Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts
Abstract
:1. Introduction
2. Results and Discussion
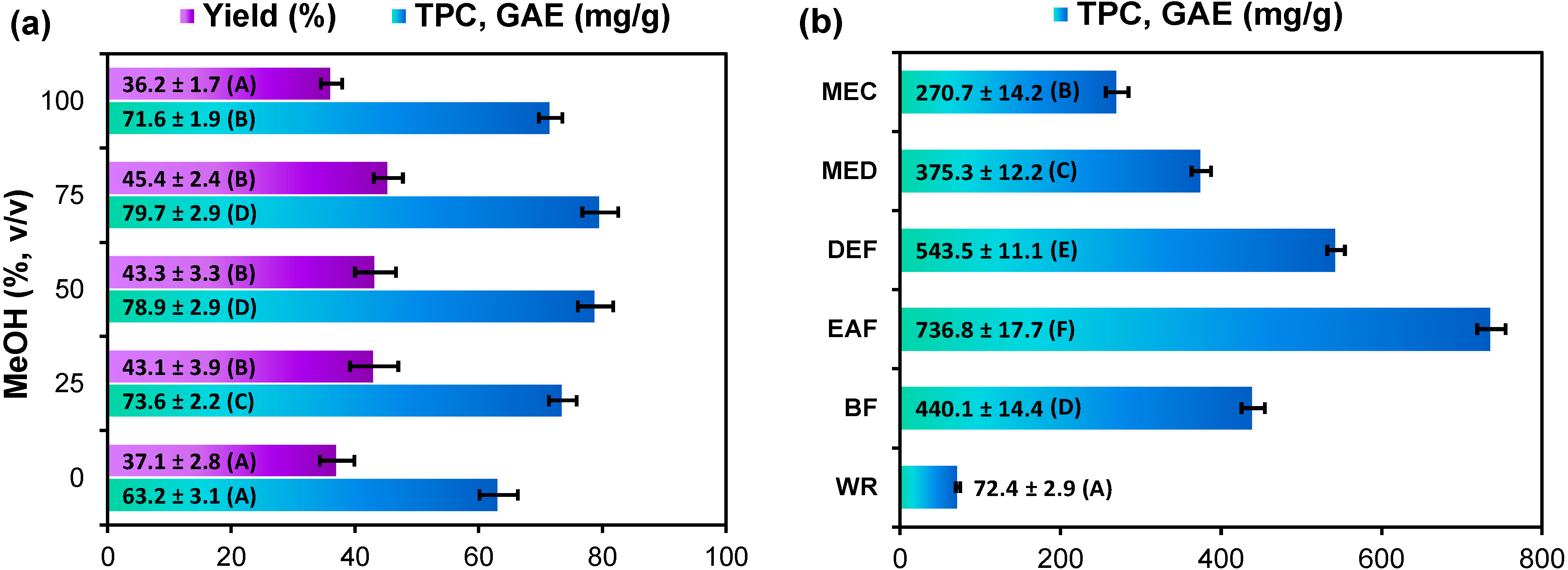
2.1. Extraction Yield and Total Phenolic Content of G. procumbens Dry Leaf Extracts
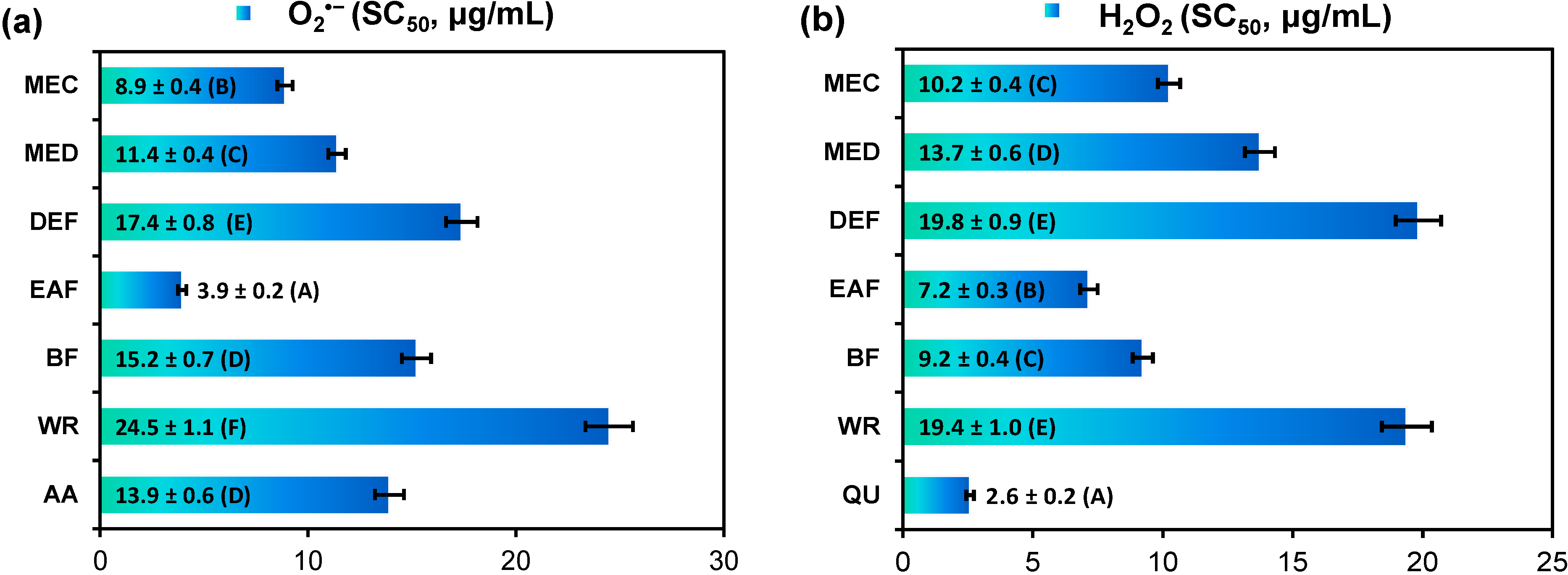
2.2. Antioxidant Activity of G. procumbens Dry Leaf Extracts
| Analyte | Radical Scavenging Activity b | Reducing Power c | LA-Peroxidation d |
|---|---|---|---|
| DPPH (EC50, µg/mL) | FRAP (mmol Fe2+/g) | (IC50, µg/mL) | |
| MEC | 8.35 ± 0.28 G | 4.58 ± 0.24 B | 175.98 ± 7.78 H |
| MED | 6.67 ± 0.43 F | 5.97 ± 0.21 C | 207.98 ± 9.47 I |
| DEF | 4.34 ± 0.24 E | 12.50 ± 0.84 F | 109.39 ± 5.36 G |
| EAF | 2.90 ± 0.15 C | 12.77 ± 0.76 F | 123.94 ± 6.11 G |
| BF | 4.94 ± 0.25 E | 8.17 ± 0.48 D | 164.77 ± 5.77 H |
| WR | 30.91 ± 1.43 H | 1.46 ± 0.08 A | 651.85 ± 20.21 J |
| CA | 2.17 ± 0.11 B | 25.37 ± 0.44 I | 69.68 ± 0.70 F |
| CHA | 4.42 ± 0.13 E | 18.04 ± 0.79 H | 52.47 ± 2.03 E |
| QU | 1.63 ± 0.07 A | 36.02 ± 1.10 J | 48.51 ± 1.74 D |
| RT | 3.44 ± 0.09 D | 11.89 ± 0.70 E,F | 67.73 ± 0.34 F |
| BHA | 2.90 ± 0.14 C | 16.13 ± 0.83 G | 14.33 ± 0.70 A |
| BHT | 6.54 ± 0.28 F | 18.89 ± 0.42 H | 21.58 ± 0.95 B |
| TBHQ | 2.73 ± 0.12 C | 15.50 ± 0.71 G | 36.53 ± 1.04 C |
| TX | 4.34 ± 0.22 E | 10.83 ± 0.32 E | 22.45 ± 1.10 B |
| r (p) for: | DPPH EC50 | FRAP | LA-Inh IC50 | O2•− SC50 | H2O2 SC50 |
|---|---|---|---|---|---|
| DPPH EC50 | ― | −0.7822 (0.066) | 0.9919 (0.000) * | 0.7450 (0.089) | 0.5579 (0.250) |
| FRAP | −0.7822 (0.066) | ― | −0.7744 (0.071) | −0.4957 (0.317) | −0.2574 (0.622) |
| LA-Inh IC50 | 0.9919 (0.000) * | −0.7744 (0.071) | ― | 0.7297 (0.100) | 0.5185 (0.292) |
| O2•− SC50 | 0.7450 (0.089) | −0.4957 (0.317) | 0.7297 (0.100) | ― | 0.8203 (0.046) * |
| H2O2 SC50 | 0.5579 (0.250) | −0.2574 (0.622) | 0.5185 (0.292) | 0.8203 (0.046) * | ― |
| TPC (GAE) | −0.8255 (0.043) * | 0.9604 (0.002) * | −0.7970 (0.058) | −0.6800 (0.137) | −0.4751 (0.341) |
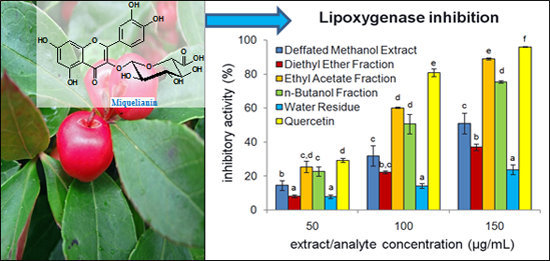
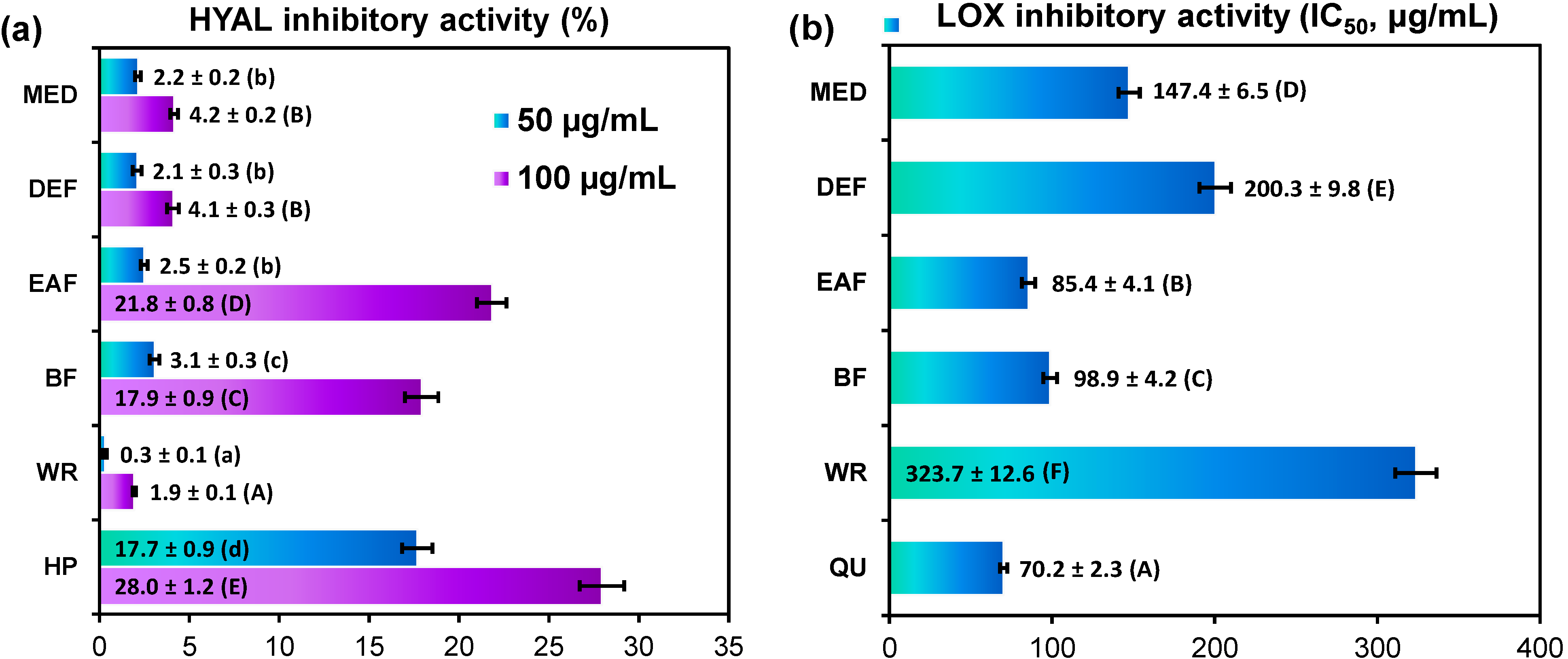
2.3. Anti-Inflammatory Activity of G. procumbens Dry Leaf Extracts
2.4. Qualitative UHPLC-PDA-ESI-MS3 Profiling of G. procumbens Leaf Phenolics
| Peak | Analyte | Rt (min) | UV λmax (nm) | [M−H]− (m/z) | Formula | Extract |
|---|---|---|---|---|---|---|
| 1 | protocatechuic acid (PCA) a | 4.4 | 295 | 153 | C7H6O4 | DEF |
| 2 | 3-O-caffeoylquinic acid (NCHA) a | 6.2 | 325 | 353 | C16H18O9 | all |
| 3 | p-hydroxybenzoic acid (pHBA) a | 7.6 | 254 | 137 | C7H6O3 | DEF |
| 4 | 3-O-p-coumaroylquinic acid a | 9.3 | 310 | 337 | C16H18O8 | DEF, EAF, BF |
| 5 | vanillic acid a | 10.3 | 260, 291 | 167 | C8H8O4 | DEF |
| 6 | 5-O-caffeoylquinic acid (CHA) a | 10.7 | 325 | 353 | C16H18O9 | MED, EAF, BF, WR |
| 7 | (+)-catechin (CA) a | 10.9 | 280 | 289 | C15H14O6 | MED, DEF |
| 8 | caffeic acid (CFA) a | 11.7 | 325 | 179 | C9H8O4 | DEF |
| 9 | 4-O-caffeoylquinic acid (CCHA) a | 12.6 | 325 | 353 | C16H18O9 | all |
| 10 | 3-O-feruloylquinic acid | 14.2 | 325 | 367 | C17H20O9 | EAF |
| 11 | procyanidin B-type dimer | 14.9 | 280 | 577 | C30H26O12 | MED, DEF, EAF, BF |
| 12 | unknown compound | 15.7 | 254 | 481 | MED, DEF, BF | |
| 13 | (−)-epicatechin (ECA) a | 16.5 | 280 | 289 | C15H14O6 | MED, DEF, EAF, BF |
| 14 | 4-O-p-coumaroylquinic acid | 16.6 | 310 | 337 | C16H18O8 | BF |
| 15 | unknown compound | 17.3 | 280 | 559 | all | |
| 16 | p-coumaric acid (pCA) a | 19.1 | 310 | 163 | C9H8O3 | DEF |
| 17 | procyanidin A-type trimer (PA) | 19.8 | 280 | 863 | C45H36O18 | MED, DEF, EAF, BF |
| 18 | procyanidin B-type trimer | 20.8 | 280 | 865 | C45H38O18 | MED, EAF, BF |
| 19 | procyanidin A-type dimer | 22.1 | 280 | 575 | C30H24O12 | MED, EAF, BF |
| 20 | unknown compound | 22.2 | 267, 298 | 639 | DEF | |
| 21 | caffeoylquinic acid derivative | 22.4 | 325 | 391 | MED, EAF | |
| 22 | unknown compound | 24.3 | 280 | 473 | DEF, EAF | |
| 23 | quercetin pentoside-glucuronide | 24.9 | 257, 356 | 609 | C26H26O17 | MED, DEF, BF, WR |
| 24 | procyanidin A-type trimer | 25.1 | 280 | 863 | C45H36O18 | EAF |
| 25 | unknown compound | 26.3 | 280 | 451 | DEF, EAF | |
| 26 | quercetin 3-O-galactoside ( HY) a | 27.2 | 254, 353 | 463 | C21H20O12 | MED, DEF, EAF, BF |
| 27 | quercetin 3-O-glucoside ( IQ) a | 28.1 | 256, 353 | 463 | C21H20O12 | all |
| 28 | quercetin 3-O-glucuronide ( MQ) a | 28.8 | 256, 356 | 477 | C21H18O13 | all |
| 29 | quercetin 3-O-arabinoside (GV) a | 30.3 | 258, 356 | 433 | C20H18O11 | MED, DEF, EAF, BF |
| 30 | quercetin derivative | 30.6 | 258, 354 | 333 | DEF | |
| 31 | kaempferol 3-O-glucuronide | 33.0 | 265, 349 | 461 | C21H18O12 | all |
| 32 | quercetin 3-O-glucuronide methyl ester | 33.9 | 265, 356 | 491 | C22H20O13 | MED, DEF, EAF, BF |
| 33 | kaempferol 3-O-glucuronide methyl ester | 38.8 | 265, 348 | 475 | C22H20O12 | EAF |
| 34 | unknown compound | 39.0 | 286, 326 | 409 | BF | |
| 35 | kaempferol 3-O-glucoside ( AG) a | 39.9 | 265, 345 | 447 | C21H20O11 | EAF |
| 36 | unknown compound | 40.3 | 280 | 451 | DEF, EAF | |
| 37 | quercetin (QU) a | 43.3 | 255, 364 | 301 | C15H10O7 | DEF, EAF |
| 38 | unknown compound | 43.7 | 286, 326 | 409 | BF | |
| 39 | unknown compound | 44.4 | 280 | 435 | DEF | |
| 40 | quercetin 3-O-pentoside-glucuronide butyl ester | 4.4 | 295 | 153 | C30H34O17 | BF |
| 41 | quercetin 3-O-glucuronide butyl ester | 6.2 | 325 | 353 | C25H26O13 | BF |
| 42 | kaempferol 3-O-pentoside-glucuronide butyl ester | 7.6 | 254 | 137 | C30H34O16 | BF |
| 43 | kaempferol (KA) a | 9.3 | 310 | 337 | C15H10O6 | DEF |
| 44 | kaempferol 3-O-glucuronide butyl ester | 10.3 | 260, 291 | 167 | C25H26O12 | BF |
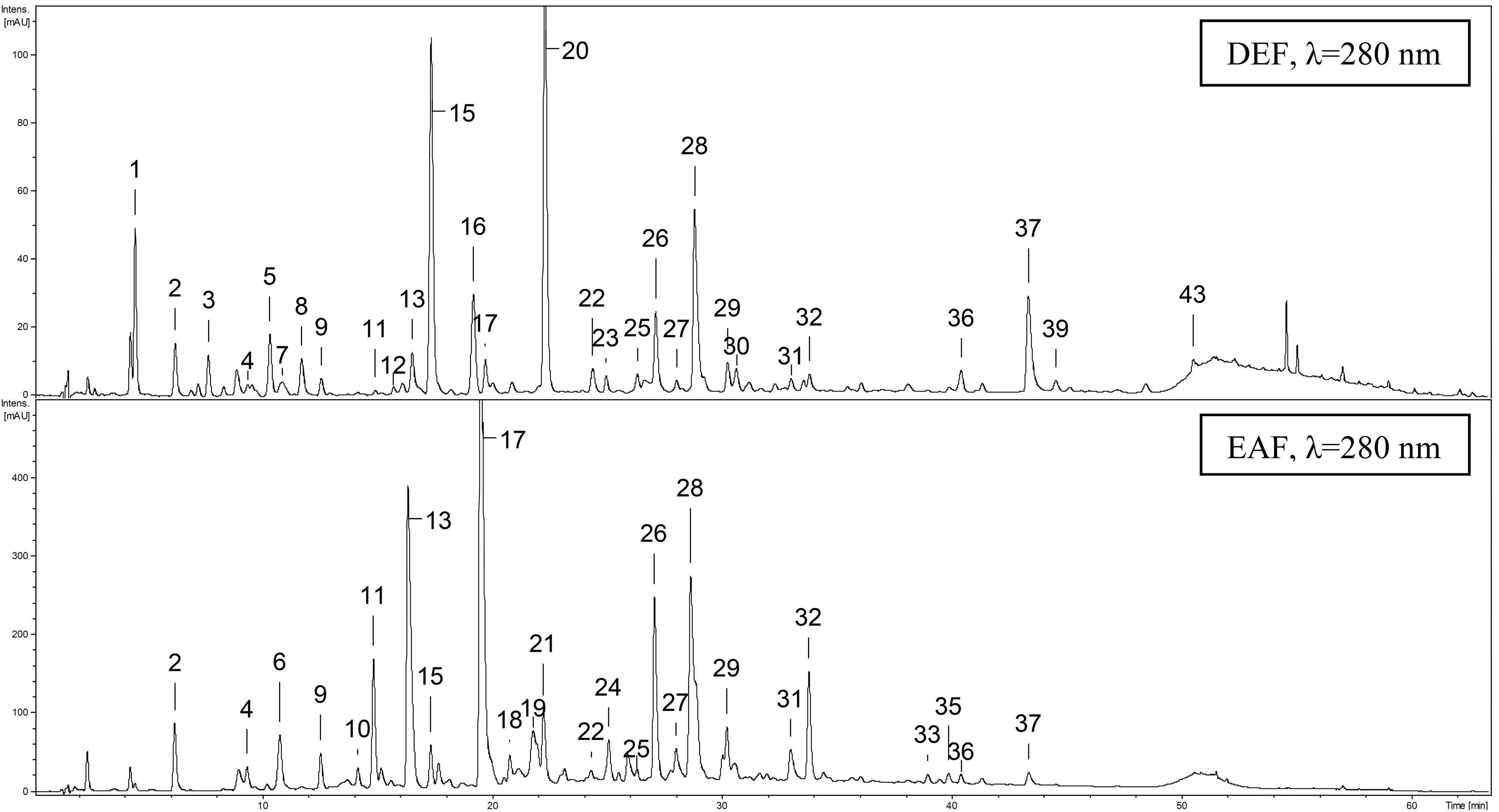
2.4.1. Phenolic Acids and Related Phenolic Compounds
2.4.2. Flavan-3-ols (Catechins and Proanthocyanidins)
2.4.3. Flavonoids
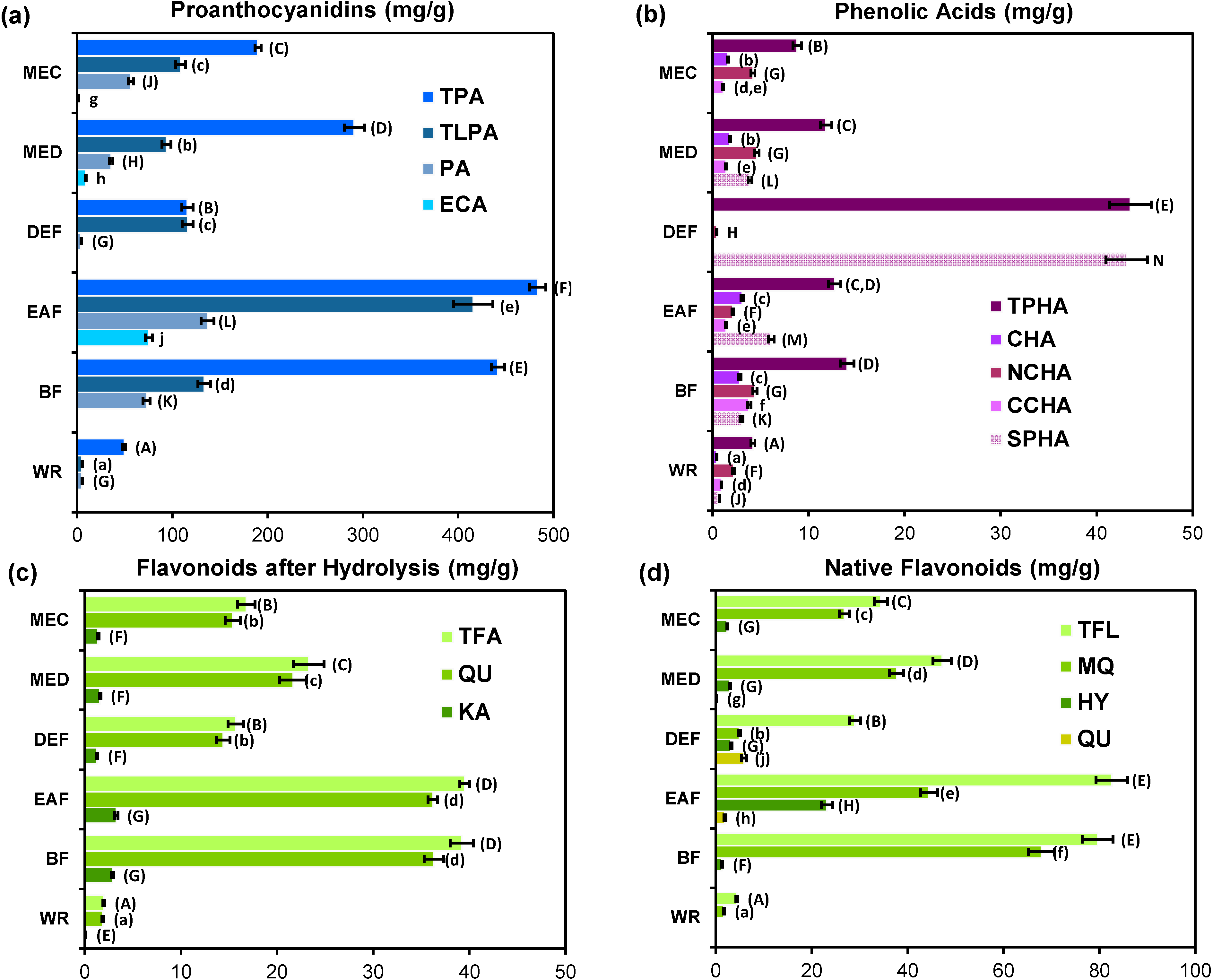
2.5. Quantitative Profiling of G. procumbens Leaf Phenolics
3. Experimental Section
3.1. General
3.2. Plant Material and Preparation of Dry Extracts
3.3. Determination of Total Phenolic Content (TPC)
3.4. Biological Activity Testing
3.4.1. Reactive Oxygen Species Scavenging Tests
3.4.2. Ferric Reducing Antioxidant Power (FRAP) Assay
3.4.3. Linoleic Acid Peroxidation Test (Ferric Thiocyanate Method, FTC)
3.4.4. Hyaluronidase and Lipoxygenase Inhibition Tests
3.5. Phytochemical Profiling
3.5.1. UHPLC-PDA-ESI-MS3 and HPLC-PDA Fingerprint Conditions
3.5.2. Determination of Total Contents of Main Groups of Polyphenols (TPA and TFA)
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prakash, D.; Kumar, N. Cost effective natural antioxidants. In Nutrients, Dietary Supplements, and Nutriceuticals: Cost Analysis Versus Clinical Benefits, 1st ed.; Watson, R.R., Gerald, J.K., Preedy, V.R., Eds.; Humana Press: New York, NY, USA, 2011; pp. 163–187. [Google Scholar]
- Giampieri, F.; Alvarez-Suarez, J.M.; Battino, M. Strawberry and human health: Effects beyond antioxidant activity. J. Agric. Food Chem. 2014, 62, 3867–3876. [Google Scholar] [CrossRef]
- Battino, M.; Beekwilder, J.; Denoyes-Rothan, B.; Laimer, M.; McDougall, G.J.; Mezzetti, B. Bioactive compounds in berries relevant to human health. Nutr. Rev. 2009, 67 (Suppl. S1), S145–S150. [Google Scholar] [CrossRef]
- Brown, E.M.; Latimer, C.; Allsopp, P.; Ternan, N.G.; McMullan, G.; McDougall, G.J.; Stewart, D.; Crozier, A.; Rowland, I.; Gill, C.I.R. In vitro and in vivo models of colorectal cancer: Antigenotoxic activity of berries. J. Agric. Food Chem. 2014, 62, 3852–3866. [Google Scholar] [CrossRef]
- Rodriquez-Mateos, A.; Heiss, C.; Borges, G.; Crozier, A. Berry (poly)phenols and cardiovascular health. J. Agric. Food Chem. 2014, 62, 3842–3851. [Google Scholar] [CrossRef]
- Sharma, J.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-inflammatory effects in humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, S.Y. Oxygen Radical Absorbing Capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. J. Agric. Food Chem. 2003, 51, 502–509. [Google Scholar] [CrossRef]
- Ehlenfeldt, M.K.; Prior, R.L. Oxygen Radical Absorbance Capacity (ORAC) and phenolic and anthocyanin concentrations in fruit and leaf tissues of highbush blueberry. J. Agric. Food Chem. 2001, 49, 2222–2227. [Google Scholar] [CrossRef]
- Pavlović, R.D.; Lakusić, B.; Doslov-Kokorus, Z.; Kovacević, N. Arbutin content and antioxidant activity of some Ericaceae species. Pharmazie 2009, 64, 656–659. [Google Scholar]
- Mechikova, G.Y.; Kuzmich, A.S.; Ponomarenko, L.P.; Kalinovsky, A.I.; Stepanova, T.A.; Fedorov, S.N.; Stonik, V.A. Cancer-preventive activities of secondary metabolites from leaves of the bilberry Vaccinium smallii A. Gray. Phytother. Res. 2010, 24, 1730–1732. [Google Scholar] [CrossRef]
- Oliveira, I.; Coelho, V.; Baltasar, R.; Pereira, J.A.; Baptista, P. Scavenging capacity of strawberry tree (Arbutus unedo L.) leaves on free radicals. Food Chem. Toxicol. 2009, 47, 1507–1511. [Google Scholar] [CrossRef]
- Middleton, D.J. Infrageneric classification of the genus Gaultheria L. (Ericaceae). Bot. J. Linn. Soc. 1991, 106, 229–258. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, R.; Sun, L.; Huang, C.; Wang, C.; Zhang, D.M.; Zhang, T.T.; Du, G.H. Anti-inflammatory activity of methyl salicylate glycosides isolated from Gaultheria yunnanensis (Franch.) Rehder. Molecules 2011, 16, 3875–3884. [Google Scholar] [CrossRef]
- Nikolić, M.; Marković, T.; Mojović, M.; Pejin, B.; Savić, A.; Perić, T.; Mojović, D.; Stević, T.; Soković, M. Chemical composition and biological activity of Gaultheria procumbens L. essential oil. Ind. Crops Prod. 2013, 49, 561–567. [Google Scholar] [CrossRef]
- Garcia-Lafuente, A.; Guillamon, E.; Villares, A.; Rostagno, M.A.; Martinez, J.A. Flavonoids as anti-inflammatory agents: Implications in cancer and cardiovascular disease. Inflamm. Res. 2009, 58, 537–552. [Google Scholar] [CrossRef]
- Li, W.G.; Zhang, X.Y.; Wu, Y.J.; Tian, X. Anti-inflammatory effect and mechanism of proanthocyanidins from grape seeds. Acta Pharmacol. Sin. 2001, 22, 1117–1120. [Google Scholar]
- Youn, J.; Lee, K.H.; Won, J.; Huh, S.J.; Yun, H.S.; Cho, W.G.; Paik, D.J. Beneficial effects of rosmarinic acid on suppression of collagen induced arthritis. J. Rheumatol. 2003, 30, 1203–1207. [Google Scholar]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Matkowski, A. Plant phenolic metabolites as antioxidants and mutagenesis inhibitors. In Cell Biology and Instrumentation: UV Radiation, Nitric Oxide and Cell Death in Plants, 1st ed.; Blume, Y., Durzan, D.J., Smertenko, P., Eds.; IOS Press: Amsterdam, The Netherlands, 2006; pp. 129–148. [Google Scholar]
- Saleem, A.; Harris, C.S.; Asim, M.; Cuerrier, A.; Martineau, L.; Haddad, P.S.; Arnason, J.T. A RP-HPLC-DAD-APCI/MSD method for the characterization of medicinal Ericaceae used by the Eeyou Istchee Cree first nations. Phytochem. Anal. 2010, 21, 328–339. [Google Scholar] [CrossRef]
- McCune, L.M.; Johns, T. Antioxidant activity in medicinal plants associated with the symptoms of diabetes mellitus used by the Indigenous Peoples of the North American boreal forest. J. Ethnopharmacol. 2002, 82, 197–205. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar]
- Capasso, F.; Gaginella, T.S.; Grandolini, G.; Izzo, A.A. Phytotherapy—A Quick Reference to Herbal Medicine, 13th ed.; Springer Verlag: Berlin/Heidelberg, Germany, 2003; pp. 45–60. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Kneifel, W.; Czech, E.; Kopp, B. Microbial contamination of medicinal plants-a review. Planta Med. 2002, 68, 5–15. [Google Scholar] [CrossRef]
- Ling, L.T.; Radhakrishnan, A.K.; Subramaniam, T.; Cheng, H.M.; Palanisamy, U.D. Assessment of antioxidant capacity and cytotoxicity of selected Malaysian plants. Molecules 2010, 15, 2139–2151. [Google Scholar] [CrossRef]
- Erol, N.T.; Sari, F.; Calikoglu, E.; Velioglu, Y.S. Green and roasted mate: phenolic profile and antioxidant activity. Turk. J. Agric. For. 2009, 33, 353–362. [Google Scholar]
- Bazylko, A.; Parzonko, A.; Jeż, W.; Osińska, E.; Kiss, A.K. Inhibition of ROS production, photoprotection, and total phenolic, flavonoids and ascorbic acid content of fresh herb juice and extractsfrom the leaves and flowers of Tropaeolum majus. Ind. Crops Prod. 2014, 55, 19–24. [Google Scholar] [CrossRef]
- Buetler, T.M.; Krauskopf, A.; Ruegg, U.T. Role of superoxide as a signaling molecule. Physiology 2004, 19, 120–123. [Google Scholar] [CrossRef]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef]
- Termentzi, A.; Kefalas, P.; Kokkalou, E. Antioxidant activities of various extracts and fractions of Sorbus domestica fruit at different maturity stages. Food Chem. 2006, 98, 599–608. [Google Scholar] [CrossRef]
- Peyrat-Maillard, M.N.; Cuvelier, M.E.; Berset, C. Antioxidant activity of phenolic compounds in 2,2'-azobis (2-amidinopropane) dihydrochloride (AAPH)-induced oxidation: Synergistic and antagonistic effects. J. Am. Oil Chem. Soc. 2003, 80, 1007–1012. [Google Scholar] [CrossRef]
- Bellick, Y.; Boukraâ, L.; Alzahrani, H.A.; Bakhotmah, B.A.; Abdellah, F.; Hammoudi, S.M.; Igeur-Ouada, M. Molecular mechanism underlying anti-inflammatory and anti-allergic activities of polyphenols: An update. Molecules 2013, 18, 322–353. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSn identification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef]
- Nagels, L.; van Dongen, W.; de Brucker, J.; de Pooter, H. High-performance liquid chromatographic separation of naturally occuring esters of phenolic acids. J. Chromatogr. A 1980, 187, 181–187. [Google Scholar] [CrossRef]
- Su, S.; Cui, W.; Zhou, W.; Duan, J.; Shang, E.; Tang, Y. Chemical fingerprint and quantitative constituent analysis of Siwu decoction categorized formule by UPLC-QTOF/MS/MS and HPLC-DAD. Chin. Med. 2013, 8, 1–15. [Google Scholar] [CrossRef]
- Li, S.; Xiao, J.; Chen, L.; Hu, C.; Chen, P.; Xie, B.; Sun, Z. Identification of A-series oligomeric procyanidins from pericarp of Litchi chinensis by FT-ICR-MS and LC-MS. Food Chem. 2012, 135, 31–38. [Google Scholar] [CrossRef]
- Fabre, N.; Rustan, I.; de Hoffmann, E.; Quetin-Leclercq, J. Determination of flavone, flavonol, and flavanone aglycones by negative ion liquid chromatography electrospray ion trap mass spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 707–715. [Google Scholar] [CrossRef]
- Perestrelo, R.; Lu, Y.; Santos, S.A.O.; Silvestre, A.J.D.; Neto, C.P.; Camara, J.S.; Rocha, S.M. Phenolic profile of Sercial and Tinta Negra Vitis vinifera L. grape skins by HPLC-DAD-ESI-MSn. Novel phenolic compounds in Vitis vinifera L. grape. Food Chem. 2012, 135, 94–104. [Google Scholar] [CrossRef]
- Li, H.; Ma, Q.; Liu, Y.; Qian, J.; Zhou, J.; Zhao, Y. Chemical constituents from Polygonum perfoliatum. Chin. J. Appl. Environ. Biol. 2009, 15, 615–620. [Google Scholar]
- Liu, W.R.; Qiao, W.L.; Liu, Z.Z.; Wang, X.H.; Jiang, R.; Li, S.Y.; Shi, R.B.; She, G.M. Gaultheria: Phytochemical and pharmacological characteristics. Molecules 2013, 18, 12071–12108. [Google Scholar] [CrossRef]
- Santos-Buelga, C.; Scalbert, A. Proanthocyanidins and tannin-like compounds—Nature, occurrence, dietary intake and effects on nutrition and health. J. Sci. Food Agric. 2000, 80, 1094–1117. [Google Scholar] [CrossRef]
- Yokozawa, T.; Chen, C.P.; Dong, E.; Tanaka, T.; Nonaka, G.I.; Nishioka, I. Study on the inhibitory effect of tannins and flavonoids against the 1,1-diphenyl-2-picrylhydrazyl radical. Biochem. Pharmacol. 1998, 56, 231–222. [Google Scholar] [CrossRef]
- Guo, X.D.; Zhang, D.Y.; Gao, X.J.; Parry, J.; Liu, K.; Liu, B.L.; Wang, M. Quercetin and quercetin-3-O-glucuronide are effective in ameliorating endothelial insulin resistance through inhibition of reactive oxygen species-associated inflammation. Mol. Nutr. Food Res. 2013, 57, 1037–1045. [Google Scholar] [CrossRef]
- Olszewska, M.; Wolbiś, M. Further flavonoids from the flowers of Prunus spinosa L. Acta Pol. Pharm. 2002, 59, 133–137. [Google Scholar]
- Olszewska, M.A.; Michel, P. Antioxidant activity of inflorescences, leaves and fruits of three Sorbus species in relation to their polyphenolic composition. Nat. Prod. Res. 2009, 23, 1507–1521. [Google Scholar] [CrossRef]
- Olszewska, M.A.; Presler, A.; Michel, P. Profiling of phenolic metabolites and antioxidant activity of dry extracts from the selected Sorbus species. Molecules 2012, 17, 3093–3113. [Google Scholar] [CrossRef]
- Granica, S.; Czerwińska, M.E.; Piwowarski, J.P.; Ziaja, M.; Kiss, A.K. Chemical composition, antioxidative and anti-inflammatory activity of extracts prepared from aerial parts of Oenothera biennis L. and Oenothera paradoxa Hudziok obtained after seeds cultivation. J. Agric. Food Chem. 2013, 61, 801–810. [Google Scholar]
- Schepetkin, I.A.; Kirpotina, L.N.; Jakiw, L.; Khlebnikov, A.I.; Blaskovich, C.L.; Jutila, M.A.; Quinn, M.T. Immunomodulatory activity of oenothein B isolated from Epilobium angustifolium. J. Immunol. 2009, 83, 6754–6766. [Google Scholar] [CrossRef]
- Piwowarski, J.P.; Kiss, A.K.; Kozłowska-Wojciechowska, M. Anti-hyaluronidase and anti-elastase activity screening of tannin-rich plant materials used in traditional Polish medicine for external treatment of diseases with inflammatory background. J. Ethnopharmacol. 2011, 137, 937–941. [Google Scholar] [CrossRef]
- Olszewska, M. Quantitative HPLC analysis of flavonoids and chlorogenic acid in the leaves and inflorescences of Prunus serotina Ehrh. Acta Chromatogr. 2007, 19, 253–269. [Google Scholar]
- Sample Availability: Samples of the dry leaf extracts are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michel, P.; Dobrowolska, A.; Kicel, A.; Owczarek, A.; Bazylko, A.; Granica, S.; Piwowarski, J.P.; Olszewska, M.A. Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts. Molecules 2014, 19, 20498-20520. https://doi.org/10.3390/molecules191220498
Michel P, Dobrowolska A, Kicel A, Owczarek A, Bazylko A, Granica S, Piwowarski JP, Olszewska MA. Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts. Molecules. 2014; 19(12):20498-20520. https://doi.org/10.3390/molecules191220498
Chicago/Turabian StyleMichel, Piotr, Anna Dobrowolska, Agnieszka Kicel, Aleksandra Owczarek, Agnieszka Bazylko, Sebastian Granica, Jakub P. Piwowarski, and Monika A. Olszewska. 2014. "Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts" Molecules 19, no. 12: 20498-20520. https://doi.org/10.3390/molecules191220498
APA StyleMichel, P., Dobrowolska, A., Kicel, A., Owczarek, A., Bazylko, A., Granica, S., Piwowarski, J. P., & Olszewska, M. A. (2014). Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts. Molecules, 19(12), 20498-20520. https://doi.org/10.3390/molecules191220498