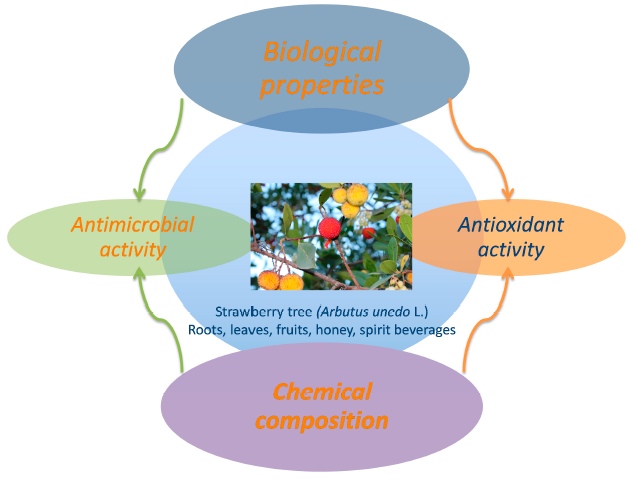
Arbutus unedo L.: Chemical and Biological Properties
Abstract
:1. Introduction
2. Chemical Composition of A. unedo
2.1. Fruits
2.1.1. Phenols
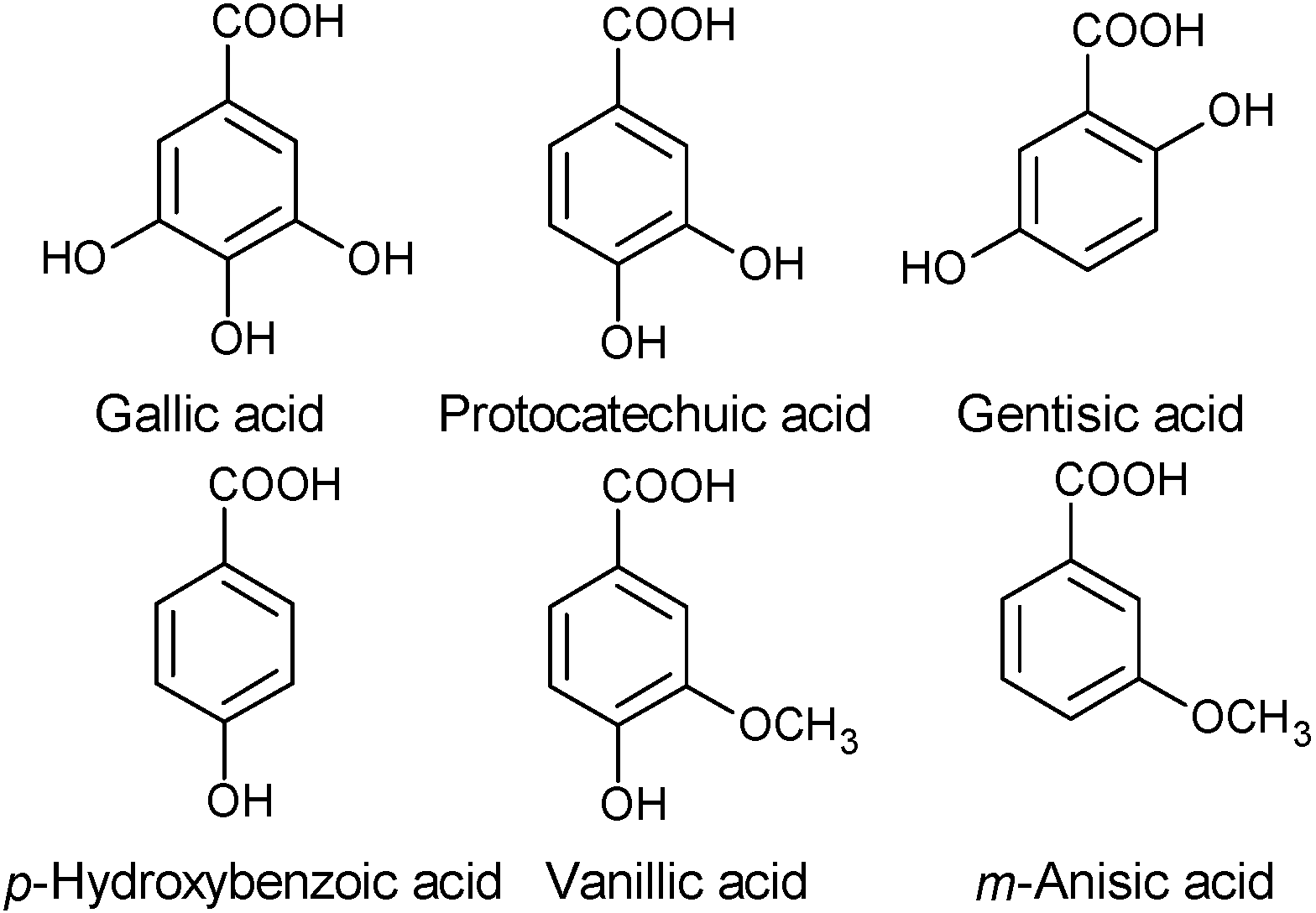
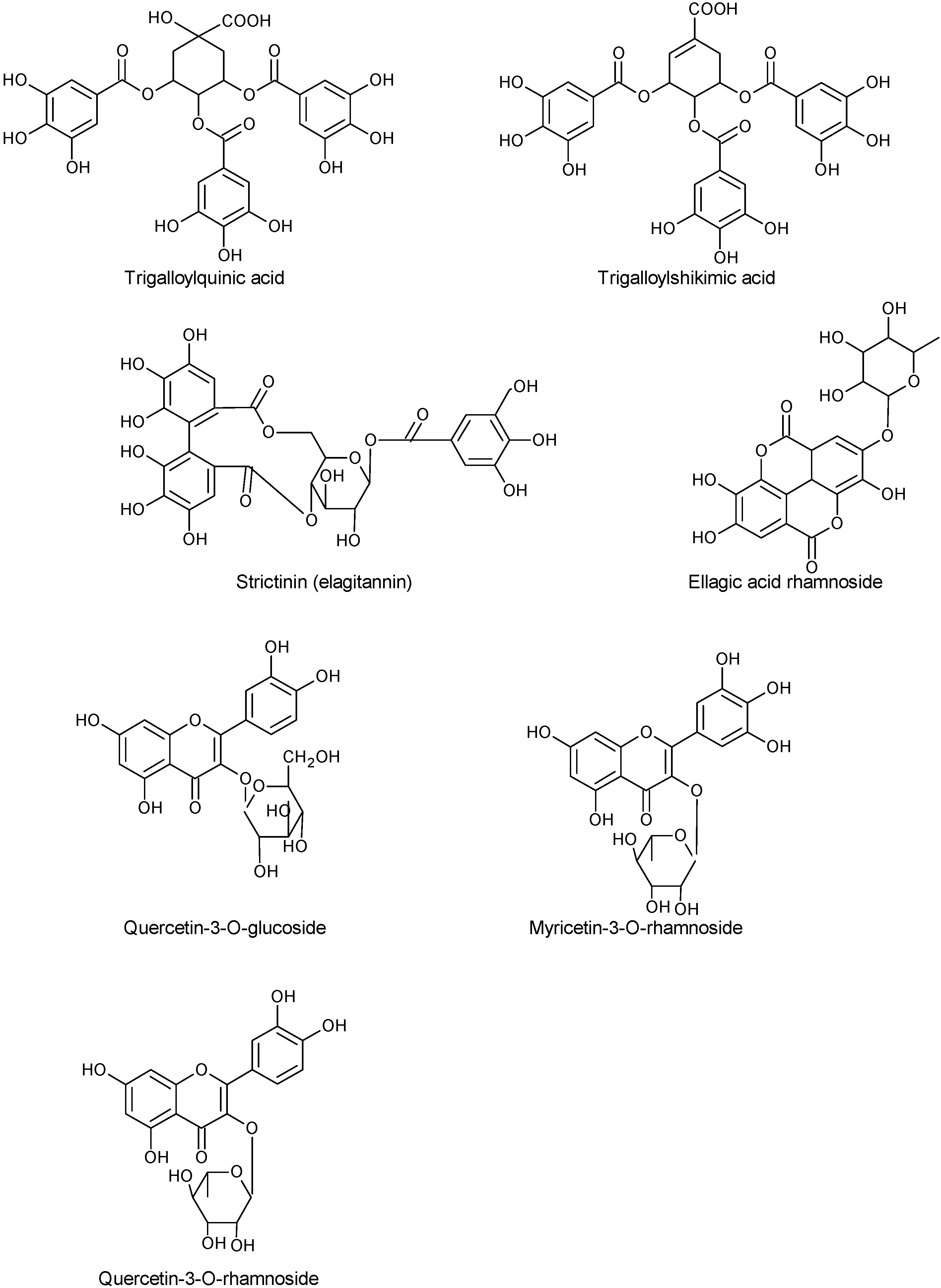
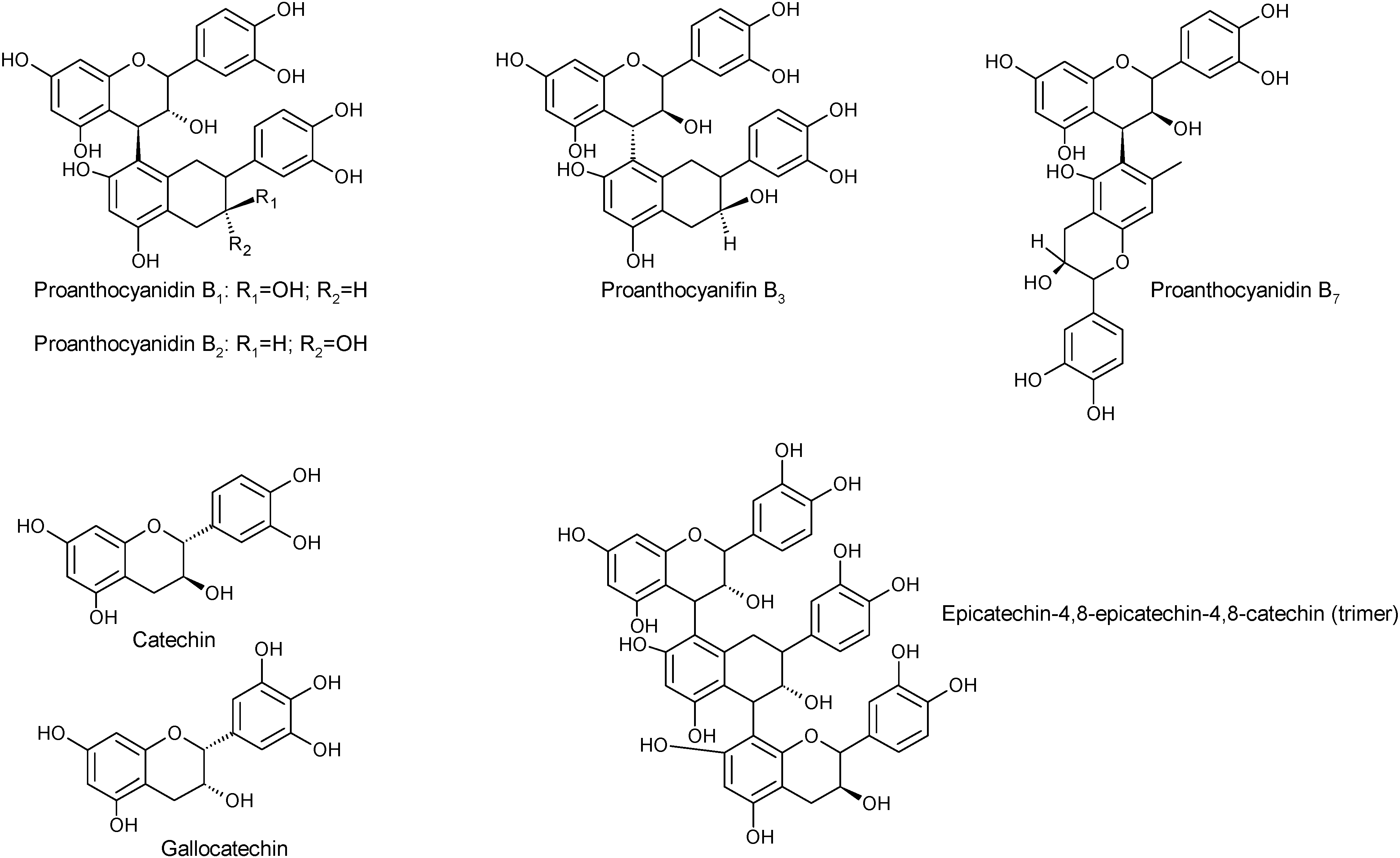
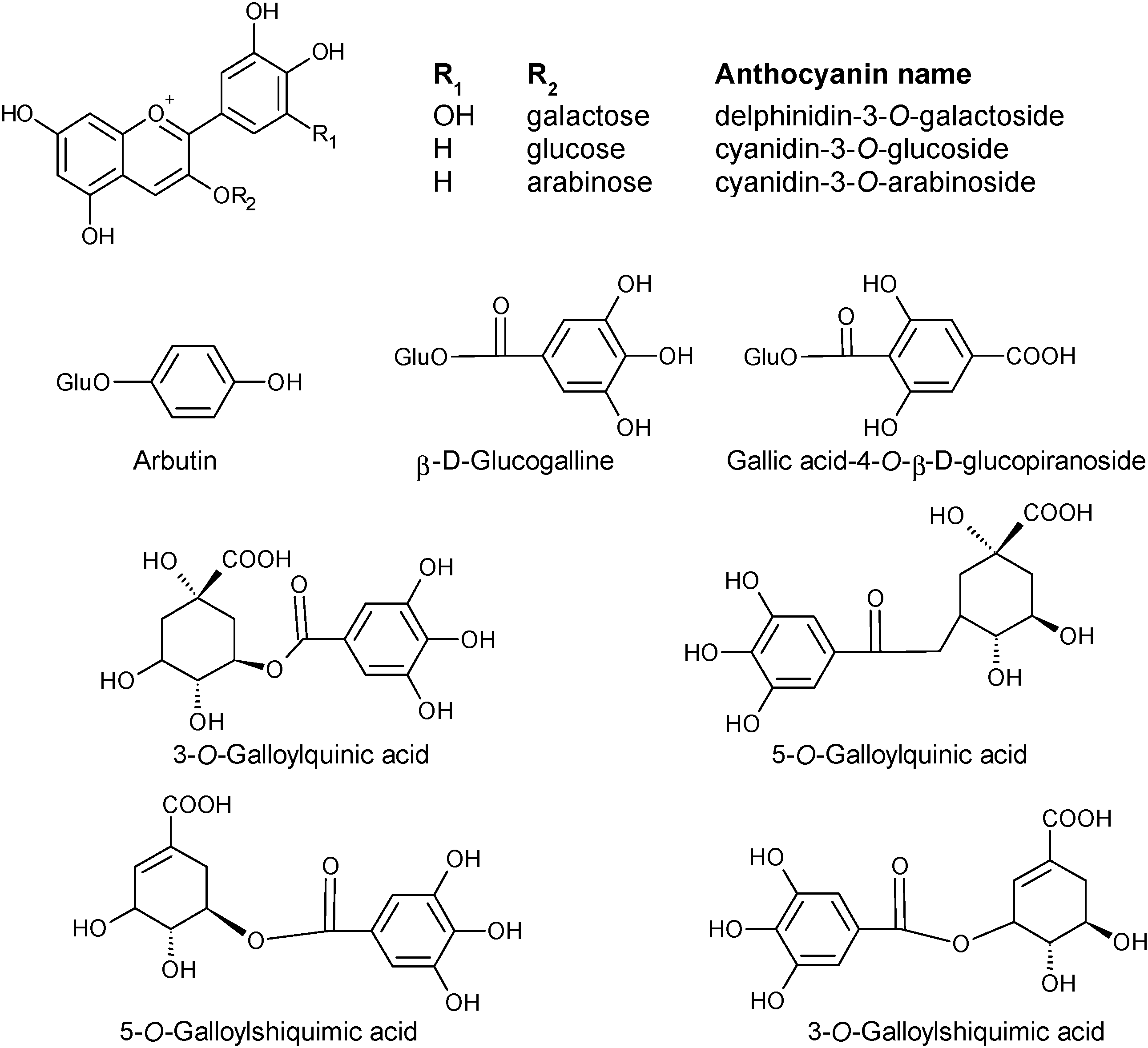
2.1.2. Fatty Acids
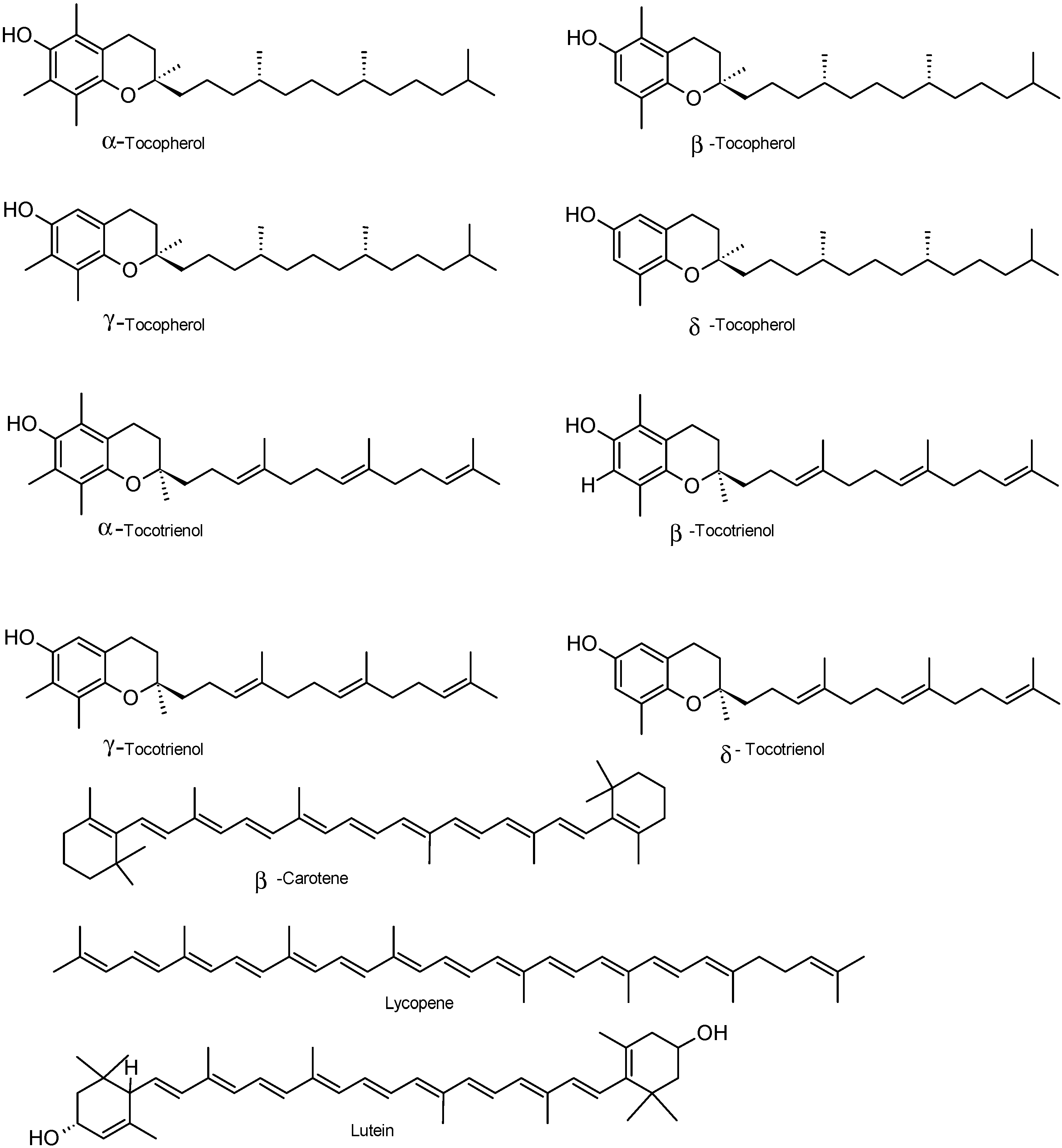
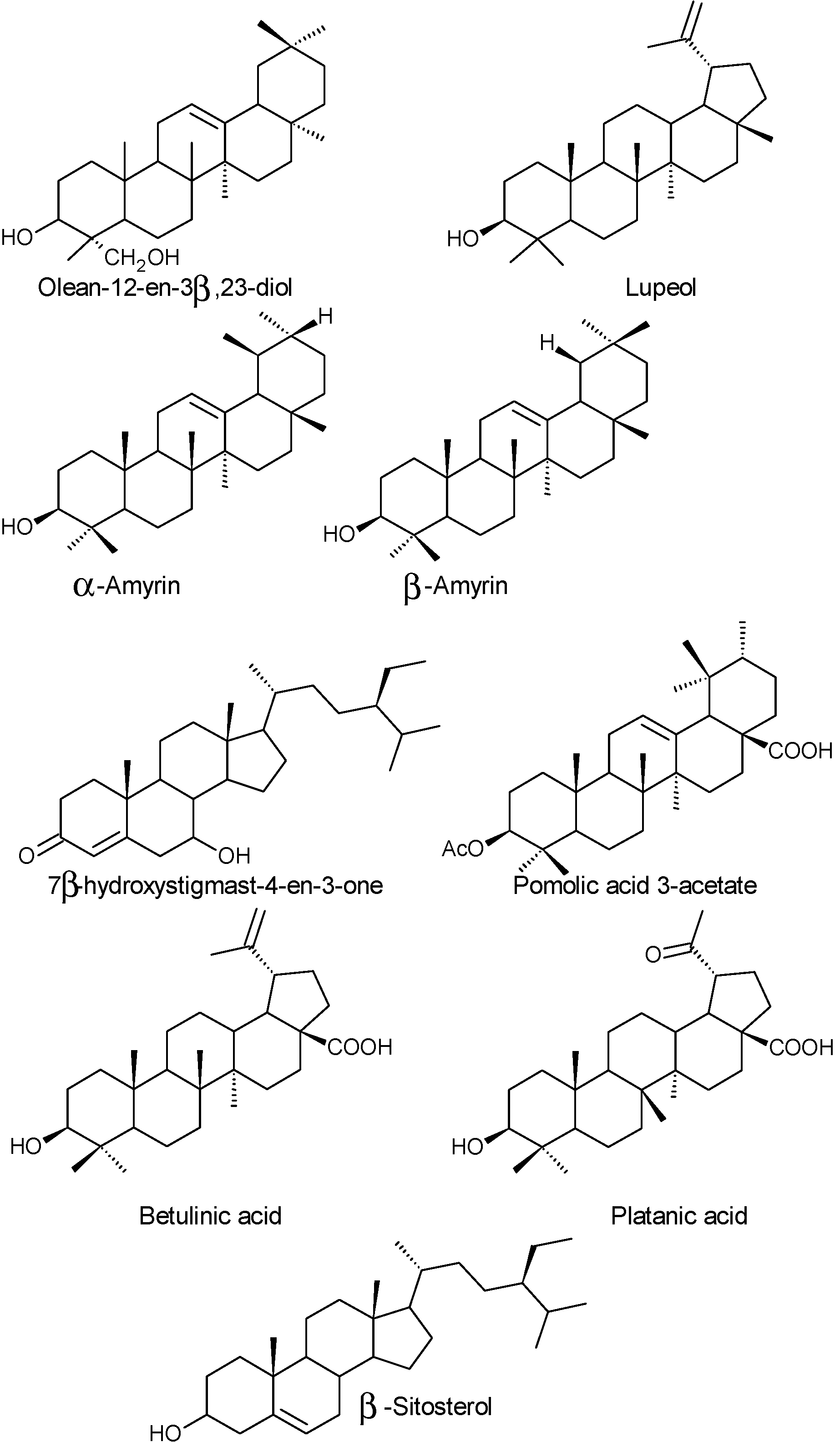
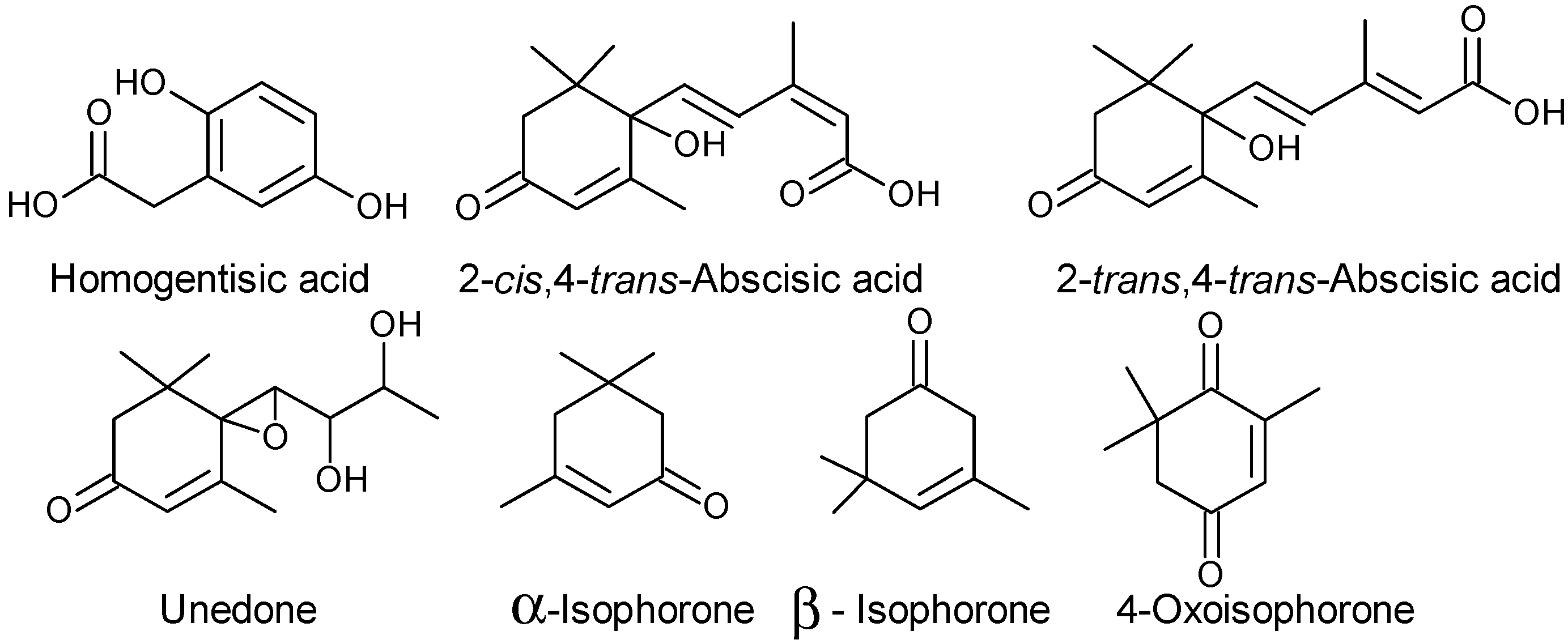
2.1.3. Vitamins and Others
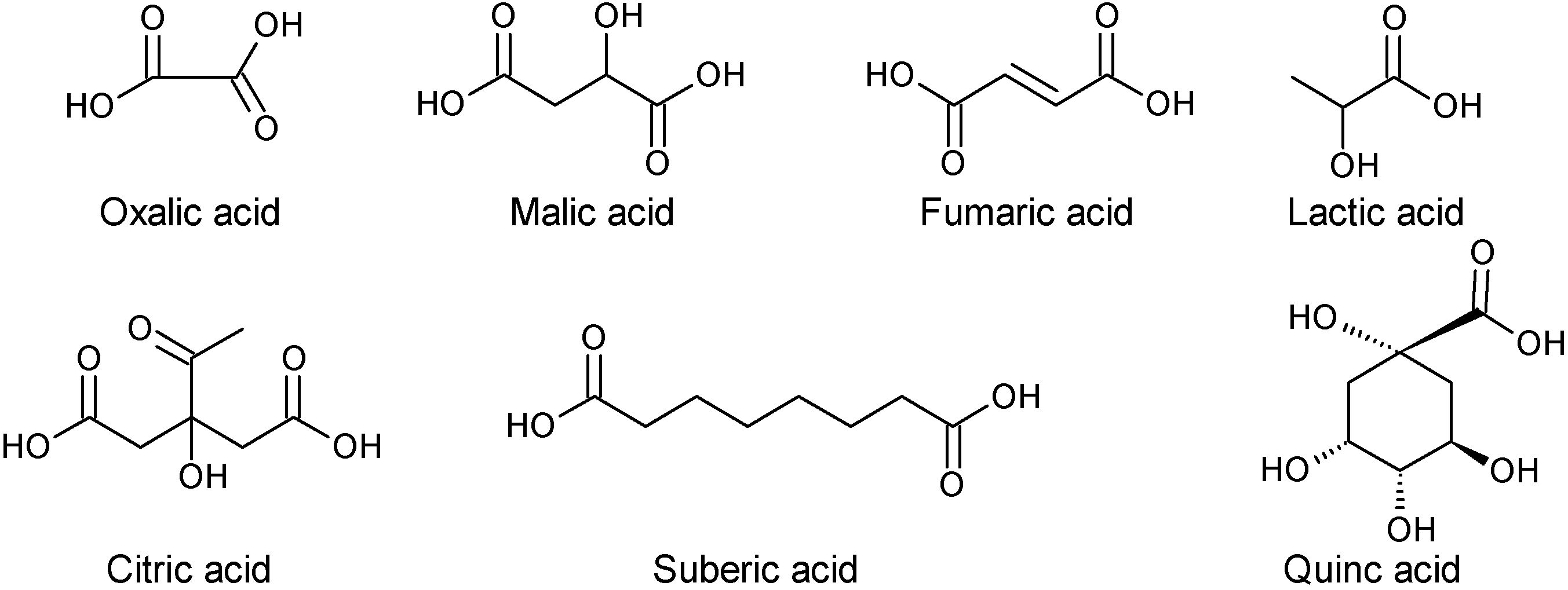
2.1.4. Organic Acids
2.1.5. Sugars
2.1.6. Volatiles
2.1.7. Minerals
2.2. Leaves
2.3. Stems
2.4. Roots
2.5. Seeds
2.6. Entire Plant
3. Biological Properties and Applications
3.1. Fruits
3.2. Leaves
| Microorganism | Extract | Activity | Ref. | |
|---|---|---|---|---|
| Gram Positive | ||||
| Staphylococcus aureus | Aqueous | Inhibition zone diameter | 10.5 mm (10 μL/disc) | [68] |
| 13.8 mm (20 μL/disc) | ||||
| 21.1 mm (40 μL/disc) | ||||
| Bacillus cereus | Aqueous | Minimum inhibitory concentration (MIC) | 1 mg/mL | [69] |
| B. subtilis | 1 mg/mL | |||
| Staphylococcus epidermis | 1 mg/mL | |||
| S. aureus | 2.5 mg/mL | |||
| B. cereus ATCC 11771 | Acetone/water (60:40) | Inhibition zone diameter | 10.0 mm (2 mg/disc) | [71] |
| Ethanol 95% | 11.3 mm | |||
| Methanol (Soxhlet) | 10.0 mm | |||
| Methanol (maceration) | 10.7 mm | |||
| Ethanol (maceration) | 9.3 mm | |||
| S. aureus ATCC 25923 | Acetone/water (60:40) | Inhibition zone diameter | 9.3 mm | [71] |
| Ethanol 95% | 12.0 mm | |||
| Methanol (Soxhlet) | 10.3 mm | |||
| Methanol (maceration) | 9.3 mm | |||
| Ethanol (maceration) | 9.3 mm | |||
| Enterococcus faecalis ATCC 29212 | Acetone/water (60:40) | Inhibition zone diameter | 8.0 mm | [71] |
| Ethanol 95% | 6.5 mm | |||
| Methanol (Soxhlet) | 7.0 mm | |||
| Methanol (maceration) | 7.3 mm | |||
| Ethanol (maceration) | 7.3 mm | |||
| Clinical Methicillin-Resistant Staphylococcus aureus 10/8 | Acetone/water (60:40) | Inhibition zone diameter | 7.8 mm | [71] |
| Ethanol 95% | 8.8 mm | |||
| Methanol (Soxhlet) | -- | |||
| Methanol (maceration) | 8.0 mm | |||
| Ethanol (maceration) | 8.0 mm | |||
| Clinical Methicillin-Resistant Staphylococcus aureus 12/8 | Acetone/water (60:40) | Inhibition zone diameter | 7.0 mm | [71] |
| Ethanol 95% | 9.5 mm | |||
| Methanol (Soxhlet) | -- | |||
| Methanol (maceration) | -- | |||
| Ethanol (maceration) | -- | |||
| Mycobacterium smegmatis | Aqueous | MIC | 6.02 mg/mL | [70] |
| M. aurum A+ | 5.59 mg/mL | |||
| M. bovis PPI | 6.02 mg/mL | |||
| Gram negative | ||||
| Pseudomonas aeruginosa | Aqueous | MIC | 5 mg/mL | [69] |
| Escherichia coli | 5 mg/mL | |||
| Klebsiella pneumoniae ATCC 13833 | Acetone/water (60:40) | Inhibition zone diameter | 9.0 mm (2 mg/disc) | [71] |
| Ethanol 95% | 10.8 mm | |||
| Methanol (Soxhlet) | 9.8 mm | |||
| Methanol (maceration) | 9.3 mm | |||
| Ethanol (maceration) | 8.7 mm | |||
| Helicobacter pylori 104/98 | Acetone/water (60:40) | Inhibition zone diameter | 18.3 mm | [71] |
| Ethanol 95% | 14.6 mm | |||
| Methanol (Soxhlet) | 17.7 mm | |||
| Methanol (maceration) | 16.7 mm | |||
| Ethanol (maceration) | 13.5 mm | |||
| Helicobacter pylori P10/92 | Acetone/water (60:40) | Inhibition zone diameter | 17.8 mm | [71] |
| Ethanol 95% | 14.6 mm | |||
| Methanol (Soxhlet) | 15.5 mm | |||
| Methanol (maceration) | 14.6 mm | |||
| Ethanol (maceration) | 14.9 mm | |||
| Helicobacter pylori 93/00 | Acetone/water (60:40) | Inhibition zone diameter | 12.9 mm | [71] |
| Ethanol 95% | 11.1 mm | |||
| Methanol (Soxhlet) | 12.3 mm | |||
| Methanol (maceration) | 11.8 mm | |||
| Ethanol (maceration) | 11.5 mm | |||
| Helicobacter pylori B22/96 | Acetone/water (60:40) | Inhibition zone diameter | 18.0 mm | [71] |
| Ethanol 95% | 17.5 mm | |||
| Methanol (Soxhlet) | 18.2 mm | |||
| Methanol (maceration) | 18.3 mm | |||
| Ethanol (maceration) | 15.8 mm | |||
| Helicobacter pylori ATCC 710392 | Acetone/water (60:40) | Inhibition zone diameter | 10.4 mm | [71] |
| Ethanol 95% | 7.6 mm | |||
| Methanol (Soxhlet) | 9.1 mm | |||
| Methanol (maceration) | 9.0 mm | |||
| Ethanol (maceration) | 8.1 mm | |||
| Fungi | ||||
| Aspergillus parasiticus NRRL 2999 (0.075%–0.3%) | Aqueous | Inhibitory effect | 30.15%–42.70% | [68] |
| Aspergillus parasiticus NRRL 465 (0.075%–0.3%) | 12.79%–29.76% | |||
| Yeasts | ||||
| Candida tropicalis ATCC 750 | Acetone/water (60:40) | Inhibition zone diameter | 9.3 mm (2 mg/disc) | [71] |
| Ethanol 95% | 13.3 mm | |||
| Methanol (Soxhlet) | 10.7 mm | |||
| Methanol (maceration) | 12.0 mm | |||
| Ethanol (maceration) | 12.7 mm | |||
3.3. Roots
3.4. Entire Plant
4. Other Products Obtained from A. unedo
4.1. Honey
4.2. Spirit Beverages
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torres, J.A.; Valle, F.; Pinto, C.; Garcia-Fuentes, A.; Salazar, C.; Cano, E. Arbutus unedo L. communities in southern Iberian Peninsula mountains. Plant Ecol. 2002, 160, 207–223. [Google Scholar] [CrossRef]
- Oliveira, I.; Baptista, P.; Bento, A.; Pereira, J.A. Arbutus unedo L. and its benefits on human health. J. Food Nutr. Res. 2011, 50, 73–85. [Google Scholar]
- Kim, T.L. Arbutus unedo. In Edible Medicinal and Noin-Medicinal Plants; Springer: Dordrecht, The Netherlands; Heidelberg, Germany; London, UK; New York, NY, USA, 2012; Volume 2, pp. 444–451. [Google Scholar]
- Celikel, G.; Demirsoy, L.; Demirsoy, H. The strawberry tree (Arbutus unedo L.) selection in Turkey. Sci. Hort. 2008, 118, 115–119. [Google Scholar] [CrossRef]
- Maleš, Ž.; Plazibat, M.; Vundać, V.B.; Žuntar, I. Qualitative and quantitative analysis of flavonoids of the strawberry tree—Arbutus unedo L. (Ericaceae). Acta Pharm. 2006, 56, 245–250. [Google Scholar] [PubMed]
- Gomes, M.F.F.N. Strategies for the Improvement of Arbutus unedo L. (Strawberry Tree): In vitro Propagation, Mycorrhization and Diversity Analysis. Ph.D. Thesis, Universidade de Coimbra, Coimbra, Portugal, 2011. [Google Scholar]
- Takrouni, M.M.; Ali, I.B.H.; Messaoued, C.; Boussaid, M. Genetic variability of Tunisian wild strawberry tree (Arbutus unedo L.) populations interfered from isozyme markers. Sci. Hort. 2012, 146, 92–98. [Google Scholar] [CrossRef]
- Godinho-Ferreira, P.G.; Azevedo, A.M.; Rego, F. Carta da tipologia florestal de Portugal Continental. Silva Lusit. 2005, 13, 1–34. [Google Scholar]
- Gomes, F.; Canhoto, J. Micropropagation of strawberry tree (Arbutus unedo L.) from adult plants. In Vitro Cell. Dev. Biol. Plant 2009, 45, 72–82. [Google Scholar] [CrossRef]
- Takrouni, M.M.; Boussaid, M. Genetic diversity and population's structure in Tunisian strawberry tree (Arbutus undo L.). Sci. Hort. 2010, 126, 330–337. [Google Scholar] [CrossRef]
- Lopes, L.; Sá, O.; Pereira, J.A.; Baptista, P. Genetic diversity of Portuguese Arbutus unedo L. populations using leaf traits and molecular markers: An approach for conservation purposes. Sci. Hort. 2012, 142, 57–67. [Google Scholar] [CrossRef]
- Gomes, F.; Costa, R.; Ribeiro, M.M.; Figueiredo, E.; Canhoto, J.M. Analysis of genetic relationship among Arbutus unedo L. genotypes using RAPD and SSR markers. J. For. 2013, 24, 227–236. [Google Scholar]
- Ogaya, R.; Peñuelas, J. Phenological patterns of Quercus ilex, Phillyrea latifolia, and Arbutus unedo growing under a field experimental drought. Ecoscience 2004, 11, 263–270. [Google Scholar]
- Molina, M.; Pardo-de-Santayana, M.; Aceituno, L.; Morales, R.; Tardío, J. Fruit production of strawberry tree (Arbutus unedo L.) in two Spanish forests. Forestry 2011, 84, 419–429. [Google Scholar] [CrossRef]
- Menedez-Baceta, G.; Aceituno-Mata, L.; Tardío, J.; Reyes-García, V.; Pardo-de-Santayana, M. Wild edible plants traditionally gathered in Gorbeialdea (Biscay, Basque Country). Genet. Resour. Crop Evol. 2012, 59, 1329–1347. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Kucukislamoglu, M.; Reunanen, M. Sugar, non-volatile and phenolic acids composition of strawberry tree (Arbutus unedo L. var. ellipsoidea) Fruits. J. Food Compos. Anal. 2000, 13, 171–177. [Google Scholar] [CrossRef]
- Alarcão-e-Silva, M.L.C.M.M.; Leitão, A.E.B.; Azinheira, H.G.; Leitão, M.C.A. The Arbutus berry: Studies on its color and chemical characteristics at two mature stages. J. Food Compos. Anal. 2001, 14, 27–35. [Google Scholar] [CrossRef]
- Soufleros, E.H.; Mygdalia, S.A.; Natskoulis, P. Production process and characterization of the traditional Greek fruit distillate “koumaro” by aromatic and mineral composition. J. Food Compos. Anal. 2005, 18, 699–716. [Google Scholar] [CrossRef]
- Ziyyat, A.; Legssyer, A.; Mekhfi, H.; Dassouli, A.; Serhouchni, M.; Benjelloun, W. Phytotherapy of hypertension and diabetes in oriental Morocco. J. Ethnopharmacol. 1997, 58, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Pabuçcuoğlu, A.; Kıvcak, B.; Bas, M.; Mert, T. Antioxidant activity of Arbutus unedo leaves. Fitoterapia 2003, 74, 597–599. [Google Scholar] [CrossRef] [PubMed]
- Pawlowska, A.M.; de Leo, M.; Braca, A. Phenolics of Arbutus unedo L. (Ericaceae) fruits: Identification of anthocyanins and gallic acid derivatives. J. Agric. Food Chem. 2006, 54, 10234–10238. [Google Scholar] [CrossRef] [PubMed]
- Pallauf, K.; Rivas-Gonzalo, J.C.; del Castillo, M.D.; Cano, M.P.; de Pascual-Teresa, S. Characterization of the antioxidant composition of strawberry tree (Arbutus unedo L.) fruits. J. Food Compos. Anal. 2008, 21, 273–281. [Google Scholar] [CrossRef]
- El Haouari, M.; Lopez, J.J.; Mekhfi, H.; Rosado, J.A.; Salido, M.G. Antiaggregant effects of Arbutus unedo extracts. J. Ethnopharmacol. 2007, 113, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Sanjust, E.; Mocci, G.; Zucca, P.; Rescigno, A. Mediterranean shrubs as potential antioxidant sources. Nat. Prod. Res. 2008, 22, 689–708. [Google Scholar] [CrossRef]
- Andrade, D.; Gil, C.; Breitenfeld, L.; Domingues, F.; Duarte, A.P. Bioactive extracts from Cistus ladanifer and Arbutus unedo L. Ind. Crops Prod. 2009, 30, 165–167. [Google Scholar] [CrossRef]
- Fortalezas, S.; Tavares, L.; Pimpao, R.; Tyagi, M.; Pontes, V.; Alves, P.M.; McDougall, G.; Stewart, D.; Ferreira, R.B.; Santos, C.N. Antioxidant properties and neuroprotective capacity of strawberry tree fruit (Arbutus unedo). Nutrients 2010, 2, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.; de Freitas, V.; Baptista, P.; Carvalho, P. Comparative antihemolytic and radical scavenging activities of strawberry tree (Arbutus unedo L.) leaf and fruit. Food Chem. Toxicol. 2011, 49, 2285–2291. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, I.; Coelho, V.; Baltasar, R.; Pereira, J.A.; Baptista, P. Scavenging capacity of strawberry tree (Arbutus unedo L.) leaves on free radicals. Food Chem. Toxicol. 2009, 47, 1507–1511. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, I.; Baptista, P.; Malheiro Casal, R.S.A.B.; Pereira, J.A. Influence of strawberry tree (Arbutus unedo L.) fruit ripening stage on chemical composition and antioxidant activity. Food Res. Int. 2011, 44, 1401–1407. [Google Scholar] [CrossRef]
- Pavlović, D.R.; Branković, S.; Kovačević, N.; Kitić, D.; Veljković, S. Comparative study of spasmolytic properties, antioxidant activity and phenolic content of Arbutus unedo from Montenegro and Greece. Phytother. Res. 2011, 25, 749–754. [Google Scholar] [PubMed]
- Ruiz-Rodriguez, B.M.; Morales, P.; Fernandez-Ruiz, V.; Sanchez-Mata, M.C.; Camara, M.; Diez-Marques, C.; Pardo-de-Santayana, M.; Molina, M.; Tardio, J. Valorization of wild strawberry-tree fruits (Arbutus unedo L.) through nutritional assessment and natural production data. Food Res. Int. 2011, 44, 1244–1253. [Google Scholar] [CrossRef]
- Boulanouar, B.; Abdelaziz, G.; Aazza, S.; Gago, C.; Miguel, M.G. Antioxidant activities of eight Algerian plant extracts and two essential oils. Ind. Crop Prod. 2013, 46, 85–96. [Google Scholar] [CrossRef]
- Ganhão, R.; Estévez, M.; Kylli, P.; Heinonen, M.; Morcuende, D. Characterization of selected wild Mediterranean fruits and comparative efficacy as inhibitors of oxidative reactions in emulsified raw pork burger patties. J. Agric. Food Chem. 2010, 58, 8854–8861. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.; Barros, L.; Dueñas, M.; Carvalho, A.M.; Queiroz, M.J.R.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterization of phenolic compounds in wild fruits from Northeastern Portugal. Food Chem. 2013, 141, 3721–3730. [Google Scholar] [CrossRef]
- Pimpão, R.C.; Dew, T.; Oliveira, P.B.; Williamson, G.; Ferreira, R.B.; Santos, C.N. Analysis of phenolic compounds in Portuguese wild and commercial berries after multienzyme hydrolysis. J. Agric. Food Chem. 2013, 61, 4053–4062. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.; Carvalho, A.M.; Morais, J.S.; Ferreira, I.C.F.R. Strawberry-tree, blackthorn and rose fruits: Detailed characterisation in nutrients and phytochemicals with antioxdant properties. Food Chem. 2010, 120, 247–254. [Google Scholar] [CrossRef]
- Morales, P.; Ferreira, I.C.F.R.; Carvalho, A.M.; Fernández-Ruiz, V.; Sánchez-Mata, M.C.; Câmara, M.; Morales, R.; Tardio, J. Wild edible fruits as a potential source of phytochemicals with capacity to inhibit lipid peroxidation. Eur. J. Lipid Sci. Technol. 2013, 115, 176–185. [Google Scholar] [CrossRef]
- Vidrih, R.; Hribar, J.; Prgomet, Ž.; Ulrich, N.P. The physico-chemical properties of strawberry tree (Arbutus unedo L.) fruits. Croat. J. Food Sci. Technol. 2013, 5, 29–33. [Google Scholar]
- Aslantas, R.; Pirlak, L.; Güleryüz, M. The nutritional value of wild fruits from the North Eastern Anatolia region of Turkey. Asian J. Chem. 2007, 19, 3072–3078. [Google Scholar]
- Şeker, M.; Toplu, C. Determination and comparison of chemical characteristics of Arbutus unedo L. and Arbutus andrachnae L. (Family Ericaceae) fruits. J. Med. Food 2010, 13, 1013–1018. [Google Scholar] [PubMed]
- Gaspar, E.M.S.M.; Neves, H.J.C.; Noronha, J.P. Application of HPLC-PBMS to the identification of unknown components in a triterpenoid fraction of Arbutus unedo fruits. J. High Resolut. Chromatogr. 1997, 20, 417–420. [Google Scholar]
- Oliveira, I.; Pinho, P.G.; Malheiro, R.; Baptista, P.; Pereira, J.A. Volatile profile of Arbutus unedo L. fruits through ripening stage. Food Chem. 2011, 128, 667–673. [Google Scholar] [CrossRef]
- Kahriman, N.; Albay, C.G.; Dogan, N.; Usta, A.; Karaoglu, S.A.; Yayli, N. Volatile constituents and antimicrobial activities from flower and fruit of Arbutus unedo L. Asian J. Chem. 2010, 22, 6437–6442. [Google Scholar]
- Özcan, M.M.; Haciseferoğullari, H. The strawberry (Arbutus unedo L.) fruits: Chemical composition, physical properties and mineral contents. J. Food Eng. 2007, 78, 1022–1028. [Google Scholar] [CrossRef]
- Ekinci, N.; Sakaldas, A.; Seker, M.; Gundogdu, M.A.; Ekinci, H. Plant and fruit characteristics of Arbutus unedo L. and Arbutus andrachnae L. from the highlands of Northwestern Turkey. Acta Hort. 2013, 97, 231–236. [Google Scholar]
- Pavlović, D.R.; Lakušić, B.; Došlov-Kokoruš, Z.; Kovaćević, N. Arbutin content and antioxidant activity of some Ericaceae species. Pharmazie 2009, 64, 656–659. [Google Scholar] [PubMed]
- Maleš, Ž.; Šarić, D.; Bojić, M. Quantitative determination of flavonoids and chlorogenic acid in the leaves of Arbutus unedo L. using thin layer chromatography. J. Anal. Methods Chem. 2010, 2013. [Google Scholar] [CrossRef]
- Kivcak, B.; Mert, T. Quantitative determination of α-tocopherol in Arbutus unedo by TLC-densitometry and colorimetry. Fitoterapia 2001, 72, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Kivcak, B.; Mert, T.; Demirci, B.; Baser, K.H.C. Composition of the essential oil of Arbutus unedo. Chem. Nat. Compd. 2001, 37, 445–446. [Google Scholar] [CrossRef]
- Bessah, R.; Benyoussef, E.-H. Essential oil composition of Arbutus unedo L. leaves from Algeria. J. Essent. Oil Bear. Plants 2012, 15, 678–682. [Google Scholar] [CrossRef]
- Owen, S.; Boissard, C.; Street, R.A.; Duckham, S.C.; Csiky, O.; Hewitt, C.N. Screening of 18 Mediterranean plant species for volatile organic compound emissions. Atmos. Environ. 1997, 31, 101–117. [Google Scholar] [CrossRef]
- Peñuelas, J.; Lluisà, J. Seasonal patterns of non-terpenoid C6–C10 VOC emission from seven Mediterranean woody species. Chemosphere 2001, 45, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Dib, M.A.; Allali, H.; Tabti, B.; Bendiabdellah, A.; Djabou, N. A new proanthocyanidins from Arbutus unedo L. stems. Asian J. Chem. 2008, 20, 3926–3934. [Google Scholar]
- Dib, M.A.; Djabou, N.; Allali, H.; Tabti, B. Identification of phenolic compounds and antimicrobial activity of roots of Arbutus unedo L. Asian J. Chem. 2010, 22, 4045–4053. [Google Scholar]
- Simonetti, M.S.; Damiani, F.; Gabrielli, L.; Cossignani, L.; Blasi, F.; Marini, F.; Montesano, D.; Maurizi, A.; Ventura, F.; Bosi, A.; et al. Characteriztion of triacylglycerols in Arbutus unedo L. seeds. Ital. J. Food Sci. 2008, 20, 49–56. [Google Scholar]
- Fiorentino, A.; Castaldi, S.; d’Abrosca, B.; Natale, A.; Carfora, A.; Messere, A.; Monaco, P. Polyphenols from the hydroalcoholic extract of Arbutus unedo living in a monospecific Mediterranean woodland. Biochem. Syst. Ecol. 2007, 35, 809–811. [Google Scholar] [CrossRef]
- Carcache-Blanco, J.F.; Cuendet, M.; Park, E.J.; Su, B.-N.; Rivero-Cruz, J.F.; Farnsworth, N.R.; Pezzuto, J.M.; Kinghorn, A.D. Potential cancer chemopreventive agents from Arbutus unedo. Nat. Prod. Res. 2006, 20, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Akay, S.; Alpak, I.; Yesil-Celiktas, O. Effects of process parameters on supercritical CO2 extraction of total phenols from strawberry (Arbutus unedo L.) fruits: An optimization study. J. Sep. Sci. 2011, 34, 1925–1931. [Google Scholar] [CrossRef]
- Isbilir, S.S.; Orak, H.H.; Yagar, H.; Ekinci, N. Determination of antioxidant activities of strawberry tree (Arbutus unedo L.) flowers and fruits at different ripening stages. Acta Sci. Pol. 2012, 11, 223–237. [Google Scholar]
- Orak, H.H.; Aktas, T.; Yagar, H.; Isbilir, S.S.; Ekinci, N.; Sahin, F.H. Effects of hot air and freeze drying methods on antioxidant activity, colour and some nutritional characteristics of strawberry tree (Arbutus unedo L.) fruit. Food Sci. Technol. Int. 2012, 18, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, A.C.; Gago, C.M.L.; Miguel, M.G.C.; Antunes, M.D.C. The effect of temperature and film covers on the storage ability of Arbutus unedo L. fresh fruit. Sci. Hort. 2013, 159, 96–102. [Google Scholar] [CrossRef]
- Guimarães, R.; Barros, L.; Calhelha, R.C.; Carvalho, A.M.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Bioactivity of different enriched phenolic extracts of wild fruits from Northeastern Portugal: A comparative study. Plant Foods Hum. Nutr. 2014, 69, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Demirsoy, H.; Demirsoy, L.; Çelikel, G.; Koyuncu, T. Effects of dried on some properties of strawberry tree fruits. Asian J. Chem. 2007, 19, 1777–1782. [Google Scholar]
- Armenteros, M.; Morcuende, D.; Ventanas, S.; Estévez, M. Application of natural antioxidants from strawberry tree (Arbutus unedo L.) and dog rose (Rosa canina L.) to frankfurters subjected to refrigerated storage. J. Integr. Agric. 2013, 12, 1972–1981. [Google Scholar]
- Ganhão, R.; Morcuende, D.; Estévez, M. Protein oxidation in emulsified cooked burger patties with added fruit extarcts: Influence on colour and texture deterioration during chill storage. Meat Sci. 2010, 85, 402–409. [Google Scholar] [CrossRef]
- Ganhão, R.; Estévez, M.; Armenteros, M.; Morcuende, D. Mediterranean berries as inhibitors of lipid oxidation in porcine burger patties subjected to cooking and chilled storage. J. Integr. Agric. 2013, 12, 1982–1992. [Google Scholar] [CrossRef]
- Cossu, M.; Juliano, C.; Pisu, R.; Alamanni, M.C. Effects of enrichment with polyphenol extracts from Sardinian plants on physico-chemical, antioxidant and microbiological properties of yougurt. Ital. J. Food Sci. 2009, 21, 447–459. [Google Scholar]
- Orak, H.H.; Yagar, H.; Isbilir, S.S.; Demirci, A.Ş.; Gümüş, T.; Ekinci, N. Evaluation of antioxidant and antimicrobial potential of strawberry tree (Arbutus unedo L.) leaf. Food Sci. Biotechnol. 2011, 20, 1249–1256. [Google Scholar] [CrossRef]
- Malheiro, R.; Sá, O.; Pereira, E.; Aguiar, C.; Baptista, P.; Pereira, J.A. Arbutus unedo L. leaves as source of phytochemicals with bioactive properties. Ind. Crops Prod. 2012, 37, 473–478. [Google Scholar] [CrossRef]
- El Ouarti, A.; Haouat, A.C.; Sqalli, H.; Haggoud, A.; Ennabili, A.; Ibnsouda, S.; Iachagar, M.; Iraqui, M. Extra- and intracellular antimycobacterial activity of Arbutus unedo L. Afr. J. Microbiol. Res. 2012, 6, 1283–1290. [Google Scholar]
- Ferreira, S.; Santos, J.; Duarte, A.; Duarte, A.P.; Queiroz, J.A. Screening of antimicrobial activity of Cistus ladanifer and Arbutus unedo extracts. Nat. Prod. Res. 2012, 26, 1558–1560. [Google Scholar] [CrossRef] [PubMed]
- Kivcak, B.; Mert, T.; Ertabaklar, H.; Balcioğlu, I.C.; Töz, S.O. In vitro activity of Arbutus unedo agaisnt Leishmania tropica promastigotes. Turk. Soc. Parasitol. 2009, 33, 114–115. [Google Scholar]
- Ertabaklar, H.; Kivçak, B.; Mert, T.; Töz, S.Ö. In vitro activity of Arbutus unedo in leaf extracts against Trichomones vaginalis trophozoites. Turk. Soc. Parasitol. 2009, 33, 263–265. [Google Scholar]
- Mariotto, S.; Esposito, E.; Paola, R.D.; Ciampa, A.; Mazzon, E.; Prati, A.C.; Darra, E.; Vincenzi, S.; Cucinotta, G.; Caminiti, R.; et al. Protective effect of Arbutus unedo aqueous extract in carrageenan-induced lung inflammation in mice. Pharmacol. Res. 2008, 57, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, S.; Prati, A.C.; Darra, E.; Vincenzi, S.; Sega, M.; Cavalieri, E.; Shogi, K.; Suzuki, H. Aqueous extract of Arbutus unedo inhibits STAT1 activation in human breast cancer cell line MDA-MB-231 and human fibroblasts through SHP2 activation. Med. Chem. 2008, 4, 219–228. [Google Scholar] [PubMed]
- Mekhifi, H.; El-Haouari, M.; Bnouham, M.; Aziz, M.; Ziyyat, A.; Legssyer, A. Effects of extracts and tannins from Arbutus unedo leaves on rat platelet aggregation. Phytother. Res. 2006, 20, 135–139. [Google Scholar] [CrossRef]
- Legssyer, A.; Ziyyat, A.; Mekhifi, H.; Bnouham, M.; Herrenknecht, C.; Roumy, V.; Fourneau, C.; Laurens, A.; Hoerter, J.; Fischmeister, R. Tannins and catechin gallate mediate the vasorelaxant effect of Arbutus unedo on the rat isolated aorta. Phytother. Res. 2004, 18, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Dib, M.A.; Allali, H.; Bendiabdellah, A.; Meliani, N.; Tabti, B. Antimicrobial activity and phytochemical screening of A. unedo L. J. Saudi Chem. Soc. 2013, 17, 381–395. [Google Scholar] [CrossRef]
- Bnouham, M.; Merhfour, F.Z.; Ziyyat, A.; Aziz, M.; Legssyer, A.; Mekhfi, H. Antidiabetic effect of some medicinal plants of Oriental Morocco in neonatal non-insulin-dependent diabetes mellitus rats. Hum. Exp. Toxicol. 2010, 29, 865–871. [Google Scholar] [CrossRef] [PubMed]
- Afkir, S.; Nguelefack, T.B.; Aziz, M.; Zoheir, J.; Cuisinaud, G.; Bnouham, M.; Mekhfi, H.; Legssyer, A.; Lahlou, S.; Ziyyat, A. Arbutus unedo prevents cardiovascular and morphological alterations in L-NAME-induced hypertensive rats. Part I: Cardiovascular and renal hemodynamic effects of Arbutus unedo in L-NAME-induced hypertensive rats. J. Ethnopharmacol. 2008, 116, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Spano, N.; Casula, L.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Scanu, R.; Tapparo, A.; Sanna, G. A RP-HPLC determination of 5-hydroxymethylfurfural in honey. The case of strawberry tree honey. Talanta 2006, 68, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Cabras, P.; Angioni, A.; Tuberoso, C.; Floris, I.; Reniero, F.; Guillou, C.; Ghelli, S. Homogentisic acid: A phenolic acid as a marker of strawberry-tree (Arbutus unedo) honey. J. Agric. Food Chem. 1999, 47, 4064–4067. [Google Scholar]
- Scanu, R.; Spano, N.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Sanna, G.; Tapparo, A. Direct chromatographic methods for the rapid determiantion of homogentisic acid in strawberry tree (Arbutus unedo L.) honey. J. Chromatogr. A 2005, 1090, 76–80. [Google Scholar] [PubMed]
- Tuberoso, C.I.G.; Bifulco, E.; Caboni, P.; Cottiglia, F.; Cabras, P.; Floris, I. Floral markers of strawberry tree (Arbutus unedo L.) honey. J. Agric. Food Chem. 2010, 58, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Careri, M.; Musci, M. Volatile norisoprenoids as markers of botanical origin of Sardinian strawberry-tree (Arbutus unedo L.) honey: Characterization of aroma compounds by dynamic headspace extraction and gas chromatography-mass spectrometry. Food Chem. 2005, 89, 527–532. [Google Scholar]
- De la Fuente, E.; Sanz, M.L.; Martínez-Castro, I.; Sanz, J.; Ruiz-Matute, A.I. Volatile and carbohydrate composition of rare unifloral honeys from Spain. Food Chem. 2007, 105, 84–93. [Google Scholar] [CrossRef]
- Spano, N.; Piras, I.; Ciulu, M.; Floris, I.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Sanna, G. Reversed-phase liquid chromatographic profile of free amino acids in strawberry-tree (Arbutus unedo L.) honey. J. AOAC Int. 2009, 92, S1145–S1156. [Google Scholar]
- Aazza, S.; Lyoussi, B.; Antunes, D.; Miguel, M.G. Physicochemical characterization and antioxidant activity of commercial Portuguese honeys. J. Food Sci. 2013, 78, C1159–C1165. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.; Ramos, A.; Gonçalves, M.M.; Bernardo, M.; Mendes, B. Antioxidant activity, quality parameters and mineral content of Portuguese monofloral honeys. J. Food Compos. Anal. 2013, 30, 130–138. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Boban, M.; Bifulco, E.; Budimir, D.; Pirisi, F.M. Antioxidant capacity and vasodilatory properties of Mediterranean food: The case of Cannonau wine, myrtle berries liqueur and strawberry-tree honey. Food Chem. 2013, 140, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.; Tuberoso, C.I.G.; Atzeri, A.; Melis, M.P.; Bifulco, E.; Dessì, M.A. Antioxidant profile of strawberry tree honey and its marker homogentisic acid in several models of oxidative stress. Food Chem. 2011, 129, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Cavaco, T.; Longuinho, C.; Quintas, C.; Carvalho, I.S. Chemical and microbial changes during the natural fermentation of strawberry tree (Arbutus unedo L.) fruits. J. Food Biochem. 2007, 31, 715–725. [Google Scholar] [CrossRef]
- Santos, D.E.; Galego, L.; Gonçalves, T.; Quintas, C. Yeast diversity in the Mediterranean strawberry tree (Arbutus unedo L.) fruits’ fermentations. Food Res. Int. 2012, 47, 45–50. [Google Scholar] [CrossRef]
- Versini, G.; Moser, S.; Franco, M.A.; Manca, G. Characterisation of strawberry tree distillate (Arbutus unedo L.) produced in Sardinia. J. Commod. Sci. Technol. Qual. 2011, 50, 197–206. [Google Scholar]
- Versini, G.; Seeber, R.; Serra, A.D.; Sferlazzo, G.; Carvalho, B.; Reniero, F. Aroma compounds of arbutus distillates. In Food Flavors: Generation, Analysis and Process Influence; Charalambous, G., Ed.; Elsevier Science B.V.: Amsterdam, The Netherlands, 1995; pp. 1779–1790. [Google Scholar]
- González, E.A.; Agrasar, A.T.; Castro, L.M.P.; Fernández, I.O.; Guerra, N.P. Solid-state fermentation of red raspberry (Rubus ideaus L.) and arbutus berry (Arbutus unedo L.) and characterization of their distillates. Food Res. Intern. 2011, 44, 1419–1426. [Google Scholar] [CrossRef]
- Galego, L.R.; Jockusch, S.; Silva, J.P. Polyphenol and volatile profiles of pomegranate (Punica granatum L.) fruit extracts and liquors. Int. J. Food Sci. Technol. 2013, 48, 693–700. [Google Scholar]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miguel, M.G.; Faleiro, M.L.; Guerreiro, A.C.; Antunes, M.D. Arbutus unedo L.: Chemical and Biological Properties. Molecules 2014, 19, 15799-15823. https://doi.org/10.3390/molecules191015799
Miguel MG, Faleiro ML, Guerreiro AC, Antunes MD. Arbutus unedo L.: Chemical and Biological Properties. Molecules. 2014; 19(10):15799-15823. https://doi.org/10.3390/molecules191015799
Chicago/Turabian StyleMiguel, Maria G., Maria L. Faleiro, Adriana C. Guerreiro, and Maria D. Antunes. 2014. "Arbutus unedo L.: Chemical and Biological Properties" Molecules 19, no. 10: 15799-15823. https://doi.org/10.3390/molecules191015799
APA StyleMiguel, M. G., Faleiro, M. L., Guerreiro, A. C., & Antunes, M. D. (2014). Arbutus unedo L.: Chemical and Biological Properties. Molecules, 19(10), 15799-15823. https://doi.org/10.3390/molecules191015799