Ethanol Extract of Atractylodes macrocephala Protects Bone Loss by Inhibiting Osteoclast Differentiation
Abstract
:1. Introduction
2. Results and Discussion
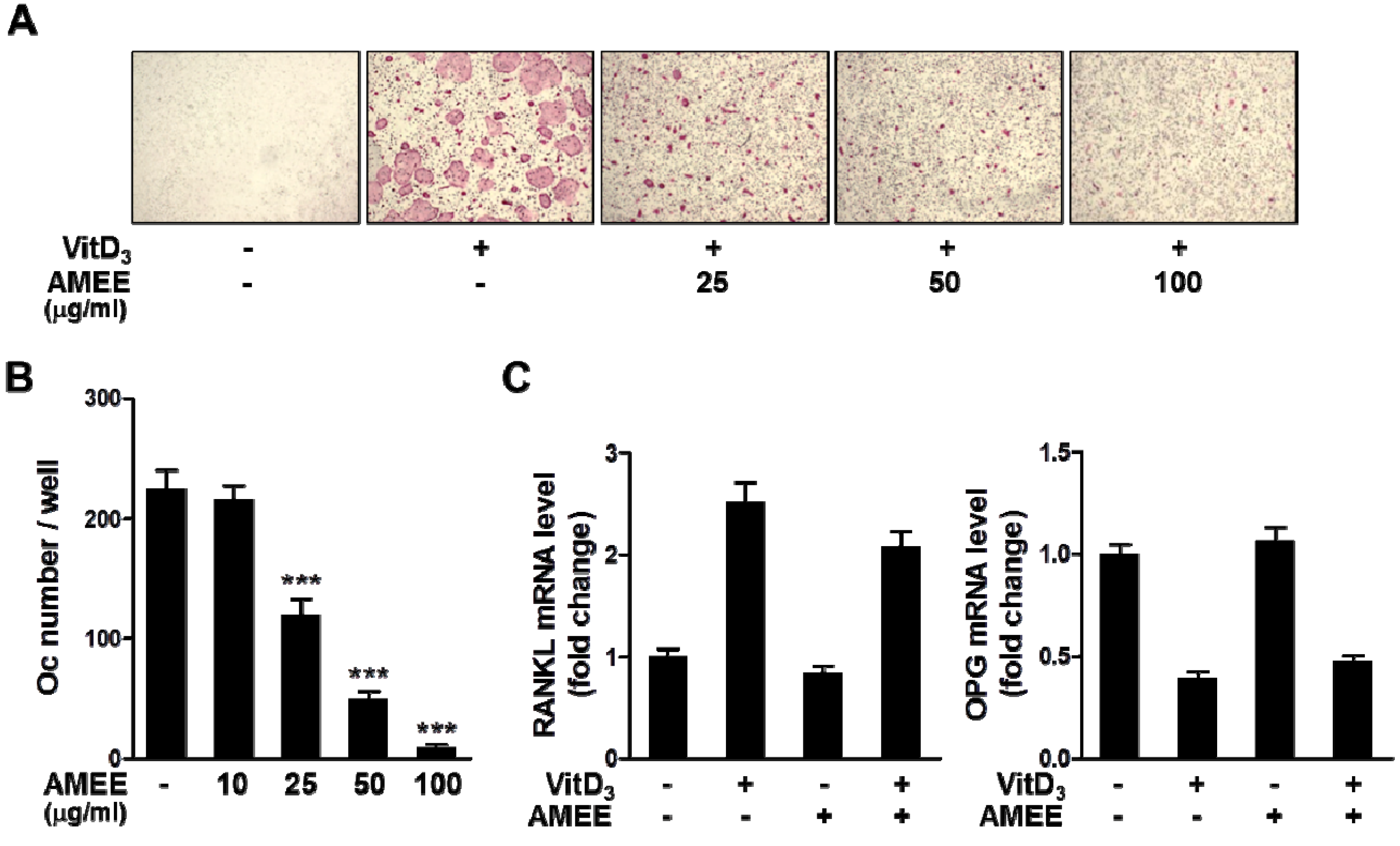
2.1. AMEE Inhibits Osteoclast Differentiation in Bone Marrow Cell-Osteoblast Coculture
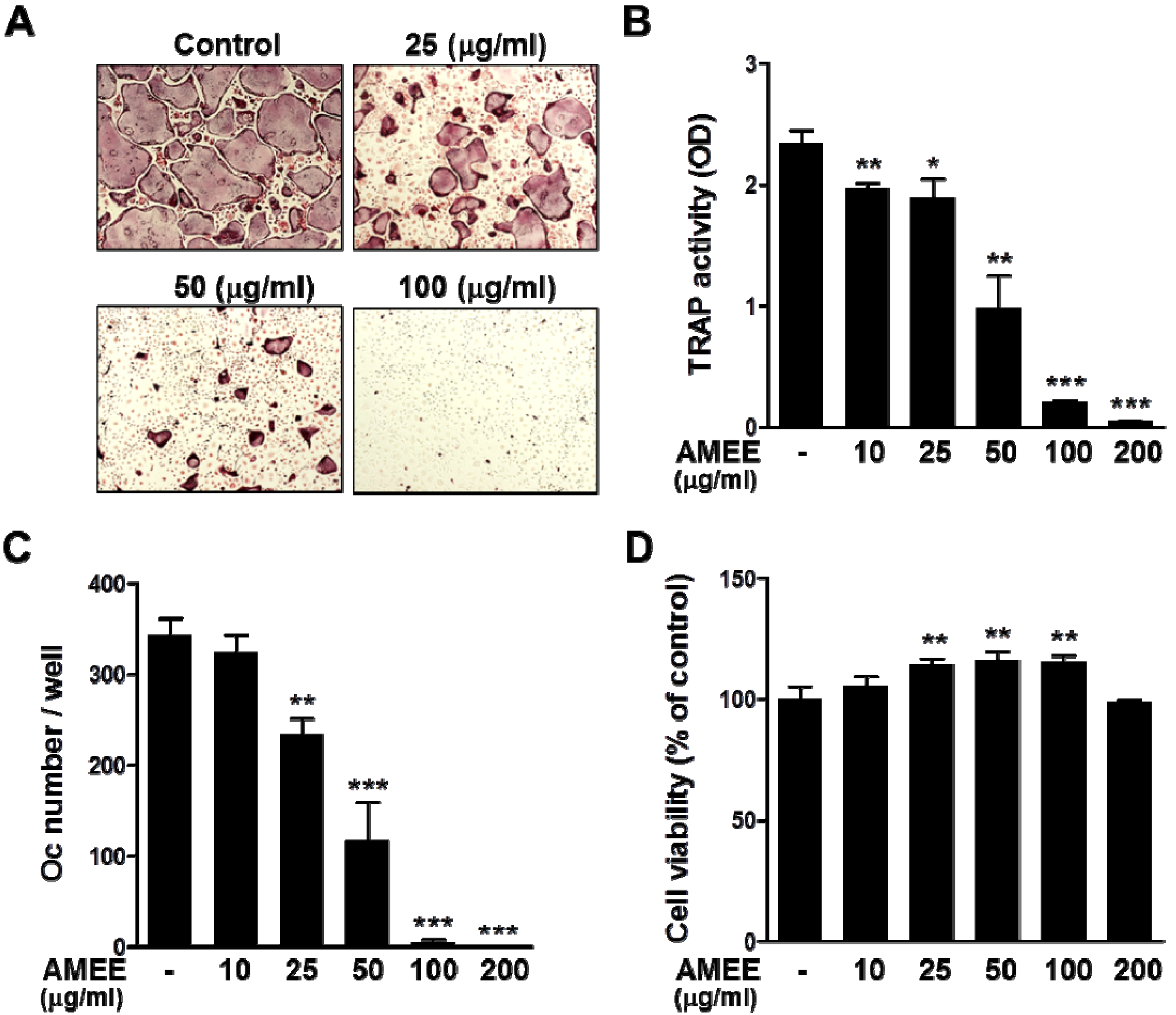
2.2. AMEE Inhibits RANKL-Induced Osteoclastogenesis from Osteoclast Precursors
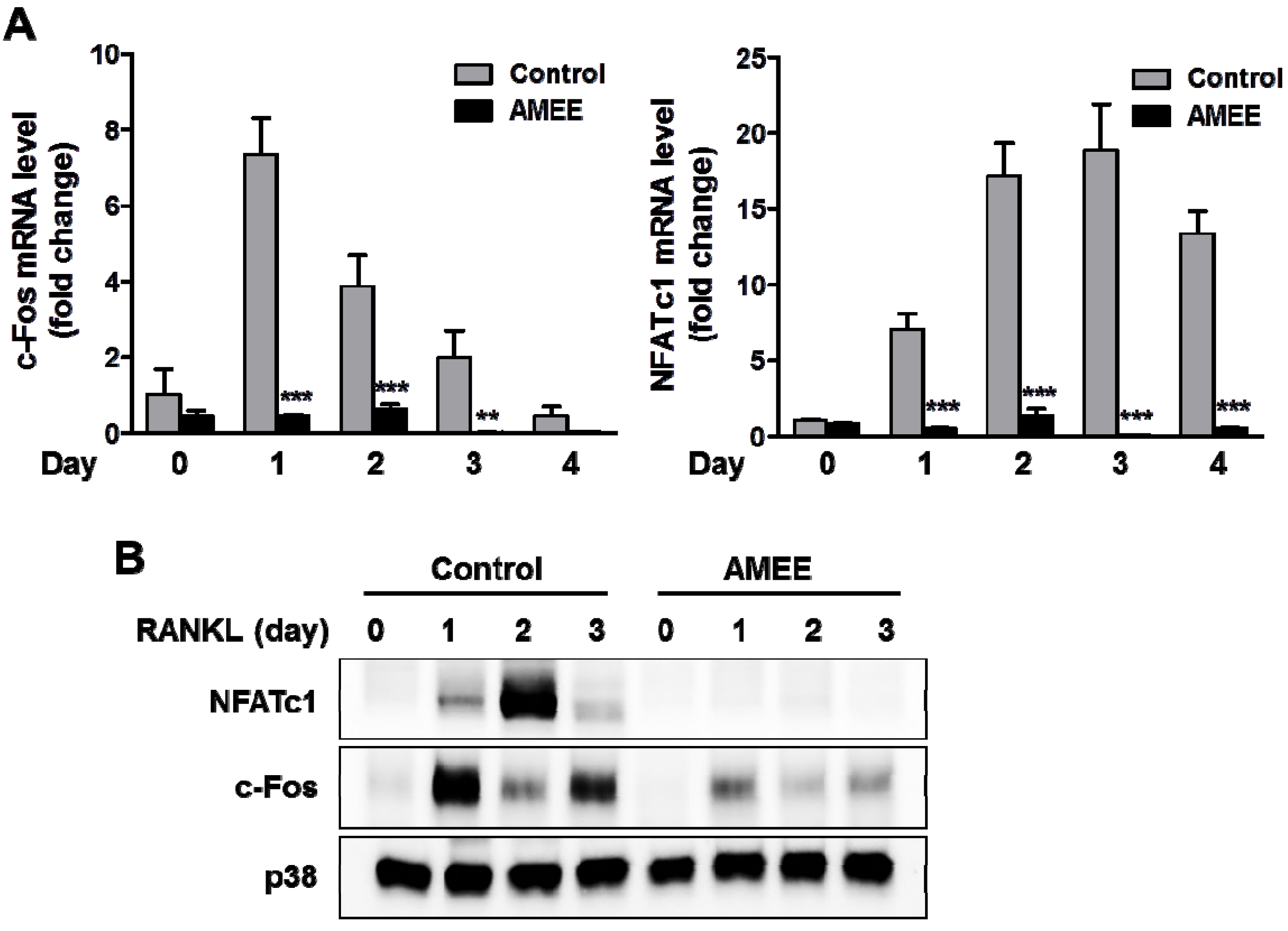
2.3. AMEE Inhibits RANKL-Induced c-Fos and NFATc1 Expression in Osteoclast Precursors
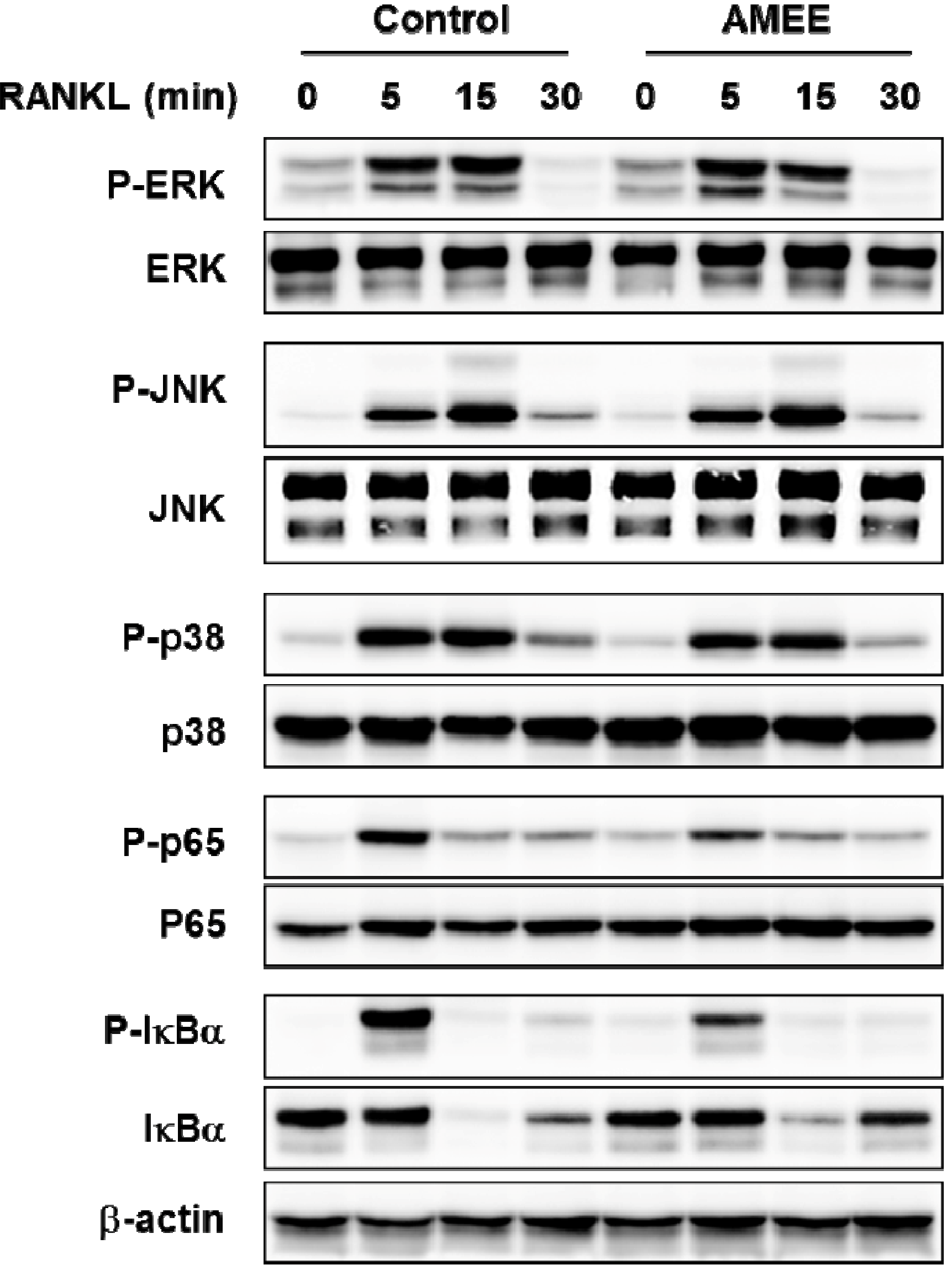
2.4. AMEE Attenuates RANKL-Induced NF-κB Activation in Osteoclast Precursors
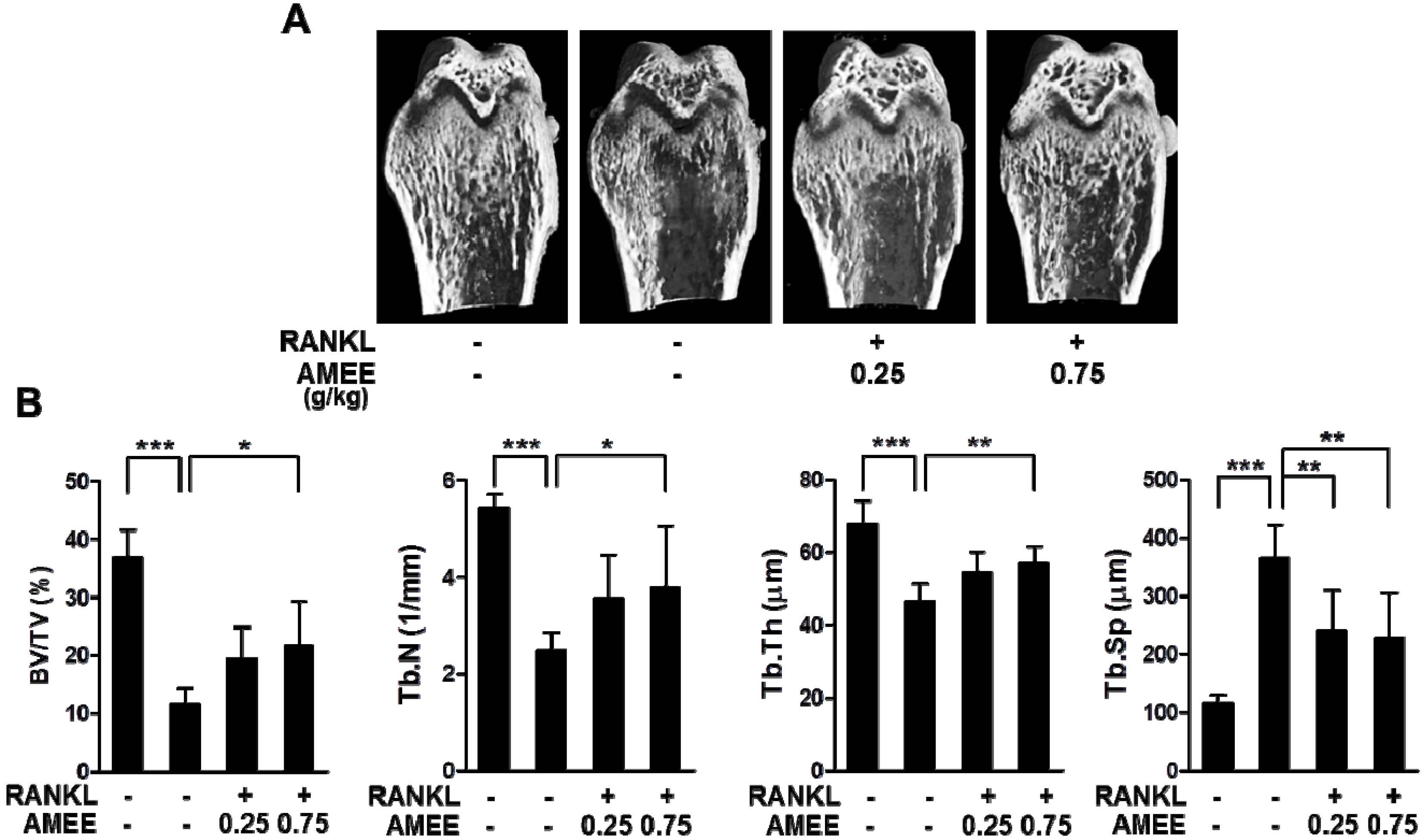
2.5. AMEE Attenuates RANKL-Induced Bone Loss in Mice
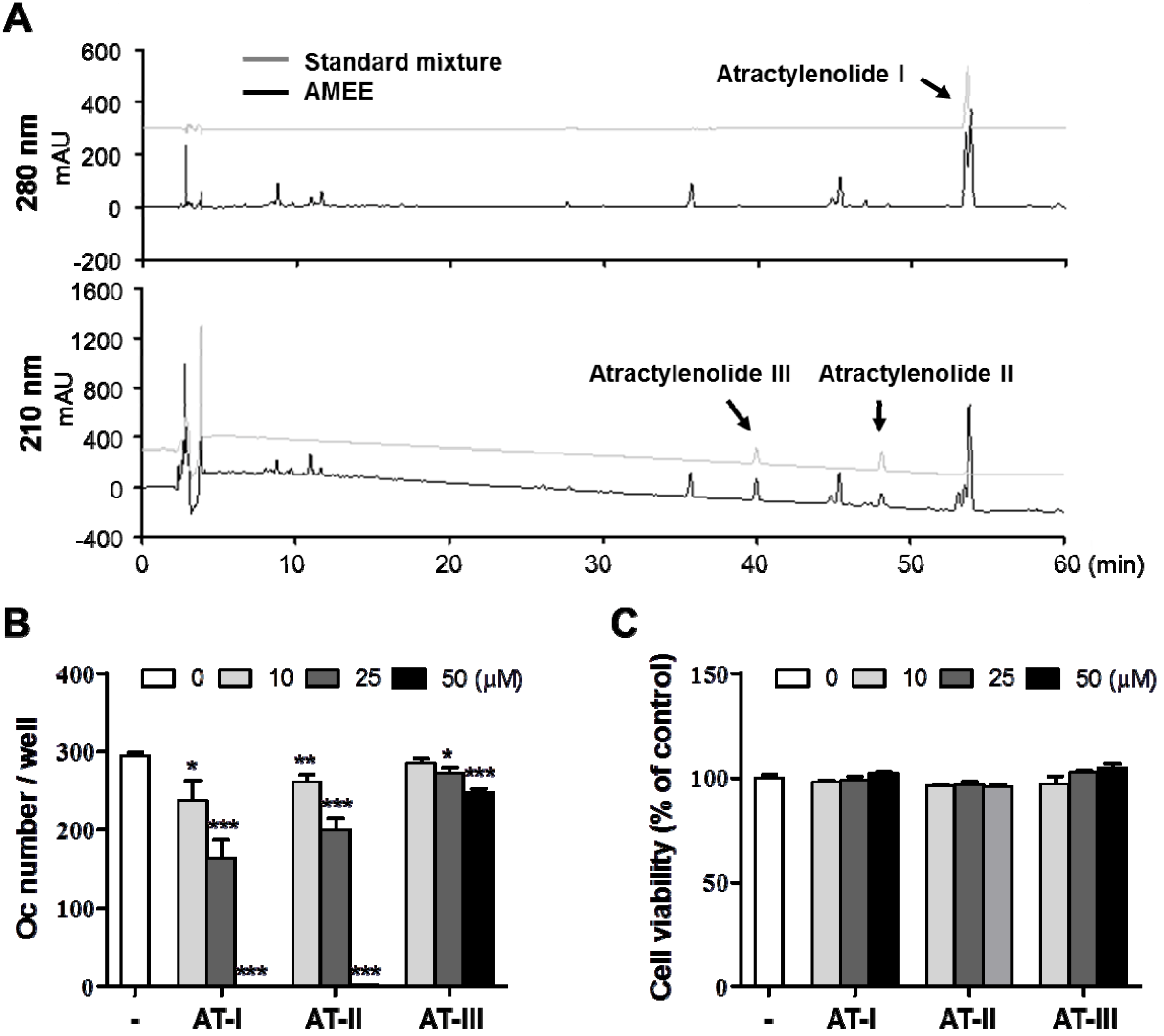
2.6. Atractylenolide I Inhibits RANKL-Induced Osteoclast Differentiation
3. Experimental
3.1. Reagents and Antibodies
3.2. Preparation of AMEE
3.3. Cell Culture
3.4. TRAP Activity and Staining
3.5. QPCR Analysis
3.6. Western Blot Analysis
3.7. Animal Experiments and Micro-CT Analysis
3.8. HPLC Analysis
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Banu, J.; Varela, E.; Fernandes, G. Alternative therapies for the prevention and treatment of osteoporosis. Nutr. Rev. 2012, 70, 22–40. [Google Scholar] [CrossRef]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342. [Google Scholar] [CrossRef]
- Lacey, D.L.; Timms, E.; Tan, H.L.; Kelley, M.J.; Dunstan, C.R.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93, 165–176. [Google Scholar] [CrossRef]
- Kong, Y.Y.; Yoshida, H.; Sarosi, I.; Tan, H.L.; Timms, E.; Capparelli, C.; Morony, S.; Oliveira-dos-Santos, A.J.; Van, G.; Itie, A.; et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 1999, 397, 315–323. [Google Scholar] [CrossRef]
- Gohda, J.; Akiyama, T.; Koga, T.; Takayanagi, H.; Tanaka, S.; Inoue, J. RANK-mediated amplification of TRAF6 signaling leads to NFATc1 induction during osteoclastogenesis. EMBO J. 2005, 24, 790–799. [Google Scholar] [CrossRef]
- Yamashita, T.; Yao, Z.; Li, F.; Zhang, Q.; Badell, I.R.; Schwarz, E.M.; Takeshita, S.; Wagner, E.F.; Noda, M.; Matsuo, K.; et al. NF-kappaB p50 and p52 regulate receptor activator of NF-kappaB ligand (RANKL) and tumor necrosis factor-induced osteoclast precursor differentiation by activating c-Fos and NFATc1. J. Biol. Chem. 2007, 282, 18245–18253. [Google Scholar] [CrossRef]
- Iotsova, V.; Caamano, J.; Loy, J.; Yang, Y.; Lewin, A.; Bravo, R. Osteopetrosis in mice lacking NF-kappaB1 and NF-kappaB2. Nat. Med. 1997, 3, 1285–1289. [Google Scholar] [CrossRef]
- Grigoriadis, A.E.; Wang, Z.Q.; Cecchini, M.G.; Hofstetter, W.; Felix, R.; Fleisch, H.A.; Wagner, E.F. c-Fos: A key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 1994, 266, 443–448. [Google Scholar]
- Takayanagi, H.; Kim, S.; Koga, T.; Nishina, H.; Isshiki, M.; Yoshida, H.; Saiura, A.; Isobe, M.; Yokochi, T.; Inoue, J.; et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 2002, 3, 889–901. [Google Scholar] [CrossRef]
- Putnam, S.E.; Scutt, A.M.; Bicknell, K.; Priestley, C.M.; Williamson, E.M. Natural products as alternative treatments for metabolic bone disorders and for maintenance of bone health. Phytother. Res. 2007, 21, 99–112. [Google Scholar] [CrossRef]
- Hou, J.; Jin, Y. The Healing Power of Chinese Herbs And Medicinal Recipes; The Haworth Integrative Healing Press: Binghamton, NY, USA, 2005. [Google Scholar]
- Kim, C.K.; Kim, M.; Oh, S.D.; Lee, S.M.; Sun, B.; Choi, G.S.; Kim, S.K.; Bae, H.; Kang, C.; Min, B.I. Effects of Atractylodes macrocephala Koidzumi rhizome on 3T3-L1 adipogenesis and an animal model of obesity. J. Ethnopharmacol. 2011, 137, 396–402. [Google Scholar] [CrossRef]
- Li, C.Q.; He, L.C.; Dong, H.Y.; Jin, J.Q. Screening for the anti-inflammatory activity of fractions and compounds from Atractylodes macrocephala koidz. J. Ethnopharmacol. 2007, 114, 212–217. [Google Scholar] [CrossRef]
- Li, X.; Lin, J.; Han, W.; Mai, W.; Wang, L.; Li, Q.; Lin, M.; Bai, M.; Zhang, L.; Chen, D. Antioxidant ability and mechanism of rhizoma Atractylodes macrocephala. Molecules 2012, 17, 13457–13472. [Google Scholar] [CrossRef]
- Gao, Q.; Ji, Z.H.; Yang, Y.; Cheng, R.; Yu, X.Y. Neuroprotective effect of Rhizoma Atractylodis macrocephalae against excitotoxicity-induced apoptosis in cultured cerebral cortical neurons. Phytother. Res. 2012, 26, 557–561. [Google Scholar] [CrossRef]
- Lee, J.C.; Lee, K.Y.; Son, Y.O.; Choi, K.C.; Kim, J.; Kim, S.H.; Chung, G.H.; Jang, Y.S. Stimulating effects on mouse splenocytes of glycoproteins from the herbal medicine Atractylodes macrocephala Koidz. Phytomedicine 2007, 14, 390–395. [Google Scholar] [CrossRef]
- Kim, S.H.; Jung, H.N.; Lee, K.Y.; Kim, J.; Lee, J.C.; Jang, Y.S. Suppression of Th2-type immune response-mediated allergic diarrhea following oral administration of traditional Korean medicine: Atractylodes macrocephala Koidz. Immunopharmacol. Immunotoxicol. 2005, 27, 331–343. [Google Scholar] [CrossRef]
- Matsuo, K.; Galson, D.L.; Zhao, C.; Peng, L.; Laplace, C.; Wang, K.Z.; Bachler, M.A.; Amano, H.; Aburatani, H.; Ishikawa, H.; et al. Nuclear factor of activated T-cells (NFAT) rescues osteoclastogenesis in precursors lacking c-Fos. J. Biol. Chem. 2004, 279, 26475–26480. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, Y.; Chang, E.J.; Kim, H.M.; Hong, S.P.; Lee, Z.H.; Ryu, J.; Kim, H.H. Suppression of osteoclastogenesis by N,N-dimethyl-d-erythro-sphingosine: A sphingosine kinase inhibition-independent action. Mol. Pharmacol. 2007, 72, 418–428. [Google Scholar] [CrossRef]
- Huang, H.; Chang, E.J.; Ryu, J.; Lee, Z.H.; Lee, Y.; Kim, H.H. Induction of c-Fos and NFATc1 during RANKL-stimulated osteoclast differentiation is mediated by the p38 signaling pathway. Biochem. Biophys. Res. Commun. 2006, 351, 99–105. [Google Scholar] [CrossRef]
- Ikeda, F.; Nishimura, R.; Matsubara, T.; Tanaka, S.; Inoue, J.; Reddy, S.V.; Hata, K.; Yamashita, K.; Hiraga, T.; Watanabe, T.; et al. Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation. J. Clin. Invest. 2004, 114, 475–484. [Google Scholar]
- Hayden, M.S.; Ghosh, S. NF-kappaB, the first quarter-century: Remarkable progress and outstanding questions. Genes. Dev. 2012, 26, 203–234. [Google Scholar] [CrossRef]
- Yang, F.; Tang, E.; Guan, K.; Wang, C.Y. IKK beta plays an essential role in the phosphorylation of RelA/p65 on serine 536 induced by lipopolysaccharide. J. Immunol. 2003, 170, 5630–5635. [Google Scholar]
- Doyle, S.L.; Jefferies, C.A.; O’Neill, L.A. Bruton’s tyrosine kinase is involved in p65-mediated transactivation and phosphorylation of p65 on serine 536 during NFkappaB activation by lipopolysaccharide. J. Biol. Chem. 2005, 280, 23496–23501. [Google Scholar] [CrossRef]
- Tomimori, Y.; Mori, K.; Koide, M.; Nakamichi, Y.; Ninomiya, T.; Udagawa, N.; Yasuda, H. Evaluation of pharmaceuticals with a novel 50-hour animal model of bone loss. J. Bone Miner. Res. 2009, 24, 1194–1205. [Google Scholar] [CrossRef]
- Dong, H.; He, L.; Huang, M.; Dong, Y. Anti-inflammatory components isolated from Atractylodes macrocephala Koidz. Nat. Prod. Res. 2008, 22, 1418–1427. [Google Scholar] [CrossRef]
- Shou, D.; Dai, S.; Zhang, J.; Li, H.; Yu, Z. Simultaneous determination of atractylenolide III, atractylenolide I and atractylon in Artactylodis macrocephala using microbore liquid chromatography. Se. Pu. 2008, 26, 637–639. [Google Scholar]
- Jiang, H.; Shi, J.; Li, Y. Screening for compounds with aromatase inhibiting activities from Atractylodes macrocephala Koidz. Molecules 2011, 16, 3146–3151. [Google Scholar] [CrossRef]
- Li, C.Q.; He, L.C.; Jin, J.Q. Atractylenolide I and atractylenolide III inhibit Lipopolysaccharide-induced TNF-alpha and NO production in macrophages. Phytother. Res. 2007, 21, 347–353. [Google Scholar] [CrossRef]
- Lim, H.; Lee, J.H.; Kim, J.; Kim, Y.S.; Kim, H.P. Effects of the rhizomes of Atractylodes japonica and atractylenolide I on allergic response and experimental atopic dermatitis. Arch. Pharm. Res. 2012, 35, 2007–2012. [Google Scholar] [CrossRef]
- Wang, C.C.; Chen, L.G.; Yang, L.L. Cytotoxic activity of sesquiterpenoids from Atractylodes ovata on leukemia cell lines. Planta. Med. 2002, 68, 204–208. [Google Scholar] [CrossRef]
- Wang, C.; Geng, Q.; Wang, Y. Protective effect of atractylenolide I on immunological liver injury. Zhongguo Zhong Yao Za Zhi 2012, 37, 1809–1813. [Google Scholar]
- Wang, K.T.; Chen, L.G.; Wu, C.H.; Chang, C.C.; Wang, C.C. Gastroprotective activity of atractylenolide III from Atractylodes ovata on ethanol-induced gastric ulcer in vitro and in vivo. J. Pharm. Pharmacol. 2010, 62, 381–388. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, H.N.; Yang, D.; Jung, K.; Kim, H.M.; Kim, H.H.; Ha, H.; Lee, Z.H. Trolox prevents osteoclastogenesis by suppressing RANKL expression and signaling. J. Biol. Chem. 2009, 284, 13725–13734. [Google Scholar] [CrossRef]
- Sample Availability: Sample of AMEE is available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ha, H.; An, H.; Shim, K.-S.; Kim, T.; Lee, K.J.; Hwang, Y.-H.; Ma, J.Y. Ethanol Extract of Atractylodes macrocephala Protects Bone Loss by Inhibiting Osteoclast Differentiation. Molecules 2013, 18, 7376-7388. https://doi.org/10.3390/molecules18077376
Ha H, An H, Shim K-S, Kim T, Lee KJ, Hwang Y-H, Ma JY. Ethanol Extract of Atractylodes macrocephala Protects Bone Loss by Inhibiting Osteoclast Differentiation. Molecules. 2013; 18(7):7376-7388. https://doi.org/10.3390/molecules18077376
Chicago/Turabian StyleHa, Hyunil, Hyosun An, Ki-Shuk Shim, Taesoo Kim, Kwang Jin Lee, Youn-Hwan Hwang, and Jin Yeul Ma. 2013. "Ethanol Extract of Atractylodes macrocephala Protects Bone Loss by Inhibiting Osteoclast Differentiation" Molecules 18, no. 7: 7376-7388. https://doi.org/10.3390/molecules18077376
APA StyleHa, H., An, H., Shim, K.-S., Kim, T., Lee, K. J., Hwang, Y.-H., & Ma, J. Y. (2013). Ethanol Extract of Atractylodes macrocephala Protects Bone Loss by Inhibiting Osteoclast Differentiation. Molecules, 18(7), 7376-7388. https://doi.org/10.3390/molecules18077376



