 2009, 57, 207–2013. [Google Scholar] [CrossRef] [PubMed]
- Anand-Apte, B.; Pepper, M.S.; Voest, E.; Montesano, R.; Olsen, B.; Murphy, G.; Apte, S.S.; Zetter, B. Inhibition of angiogenesis by tissue inhibitor of metalloproteinase-3. Investig. Ophthalmol. Vis. Sci. 1997, 38, 817–822. [Google Scholar]
- Kermer, P.; Klocker, N.; Labes, M.; Bahr, M. Inhibition of CPP32-Like Proteases Rescues Axotomized Retinal Ganglion Cells from Secondary Cell Death In Vivo. J. Neurosci. 1998, 18, 4656–4662. [Google Scholar] [PubMed]
- Ma, M.; Shofer, F.S.; Neumar, R.W. Calpastatin Overexpression Protects Axonal Transport in an In Vivo Model of Traumatic Axonal Injury. J. Neurotrauma 2012, 29, 2555–2563. [Google Scholar] [CrossRef] [PubMed]
- Shanab, A.Y.; Nakazawa, T.; Ryu, M.; Tanaka, Y.; Himori, N.; Taguchi, K.; Yasuda, M.; Watanabe, R.; Takano, J.; Saido, T.; et al. Metabolic stress response implicated in diabetic retinopathy: The role of calpain, and the therapeutic impact of calpain inhibitor. Neurobiol. Dis. 2012, 48, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Guyton, M.-K.; Smith, A.; Wallace, G.; McDowell, M.L.; Matzelle, D.D.; Ray, S.K.; Banik, N.L. Calpain inhibitor attenuated optic nerve damage in acute optic neuritis in rats. J. Neurochem. 2013, 124, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Vigneswara, V.; Berry, M.; Logan, A.; Ahmed, Z. Pharmacological Inhibition of Caspase-2 Protects Axotomised Retinal Ganglion Cells from Apoptosis in Adult Rats. PLoS One 2012, 7, e53473. [Google Scholar] [CrossRef]
- Malagola, R.; Giannotti, R.; Arrico, L. OCT: Macular thickness in insulin-dependent patients without evident retinopathy: A preliminary study. Clin. Ter. 2012, 163, e393–e396. [Google Scholar] [PubMed]
- Nebbioso, M.; Scarsella, G.; Tafani, M.; Pescosolido, N. Mechanisms of ocular neuroprotection by antioxidant molecole in animal models. J. Biol. Regul. Homeost. Agents 2013, 27, 197–209. [Google Scholar] [PubMed]
- Persson, H.; Kawashima, S.; Karlsson, J.O. Immunohistochemical localization of calpains and calpastatin in the rabbit eye. Brain Res. 1993, 611, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Caberoy, N.B.; Alvarado, G.; Li, W. Identification of Calpain Substrates by ORF Phage Display. Molecules 2011, 16, 1739–1748. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.E.; Ollivier, F.J. Matrix metalloproteinase inhibition in corneal ulceration. Vet. Clin. N. Am. Small Anim. Pract. 2004, 34, 611–622. [Google Scholar] [CrossRef]
- Reviglio, V.E.; Grenat, A.; Pegoraro, F.; Sambuelli, R.H.; Rana, T.; Kuo, I.C. Secretory leukoprotease inhibitor: A native antimicrobial protein in the innate immune response in a rat model of S. aureus Keratitis. J. Ophthalmol. 2009, 2009, 259393. [Google Scholar] [CrossRef] [PubMed]
- Reviglio, V.E.; Sambuelli, R.H.; Olmedo, A.; Falco, M.; Echenique, J.; O’Brien, T.P.; Kuo, I.C. Secretory leukocyte protease inhibitor is an inducible antimicrobial peptide expressed in staphylococcus aureus endophthalmitis. Mediat. Inflamm. 2007, 2007, 93857. [Google Scholar]
- Smith, A.W.; Das, A.; Guyton, K.; Ray, S.K.; Rohrer, B.; Banik, N.L. Calpain inhibition attenuates apoptosis of retinal ganglion cells in acute optic neuritis. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4935–4941. [Google Scholar] [CrossRef]
- Das, A.; Garner, D.P.; Del Re, A.M.; Woodward, J.J.; Maneesh Kumar, D.; Agarwal, N.; Banik, N.L.; Ray, S.K. Calpeptin provides functional neuroprotection to rat retinal ganglion cells following Ca2+ influx. Brain Res. 2006, 1084, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Kuro, M.; Yoshizawa, K.; Uehara, N.; Miki, H.; Takahashi, K.; Tsubura, A. Calpain inhibition restores basal autophagy and suppresses MNU-induced photoreceptor cell death in mice. In Vivo 2011, 25, 617–623. [Google Scholar] [PubMed]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pescosolido, N.; Barbato, A.; Pascarella, A.; Giannotti, R.; Genzano, M.; Nebbioso, M. Role of Protease-Inhibitors in Ocular Diseases. Molecules 2014, 19, 20557-20569. https://doi.org/10.3390/molecules191220557
Pescosolido N, Barbato A, Pascarella A, Giannotti R, Genzano M, Nebbioso M. Role of Protease-Inhibitors in Ocular Diseases. Molecules. 2014; 19(12):20557-20569. https://doi.org/10.3390/molecules191220557
Chicago/Turabian StylePescosolido, Nicola, Andrea Barbato, Antonia Pascarella, Rossella Giannotti, Martina Genzano, and Marcella Nebbioso. 2014. "Role of Protease-Inhibitors in Ocular Diseases" Molecules 19, no. 12: 20557-20569. https://doi.org/10.3390/molecules191220557
APA StylePescosolido, N., Barbato, A., Pascarella, A., Giannotti, R., Genzano, M., & Nebbioso, M. (2014). Role of Protease-Inhibitors in Ocular Diseases. Molecules, 19(12), 20557-20569. https://doi.org/10.3390/molecules191220557