Design, Synthesis, Biological Activities and 3D-QSAR of New N,N'-Diacylhydrazines Containing 2,4-Dichlorophenoxy Moieties
Abstract
:1. Introduction
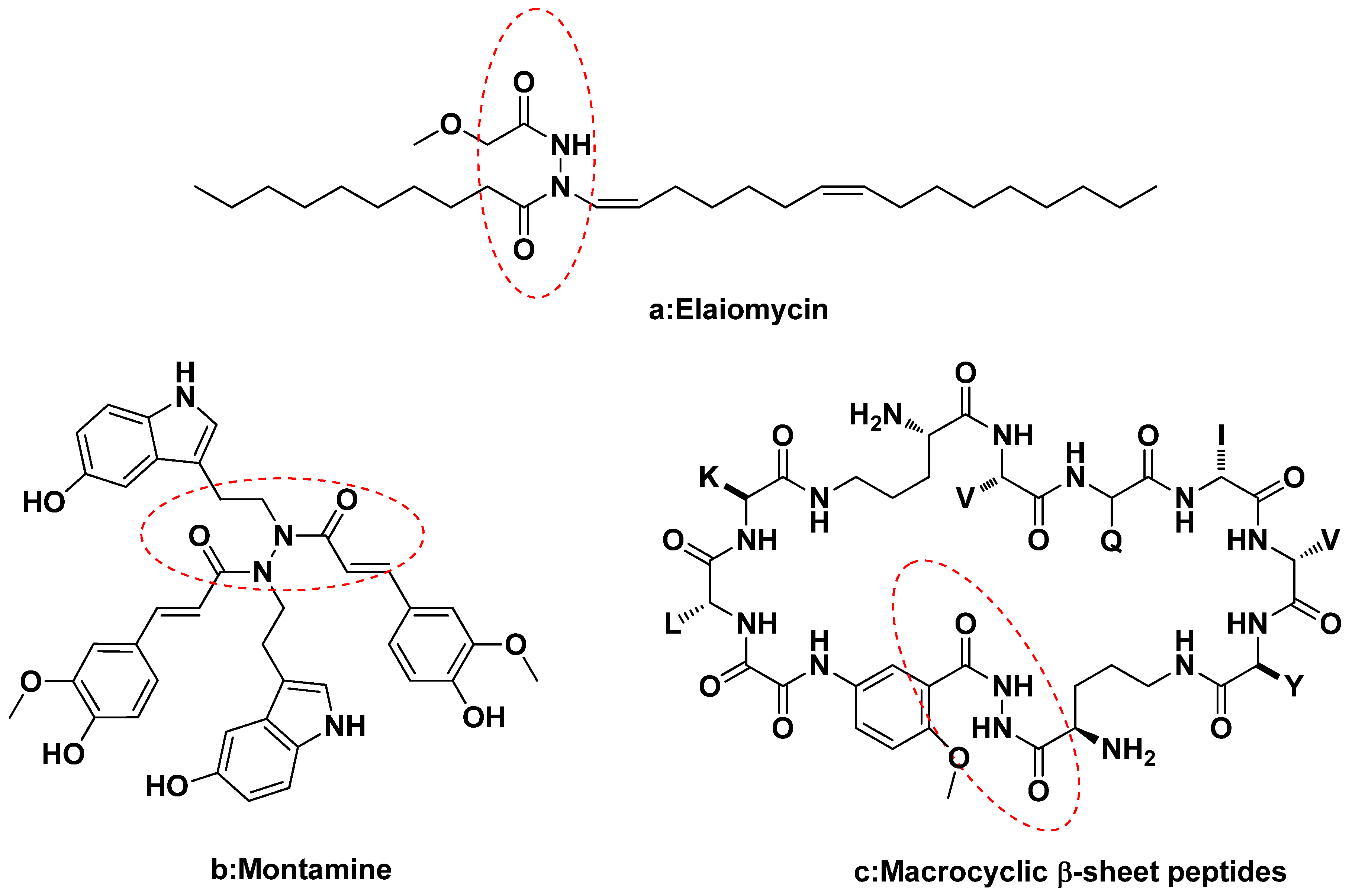
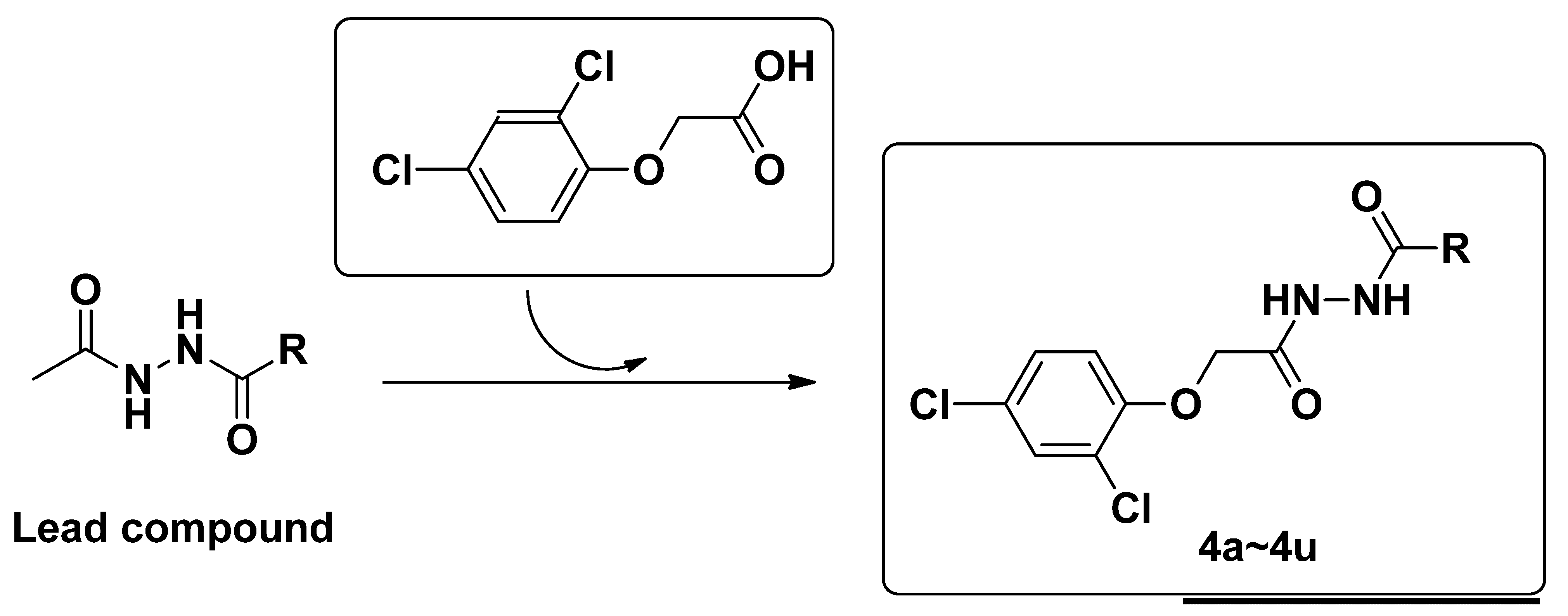
2. Results and Discussion
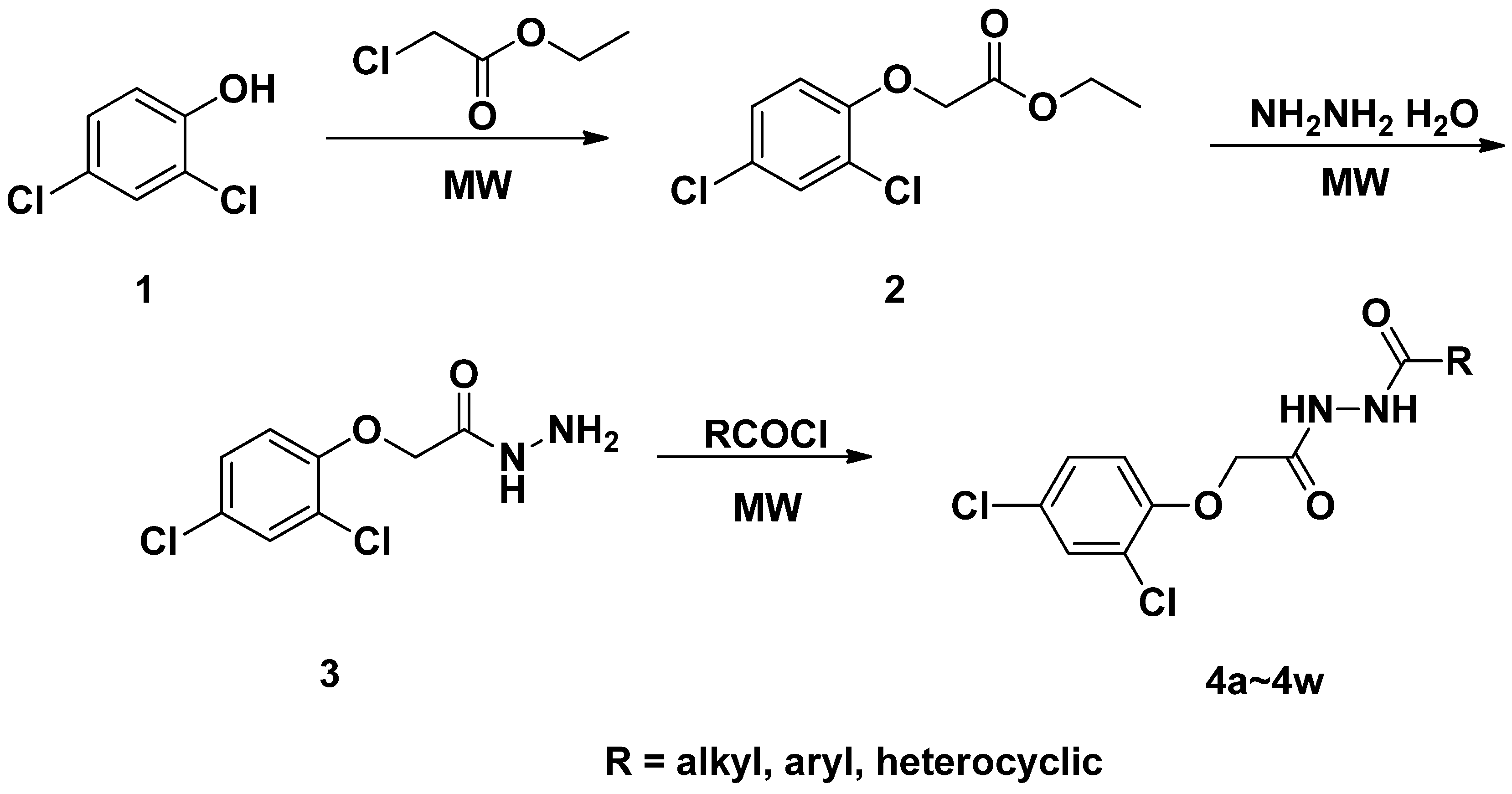
2.1. Synthesis and Characterization
| No. | Method | Time | condition | Yield/% |
|---|---|---|---|---|
| 2 | No-MW | 24 h | r.t. | 88 |
| MW | 4 min | 200 W | 95 | |
| 3 | No-MW | 5 h | reflux | 90 |
| MW | 1 min | 500 W | 96 |
2.2. Herbicidal Activities
| No. | R | CRC | Ech. | Bra. | Dig. | Ama. | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre- | Post- | Pre- | Post- | Pre- | Post- | Pre- | Post- | |||
| 4a | cyclopropyl | −100 | 6.2 | 15.0 | 92.9 | 100 | 72.5 | 30.0 | 100 | 100 |
| 4b | phenyl | −100 | 5.4 | 26.3 | 86.9 | 100 | 52.9 | 20.0 | 100 | 100 |
| 4c | p-nitrophenyl | −100 | 0 | 10.0 | 71.7 | 100 | 21.6 | 0 | 100 | 100 |
| 4d | p-chlorophenyl | −100 | 19.5 | 18.9 | 11.2 | 100 | 53.7 | 10.7 | 91.6 | 100 |
| 4e | p-fluorophenyl | −100 | 0 | 28.5 | 12.4 | 100 | 100 | 0 | 95.8 | 100 |
| 4f | m-methylphenyl | −100 | 12.1 | 27.3 | 32.0 | 100 | 11.1 | 7.1 | 91.6 | 100 |
| 4g | m-chlorophenyl | −100 | 0 | 6.5 | 33.7 | 100 | 16.7 | 21.4 | 87.4 | 100 |
| 4h | o-fluorophenyl | −100 | 11.6 | 19.4 | 100 | 100 | 37.0 | 21.5 | 89.5 | 100 |
| 4i | o-chlorophenyl | −100 | 9.1 | 22.3 | 64.5 | 100 | 64.8 | 7.1 | 91.6 | 100 |
| 4j | 2,4-dichlorophenyl | −100 | 21.0 | 0 | 56.8 | 100 | 50.0 | 0 | 78.9 | 100 |
| 4k | o-methoxyphenyl | −100 | 0 | 21.7 | 49.1 | 100 | 22.2 | 3.6 | 81.1 | 100 |
| 4l | p-methoxyphenyl | −100 | 16.6 | 20.0 | 62.1 | 100 | 48.1 | 21.4 | 100 | 100 |
| 4m | p-iodophenyl | −100 | 23.5 | 0 | 52.1 | 100 | 3.7 | 3.6 | 70.5 | 100 |
| 4n | 5-methylisoxazole-4-yl | −100 | 8.1 | 9.1 | 100 | 100 | 38.9 | 0 | 78.9 | 100 |
| 4o | 1-CN-cyclopropyl | −100 | 20.5 | 14.9 | 14.8 | 100 | 0 | 17.9 | 49.5 | 100 |
| 4p | propyl | −100 | 100 | 27.9 | 100 | 100 | 87.0 | 28.6 | 100 | 100 |
| 4q | isopropyl | −100 | 100 | 20.0 | 100 | 100 | 70.4 | 21.4 | 83.2 | 100 |
| 4r | 2,4-dichlorophenoxymethyl | −63.5 | 100 | 7.6 | 18.3 | 58.6 | 25.9 | 0 | 78.9 | 100 |
| 4s | (2-(2,4-dichlorophenoxy)-acetyl)propyl | −100 | 5.6 | 0 | 56.2 | 100 | 87.0 | 3.6 | 87.4 | 100 |
| 4t | furan | −100 | 0 | 7.6 | 100 | 100 | 63.0 | 17.9 | 85.3 | 100 |
| 4u | (2 E,4Z)-hexa-2,4-diene- | −100 | 100 | 31.8 | 100 | 100 | 31.5 | 0 | 93.7 | 100 |
| 4v | 3-pyridine | −100 | 100 | 20.0 | 100 | 100 | 25.9 | 0 | 100 | 100 |
| 4w | 4-pyridine | −100 | 100 | 17.2 | 100 | 100 | 66.7 | 10.7 | 83.2 | 100 |
| 4x | Methyl | −100 | 68.2 | 26.2 | 88.8 | 100 | 87.0 | 0 | 70.5 | 100 |
| 2,4-D | 65.7 | 100 | 85.4 | 100 | 100 | 100 | 100 | 81.1 | 100 | |
| No. | Rate g/ha | Ech. | Bra. | Dig. | Ama. | ||||
|---|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre | Post | Pre | Post | ||
| 4a | 187.5 | 0 | 37.7 | 55.7 | 100 | 0 | 8.3 | 100 | 100 |
| 375 | 0 | 50.4 | 53.3 | 100 | 0 | 12.7 | 100 | 100 | |
| 750 | 0 | 61.2 | 59.1 | 100 | 89.0 | 31.5 | 100 | 100 | |
| 2,4-D | 187.5 | 0 | 0 | 0 | 100 | 0 | 0 | 0 | 100 |
| 375 | 0 | 0 | 0 | 100 | 0 | 0 | 52.4 | 100 | |
| 750 | 32.6 | 32.4 | 0 | 100 | 50.0 | 0 | 78.6 | 100 | |
2.3. Plant Growth Regulatory Activity
2.4. CoMFA Analysis
| method | q2 | r2 | S | F | No. | Contributor (%) | |
|---|---|---|---|---|---|---|---|
| Steric | Electrostatic | ||||||
| CoMFA | 0.57 | 0.886 | 0.435 | 97.628 | 4f | 66.9% | 33.1% |

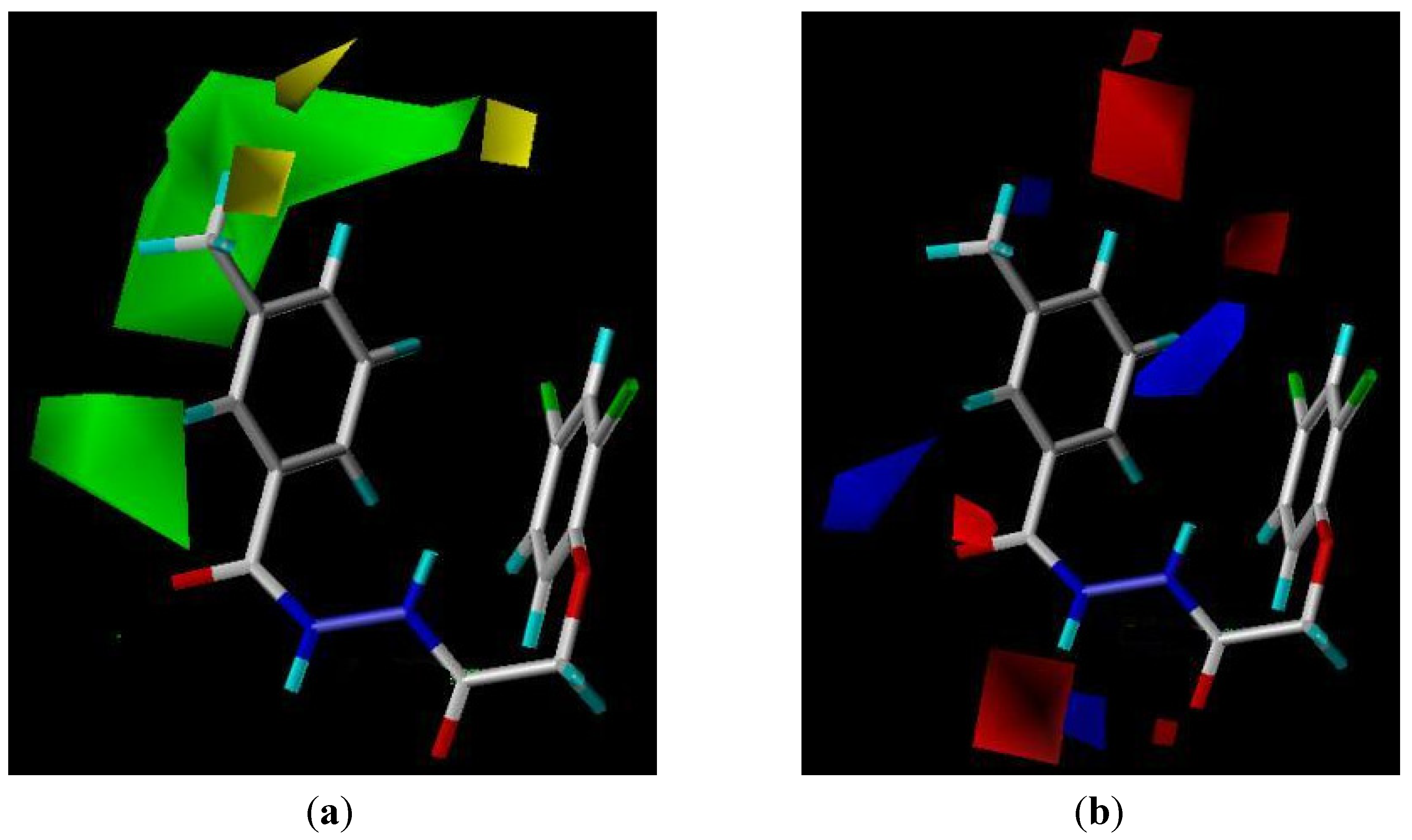
3. Experimental
3.1. Instruments
3.2. General Procedure
3.3. 3D-QSAR Analysis
| No. | R | pIC50 | pIC50' | Residue |
|---|---|---|---|---|
| 4a | cyclopropyl | −2.06064 | −2.1257 | 0.06506 |
| 4b | phenyl | −2.47998 | −2.5328 | 0.05282 |
| 4c | p-nitrophenyl | −3.14439 | −3.0982 | −0.04619 |
| 4d | p-chlorophenyl | −2.50804 | −2.5895 | 0.08146 |
| 4e | p-fluorophenyl | −0.55723 | −0.6172 | 0.05997 |
| 4f * | m-methylphenyl | −3.4516 | −3.5621 | 0.1105 |
| 4g | m-chlorophenyl | −3.27036 | −3.5229 | 0.25254 |
| 4h | o-fluorophenyl | −2.784 | −2.8013 | 0.0173 |
| 4i | o-chlorophenyl | −2.3074 | −2.4085 | 0.1011 |
| 4j # | 2,4-dichlorophenyl | −2.61072 | −2.8691 | 0.25838 |
| 4k | o-methoxyphenyl | −3.11189 | −3.0097 | −0.10219 |
| 4l | p-methoxyphenyl | −2.60028 | −2.4112 | −0.18908 |
| 4m | p-iodophenyl | −4.08294 | −4.1954 | 0.11246 |
| 4n | 5-methylisoxazole-4-yl | −2.73284 | −2.6376 | −0.09524 |
| 4o | 1-CN-cyclopropyl | −4.51171 | −4.2652 | −0.24651 |
| 4p # | propyl | −1.65895 | −1.6622 | 0.00325 |
| 4q | isopropyl | −2.10825 | −2.3056 | 0.19735 |
| 4r | 2,4-dichlorophenoxymethyl | −3.09808 | −3.2077 | 0.10962 |
| 4s | (2-(2,4-dichlorophenoxy)acetyl)propyl | −1.82968 | −1.9661 | 0.13642 |
| 4t | furan | −2.28624 | −2.1627 | −0.12354 |
| 4u | (2E,4Z)- hexa-2,4-diene- | −2.85481 | −2.6382 | −0.21661 |
| 4v # | 3-pyridine | −2.9882 | −2.7025 | −0.2857 |
| 4w | 4-pyridine | −2.23 | −2.3669 | 0.1369 |

3.4. Herbicidal Activities Assay
3.5. Plant Growth Regulatory Activity Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Kadhum, A.A.H.; Al-Amiery, A.A.; Musa, A.Y.; Mohamad, A.B. The antioxidant activity of new coumarin derivatives. Int. J. Mol. Sci. 2011, 12, 5747–5761. [Google Scholar] [CrossRef]
- Wang, H.; Fu, Y.F.; Fan, Z.J.; Song, H.B.; Wu, Q.J.; Zhang, Y.J.; Belskaya, N.P.; Bakulev, V.A. Synthesis, crystal structure and biological activity of N-tert-butyl-N-(4-methyl-1,2,3-thiadiazole)-5-yl-N'-(4-methyl-1,2,3-thiadiazole)-5-formyl-N'-3,5-dichloropyridin-2-yl-diacylhydrazine. Chin. J. Struct. Chem. 2011, 30, 412–416. [Google Scholar]
- Abdel-Aziz, H.A.; Elsaman, T.; Attia, M.I.; Alanazi, A.M. The reaction of ethyl 2-oxo-2H-chromene-3-carboxylate with hydrazine hydrate. Molecules 2013, 18, 2084–2095. [Google Scholar] [CrossRef]
- Ke, S.Y.; Qian, X.H.; Liu, F.Y.; Wang, N.; Fan, F.; Li, Z.; Yang, Q. Diacylhydrazine derivatives as novel potential chitin biosynthesis inhibitors: Design, synthesis, and structure-activity relationship. Eur. J. Med. Chem. 2009, 44, 2985–2993. [Google Scholar] [CrossRef]
- Zhao, Q.Q.; Shang, J.; Liu, Y.X.; Wang, K.Y.; Bi, F.C.; Huang, R.Q.; Wang, Q.M. Synthesis and insecticidal activities of novel N-Sulfenyl-N'-tert-butyl-N,N'-diacylhydrazines. 1. N-alkoxysulfenate derivatives. J. Agric. Food Chem. 2007, 55, 9614–9619. [Google Scholar]
- Zhao, Q.Q.; Ou, X.M.; Huang, Z.Q.; Bi, F.C.; Huang, R.Q.; Wang, Q.M. Synthesis and insecticidal activities of novel N-sulfenyl-N'-tert-butyl-N,N'-diacylhydrazines. 3. N-(alkyldithio), N-(aminothio), and N,N-dithio derivatives. J. Agric. Food Chem. 2008, 56, 10799–10804. [Google Scholar] [CrossRef]
- Zhao, Q.Q.; Shang, J.; Huang, Z.Q.; Wang, K.Y.; Bi, F.; Huang, R.Q.; Wang, Q.M. Synthesis and insecticidal activities of novel N-sulfenyl-N'-tert-butyl-N,N'-diacylhydrazines. 2. N-substituted phenoxysulfenate derivatives. J. Agric. Food Chem. 2008, 56, 5254–5259. [Google Scholar]
- Cui, Z.N.; Ling, Y.; Li, B.J.; Li, Y.Q.; Rui, C.H.; Cui, J.R.; Shi, Y.X.; Yang, X.L. Synthesis and bioactivity of N-benzoyl-N'-[5-(2'-substituted phenyl)-2-furoyl] semicarbazide derivatives. Molecules 2010, 15, 4267–4282. [Google Scholar] [CrossRef]
- Liu, X.H.; Shi, Y.X.; Ma, Y.; He, G.R.; Dong, W.L.; Zhang, C.Y.; Wang, B.L.; Wang, S.H.; Li, B.J.; Li, Z.M. Synthesis of some N,N'-diacylhydrazine derivatives with radical-scavenging and antifungal activity. Chem. Biol. Drug Des. 2009, 73, 320–327. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Li, Y.S.; Weng, J.Q.; Tan, C.X. Synthesis and herbicidal activity of some new diacylhydrazine derivatives fluoride-containing pyrazolyl moiety. Chin. J. Org. Chem. 2011, 31, 1295–1299. [Google Scholar]
- Zhao, H.; Neamati, N.; Sunder, S.; Hong, H.X.; Wang, S.M.; Milne, G.W.A.; Pommier, Y.; Burke, T.R. Hydrazide-containing inhibitors of HIV-1 integrase. J. Med. Chem. 1997, 40, 937–941. [Google Scholar] [CrossRef]
- Li, X.C.; Yang, X.L.; Cui, Z.N.; Li, Y.; He, H.W.; Ling, Y. Synthesis and bioactivity of novel N,N '-diacylhydrazine derivatives containing furan (II). Chin. J. Org. Chem. 2010, 28, 1233–1239. [Google Scholar] [CrossRef]
- Kim, B.Y.; Willbold, S.; Kulik, A.; Helaly, S.E.; Zinecker, H.; Wiese, J.; Imhoff, J.F.; Goodfellow, M.; Sussmuth, R.D.; Fiedler, H.P. Elaiomycins B and C, novel alkylhydrazides produced by Streptomyces sp BK 190. J. Antibiot. 2011, 64, 595–597. [Google Scholar] [CrossRef]
- Shoeb, M.; MacManus, S.M.; Jaspars, M.; Trevidu, J.; Nahar, L.; Kong-Thoo-Lin, P.; Sarker, S.D. Montamine, a unique dimeric indole alkaloid, from the seeds of Centaurea montana (Asteraceae), and its in vitro cytotoxic activity against the CaCo2 colon cancer cells. Tetrahedron 2006, 62, 11172–11177. [Google Scholar] [CrossRef]
- Zheng, J.; Liu, C.; Sawaya, M.R.; Vadla, B.; Khan, S.; Woods, R.J.; Eisenberg, D.; Goux, W.J.; Nowick, J.S. Macrocyclic beta-sheet peptides that inhibit the aggregation of a tau-protein-derived hexapeptide. J. Am. Chem. Soc. 2011, 133, 3144–3157. [Google Scholar] [CrossRef]
- Moanta, A.; Radu, S. New phenoxyacetic acid analogues with antimicrobial activity. Rev. Chim. 2008, 59, 708–711. [Google Scholar]
- Evans, K.A.; Shearer, B.G.; Wisnoski, D.D.; Shi, D.C.; Sparks, S.M.; Sternbach, D.D.; Winegar, D.A.; Billin, A.N.; Britt, C.; Way, J.M.; et al. Phenoxyacetic acids as PPAR delta partial agonists: Synthesis, optimization, and in vivo efficacy. Bioorg. Med. Chem. Lett. 2011, 21, 2345–2350. [Google Scholar] [CrossRef]
- Luker, T.; Bonnert, R.; Schmidt, J.; Sargent, C.; Paine, S.W.; Thom, S.; Pairaudeau, G.; Patel, A.; Mohammed, R.; Akam, E.; et al. Switching between agonists and antagonists at CRTh2 in a series of highly potent and selective biaryl phenoxyacetic acids. Bioorg. Med. Chem. Lett. 2011, 21, 3616–3621. [Google Scholar] [CrossRef]
- Liu, H.; Wang, H.Q.; Liu, Z.J. Synthesis and herbicidal activity of novel pyrazolo[3,4-d]pyrimidin-4-one derivatives containing aryloxyphenoxypropionate moieties. Bioorg. Med. Chem. Lett. 2007, 17, 2203–2209. [Google Scholar] [CrossRef]
- Han, L.; Gao, J.R.; Li, Z.M.; Zhang, Y.; Guo, W.M. Synthesis of new plant growth regulator: N-(Fatty acid)O-aryloxyacetyl ethanolamine. Bioorg. Med. Chem. Lett. 2007, 17, 3231–3234. [Google Scholar] [CrossRef]
- Yamada, K.; Yoshizawa, Y.; Oh, K. Synthesis of 2RS,4RS-1-[2-Phenyl-4-[2-(2-trifluromethoxy-phenoxy)-ethyl]-1,3-dioxolan-2-yl-methyl]-1H-1,2,4-triazole derivatives as potent inhibitors of brassinosteroid biosynthesis. Molecules 2012, 17, 4460–4473. [Google Scholar] [CrossRef]
- Voskienė, A.; Sapijanskaitė, B.; Mickevičius, V.; Jonuškienė, I.; Stasevych, M.; Komarovska-Porokhnyavets, O.; Musyanovych, R.; Novikov, V. Synthesis and microbiological evaluation of new 2- and 2,3-diphenoxysubstituted naphthalene-1,4-diones with 5-oxopyrrolidine moieties. Molecules 2012, 17, 14434–14448. [Google Scholar] [CrossRef]
- Sun, N.-B.; Shi, Y.-X.; Liu, X.-H.; Ma, Y.; Tan, C.-X.; Weng, J.-Q.; Jin, J.-Z.; Li, B.-J. Design, synthesis, antifungal activities and 3D-QSAR of new N,N'-diacylhydrazines containing 2,4-dichlorophenoxy moiety. Int. J. Mol. Sci. 2013, 14, 21741–21756. [Google Scholar] [CrossRef]
- Aibibuli, Z.; Wang, Y.; Tu, H.; Huang, X.; Zhang, A. Facile synthesis and herbicidal evaluation of 4H-3,1-benzoxazin-4-ones and 3H-quinazolin-4-ones with 2-phenoxymethyl substituents. Molecules 2012, 17, 3181–3201. [Google Scholar] [CrossRef]
- Ang, W.; Lin, Y.N.; Yang, T.; Yang, J.Z.; Pi, W.Y.; Yang, Y.H.; Luo, Y.F.; Deng, Y.; Wei, Y.Q. Synthesis and biological evaluation of 2-(3-fluoro-4-nitro phenoxy)-N-phenylacetamide derivatives as novel potential affordable antitubercular agents. Molecules 2012, 17, 2248–2258. [Google Scholar] [CrossRef]
- Liu, X.H.; Pan, L.; Ma, Y.; Weng, J.Q.; Tan, C.X.; Li, Y.H.; Shi, Y.X.; Li, B.J.; Li, Z.M.; Zhang, Y.G. Design, synthesis, biological activities, and 3D-QSAR of new N,N'-diacylhydrazines containing 2-(2,4-dichlorophenoxy)propane moiety. Chem. Biol. Drug Des. 2011, 78, 689–694. [Google Scholar] [CrossRef]
- SYBYL, version 6.91; Tripos Inc.: St. Louis, MO, USA, 2004.
- Liu, X.H.; Shi, Y.X.; Ma, Y.; Zhang, C.Y.; Dong, W.L.; Pan, L.; Wang, B.L.; Li, B.J.; Li, Z.M. Synthesis, antifungal activities and 3D-QSAR study of N-(5-substituted-1,3,4-thiadiazol-2-yl)cyclopropanecarboxamides. Eur. J. Med. Chem. 2009, 44, 2782–2786. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, G.-X.; Sun, Z.-H.; Yang, M.-Y.; Liu, X.-H.; Ma, Y.; Wei, Y.-Y. Design, Synthesis, Biological Activities and 3D-QSAR of New N,N'-Diacylhydrazines Containing 2,4-Dichlorophenoxy Moieties. Molecules 2013, 18, 14876-14891. https://doi.org/10.3390/molecules181214876
Sun G-X, Sun Z-H, Yang M-Y, Liu X-H, Ma Y, Wei Y-Y. Design, Synthesis, Biological Activities and 3D-QSAR of New N,N'-Diacylhydrazines Containing 2,4-Dichlorophenoxy Moieties. Molecules. 2013; 18(12):14876-14891. https://doi.org/10.3390/molecules181214876
Chicago/Turabian StyleSun, Guo-Xiang, Zhao-Hui Sun, Ming-Yan Yang, Xing-Hai Liu, Yi Ma, and Yun-Yang Wei. 2013. "Design, Synthesis, Biological Activities and 3D-QSAR of New N,N'-Diacylhydrazines Containing 2,4-Dichlorophenoxy Moieties" Molecules 18, no. 12: 14876-14891. https://doi.org/10.3390/molecules181214876
APA StyleSun, G.-X., Sun, Z.-H., Yang, M.-Y., Liu, X.-H., Ma, Y., & Wei, Y.-Y. (2013). Design, Synthesis, Biological Activities and 3D-QSAR of New N,N'-Diacylhydrazines Containing 2,4-Dichlorophenoxy Moieties. Molecules, 18(12), 14876-14891. https://doi.org/10.3390/molecules181214876




