Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
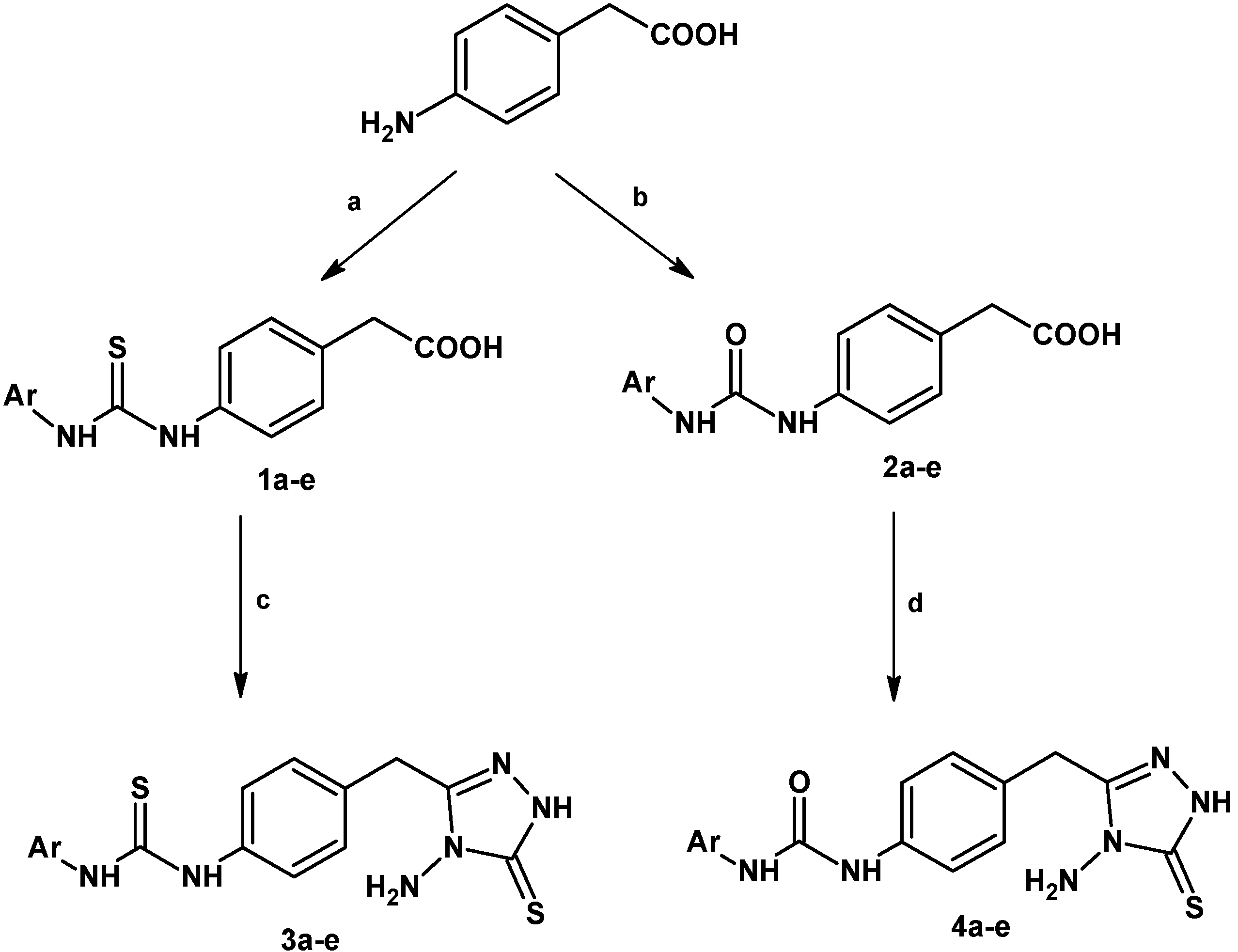
| Compd. | Ar | M.P. (°C) | M.F. | M.W. | Yield (%) |
|---|---|---|---|---|---|
| 1a | 2,4,6-Cl-C6H2 | 214–215 | C15H11Cl3N2O2S | 389.68 | 50.5 |
| 1b | 2,6-Cl2-C6H4 | 206–207 | C15H13ClN2O2S | 320.79 | 48.3 |
| 1c | 4-CH3S-C6H4 | 193–194 | C16H16N2O3S | 316.37 | 61.1 |
| 1d | 4-CF3-C6H5 | 240–241 | C15H13N3O4S | 331.34 | 45.5 |
| 1e | 4-NO2-C6H5 | 200–201 | C15H13FN2O2S | 304.34 | 67.2 |
| 2a | 2,4,6-Cl-C6H2 | 276–277 | C15H11Cl3N2O3 | 373.62 | 77.3 |
| 2b | 2,6-Cl2-C6H4 | 258–259 | C15H13ClN2O3 | 304.73 | 78.0 |
| 2c | 4-CH3S-C6H4 | 234–235 | C16H16N2O4 | 300.31 | 83.5 |
| 2d | 4-CF3-C6H5 | 250–251 | C15H13N3O5 | 315.28 | 67.4 |
| 2e | 4-NO2-C6H5 | 245–246 | C15H13FN2O3 | 288.27 | 80.9 |
| 3a | 2,4,6-Cl-C6H2 | 227–228 | C16H13Cl3N6S2 | 459.80 | 46.4 |
| 3b | 2,6-Cl2-C6H4 | 206–207 | C16H15ClN6S2 | 390.85 | 50.8 |
| 3c | 4-CH3S-C6H4 | 228–230 | C17H18N6OS2 | 386.56 | 59.3 |
| 3d | 4-CF3-C6H5 | 240–241 | C15H17N7O2S2 | 401.47 | 55.4 |
| 3e | 4-NO2-C6H5 | 234–236 | C16H15FN6S2 | 374.46 | 52.2 |
| 4a | 2,4,6-Cl-C6H2 | 237–238 | C16H13Cl3N6OS | 443.74 | 58.0 |
| 4b | 2,6-Cl2-C6H4 | 258–259 | C16H15ClN6OS | 374.85 | 55.9 |
| 4c | 4-CH3S-C6H4 | 180–182 | C17H19N6O2S | 371.43 | 51.8 |
| 4d | 4-CF3-C6H5 | 214–216 | C16H15N7O3S | 385.40 | 50.2 |
| 4e | 4-NO2-C6H5 | 240–241 | C16H15FN6OS | 358.39 | 48.8 |
2.2. Biological Activity

| Compounds | LD50 (95% CI) * | LD90 (95% CI) * | Chi square | DF |
|---|---|---|---|---|
| 1c | 118.8 (105.3–135.2) | 216.4 (182.4–280.4) | 61.7 | 38 |
| 3d | 67.4 (59.0–77.0) | 139.4 (116.4–180.6) | 75.0 | 38 |
| 3e | 165.6 (141.7–205.2) | 370.95 (278.5–619.3) | 40.3 | 38 |
3. Experimental
3.1. General
3.1.1. General Procedure for the Preparation of 2a–e
3.1.2. General Procedure for the Preparation of 3a–e and 4a–e
3.2. Biological Activity
3.2.1. Antifungal Activity
3.2.2. Mosquito Larval Bioassay
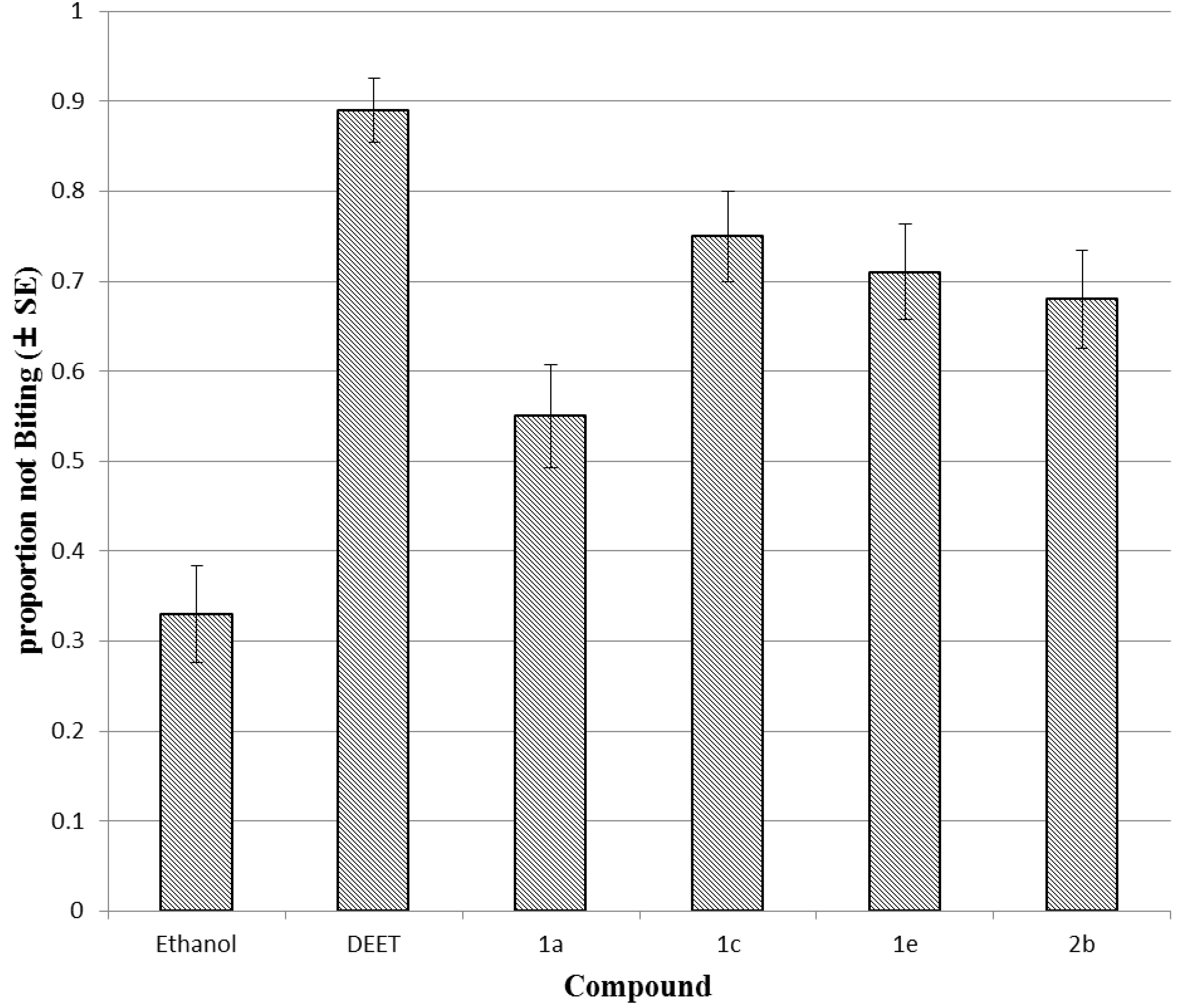
3.2.3. Mosquito Biting Bioassays
3.2.4. Cytotoxicity Activity
3.2.5. Anti-Inflammatory Activity
4. Conclusions
Acknowledgments
References
- Vega-Pérez, J.M.; Periñán, I.; Argandoña, M.; Vega-Holm, M.; Palo-Nieto, C.; Burgos-Morón, E.; López-Lázaro, M.; Vargas, C.; Nieto, J.J.; Iglesias-Guerra, F. Isoprenyl-thiourea and urea derivatives as new farnesyl diphosphate analogues: Synthesis and in vitro antimicrobial and cytotoxic activities. Eur. J. Med. Chem. 2012, 58, 591–612. [Google Scholar] [CrossRef]
- Yao, J.; Chen, J.; He, Z.; Sun, W.; Xu, W. Design, synthesis and biological activities of thiourea containing sorafenib analogs as antitumor agents. Bioorg. Med. Chem. 2012, 20, 2923–2929. [Google Scholar] [CrossRef]
- Shantharam, C.S.; Suyoga, V.D.M.; Suhas, R.; Sridhara, M.B.; Channe, G.D. Inhibition of protein glycation by urea and thiourea derivatives of glycine/proline conjugated benzisoxazole analogue—Synthesis and structure-activity studies. Eur. J. Med. Chem. 2013, 60, 325–332. [Google Scholar] [CrossRef]
- Yang, W.; Hu, Y.; Yang, Y.S.; Zhang, F.; Zhang, Y.B.; Wang, X.L.; Tang, J.F.; Zhong, W.Q.; Zhu, H.L. Design, modification and 3D QSAR studies of novel naphthalin-containing pyrazoline derivatives with/without thiourea skeleton as anticancer agents. Bioorg. Med. Chem. 2013, 21, 1050–1063. [Google Scholar] [CrossRef]
- Keche, A.P.; Hatnapure, G.D.; Tale, R.H.; Rodge, A.H.; Kamble, V.M. Synthesis, anti-inflammatory and antimicrobial evaluation of novel 1-acetyl-3,5-diaryl-4,5-dihydro (1H) pyrazole derivatives bearing urea, thiourea and sulfonamide moieties. Bioorg. Med. Chem. 2012, 22, 6611–6615. [Google Scholar] [CrossRef]
- Burgeson, J.R.; Moore, A.L.; Boutilier, J.K.; Cerruti, N.R.; Gharaibeh, D.N.; Lovejoy, C.E.; Amberg, S.M.; Hruby, D.E.; Tyavanagimatt, S.R.; Allen, R.D.; et al. Analysis of a series of acylthiourea derivatives possessing broad-spectrum antiviral. Bioorg. Med. Chem Lett. 2012, 22, 4263–4272. [Google Scholar] [CrossRef]
- Upadhayaya, R.S.; Kulkarni, G.M.; Vasireddy, N.R.; Vandavasi, J.K.; Dixit, S.S.; Sharma, V. Chattapadhayaya, Design, synthesis and biological evaluation of novel triazole, urea and thiourea derivatives of quinoline against Mycobacterium tuberculosis. Bioorg. Med. Chem. 2009, 17, 4681–4692. [Google Scholar] [CrossRef]
- Khan, S.A.; Singh, N.; Saleem, K. Synthesis, characterization and in vitro antibacterial activity of thiourea and urea derivatives of stereoids. Eur. J. Med. Chem. 2008, 43, 2272–2277. [Google Scholar] [CrossRef]
- Sett, P.P.; Ranken, R.; Robinson, D.E.; Osgood, S.A.; Risen, L.M.; Rodgers, E.L.; Migawa, M.T.; Jefferson, E.A.; Swayze, E.E. Aryl urea analogs with broad-spectrum antibacterial activity. Bioorg. Med. Chem. Lett. 2004, 14, 5569–5572. [Google Scholar] [CrossRef]
- Koçyiğit-Kaymakçıoğlu, B.; Rollas, S.; Körceğez, E.; Arıcıoğlu, F. Synthesis and bological evaluation of new N-substituted-N′-(3,5-di/1,3,5-trimethylpyrazole-4-yl)thiourea/urea derivatives. Eur. J. Pharm. Sci. 2005, 26, 97–103. [Google Scholar] [CrossRef]
- Holla, B.S.; Veerenda, B.; Shivananda, K.; Boja, P. Synthesis characterization and anticancer activity studies on some Mannich bases derived from 1,2,4-triazoles. Eur. J.Med. Chem. 2003, 38, 759–767. [Google Scholar] [CrossRef]
- Abdel-Megeed, A.M.; Abdel-Rahman, H.M.; Alkaramany, G.E.; El-Gendy, M.A. Design, synthesis and molecular modeling study of acylated 1,2,4-triazole-3-acetates with potential anti-inflammatory activity. Eur. J. Med. Chem. 2009, 44, 117–123. [Google Scholar] [CrossRef]
- Kaplancikli, Z.A.; Turan-Zitouni, G.; Ozdemir, A.; Revial, G. New triazole and triazolothiadiazine derivatives as possible antimicrobial agents. Eur. J. Med. Chem. 2008, 43, 155–159. [Google Scholar] [CrossRef]
- Koçyiğit-Kaymakçıoğlu, B.; Çalışır, M.M.; Özbek, B.; Ötük, G. Synthesis and antimicrobial activities of schiff bases derived from 4-amino-5-(1-phenylethyl)-2,4-dihydro-3H-1,2,4-triazole-3-thione. E. J. Chem. 2010, 7, 458–464. [Google Scholar] [CrossRef]
- Ashok, M.; Holla, B.S.; Boja, P. Convenient one pot synthesis and antimicrobial evaluation of some new Mannich bases carrying 4-methylthiobenzyl moiety. Eur. J. Med. Chem. 2007, 42, 1095–1101. [Google Scholar] [CrossRef]
- Sabatelli, F.; Patel, R.; Mann, P.A.; Mendrick, C.A.; Norris, C.C.; Hare, R.; Loebenberg, D.; Black, T.A.; McNicholas, P.M. In vitro activities of posaconazole, fluconazole, itraconazole, voriconazole, and amphotericin B against a large collection of clinically important molds and yeats. Antimicrob. Agents Chemother. 2006, 50, 2009–2015. [Google Scholar] [CrossRef]
- Johnson, E.; Espinel-Ingroff, A.; Szekely, A.; Hockey, H.; Troke, P. Activity of voriconazole, itraconazole, fluconazole and amphotericin B in vitro against 1763 yeasts from 472 patients in the voriconazole phase III clinical studies. Int. J. Antimic. Agents 2008, 32, 511–514. [Google Scholar] [CrossRef]
- Rodriguez-Fernandez, E.; Manzano, J.L.; Benito, J.J.; Hermosa, R.; Monte, E.; Criado, J.J. Thiourea, trizole and thiadiazine compounds and their metal complexes as antifungal agents. J. Inorg. Biochem. 2005, 99, 1558–1572. [Google Scholar] [CrossRef]
- Rauf, M.K.; Ebihara, M.; Imtiaz-ud-Din, B.A. 1-Benzoyl-3-(2,4,5-trichloro-phenyl)thiourea. Acta Cryst. 2012. [Google Scholar] [CrossRef]
- Cao, X.; Li, F.; Hu, M.; Li, M.; Yu, G.A.; Liu, S.H. Chiral γ-Aryl-1H-1,2,4-triazole derivatives as highly potential antifungal agents: Design, synthesis, structure, and in vitro fungicidal activities. J. Agric. Food Chem. 2008, 56, 11367–11375. [Google Scholar] [CrossRef]
- Cao, X.; Li, F.; Hu, M.; Zhang, J.; Li, F.; Yang, Y.; Liu, D.; Liu, S.H. Determination of stereoselective ınteraction between enantiomers of chiral γ-Aryl-1H-1,2,4-triazole derivatives and Penicillium digitatum. J. Agric. Food Chem. 2009, 57, 6914–6919. [Google Scholar] [CrossRef]
- Crank, G.; Neville, M.; Ryden, R. Thiourea derivatives of 2-aminooxazoles showing antibacterial and antifungal activity. J. Med. Chem. 1973, 16, 1402–1405. [Google Scholar] [CrossRef]
- Celen, A.Ö.; Koçyiğit-Kaymakçıoğlu, B.; Gümrü, S.; Toklu, H.Z.; Arıcıoğlu, F. Synthesis and anticonvulsant activity of substituted thiourea derivatives. Marmara Pharm. J. 2011, 15, 43–47. [Google Scholar]
- Wedge, D.E.; Smith, B.J.; Quebedeaux, J.P.; Constantin, R.J. Fungicide management strategies for control of strawberry fruit rot diseases in Louisiana and Mississippi. Crop Protect. 2007, 26, 1449–1458. [Google Scholar] [CrossRef]
- Denny, W.A.; Cain, B.F.; Atwell, G.J.; Hansch, C.; Augustine Leo, P.A. Potential antitumor agents. 36. Quantitative relationships between experimental antitumor activity, toxicity, and structure for the general class of 9-anilinoacridine antitumor agents. J. Med. Chem. 1982, 25, 276–315. [Google Scholar] [CrossRef]
- Wedge, D.E.; Kuhajek, J. Microbioassay for fungicide discovery. SAAS Bull. Biochem. Biotechnol. 1998, 11, 1–7. [Google Scholar]
- Sobolev, V.S.; Khan, S.I.; Tabanca, N.; Wedge, D.E.; Mainly, S.P.; Cutler, S.J.; Coy, M.R.; Neff, S.A.; Gloer, J.B. Biological activity of peanut (Arachis hypogaea) phytoalexins and selected natural and synthetic stilbenoids. J. Agric. Food Chem. 2011, 59, 1673–1682. [Google Scholar] [CrossRef]
- SAS Online Document Version 9.2; SAS Institute: Cary, NC, USA, 2007.
- Steel, R.G.D.; Torrie, J.H. Principles and Procedures of Statistics: A Biometrical Approach, 2nd ed.; McGraw Hill Book Company: New York, NY, USA, 1980. [Google Scholar]
- Pridgeon, J.W.; Meepagala, K.M.; Becnel, J.J.; Clark, G.G.; Pereira, R.M.; Linthicum, K.J. Structure-activity relationships of 33 piperidines as toxicants against female adults of Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2007, 44, 263–269. [Google Scholar] [CrossRef]
- Pridgeon, J.W.; Becnel, J.J.; Clark, G.G.; Linthicum, K.J. A high-throughput screening method to identify potential pesticides for mosquito control. J. Med. Entomol. 2009, 46, 335–341. [Google Scholar] [CrossRef]
- Klun, J.A.; Kramer, M.; Debboun, M. A new in vitro bioassay system for discovery of novel human-use mosquito repellents. J. Am. Mosq. Control Assoc. 2005, 21, 64–70. [Google Scholar] [CrossRef]
- Dethier, V.G.; Browne, B.L.; Smith, C.N. The Designation of chemicals in terms of the responses they elicit from insects. J. Econ. Entomol. 1960, 53, 134–136. [Google Scholar]
- Ali, A.; Cantrell, C.L.; Bernier, U.R.; Duke, S.O.; Schneider, J.C.; Khan, S.I. Aedes aegypti (Diptera: Culicidae) Biting deterrence: Structure-activity relationship of saturated and unsaturated fatty acids. J. Med. Entomol. 2012. accepted. [Google Scholar]
- Tabanca, N.; Bedir, E.; Kırımer, N.; Başer, K.H.C.; Khan, S.I.; Jacob, M.R.; Khan, I.A. Antimicrobial compounds from Pimpinella species growing in Turkey. Planta Med. 2003, 69, 933–938. [Google Scholar] [CrossRef]
- Tabanca, N.; Ma, G.; Pasco, D.S.; Bedir, E.; Kırımer, N.; Başer, K.H.C.; Khan, I.A.; Khan, S.I. Effect of essential oils and ısolated compounds from Pimpinella species on NF-kB: A target for anti-inflammatory action. Phytotherap. Res. 2007, 21, 741–745. [Google Scholar] [CrossRef]
- Koçyiğit-Kaymakçıoğlu, B.; Oruç-Emre, E.E.; Ünsalan, S.; Tabanca, N.; Wedge, D.E.; Khan, S.I.; Demirci, F.; Rollas, S. Synthesis and biological activities of hydrazide hydrazones and their corresponding 3-acetyl-2,5-disubstituted-1,3,4-oxadiazolines. Med. Chem. Res. 2012. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kocyigit-Kaymakcioglu, B.; Celen, A.O.; Tabanca, N.; Ali, A.; Khan, S.I.; Khan, I.A.; Wedge, D.E. Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties. Molecules 2013, 18, 3562-3576. https://doi.org/10.3390/molecules18033562
Kocyigit-Kaymakcioglu B, Celen AO, Tabanca N, Ali A, Khan SI, Khan IA, Wedge DE. Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties. Molecules. 2013; 18(3):3562-3576. https://doi.org/10.3390/molecules18033562
Chicago/Turabian StyleKocyigit-Kaymakcioglu, Bedia, Ahmet Ozgur Celen, Nurhayat Tabanca, Abbas Ali, Shabana I. Khan, Ikhlas A. Khan, and David E. Wedge. 2013. "Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties" Molecules 18, no. 3: 3562-3576. https://doi.org/10.3390/molecules18033562
APA StyleKocyigit-Kaymakcioglu, B., Celen, A. O., Tabanca, N., Ali, A., Khan, S. I., Khan, I. A., & Wedge, D. E. (2013). Synthesis and Biological Activity of Substituted Urea and Thiourea Derivatives Containing 1,2,4-Triazole Moieties. Molecules, 18(3), 3562-3576. https://doi.org/10.3390/molecules18033562




