Anticholinesterase Inhibitory Activity of Quaternary Alkaloids from Tinospora crispa
Abstract
:1. Introduction
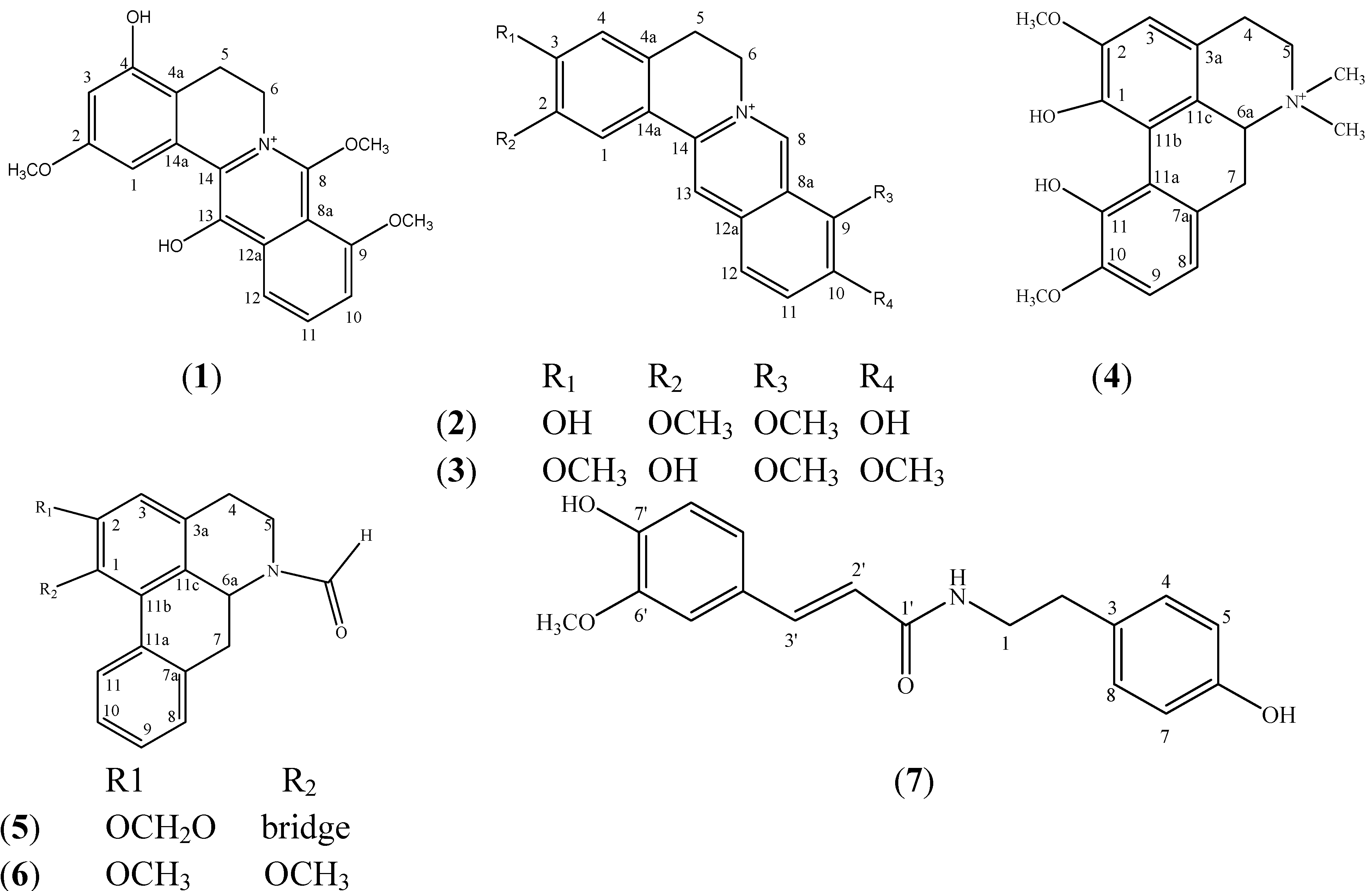
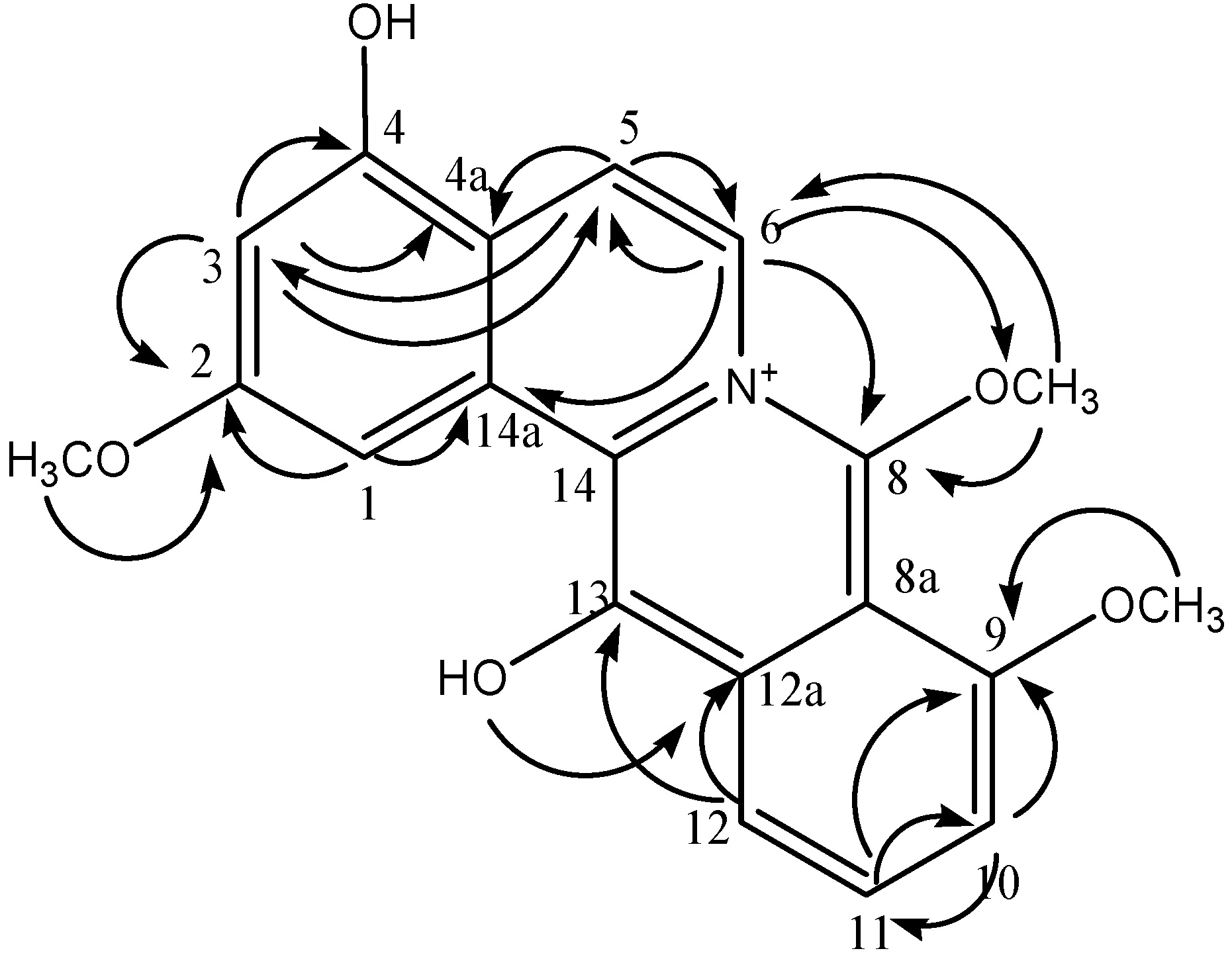
2. Results and Discussion
| Carbons | δC (ppm) | δH (ppm) Int. Mult. J | 1H-1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 107.69 | 7.28 (1H, br s) | - | C14a, C2 |
| 2 | 162.37 | - | - | - |
| 3 | 103.89 | 7.35 (1H, br s) | - | C5, C4a, C4, C2 |
| 4 | 163.81 | - | - | - |
| 4a | 127.21 | - | - | - |
| 5 | 121.33 | 7.87 (1H, d, 6.50) | H6 | C3, C4a, C6 |
| 6 | 130.32 | 7.96 (1H, d, 6.00) | H5 | C8, C14a, C5, 8-OCH3 |
| 8 | 150.46 | - | - | |
| 8a | 121.53 | - | - | - |
| 9 | 147.22 | - | - | |
| 10 | 114.86 | 6.56 (1H, d, 2.0) | H11 | C11, C9 |
| 11 | 118.52 | 6.52 (1H, dd, 8.0, 2.0) | H12 | C9, C10 |
| 12 | 111.85 | 6.86 (1H, d, 8.5) | - | C12a, C13 |
| 12a | 127.19 | - | - | - |
| 13 | 147.55 | - | - | - |
| 14 | 136.98 | - | - | - |
| 14a | 132.05 | - | - | - |
| 2-OCH3 | 55.08 | 4.07 (3H, s) | - | C2 |
| 9-OCH3 | 54.94 | 3.81 (3H, s) | - | C9 |
| 8-OCH3 | 44.17 a | 4.20 (3H, s) | - | C8, C6 |
| 13-OH | - | 4.71 (1H, s) | - | C12a |
| Compound | IC50 (μM) |
|---|---|
| 1 | 517.6 ± 5.3 |
| 2 | 276.1 ± 1.8 |
| 3 | 48.1 ± 1.3 |
| 4 | NA |
| 5 | 415.3 ± 2.7 |
| 6 | 564.6 ± 2.1 |
| 7 | NA |
| Physostigmine | 31.4 ± 0.5 |
3. Experimental
3.1. General
3.2. Plant Materials
3.3. Extraction and Chromatography
3.4. Spectroscopic Data
3.5. Acetylcholinesterase (AChE) Inhibitory Assay
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Pathak, A.K.; Jain, D.C.; Sharma, R.P. Chemistry and biological activities of the Genera tinospora. Pharmaceut. Biol. 1995, 33, 277–287. [Google Scholar] [CrossRef]
- Bisset, N.G.; Nawaiwu, J. Quaternary alkaloids of tinospora species. Planta Med. 1983, 48, 275–279. [Google Scholar] [CrossRef]
- Chang, H.M.; El-Fishawary, A.M.; Slatkin, D.J.; Schiff, P.L., Jr. Quaternary alkaloids of Tinospora capillipes. Planta Med. 1984, 50, 88–90. [Google Scholar] [CrossRef]
- Houghton, P.J.; Howes, M.-J.R. Natural products and derivatives affecting neurotransmission relevant to Alzheimer’s and Parkinson’s disease. Neurosignals 2005, 14, 6–22. [Google Scholar] [CrossRef]
- Hung, T.M.; Ngoc, T.M.; Youn, U.J. Anti-amnestic activity of pseudocoptisine from corydalis tuber. Biol. Pharm. Bull. 2008, 31, 159–162. [Google Scholar] [CrossRef]
- Jung, H.A.; Min, B.S.; Yokozawa, T.; Lee, J.H.; Kim, Y.S.; Choi, J.S. Anti-alzheimer and antioxidant activities of Coptidis rhizoma alkaloids. Biol. Pharm. Bull. 2009, 32, 1433–1438. [Google Scholar] [CrossRef]
- Dweck, A.C.; Cavin, J.P. Andawali (Tinospora crispa) — A review. Available online: http://www.dweckdata.com/published_papers/Tinospora_crispa.pdf (accessed on 28 June 2013).
- Hungerford, N.L.; Sands, D.P.A.; Kitching, W. Isolation and structure of some constituents of the Australian medicinal plant, Tinospora smilacina (Snakevine). Aust. J. Chem. 1998, 51, 1103–1111. [Google Scholar] [CrossRef]
- Le, P.M.; McCooeye, M.; Windust, A. Characterization of the alkaloids in goldenseal (Hydrastiscanadensis) root by high resolution Orbitrap LC-MS. Anal. Bioanal. Chem. 2013, 405, 4487–4498. [Google Scholar] [CrossRef]
- Contreras, H.R.; Biekofsky, R.R.; Kowalewski, D.C. Effects of electronic resonance interaction on the methoxy group NMR parameters: Theoretical and experimental study of substituted 2-methoxypyridines. J. Phys. Chem. 1993, 97, 91–93. [Google Scholar] [CrossRef]
- Li, S.; Chestnut, D.B. Intramolecular van der Waals interactions and chemical shifts: A model for β- and γ-effects. Magn. Reson. Chem. 1985, 23, 625–638. [Google Scholar] [CrossRef]
- Makhey, D.; Gatto, B.; Yu, C.; Liu, A.; Liu, L.F.; LaVoie, E.J. Coralyne and related compounds as mammalian topoisomerase I and topoisomerase II poisons. Bioorg. Med. Chem. 1996, 4, 781–791. [Google Scholar] [CrossRef]
- Thuy, T.T.; Franke, K.; Porzel, A.; Wessjohann, L.; Sung, T.V. Quaternary protoberberine alkaloids from Stephania rotunda. J. Chem. 2006, 44, 259–264. [Google Scholar]
- Patel, M.B.; Mishra, S.M. Magnoflorine from Tinospora cordifolia stem inhibits α-glucosidase and is antiglycemic in rats. J. Funct. Foods 2012, 4, 79–86. [Google Scholar] [CrossRef]
- Pachaly, P.; Adnan, A.Z.; Will, G. NMR-assignments of N-acylaporphine alkaloids from Tinospora crispa. Planta Med. 1992, 58, 184–187. [Google Scholar] [CrossRef]
- Fukuda, N.; Yonemitsu, M.; Kimura, T. Studies on the constituents of the stems of Tinospora tuberculata Beumee. I. N-trans- and N-cis-feruloyltyramine, and a new phenolic glucoside, Tinotuberide. Chem. Pharm. Bull. 1983, 31, 156–161. [Google Scholar] [CrossRef]
- Wang, B.; Mai, Y.; Li, Y.; Hou, J.; Huang, S.; Ou, T.; Tan, J. Synthesis and evaluation of novel rutaecarpine derivatives and related alkaloids derivatives as selective acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2010, 45, 1415–1423. [Google Scholar] [CrossRef]
- Adekanmi, E.; Fouche, G.; Steenkamp, V. Cytotoxicity and acetylcholinesterase inhibitory activity of an isolated crinine alkaloid from Boophane disticha (Amaryllidaceae). J. Ethnopharmacol. 2012, 143, 572–578. [Google Scholar] [CrossRef]
- Dall’Acqua, S. Plant-derived acetylcholinesterase inhibitory alkaloids for the treatment of Alzheimer’s disease. Bot.: Targets Ther. 2013, 3, 19–28. [Google Scholar] [CrossRef]
- Khalid, A.; Ghayur, M.N.; Feroz, F. Cholinesterase inhibitory and spasmolytic potential of steroidal alkaloids. J. Steroid Biochem. Mol. Biol. 2004, 92, 477–484. [Google Scholar] [CrossRef]
- Arning, J.; Stolte, S.; Böschen, A.; Stock, F.; Pitner, W.R.; Welz-Biermann, U.; Jastorff, B. Qualitative and quantitative structure activity relationships for the inhibitory effects of cationic head groups, functionalised side chains and anions of ionic liquids on acetylcholinesterase. Green Chem. 2008, 10, 47–58. [Google Scholar] [CrossRef]
- Huang, L.; Luo, Z.; He, F.; Lu, J.; Li, X. Synthesis and biological evaluation of a new series of berberine derivatives as dual inhibitors of acetylcholinesterase and butyrylcholinesterase. Bioorg. Med. Chem. 2010, 18, 4475–4484. [Google Scholar] [CrossRef]
- Israilov, I.A.; Karimova, S.U.; Yunusov, M.S. Aporphine alkaloids. Chem. Nat. Comp. 1980, 16, 197–225. [Google Scholar] [CrossRef]
- Eldeen, I.M.S.; Elgorashi, E.E.; van Staden, J. Antibacterial, anti-inflammatory, anti-cholinesterase and mutagenic effects of extracts obtained from some trees used in South African traditional medicine. J. Ethnopharmacol. 2005, 102, 457–464. [Google Scholar] [CrossRef]
- Sample Availability: Samples of all compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yusoff, M.; Hamid, H.; Houghton, P. Anticholinesterase Inhibitory Activity of Quaternary Alkaloids from Tinospora crispa. Molecules 2014, 19, 1201-1211. https://doi.org/10.3390/molecules19011201
Yusoff M, Hamid H, Houghton P. Anticholinesterase Inhibitory Activity of Quaternary Alkaloids from Tinospora crispa. Molecules. 2014; 19(1):1201-1211. https://doi.org/10.3390/molecules19011201
Chicago/Turabian StyleYusoff, Mashitah, Hazrulrizawati Hamid, and Peter Houghton. 2014. "Anticholinesterase Inhibitory Activity of Quaternary Alkaloids from Tinospora crispa" Molecules 19, no. 1: 1201-1211. https://doi.org/10.3390/molecules19011201
APA StyleYusoff, M., Hamid, H., & Houghton, P. (2014). Anticholinesterase Inhibitory Activity of Quaternary Alkaloids from Tinospora crispa. Molecules, 19(1), 1201-1211. https://doi.org/10.3390/molecules19011201




