Anti-Obesity Effects of Hispidin and Alpinia zerumbet Bioactives in 3T3-L1 Adipocytes
Abstract
:1. Introduction
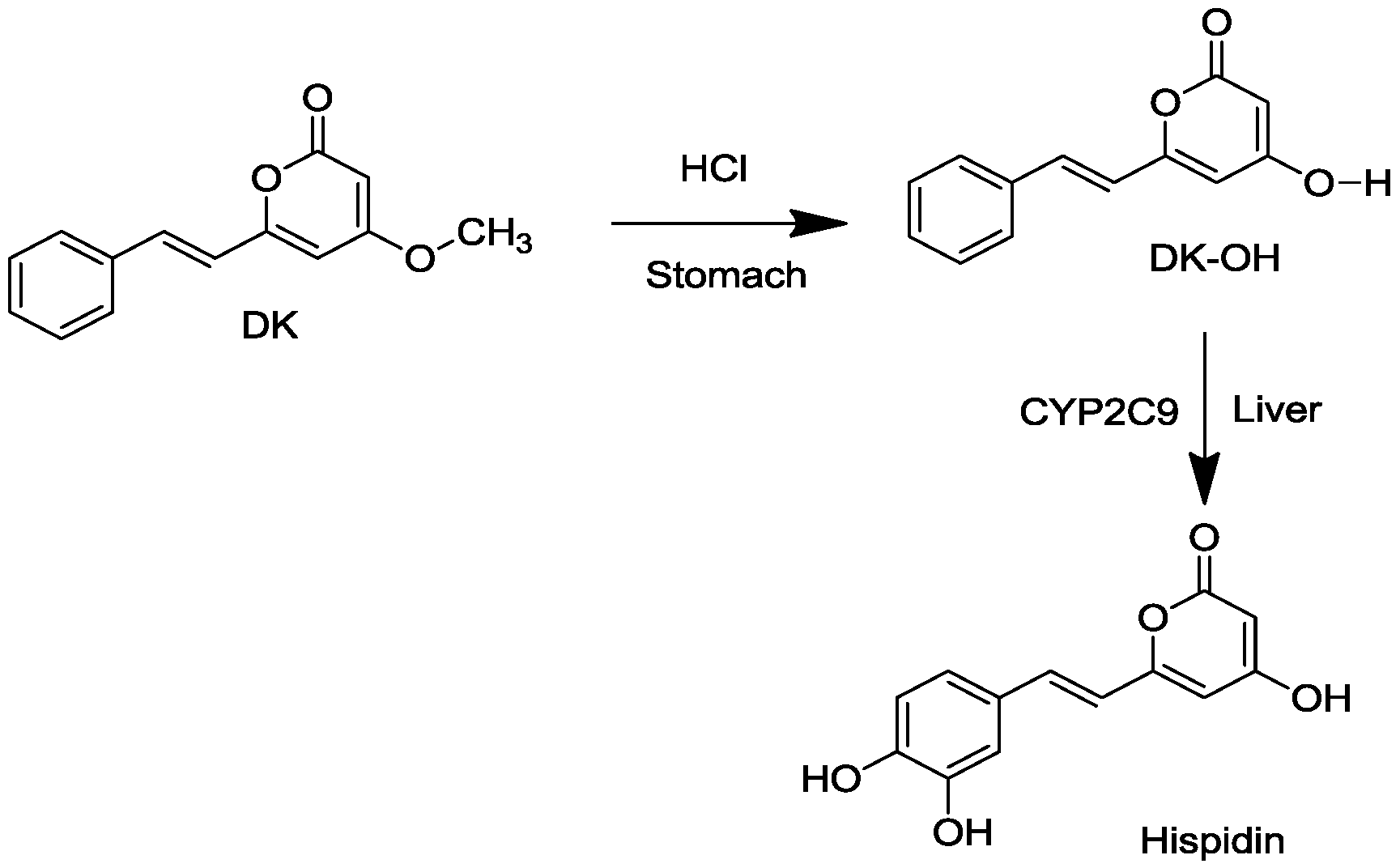
2. Results and Discussion
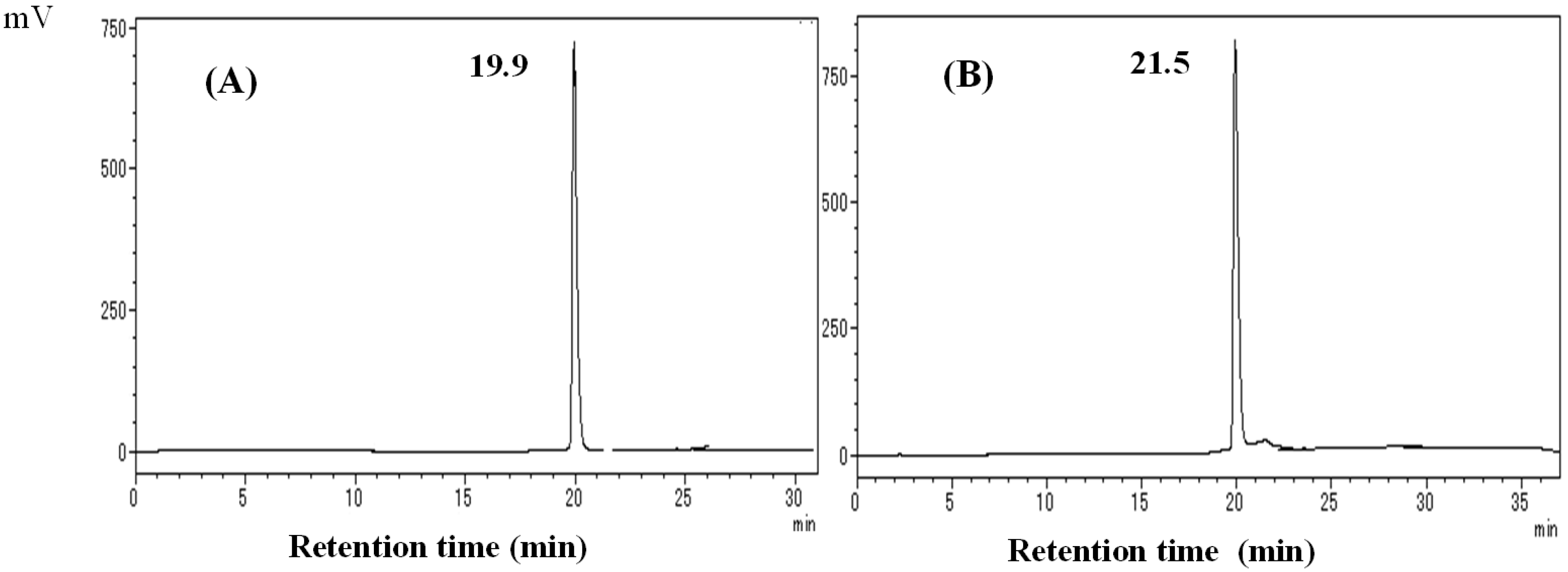
2.1. Conversion of DK to Hispidin and Purification of DK and DDK
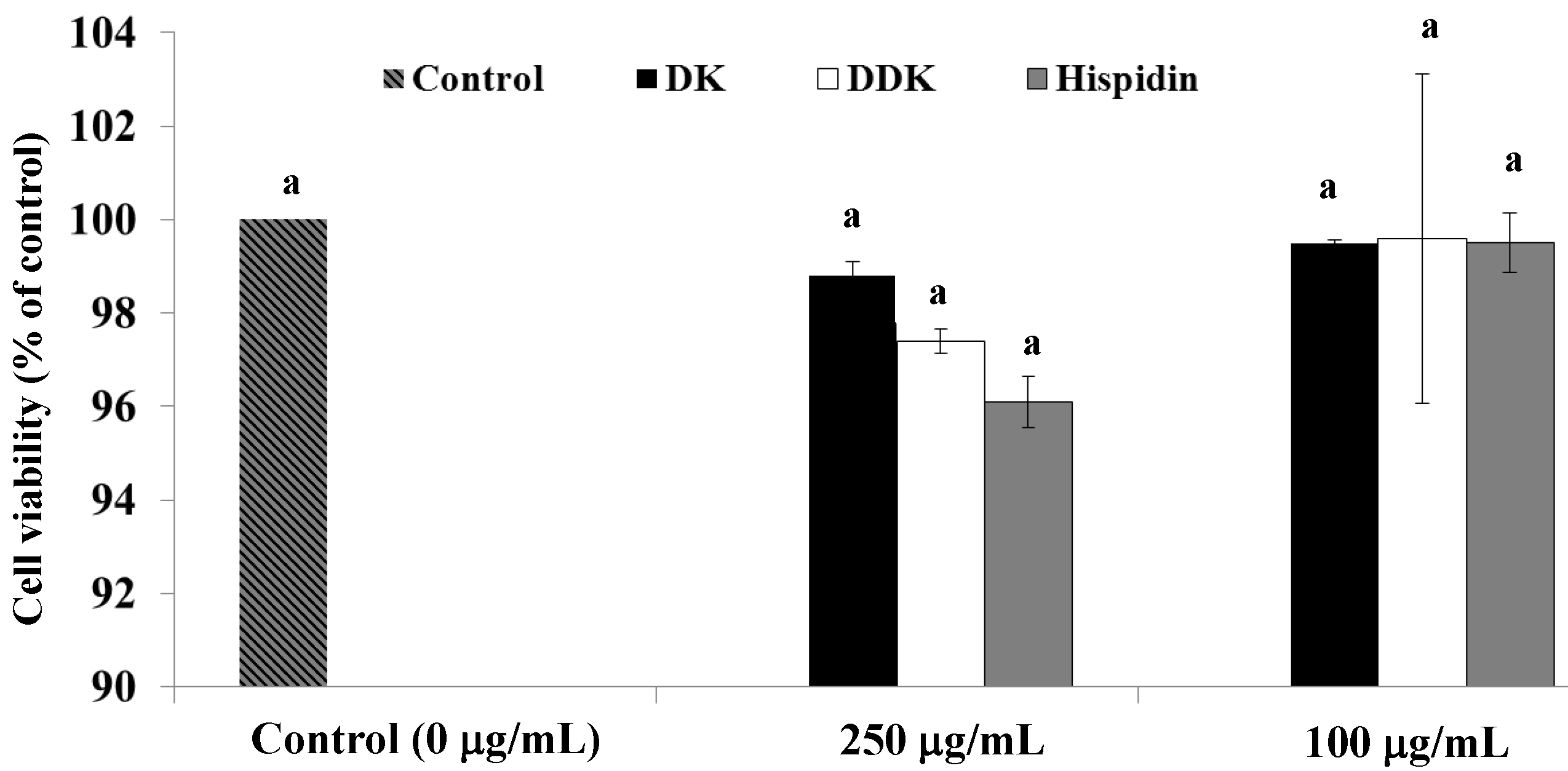
2.2. Effect of Hispidin, DK and DDK on Cell Viability
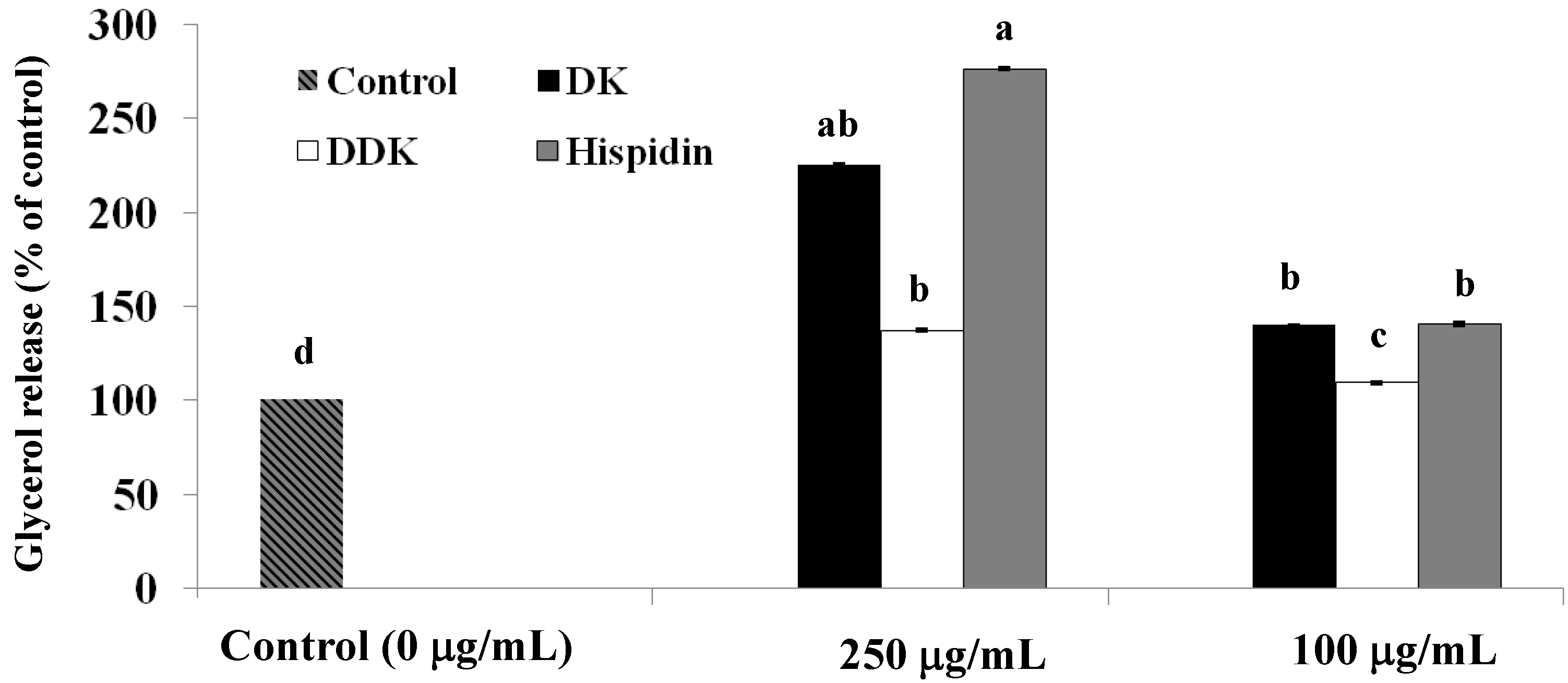
2.3. Stimulation of Glycerol Release
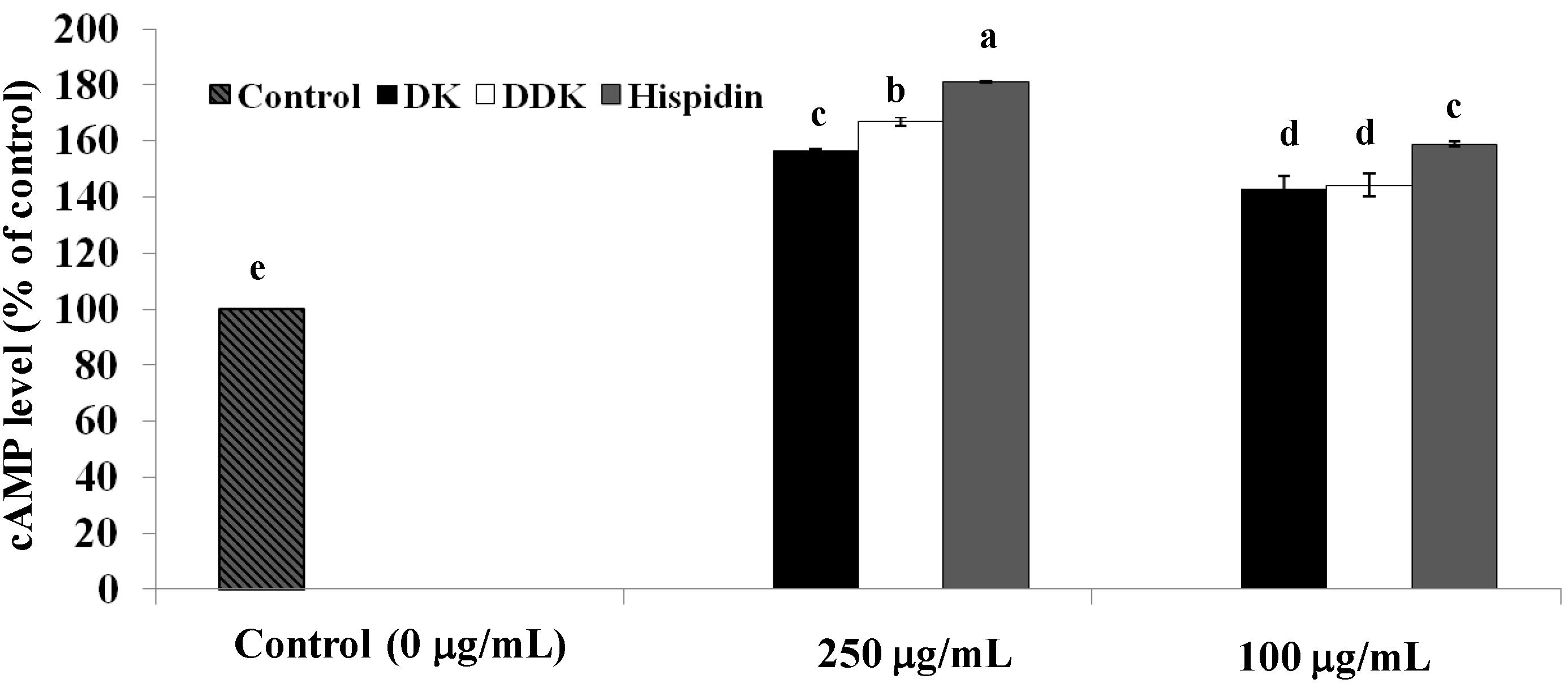
2.4. Measurement of Intracellular cAMP
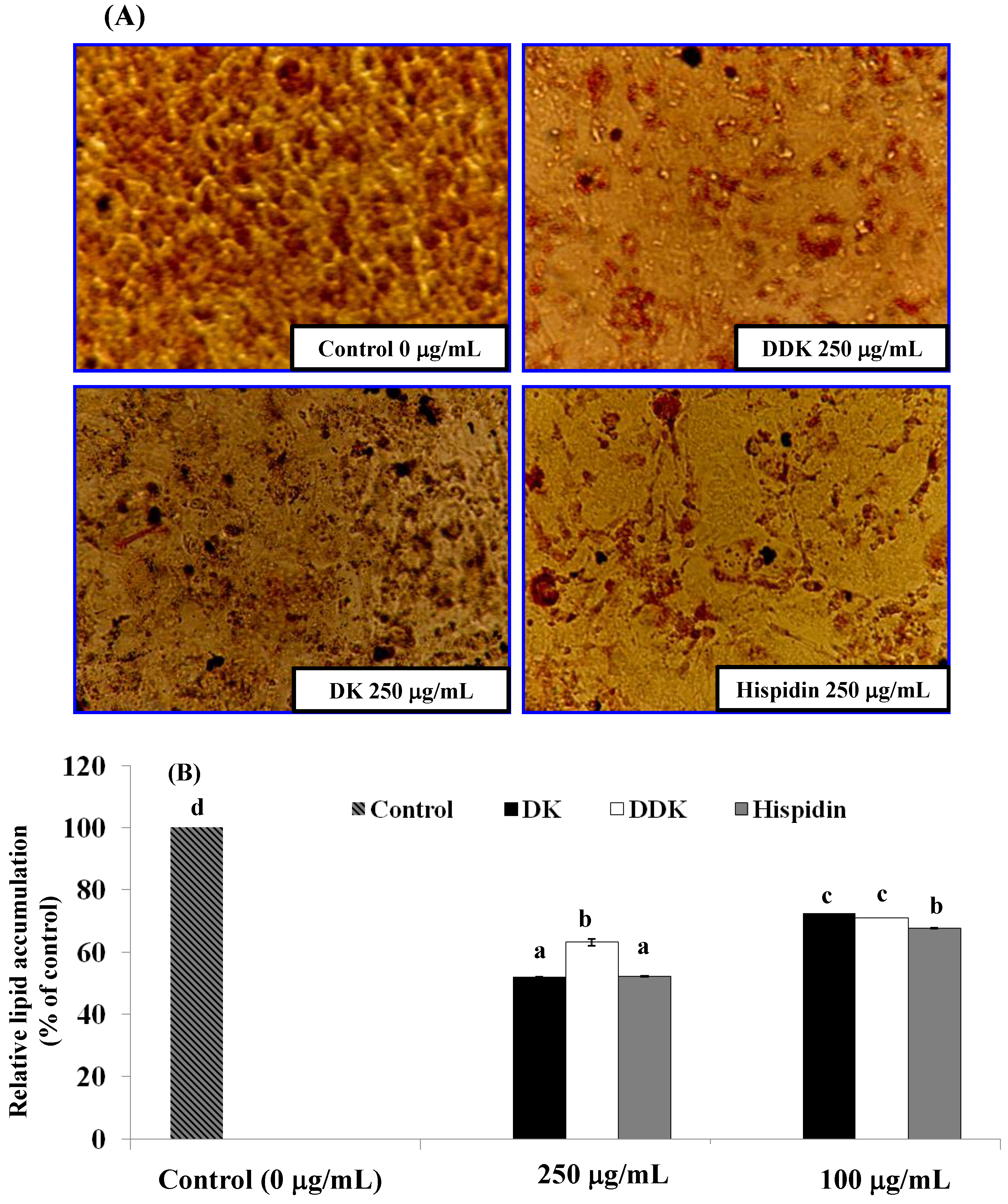
2.5. Evaluation of Lipid Accumulation
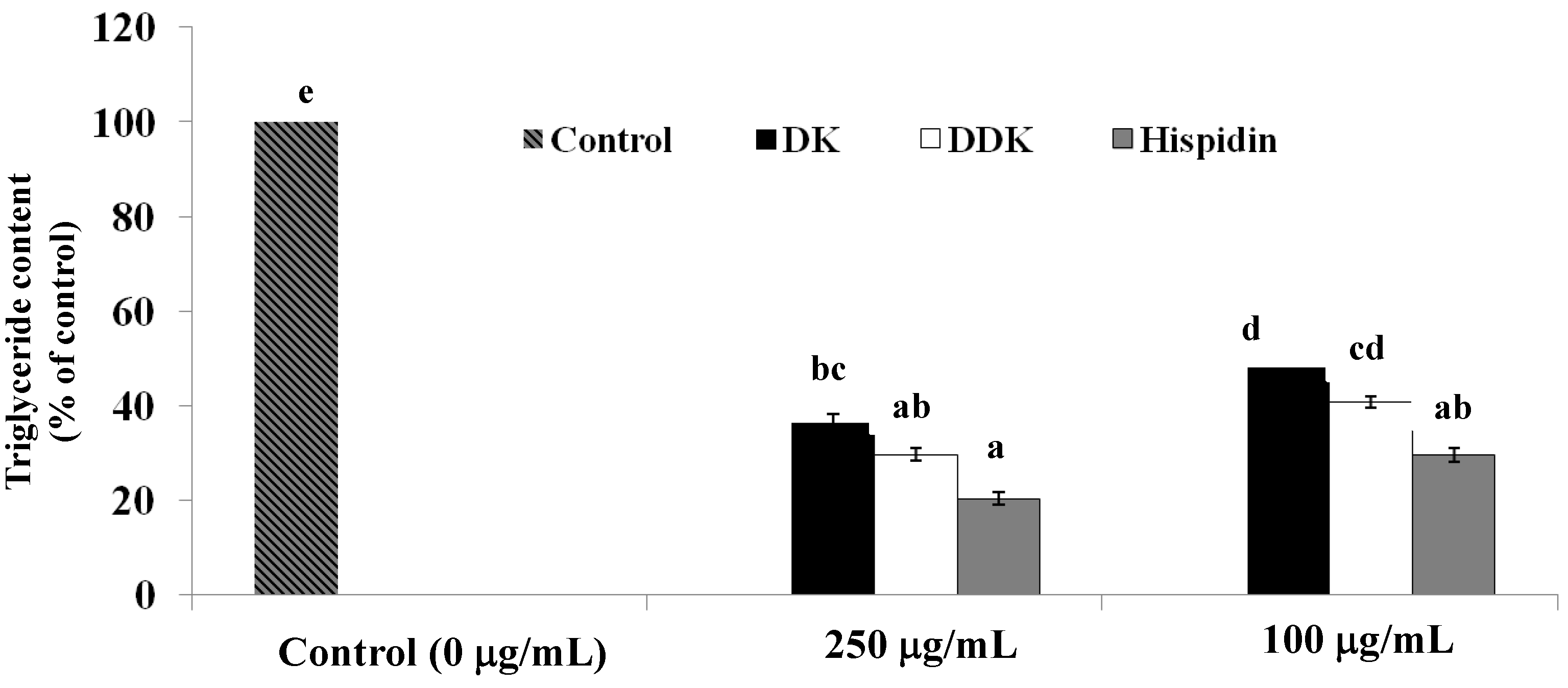
2.6. Measurement of Intracellular Triglyceride Content
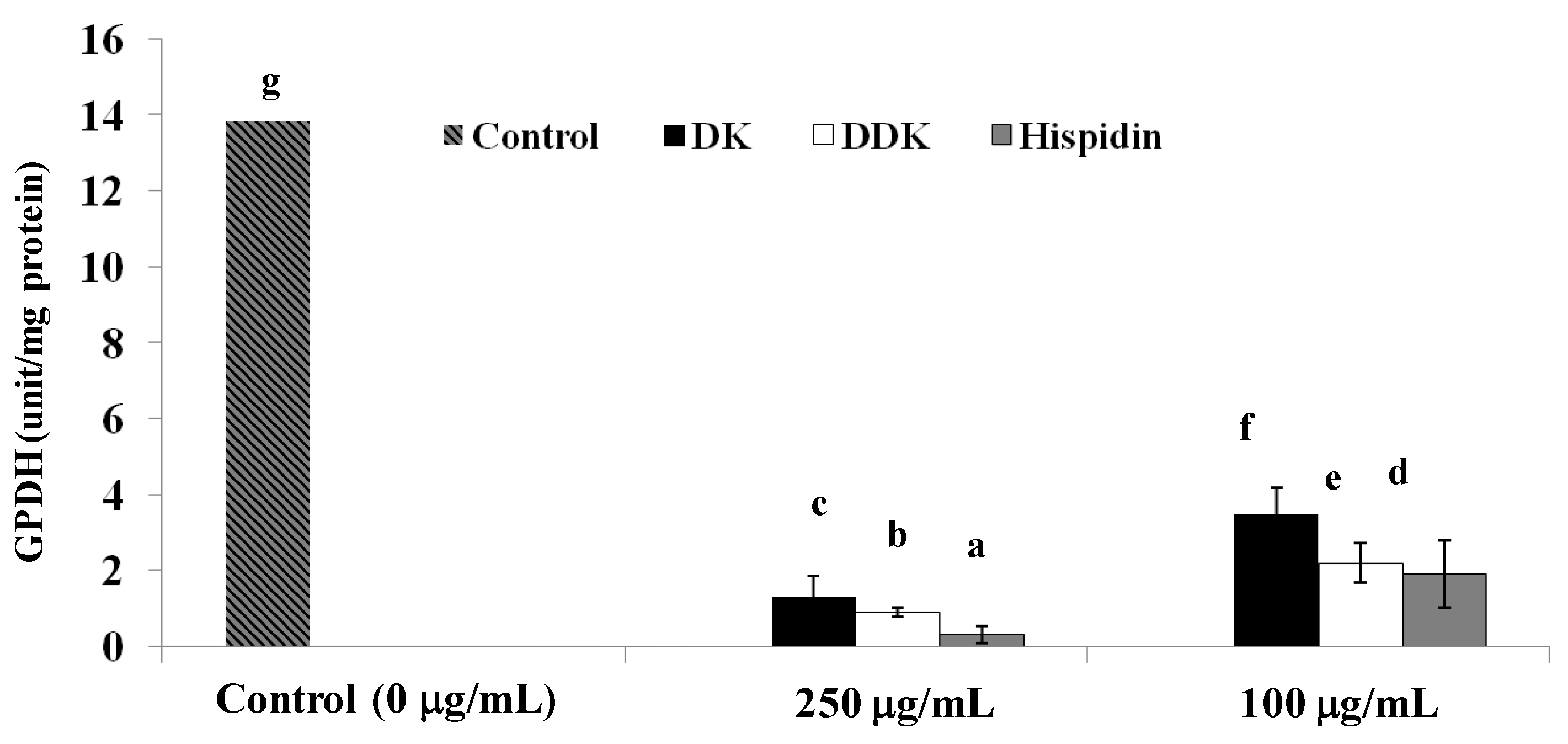
2.7. Glycerol-3-Phosphate Dehydrogenase Inhibition
2.8. Inhibition of Pancreatic Lipase Activity
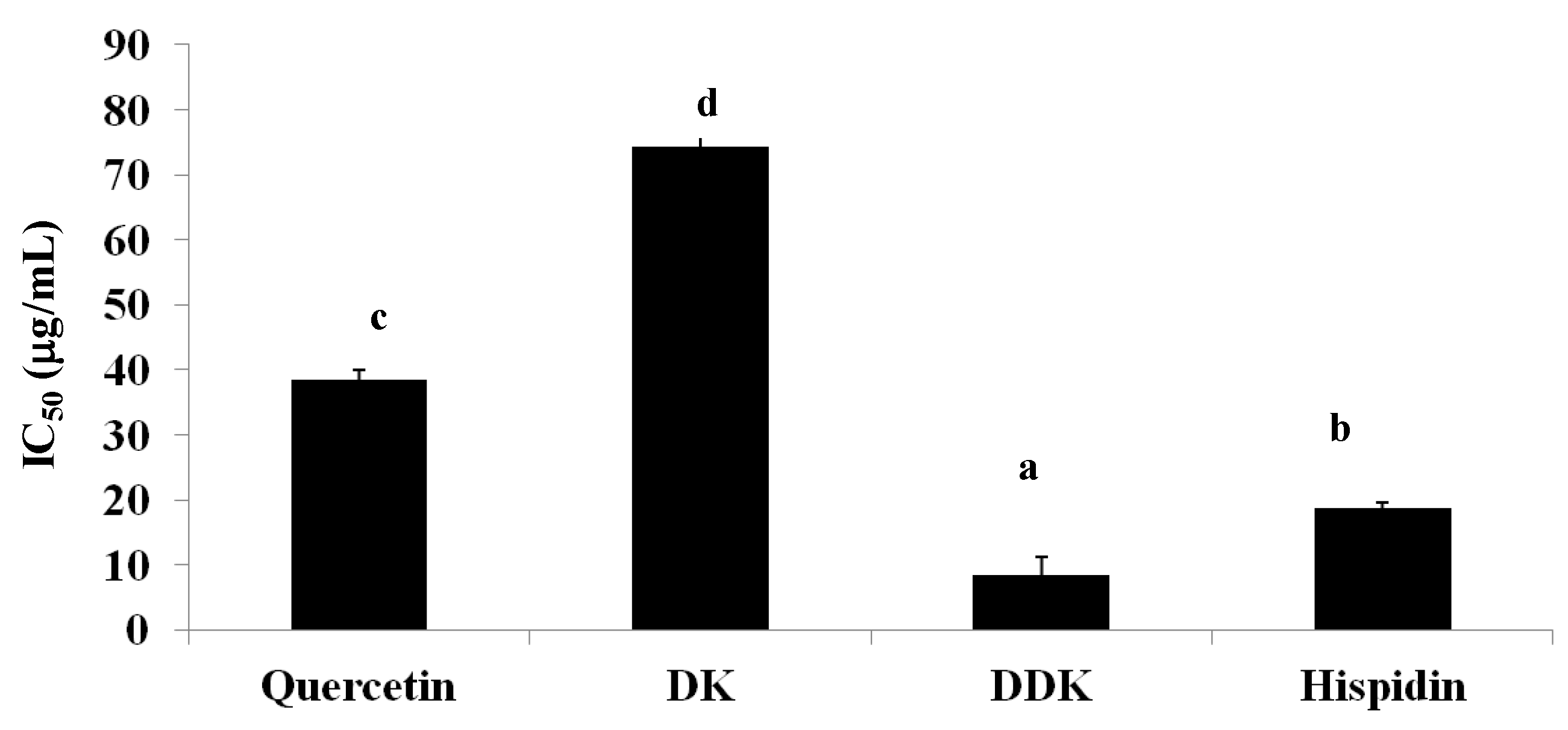
2.9. Discussion
3. Experimental Section
3.1. General
3.2. Preparation of Rhizomes Extracts and Isolation of DK and DDK
3.3. Cell Culture and Differentiation
3.4. Cell Viability Assays
3.5. Determination of Glycerol Release
3.6. Measurement of Intracellular cAMP
3.7. Evaluation of Lipid Accumulation by Oil Red O Staining
3.8. Measurement of Triglyceride Content
3.9. Glycerol-3-Phosphate Dehydrogenase Assay
3.10. Pancreatic Lipase Assay
3.11. Statistical Treatment
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hsu, C.L.; Yen, G.C. Effects of flavonoids and phenolic acids on the inhibition of adipogenesis in 3T3-L1 adipocytes. J. Agric. Food Chem. 2007, 55, 8404–8410. [Google Scholar]
- Kopelman, P.G. Obesity as a medical problem. Nature 2000, 404, 635–643. [Google Scholar]
- WHO Fact Files: Ten Facts on Obesity. Available online: http://www.who.int/features/factfiles/obesity/en/index.html (accessed on 20 March 2011).
- Gregor, M.F.; Hotamisligil, G.S. Inflammatory mechanism in obesity. Annu. Rev. Immunol. 2011, 29, 415–445. [Google Scholar]
- Williamson, D.F.; Pamuk, E.; Thun, M.; Flanders, D.; Byers, T.; Heath, C. Prospective study of international weight loss and mortality in overweight white men aged 40–64 years. Am. J. Epidemiol. 1999, 141, 1128–1141. [Google Scholar]
- Jang, W.S.; Choung, S.Y. Antiobesity effects of the ethanol extract of Laminaria japonica areshoung in high-fat-diet-induced obese rat. Evid.-Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef]
- Chen, W.; Feng, L.; Huang, Z.; Su, H. Hispidin produced from Phellinus linteus protects against peroxynitrite-mediated DNA damage and hydroxyl radical generation. Chem.-Biol. Interact. 2012, 199, 137–142. [Google Scholar]
- Jang, J.S.; Lee, J.S.; Lee, J.H.; Kwon, D.S.; Lee, K.E.; Lee, S.Y.; Hong, E.K. Hispidin produced from Phellinus linteus protects pancreatic β-cells from damage by hydrogen peroxide. Arch. Pharm. Res. 2010, 33, 853–861. [Google Scholar]
- Han, J.J.; Bao, L.; He, L.W.; Zhang, X.Q.; Yang, X.L.; Li, S.J.; Yao, Y.J.; Liu, H.W. Phaeolschidins A–E, five hispidin derivatives with antioxidant activity from the fruiting body of Phaeolus schweinitzii collected in the Tibetan Plateau. J. Nat. Prod. 2013, 76, 1448–1453. [Google Scholar]
- Park, I.H.; Jeon, S.Y.; Lee, H.J.; Kim, S.I.; Song, K.S. A β-secretase (BACE1) inhibitor hispidin from the mycelial cultures of Phellinus linteus. Planta Med. 2004, 70, 143–146. [Google Scholar]
- Yeom, J.H.; Lee, I.K.; Ki, D.W.; Lee, M.S.; Seok, S.J.; Yun, B.S. Neuraminidase inhibitors from the culture broth of Phellinus linteus. Mycobiology 2012, 40, 142–144. [Google Scholar]
- Risal, P.; Hwang, P.H.; Yun, B.S.; Yi, H.K.; Cho, B.H.; Jang, K.Y.; Jeong, Y.J. Hispidin analogue davallialactone attenuates carbon tetrachloride-induced hepatotocity in mice. J. Nat. Prod. 2012, 75, 1683–1689. [Google Scholar]
- Tawata, S.; Taira, S.; Kobamoto, N.; Ishihara, M.; Toyama, S. Syntheses and biological activities of dihydro-5,6-dehydrokawain derivatives. Biosci. Biotechnol. Biochem. 1996, 60, 1643–1645. [Google Scholar]
- Upadhyay, A.; Uezato, Y.; Tawata, S.; Ohkawa, H. CYP2C9 catalyzed bioconversion of secondary metabolites of three Okinawan plants. In Proceedings of 16th International Conference on Cytochrome P450, Okinawa, Japan, 21–25 June 2009; Shoun, H., Ohkawa, H., Eds.; Nago: Okinawa, Japan, 2009; pp. 31–34. [Google Scholar]
- Cavalcanti, B.C.; Ferreira, J.R.O.; Cabral, I.O.; Magalhaes, H.I.F.; de Olivereira, C.C.; Rodrigues, F.A.R.; Rocha, D.D.; Barros, F.W.A.; da Silva, C.R.; Junior, H.V.N.; et al. Genetic toxicology evaluation of essential oil of Alpinia zerumbet and its chemoprotective effects against H2O2-induced DNA damage in cultured human leukocytes. J. Food Chem. Toxicol. 2012, 50, 4051–4061. [Google Scholar]
- Lahlou, S.; Interaminense, L.F.L.; Leal-Cardoso, J.H.; Duarte, J.P. Antihypertensive effects of the essential oil of Alpinia zerumbet and its main constituent, terpinen-4-ol, in DOCA-salt hypertensive conscious rats. Fund. Clin. Pharm. 2003, 17, 323–330. [Google Scholar]
- De Araújo, F.Y.; de Oliveira, G.V.; Gomes, P.X.; Soares, M.A.; Silva, M.I.; Carvalho, A.F.; de Moraes, M.O.; de Moraes, M.E.; Vasconcelos, S.M.; Viana, G.S.; et al. Inhibition of ketamine-induced hyperlocomotion in mice by the essential oil of Alpinia zerumbet: Possible involvement of an antioxidant effect. J. Pharm. Pharmacol. 2011, 63, 1103–1110. [Google Scholar]
- Da Cunha, G.H.; de Moraes, M.O.; Fechine, F.V.; Bezerra, F.A.F.; Silveira, E.R.; Canuto, K.M.; de Moraes, M.E.A. Vasorelaxant and antihypertensive effects of methonilic fraction of the essential oil of Alpinia zerumbet. Vasc. Pharm. 2013, 58, 337–345. [Google Scholar]
- Chompoo, J.; Upadhyay, A.; Kishimoto, W.; Makise, T.; Tawata, S. Advanced glycation end products inhibitors from Alpinia zerumbet rhizomes. J. Food Chem. 2011, 129, 709–715. [Google Scholar]
- Junior, W.A.R.; Piato, A.L.; Conterato, G.M.M.; Wildner, S.M.; Marcon, M.; Moreira, S.; Santo, G.D.; Mocelin, R.; Emanuelli, T.; de Moreaes Antos, C.A. Psychopharmacological and antioxidant effects of hydroethanolic extract of Alpinia zerumbet leaves in mice. Pharmacogn. J. 2013, 5, 113–118. [Google Scholar]
- Chompoo, J.; Upadhyay, A.; Fukuta, M.; Tawata, S. Effect of Alpinia zerumbet components on antioxidant and skin diseases-related enzymes. BMC Complement. Altern. Med. 2012, 12, 106. [Google Scholar]
- Upadhyay, A.; Chompoo, J.; Kishimoto, W.; Makise, T.; Tawata, S. HIV-1 integrase and neuraminidase inhibitors from Alpinia zerumbet. J. Agric. Food Chem. 2011, 59, 2857–2862. [Google Scholar]
- Mpalantinos, M.A.; de Moura, R.S.; Parente, J.P.; Kuster, R.M. Biologically active flavonoids and kava pyrones from the aqueous extract of Alpinia zerumbet. Phytother. Res. 1998, 12, 442–444. [Google Scholar]
- Teng, C.M.; Hsu, S.Y.; Lin, C.H.; Yu, S.M.; Wang, K.J.; Lin, M.H.; Chen, C.F. Antiplatelet action of dehydrokawain derivatives isolated from Alpinia speciosa rhizome. Chin. J. Physiol. 1990, 33, 41–48. [Google Scholar]
- Fujioka, K. Management of obesity as a chronic disease: Nonpharmacologic, pharmacologic, and surgical options. Obes. Res. 2002, 10, 116–123. [Google Scholar]
- Seidell, J.C. Obesity, insulin resistance and diabetes-a worldwide epidemic. Br. J. Nutr. 2000, 83, S5–S8. [Google Scholar]
- Wang, Y.W.; Jones, P.J.H. Conjugated linoleic acid and obesity control: Efficacy and mechanisms. Int. J. Obes. 2004, 28, 941–955. [Google Scholar]
- Rhoh, C.; Jung, U. Screening of crude plant extracts with anti-obesity activity. Int. J. Mol. Sci. 2012, 13, 1710–1719. [Google Scholar]
- Park, I.H.; Chung, S.K.; Lee, K.B.; Yoo, Y.C.; Kim, S.K.; Kim, G.S.; Song, K.S. An antioxidant hispidin from the mycelia cultures of Phellinus linteus. Arch. Pharm. Res. 2004, 27, 615–618. [Google Scholar]
- Holm, C.; Østerlund, T.; Laurell, H.; Contreras, J.A. Molecular mechanisms regulating hormones sensitive lipase and lipolysis. Annu. Rev. Nutr. 2000, 20, 365–393. [Google Scholar]
- Hepp, K.D.; Renner, R.; Langley, J.; Haring, H.U. Effect of partially purified NSILA on adenylate cyclase, phosphodiesterase and 3′,5′-cylic AMP in fat cell. Mol. Cell. Endocrinol. 1975, 3, 309–321. [Google Scholar]
- Sugima, H.; Akazome, Y.; Shoji, T.; Yamaguchi, A.; Yasue, M.; Kanda, T.; Ohtake, Y. Oligomeric procyanidins in apple polyphenol are main active components for inhibition of pancreatic lipase and triglyceride absorption. J. Agric. Food Chem. 2007, 55, 4604–4609. [Google Scholar]
- Wise, L.S.; Green, H. Participation of one isozyme of cytosolic glycerophosphate dehydrogenase in the adipose conversion of 3T3 cells. J. Biol. Biochem. 1979, 254, 273–275. [Google Scholar]
- Tomiyama, K.; Nakata, H.; Sasa, H.; Arimuar, S.; Nishio, E.; Watanabe, Y. Wortmannin, a specific phosphatidylinositol 3-kinase inhibitor, inhibits adipocytic differentiation of 3T3-L1 cells. Biochem. Biophys. Res. Commun. 1995, 212, 263–269. [Google Scholar]
- Kim, Y.S.; Lee, Y.; Kim, J.; Sohn, E.; Kim, C.S.; Lee, Y.M.; Jo, K.; Shin, S.; Song, Y.; Kim, J.H.; et al. Inhibitory activities of Cudrania tricuspidata leaves on pancreatic lipase in vitro and lipolysis in vivo. Evid.-Based Complement. Altern. Med. 2012. [Google Scholar] [CrossRef]
- Knuckles, B.E. Effect of phytate and other myo-inositol phosphate esters on lipase activity. J. Food Sci. 1988, 53, 250–252. [Google Scholar]
- Shimura, S.; Tsuzuki, W.; Itoh, Y.; Kobayashi, S.; Suzuki, T. Inhibitory effect of tannin fraction from Cassia mimosoides L. var. nomame Makino on lipase activity. J. Jpn. Soc. Food Sci. Technol. 1994, 41, 561–564. [Google Scholar]
- Yoshizmi, K.; Hirano, K.; Ando, H.; Hirai, Y.; Ida, Y.; Tsuji, T.; Takana, T.; Satouchi, K.; Terao, J. Lupane-type saponins from leaves of Acanthopanx sessiliflorus and their inhibitory activity on pancreatic lipase. J. Agric. Food Chem. 2006, 54, 335–341. [Google Scholar]
- Masaaki, N.; Yuko, F.; Suimo, A.; Yoshiko, T.O.; Takashi, I.; Hiroshi, S.; Tohru, M.; Fumio, H.; Yoshinobu, K. Inhibitory effects of Oolong tea polyphenols on pancreatic lipase in vitro. J. Agric. Food Chem. 2005, 53, 4593–4598. [Google Scholar]
- Chompoo, J.; Upadhyay, A.; Gima, S.; Fukuta, M.; Tawata, S. Antiatherogenic properties of acetone extract of Alpinia zerumbet seeds. Molecules 2012, 17, 6237–6248. [Google Scholar]
- Lee, J.K.; Jang, J.H.; Lee, J.T.; Lee, J.S. Extraction and characteristics of anti-obesity lipase inhibitor from Phellinus linteus. Mycobiology 2010, 38, 52–57. [Google Scholar]
- Sung, J.H.; Chon, J.W.; Lee, M.A.; Park, J.K.; Woo, J.T.; Park, Y.K. The anti-obesity effect of Lethariella cladonioides in 3T3-L1 cells and obese mice. Nutr. Res. Pract. 2011, 6, 503–510. [Google Scholar]
- Kim, Y.S.; Lee, Y.M.; Kim, H.; Kim, J.; Jang, D.S.; Kim, J.H.; Kim, J.S. Anti-obesity effect of Morus bombycis root extract: Anti-lipase activity and lipolytic effect. J. Ethopharm. 2010, 130, 621–624. [Google Scholar]
- Lee, Y.S.; Kim, H.K.; Lee, K.J.; Jeon, H.W.; Cui, S.; Lee, Y.M.; Moon, B.J.; Kim, Y.H.; Lee, Y.S. Inhibitory effect of glyceollin isolated from soybean against melanogenensis in B16 melanoma cells. Biochem. Mol. Biol. Rep. 2010, 43, 461–467. [Google Scholar]
- Ramirez-zacarias, J.L.; Castro-Munozled, F.; Kuri-Harcuch, W. Quantification of adipose conversion and triglycerides by staining intracytoplasmic lipids with Oil Red O. Histochemistry 1992, 97, 493–497. [Google Scholar]
- Kim, J.; Jang, D.S.; Kim, H.; Kim, J.S. Anti-lipase and lipolytic activities of ursolic acid isolated from the roots of Actinidia arguta. Arch. Pharm. Res. 2009, 32, 983–987. [Google Scholar]
- Sample Availability: Samples of the compounds DK and DDK are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tu, P.T.B.; Tawata, S. Anti-Obesity Effects of Hispidin and Alpinia zerumbet Bioactives in 3T3-L1 Adipocytes. Molecules 2014, 19, 16656-16671. https://doi.org/10.3390/molecules191016656
Tu PTB, Tawata S. Anti-Obesity Effects of Hispidin and Alpinia zerumbet Bioactives in 3T3-L1 Adipocytes. Molecules. 2014; 19(10):16656-16671. https://doi.org/10.3390/molecules191016656
Chicago/Turabian StyleTu, Pham Thi Be, and Shinkichi Tawata. 2014. "Anti-Obesity Effects of Hispidin and Alpinia zerumbet Bioactives in 3T3-L1 Adipocytes" Molecules 19, no. 10: 16656-16671. https://doi.org/10.3390/molecules191016656
APA StyleTu, P. T. B., & Tawata, S. (2014). Anti-Obesity Effects of Hispidin and Alpinia zerumbet Bioactives in 3T3-L1 Adipocytes. Molecules, 19(10), 16656-16671. https://doi.org/10.3390/molecules191016656




