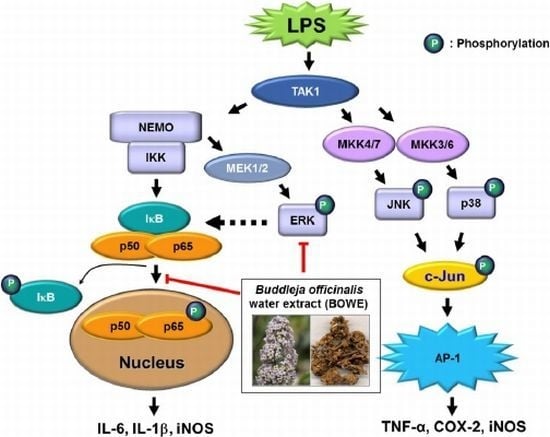
Inhibition of Lipopolysaccharide-Induced Proinflammatory Responses by Buddleja officinalis Extract in BV-2 Microglial Cells via Negative Regulation of NF-kB and ERK1/2 Signaling
Abstract
:1. Introduction
2. Results and Discussion
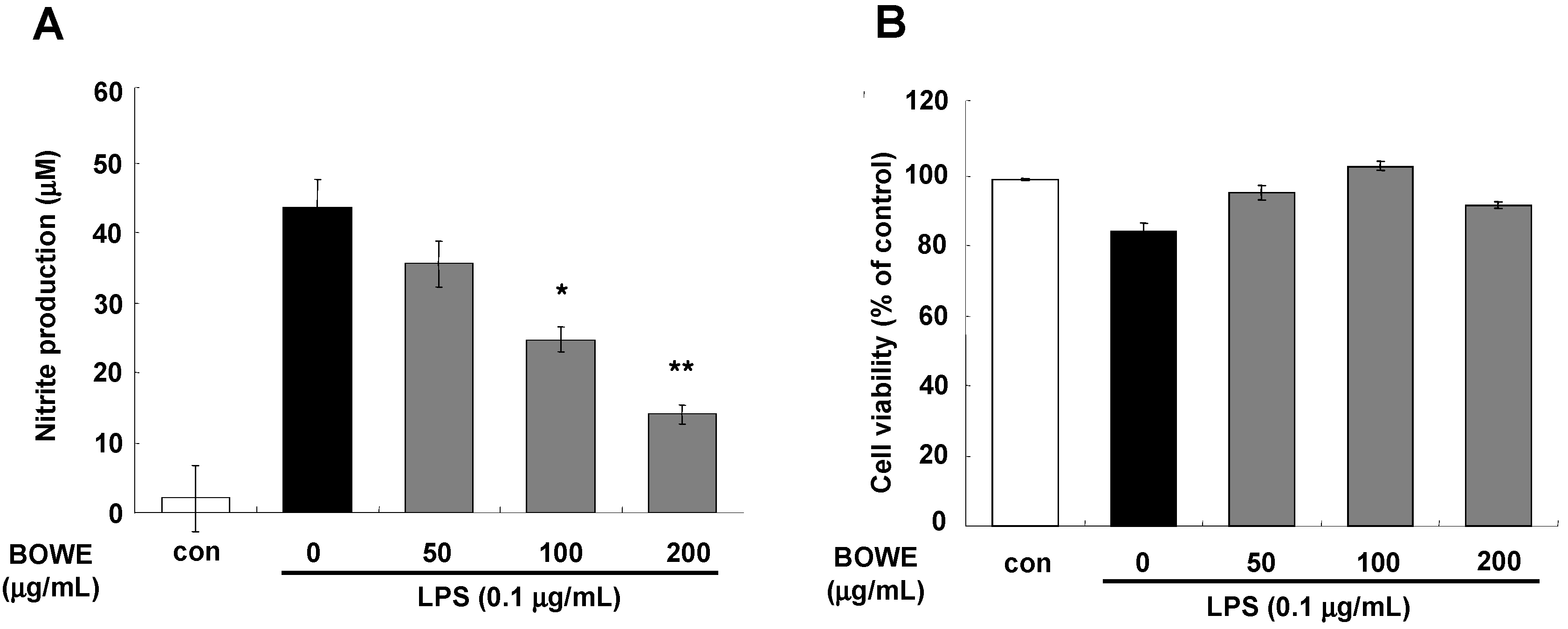
2.1. BOWE Attenuates LPS-Induced NO Production
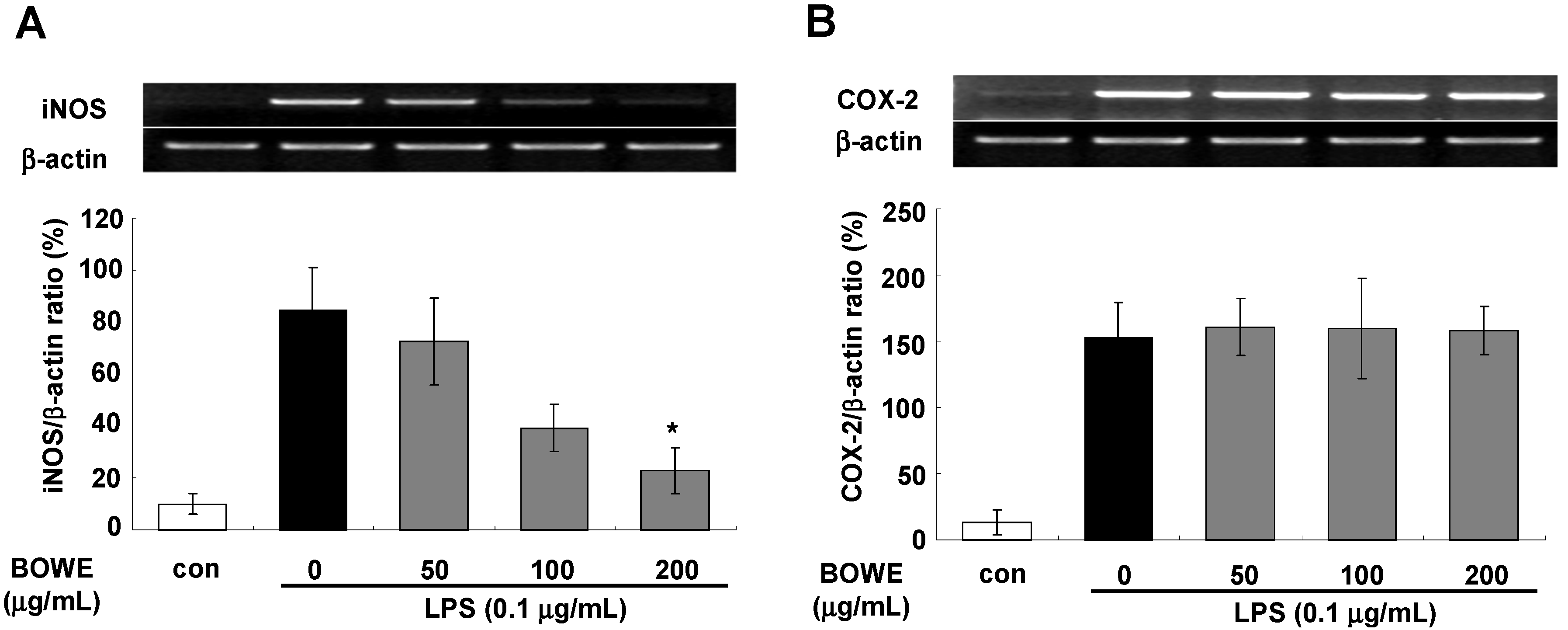
2.2. BOWE Inhibits LPS-Induced iNOS Expression, but not COX-2 Expression
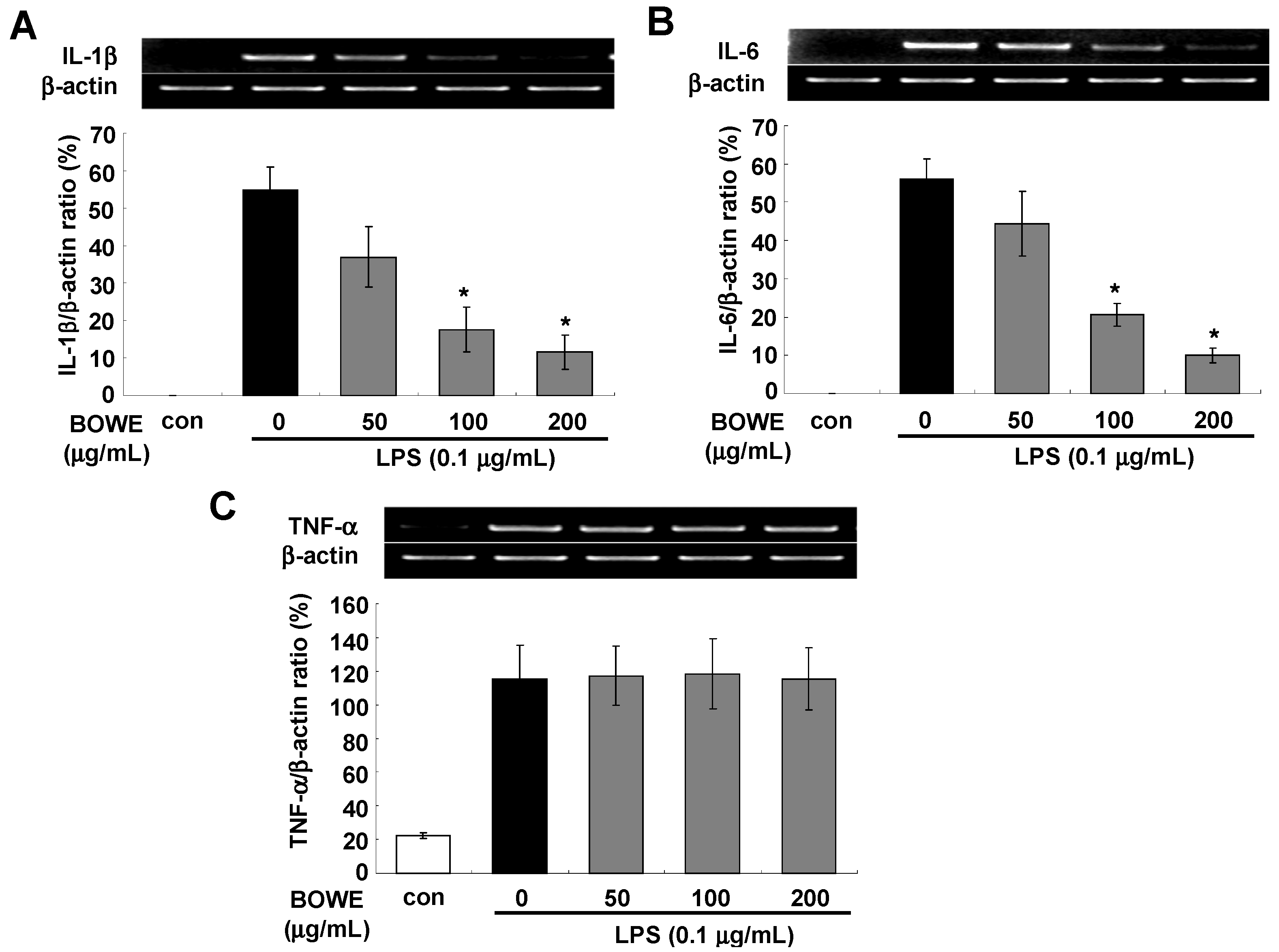
2.3. BOWE Inhibits LPS-Induced IL-1β and IL-6, but not TNF-α
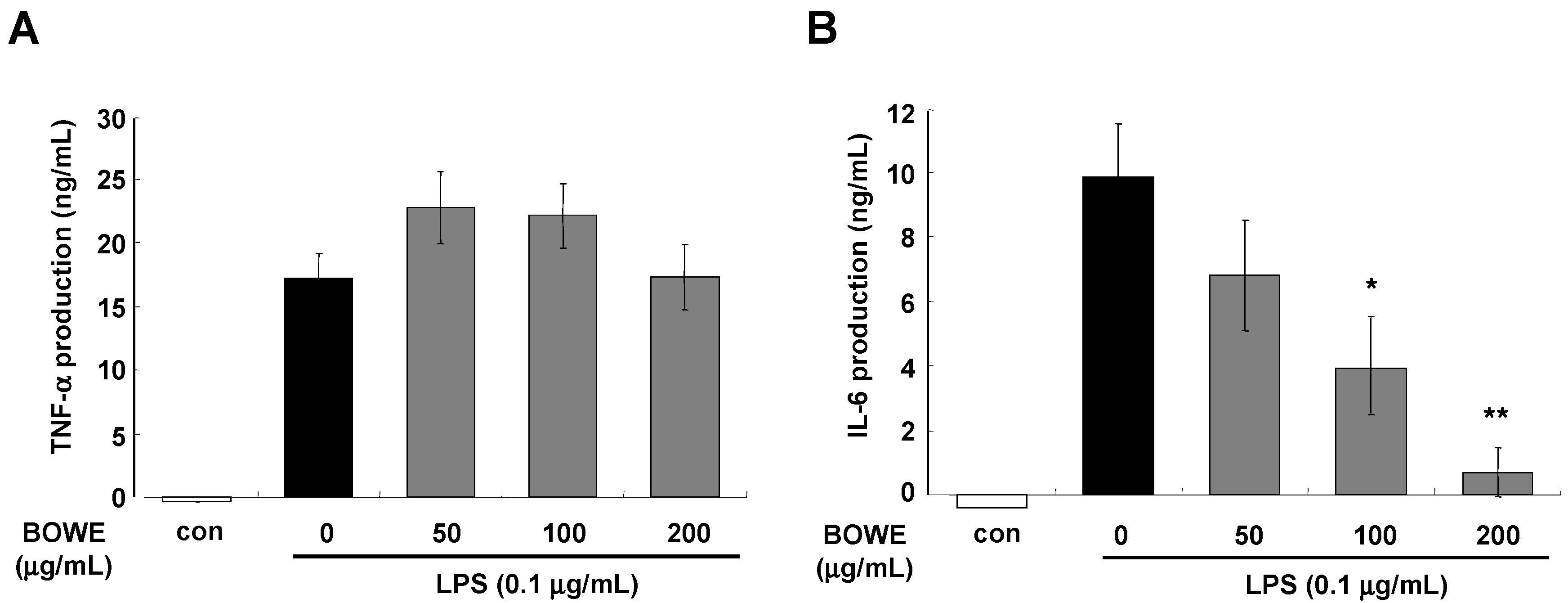
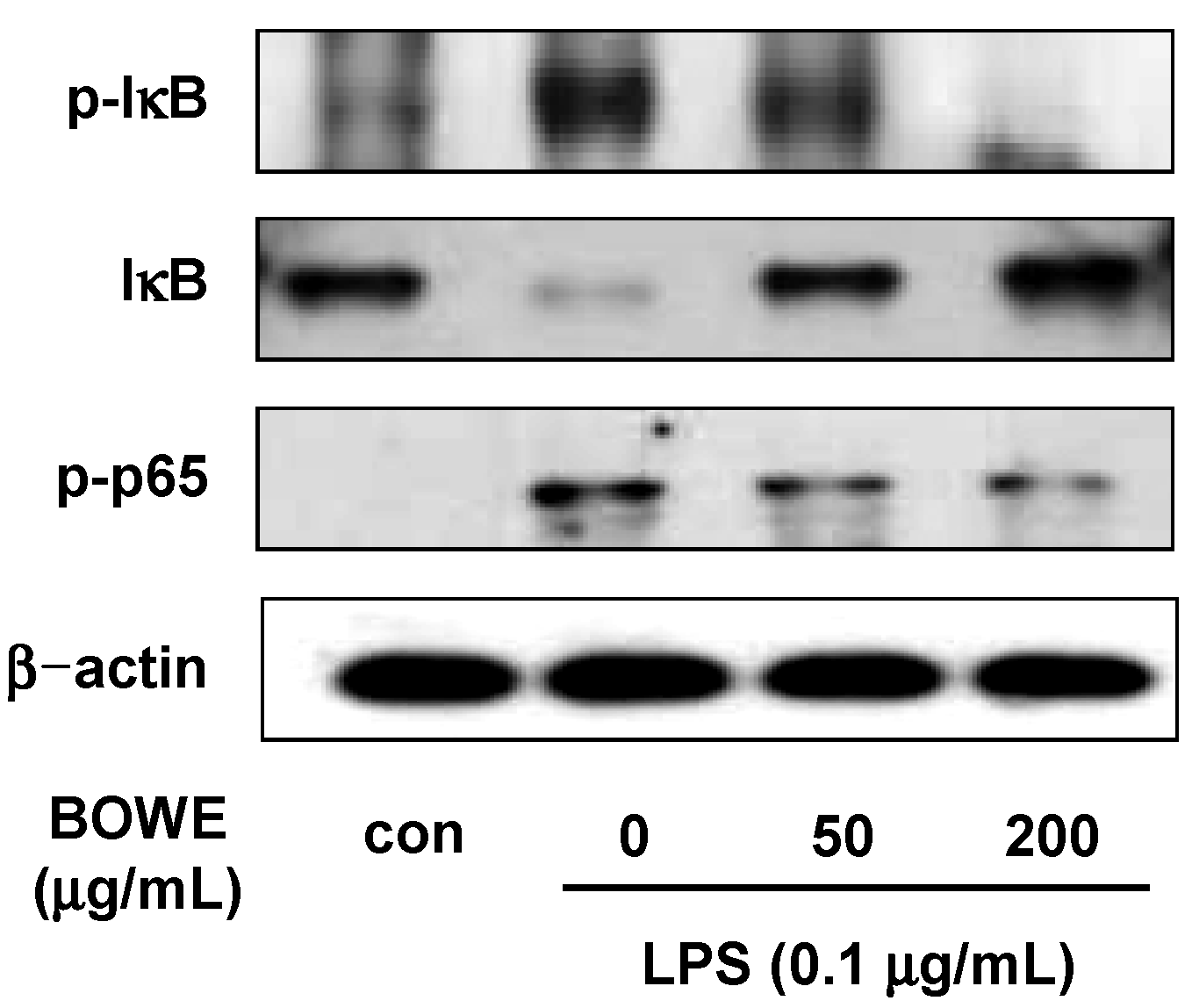
2.4. BOWE Inhibits LPS-Induced NF-κB Signaling
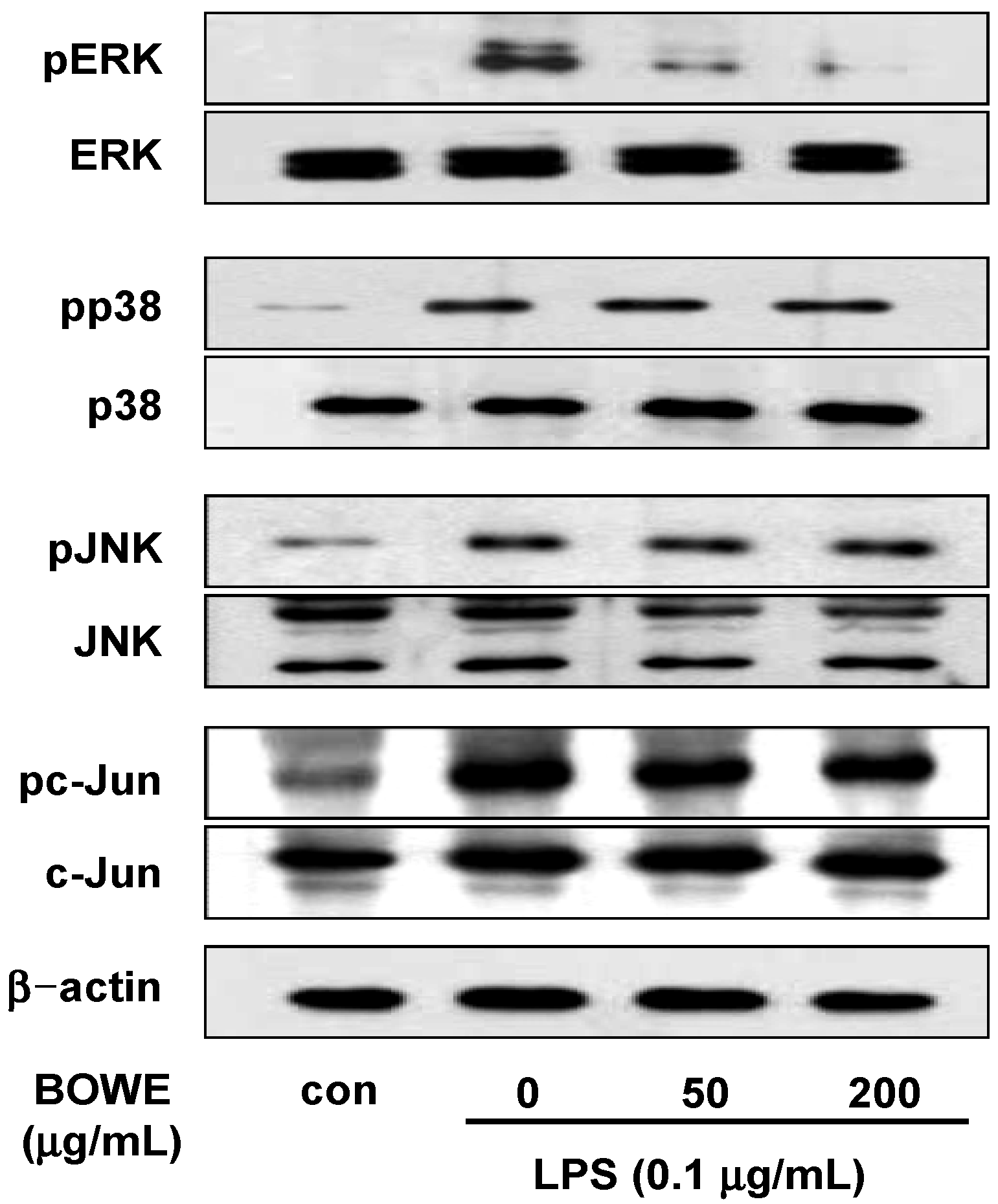
2.5. BOWE Inhibits LPS-Induced ERK1/2 Phosphorylation, but not p38 and JNK Phosphorylation

3. Experimental
3.1. Preparation of B. officinalis Water Extract
3.2. Cell Culture
3.3. Cell Viability and NO Assay
3.4. Enzyme-Linked Immunosorbent Assay (ELISA)
3.5. RNA Isolation and Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
| iNOS [29] | sense | CCG TCC ACA GTA TGT GAG GA |
| anti-sense | GAA CTC CAA GGT GGC AGC A | |
| COX-2 [30] | sense | TGA TGA CTG CCC AAC TCC CAT G |
| anti-sense | AAT GTT GAA GGT GTC CGG CAG C | |
| TNF-α [30] | sense | CAG ACC CTC ACA CTC AGA TCA TCT T |
| anti-sense | CAG AGC AAT GAC TCC AAA GTA GAC CT | |
| IL-1β [31] | sense | GAG GCT GAC AGA CCC CAA AAG AT |
| anti-sense | GCA CGA GGC ATT TTT GTT GTT CA | |
| IL-6 [22] | sense | GGA GCC CAC CAA GAA CGA TAG TCA |
| anti-sense | GAA GTA GGG AAG GCC GTG GTT |
3.6. Western Blot Analysis of c-Jun, MAPKs, and NF-κB Protein Expression
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Whitney, N.P.; Eidem, T.M.; Peng, H.; Huang, Y.; Zheng, J.C. Inflammation mediates varying effects in neurogenesis: relevance to the pathogenesis of brain injury and neurodegenerative disorders. J. Neurochem. 2009, 108, 1343–1359. [Google Scholar] [CrossRef]
- Liu, B. Role of Microglia in Inflammation-Mediated Neurodegenerative Diseases: Mechanisms and Strategies for Therapeutic Intervention. J. Pharm. Exp. Ther. 2003, 304, 1–7. [Google Scholar] [CrossRef]
- Saha, R.N.; Pahan, K. Regulation of Inducible Nitric Oxide Synthase Gene in Glial Cells. Antioxid. Redox Signal. 2006, 8, 929–947. [Google Scholar] [CrossRef]
- Kim, Y.S.; Joh, T.H. Microglia, major player in the brain inflammation: Their roles in the pathogenesis of Parkinson’s disease. Exp. Mol. Med. 2006, 38, 333–347. [Google Scholar] [CrossRef]
- McGeer, E.G.; McGeer, P.L. Inflammatory processes in Alzheimer’s disease. Prog. Neuro Psychoph. 2003, 27, 741–749. [Google Scholar] [CrossRef]
- Eikelenboom, P.; Bate, C.; van Gool, W.A.; Hoozemans, J.J.M.; Rozemuller, J.M.; Veerhuis, R.; Williams, A. Neuroinflammation in Alzheimer’s disease and prion disease. Glia 2002, 40, 232–239. [Google Scholar] [CrossRef]
- Sanders, P.; de Keyser, J. Janus faces of microglia in multiple sclerosis. Brain Res. 2007, 54, 274–285. [Google Scholar] [CrossRef]
- Sabroe, I.; Dower, S.K.; Whyte, M.K.B. The Role of Toll-Like Receptors in the Regulation of Neutrophil Migration, Activation, and Apoptosis. Clin. Infect. Dis. 2005, 41, S421–S426. [Google Scholar] [CrossRef]
- Necela, B.M.; Su, W.; Thompson, E.A. Toll-like receptor 4 mediates cross-talk between peroxisome proliferator-activated receptor γ and nuclear factor-κB in macrophages. Immunology 2008, 125, 344–358. [Google Scholar] [CrossRef]
- Kaminska, B. MAPK signalling pathways as molecular targets for anti-inflammatory therapy—From molecular mechanisms to therapeutic benefits. Biochim. Biophys. Acta 2005, 1754, 253–262. [Google Scholar] [CrossRef]
- Krens, S.F.G.; Spaink, H.P.; Snaar-Jagalska, B.E. Functions of the MAPK family in vertebrate-development. FEBS Lett. 2006, 580, 4984–4990. [Google Scholar] [CrossRef]
- Hayden, M.S.; Ghosh, S. Shared Principles in NF-κB Signaling. Cell 2008, 132, 344–362. [Google Scholar] [CrossRef]
- Hur, J. Tongeuibokam; Namsandang Publishing Co.: Seoul, Korea, 1983; p. 744. [Google Scholar]
- Tai, B.H.; Nhiem, N.X.; Quang, T.H.; Ngan, N.T.T.; Tung, N.H.; Kim, Y.; Lee, J.-J.; Myung, C.-S.; Cuong, N.M.; Kim, Y.H. A new iridoid and effect on the rat aortic vascular smooth muscle cell proliferation of isolated compounds from Buddleja officinalis. Bioorgan. Med. Chem. Lett. 2011, 21, 3462–3466. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, Y.S.; Choi, E.M. Linarin isolated from Buddleja officinalis prevents hydrogen peroxide-induced dysfunction in osteoblastic MC3T3-E1 cells. Cell. Immunol. 2011, 268, 112–116. [Google Scholar] [CrossRef]
- Lou, H.; Fan, P.; Perez, R.G.; Lou, H. Neuroprotective effects of linarin through activation of the PI3K/Akt pathway in amyloid-β-induced neuronal cell death. Bioorgan. Med. Chem. 2011, 19, 4021–4027. [Google Scholar] [CrossRef]
- Wang, Y.-C.; Huang, K.-M. In vitro anti-inflammatory effect of apigenin in the Helicobacter pylori-infected gastric adenocarcinoma cells. Food Chem. Toxicol. 2013, 53, 376–383. [Google Scholar] [CrossRef]
- Lee, D.H.; Ha, N.; Bu, Y.M.; Choi, H.I.; Park, Y.G.; Kim, Y.B.; Kim, M.Y.; Kim, H. Neuroprotective effect of Buddleja officinalis extract on transient middle cerebral artery occlusion in rats. Biol. Pharm. Bull. 2006, 29, 1608–1612. [Google Scholar] [CrossRef]
- Lala, P.K.; Chakraborty, C. Role of nitric oxide in carcinogenesis and tumour progression. Lancet Oncol. 2001, 2, 149–156. [Google Scholar] [CrossRef]
- Naseem, K. The role of nitric oxide in cardiovascular diseases. JMAM 2005, 26, 33–65. [Google Scholar]
- Pannu, R.; Singh, I. Pharmacological strategies for the regulation of inducible nitric oxide synthase: Neurodegenerative versus neuroprotective mechanisms. Neurochem. Int. 2006, 49, 170–182. [Google Scholar] [CrossRef]
- Kim, Y.H.; Koh, H.K.; Kim, D.S. Down-regulation of IL-6 production by astaxanthin via ERK-, MSK-, and NF-kappaB-mediated signals in activated microglia. Int. Immunopharmacol. 2010, 10, 1560–1572. [Google Scholar] [CrossRef]
- Song, L.; Li, J.; Hu, M.; Huang, C. Both IKKα and IKKβ are implicated in the arsenite-induced AP-1 transactivation correlating with cell apoptosis through NF-κB activity-independent manner. Exp. Cell Res. 2008, 314, 2187–2198. [Google Scholar] [CrossRef]
- Medeiros, R.; Figueiredo, C.P.; Pandolfo, P.; Duarte, F.S.; Prediger, R.D.S.; Passos, G.F.; Calixto, J.B. The role of TNF-α signaling pathway on COX-2 upregulation and cognitive decline induced by β-amyloid peptide. Behav. Brain Res. 2010, 209, 165–173. [Google Scholar] [CrossRef]
- Ramanan, S.; Kooshki, M.; Zhao, W.; Hsu, F.-C.; Robbins, M.E. PPARα ligands inhibit radiation-induced microglial inflammatory responses by negatively regulating NF-κB and AP-1 pathways. Free Radic. Bio. Med. 2008, 45, 1695–1704. [Google Scholar] [CrossRef]
- Kim, J.S.; Kim, K.D.; Na, H.S.; Jeong, S.Y.; Park, H.R.; Kim, S.; Chung, J. Tumor necrosis factor-alpha and interleukin-1beta expression pathway induced by Streptococcus mutans in macrophage cell line RAW 264.7. Mol. Oral Microbiol. 2012, 27, 149–159. [Google Scholar] [CrossRef]
- Shen, J.; Sakaida, I.; Uchida, K.; Terai, S.; Okita, K. Leptin enhances TNF-alpha production via p38 and JNK MAPK in LPS-stimulated Kupffer cells. Life Sci. 2005, 77, 1502–1515. [Google Scholar] [CrossRef]
- Svensson, C.; Part, K.; Kunnis-Beres, K.; Kaldmae, M.; Fernaeus, S.Z.; Land, T. Pro-survival effects of JNK and p38 MAPK pathways in LPS-induced activation of BV-2 cells. Biochem. Biophys. Res. Commun. 2011, 406, 488–492. [Google Scholar] [CrossRef]
- Lee, P.; Hur, J.; Lee, J.; Kim, J.; Jeong, J.; Kang, I.; Kim, S.Y.; Kim, H. 15,16-dihydrotanshinone I suppresses the activation of BV-2 cell, a murine microglia cell line, by lipopolysaccharide. Neurochem. Int. 2006, 48, 60–66. [Google Scholar] [CrossRef]
- Zhou, F.; Wu, J.Y.; Sun, X.L.; Yao, H.H.; Ding, J.H.; Hu, G. Iptakalim alleviates rotenone-induced degeneration of dopaminergic neurons through inhibiting microglia-mediated neuroinflammation. Neuropsychopharmacology 2007, 32, 2570–2580. [Google Scholar] [CrossRef]
- Tocharus, J.; Khonthun, C.; Chongthammakun, S.; Govitrapong, P. Melatonin attenuates methamphetamine-induced overexpression of pro-inflammatory cytokines in microglial cell lines. J. Pineal Res. 2010, 48, 347–352. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oh, W.-J.; Jung, U.; Eom, H.-S.; Shin, H.-J.; Park, H.-R. Inhibition of Lipopolysaccharide-Induced Proinflammatory Responses by Buddleja officinalis Extract in BV-2 Microglial Cells via Negative Regulation of NF-kB and ERK1/2 Signaling. Molecules 2013, 18, 9195-9206. https://doi.org/10.3390/molecules18089195
Oh W-J, Jung U, Eom H-S, Shin H-J, Park H-R. Inhibition of Lipopolysaccharide-Induced Proinflammatory Responses by Buddleja officinalis Extract in BV-2 Microglial Cells via Negative Regulation of NF-kB and ERK1/2 Signaling. Molecules. 2013; 18(8):9195-9206. https://doi.org/10.3390/molecules18089195
Chicago/Turabian StyleOh, Won-Jun, Uhee Jung, Hyun-Soo Eom, Hee-June Shin, and Hae-Ran Park. 2013. "Inhibition of Lipopolysaccharide-Induced Proinflammatory Responses by Buddleja officinalis Extract in BV-2 Microglial Cells via Negative Regulation of NF-kB and ERK1/2 Signaling" Molecules 18, no. 8: 9195-9206. https://doi.org/10.3390/molecules18089195
APA StyleOh, W.-J., Jung, U., Eom, H.-S., Shin, H.-J., & Park, H.-R. (2013). Inhibition of Lipopolysaccharide-Induced Proinflammatory Responses by Buddleja officinalis Extract in BV-2 Microglial Cells via Negative Regulation of NF-kB and ERK1/2 Signaling. Molecules, 18(8), 9195-9206. https://doi.org/10.3390/molecules18089195