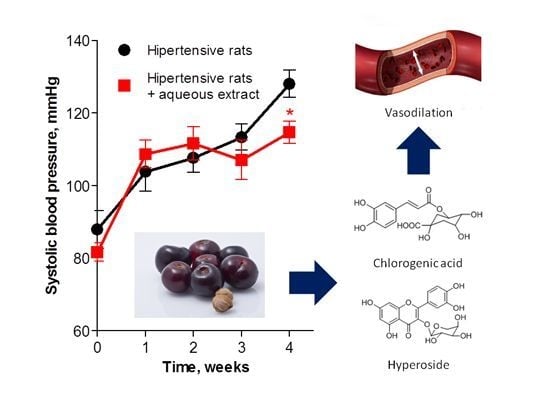
Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties
Abstract
:1. Introduction
2. Results and Discussion
| Component | Black Cherry | Plum | Grape |
|---|---|---|---|
| Moisture | 81.18 ± 0.87 a | 87.29 ± 1.12 b | 81.83 ± 0.72 a |
| Protein | 2.10 ± 0.01 a | 0.49 ± 0.04 b | 0.46 ± 0.01 b |
| Fat | 0.05 ± 0.01 a | 0.04 ± 0.01 a | 0.03 ± 0.01 a |
| Fiber | 3.58 ± 0.03 a | 3.57 ± 0.03 a | 3.21 ± 0.45 a |
| Ash | 0.86 ± 0.11 a | 0.37 ± 0.03 b | 0.49 ± 0.07 b |
| Carbohydrate + | 12.23 ± 0.79 a | 8.28 ± 1.02 b | 13.96 ± 0.33 a |
| Mineral | Black Cherry | Plum | Grape |
|---|---|---|---|
| Sodium | 22.40 ± 1.60 a | 15.2 ± 2.40 a | 12.50 ± 0.49 a |
| Potassium | 184.30 ± 3.50 a | 81.30 ± 2.00 b | 96.60 ± 3.74 b |
| Calcium | 12.90 ± 1.90 a | 4.20 ± 0.15 b | 4.41 ± 0.21 b |
| Magnesium | 21.20 ± 0.20 a | 10.40 ± 1.20 b | 5.30 ± 0.20 c |
| Phosphorous | 28.10 ± 0.40 a | 13.50 ± 0.50 b | 1.39 ± 0.40 b |
| Fruit/Part of Fruit | Total Phenolics (mg of GAE/100 g of FW) | Flavonoids (mg of CE/100 g of FW) | DPPH b (µmol TE/100 g of FW) | FRAP b (µmol TE/100 g of FW) |
|---|---|---|---|---|
| black cherry | 362.2 ± 11.6 | 201.8 ± 12.1 | 2056.7 ± 108.0 | 1455.2 ± 92.5 |
| flesh | 325.1 ± 12.0 | 146.3 ± 8.0 | 1764.6 ± 170.4 | 1100.7 ± 35.4 |
| peel | 564.9 ± 10.9 | 259.5 ± 14.5 | 2681.6 ± 180.0 | 1991.4 ± 40.1 |
| plum | 218.0 ± 10.5 | 180.2 ± 8.3 | 1137.8 ± 40.1 | 737.2 ± 68.0 |
| grape | 160.9 ± 12.1 | 121.2 ± 4.9 | 1148.6 ± 102.1 | 593.8 ± 46.9 |
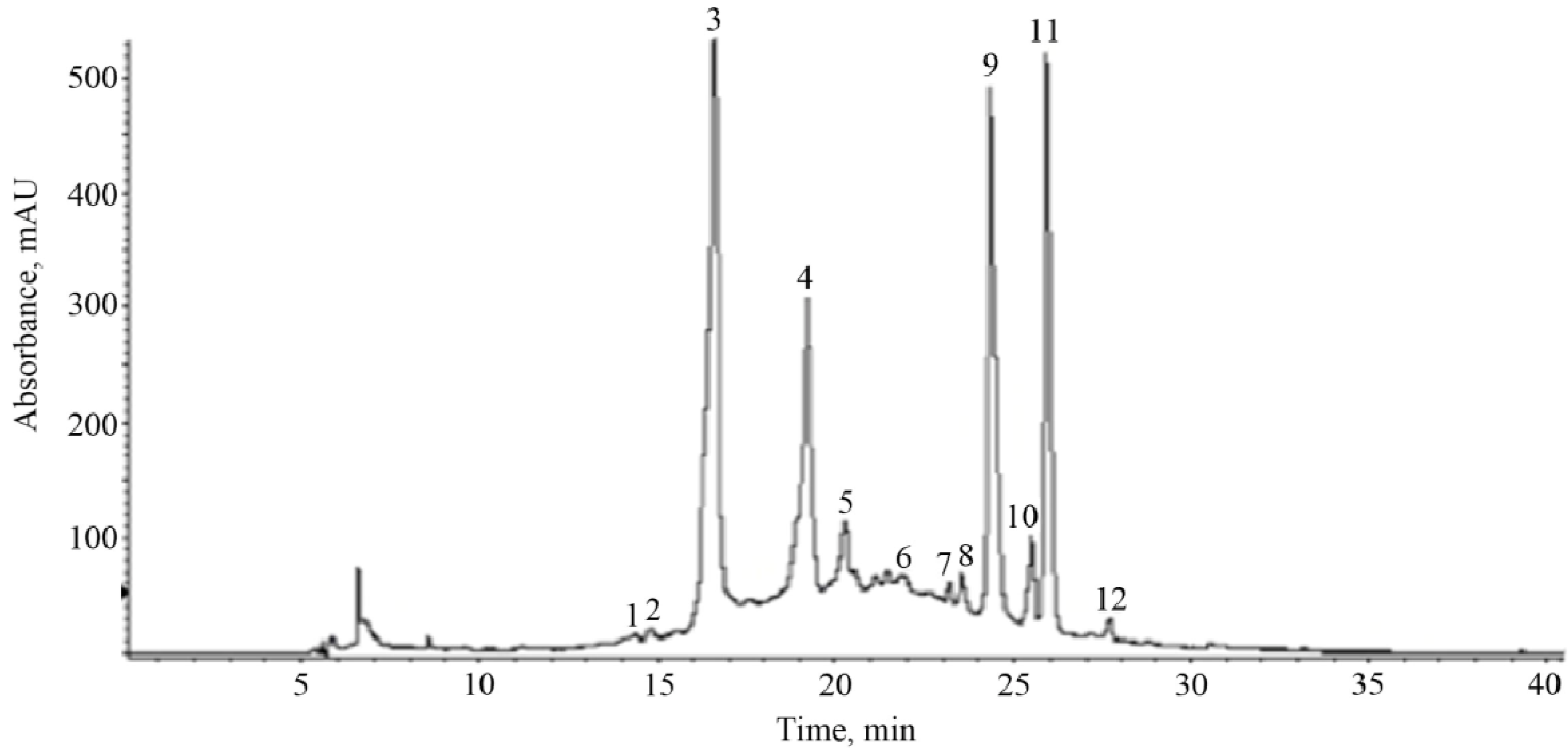
| Peak a | tR (min) | λmax (nm) | Fragmentation Pattern [M-H]− | Compound |
|---|---|---|---|---|
| 1 | 14.3 | 272 | 315 (169, 125) | gallic acid hexoside |
| 2 | 14.8 | 292 | 329 (167, 152, 123, 108) | vanillic acid hexoside |
| 3 | 16.6 | 284, 518 | 593 (465, 447, 285) | cyanidin-3-O-rutinoside |
| 4 | 19.2 | 300, 328 | 353 (191) | chlorogenic acid |
| 5 | 20.3 | 280 | 577 (425, 289) | procyanidin B (dimer) |
| 6 | 22.3 | 296, 324 | 387 (179, 161, 135) | Caffeoyl hexose-deoxyhexoside |
| 7 | 23.2 | 255, 354 | 609 (353, 301) | rutin |
| 8 | 23.5 | 282 | 577 (425, 289) | procyanidin B (dimer) |
| 9 | 24.3 | 256, 356 | 463 (301) | hyperoside |
| 10 | 25.5 | 266, 348 | 447 (285) | kaempferol hexoside |
| 11 | 25.9 | 256, 356 | 433 (301) | quercetin pentoside |
| 12 | 27.7 | 264, 348 | 417 (285) | kaempferol pentoside |
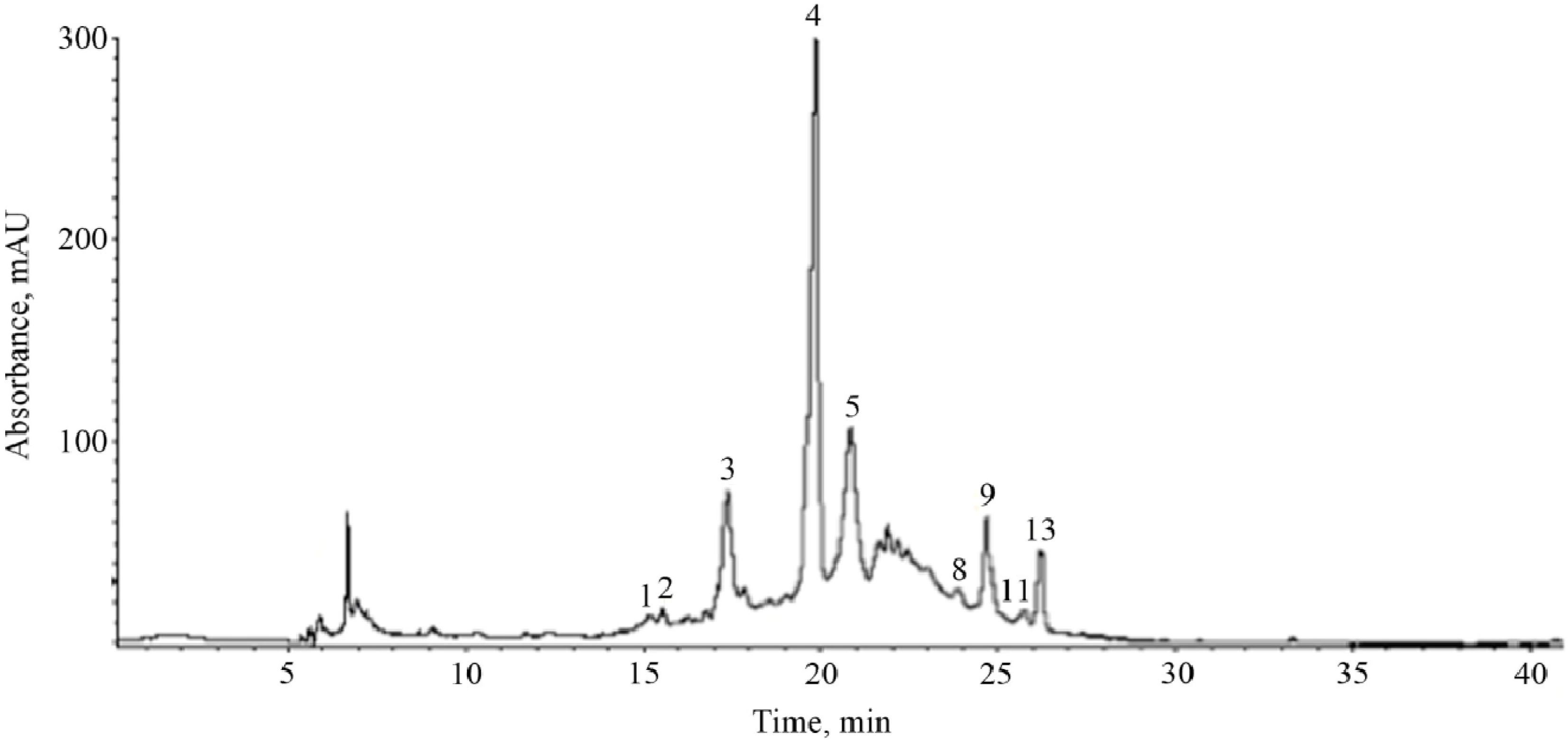
| Peak a | tR (min) | λmax (nm) | Fragmentation Pattern [M-H]− | Compound |
|---|---|---|---|---|
| 1 | 15.1 | 272 | 315 (169, 125) | gallic acid hexoside |
| 2 | 15.5 | 292 | 329 (167, 152, 123, 108) | vanillic acid hexoside |
| 3 | 17.3 | 284, 518 | 593 (465, 447, 285) | cyanidin-3-O-rutinoside |
| 4 | 19.8 | 300, 328 | 353 (191) | chlorogenic acid |
| 5 | 20.8 | 280 | 577 (425, 289) | procyanidin B (dimer) |
| 8 | 23.9 | 282 | 577 (425, 289) | procyanidin B (dimer) |
| 9 | 24.6 | 256, 356 | 463 (301) | hyperoside |
| 11 | 25.7 | 256, 356 | 433 (301) | quercetin pentoside |
| 13 | 26.2 | 256, 356 | 549 (505, 301) | quercetin malonilglucoside |

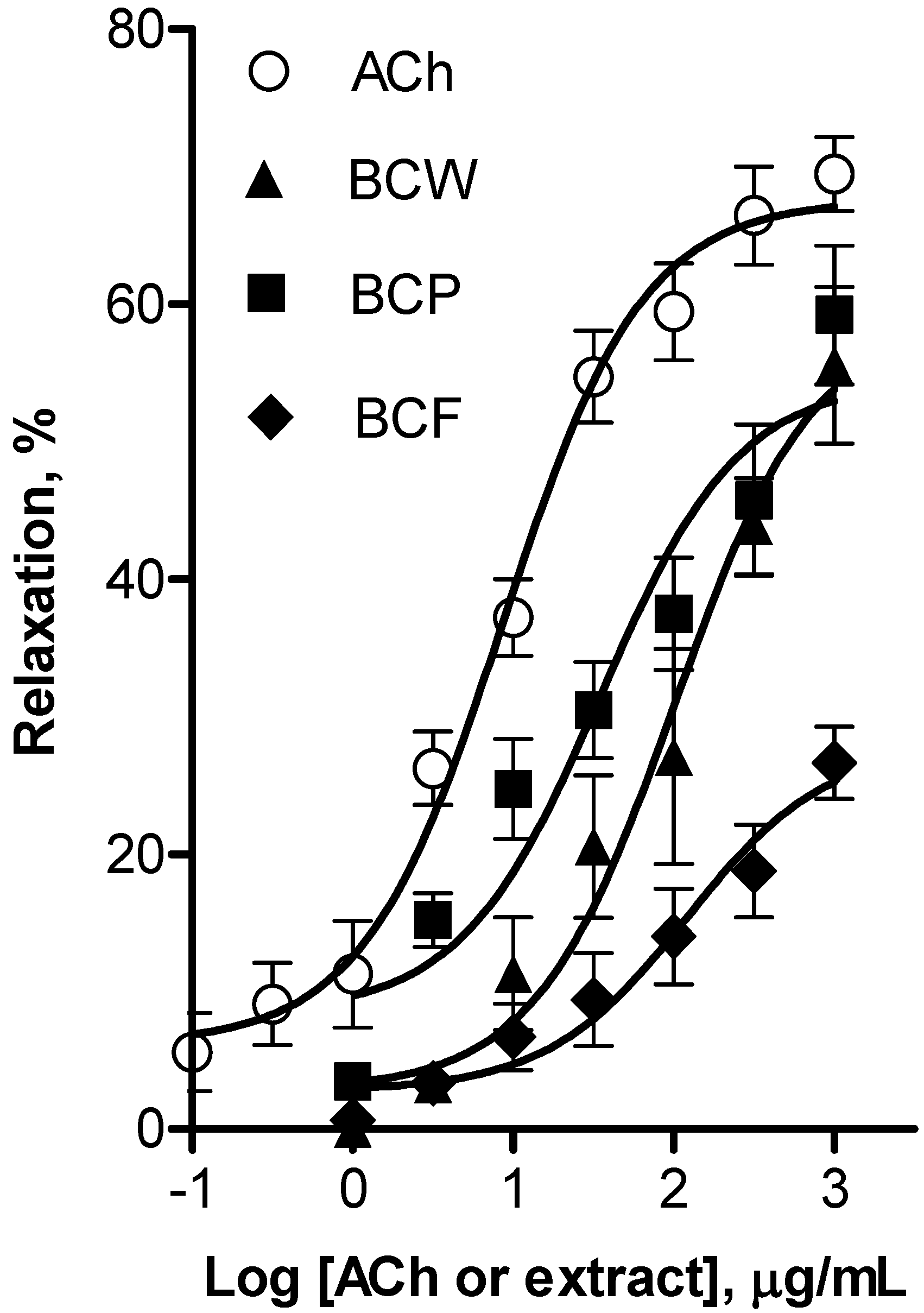
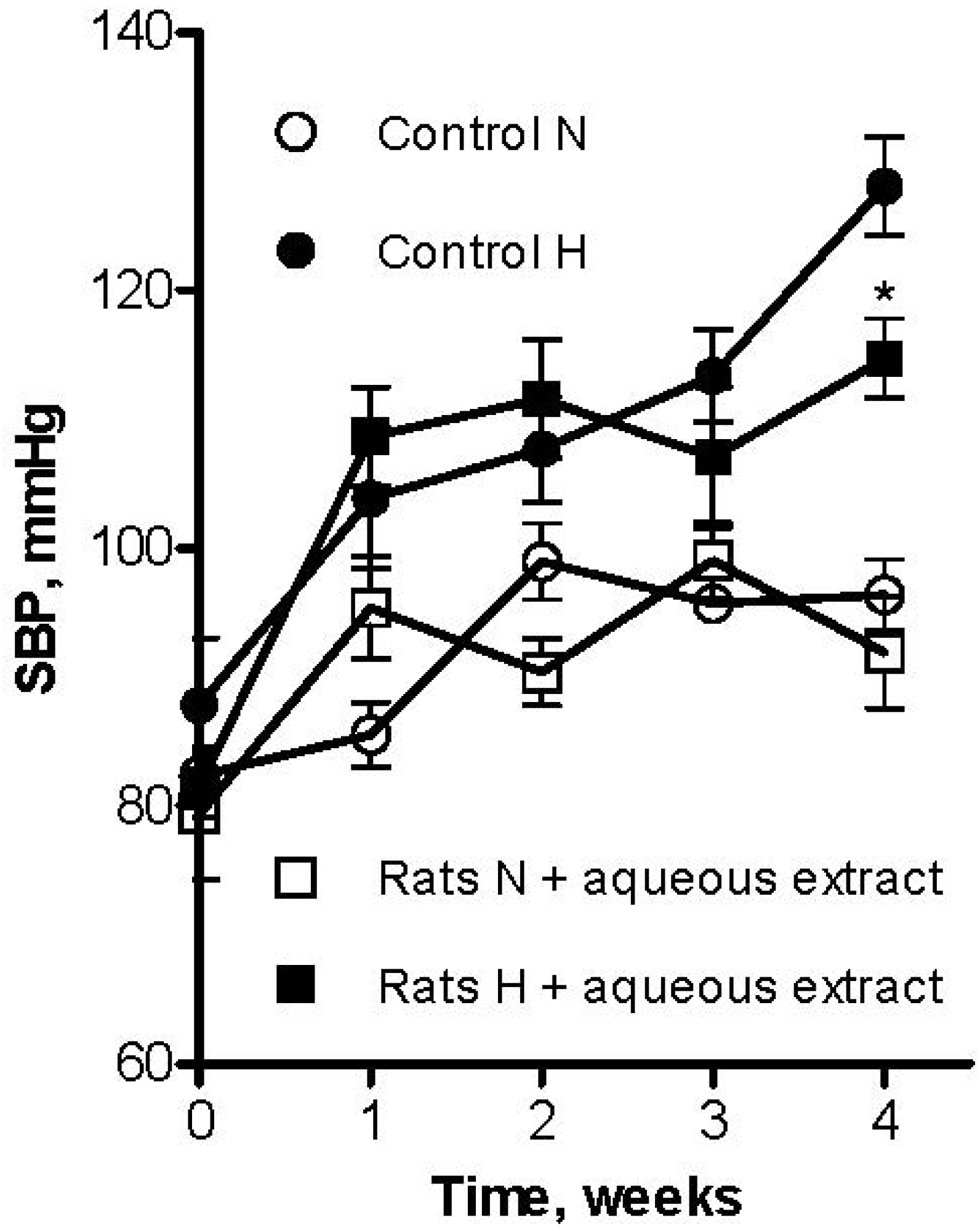
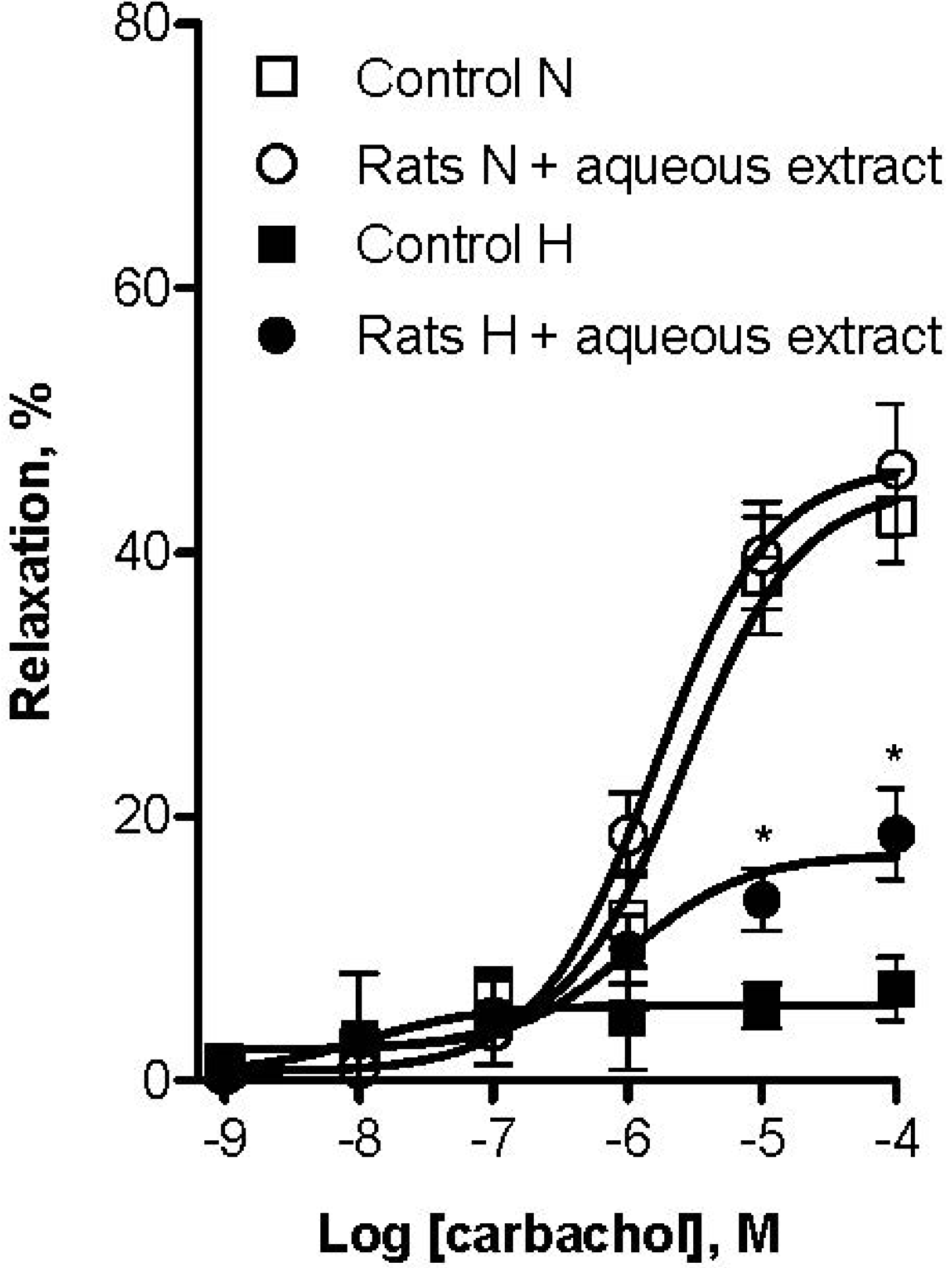
3. Experimental
3.1. Black Cherry Samples
3.2. Plum and Grape (Red Seedless) Samples
3.3. Proximate Analysis and Mineral Content
3.4. Extraction and Quantification of Total Phenolic Content
3.5. Determination of Antioxidant Capacity
3.6. HPLC/DAD/ESI-MS Analysis
3.7. Preparation of Aqueous Extracts
3.8. Determination of the Vasorelaxant Effect
3.9. Determination of the Antihypertensive Effect
3.9.1. Experimental Schedule and Blood Pressure Measurements
3.9.2. Effect of Long-Term Oral Administration of the Lyophilized Aqueous Extract (BCAE) on Vascular Reactivity: Ex Vivo Experiments
3.10. Statistical Analysis
3.10.1. Proximate Analysis, Phenolic Content and Antioxidant Capacity
3.10.2. In Vitro Assays
3.10.3. In Vivo Assays
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gaziano, T.A.; Bitton, A.; Anand, S.; Abrahams-Gessel, S.; Murphy, A. Growing epidemic of coronary heart disease in low- and middle-income countries. Curr. Probl. Cardiol. 2010, 35, 72–115. [Google Scholar] [CrossRef]
- Appel, L.J.; Giles, T.D.; Black, H.R.; Izzo, J.L.; Materson, B.J.; Oparil, S.; Weber, M.A. ASH position paper: Dietary approaches to lower blood pressure. J. Am. Soc. Hypertens. 2010, 4, 79–89. [Google Scholar] [CrossRef]
- Rosenthal, T.; Alter, A. Occupational stress and hypertension. J. Am. Soc. Hypertens. 2011, 6, 2–22. [Google Scholar] [CrossRef]
- Pessina, A.C.; Rossi, G.P. Uncontrolled hypertension: Highlights and perspectives from the European Society of Hypertension Satellite Symposium. Expert Rev. Cardiovasc. Ther. 2011, 9, 1515–1518. [Google Scholar] [CrossRef]
- Andriantsitohaina, R.; Auger, C.; Chataigneau, T.; Étienne-Selloum, N.; Li, H.; Martínez, M.C.; Schini-Kerth, V.B.; Laher, I. Molecular mechanisms of the cardiovascular protective effects of polyphenols. Br. J. Nutr. 2012, 108, 1532–1549. [Google Scholar] [CrossRef]
- Proteggente, A.R.; Pannala, A.S.; Paganga, G.; van Buren, L.; Wagner, E.; Wiseman, S.; van de Put, F.; Dacombe, C.; Rice-Evans, C. The antioxidant activity of regularly consumed fruit and vegetables reflects their phenolic and vitamin C composition. Free Radic. Res. 2002, 36, 217–233. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Niki, E. Antioxidant capacity: Which capacity and how to assess it? J. Berry Res. 2011, 1, 169–176. [Google Scholar]
- Pinchuk, I.; Shoval, H.; Dotan, Y.; Lichtenberg, D. Evaluation of antioxidants: Scope, limitations and relevance of assays. Chem. Phys. Lipids 2012, 165, 638–647. [Google Scholar]
- Prior, R.L. Fruits and vegetables in the prevention of cellular oxidative damage. Am. J. Clin. Nutr. 2003, 78, 570S–578S. [Google Scholar]
- Szajdek, A.; Borowska, E.J. Bioactive compounds and health-promoting properties of berry fruits: A review. Plant Foods Hum. Nutr. 2008, 63, 147–156. [Google Scholar] [CrossRef]
- Edirisinghe, I.; Banaszewski, K.; Cappozzo, J.; McCarthy, D.; Burton-Freeman, B.M. Effect of black currant anthocyanins on the activation of endothelial nitric oxide synthase (eNOS) in vitro in human endothelial cells. J. Agric. Food Chem. 2011, 59, 8616–8624. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Duarte, J.; Jimenez, R.; Santos-Buelga, C.; Osuna, A. Antihypertensive effects of the flavonoid quercetin. Pharmacol. Rep. 2009, 61, 67–75. [Google Scholar]
- Kawakami, K.; Aketa, S.; Sakai, H.; Watanabe, Y.; Nishida, H.; Hirayama, M. Antihypertensive and vasorelaxant effects of water-soluble proanthocyanidins from Persimmom leaf tea in spontaneously hypertensive rats. Biosci. Biotechnol. Biochem. 2011, 75, 1435–1439. [Google Scholar] [CrossRef]
- Suri, S.; Rayment, D.A.; Kroon, P.A.; Needs, P.W.; Taylor, M.A.; Tribolo, S.; Wilson, V.G. Quercetin and its major metabolites selectively modulate cyclic GMP-dependent relaxations and associated tolerance in pig isolated coronary artery. Br. J. Pharmacol. 2010, 159, 566–575. [Google Scholar]
- Mac Vaugh, R. A revision of the North American Black cherries (Prunus serotina ehrh) and relatives. Brittonia 1951, 7, 279–315. [Google Scholar] [CrossRef]
- Martínez, M. Plantas Medicinales de México; Editorial Botas: México, D.F., México, 1991; pp. 61–62. [Google Scholar]
- Argueta, A. Atlas de las Plantas de la Medicina Tradicional Mexicana; Instituto Nacional Indigenista: México, D.F., Mexico, 1994; Volume 1, pp. 319–320. [Google Scholar]
- Ordaz-Galindo, A.; Wesche-Ebeling, P.; Wrolstad, R.E.; Rodriguez-Saona, L.; Argaiz-Jamet, A. Purification and identification of capulin (Prunus serotina Ehrh) anthocyanins. Food Chem. 1999, 65, 201–206. [Google Scholar]
- Vasco, C.; Ruales, J.; Kamal-Eldin, A. Total phenolic compounds and antioxidant capacities of major fruits from Ecuador. Food Chem. 2008, 111, 816–823. [Google Scholar] [CrossRef]
- Vasco, C.; Riihinen, K.; Ruales, J.; Kamal-Eldin, A. Phenolic compounds in Rosaceae fruits from Ecuador. J. Agric. Food Chem. 2009, 57, 1204–1212. [Google Scholar] [CrossRef]
- Ibarra-Alvarado, C.; Rojas, A.; Mendoza, S.; Bah, M.; Gutiérrez, D.M.; Hernández-Sandoval, L.; Martínez, M. Vasoactive and antioxidant activities of plants used in Mexican traditional medicine for the treatment of cardiovascular diseases. Pharm. Biol. 2010, 48, 732–739. [Google Scholar] [CrossRef]
- Ibarra-Alvarado, C.; Rojas, A.; Luna, F.; Rojas, J.I.; Rivero-Cruz, B.; Rivero-Cruz, F. Vasorelaxant constituents of the leaves of Prunus serotina “capulín”. Rev. Latinoam. Quim. 2009, 37, 164–173. [Google Scholar]
- Nutrient Data Laboratory (2012). National Nutrient Database for Standard Reference, Release 24. Available online: http://ndb.nal.usda.gov/ndb/foods/list/ (accessed on 15 July 2012).
- Morton, J. Fruits of Warm Climates. Available online: http://www.hort.purdue.edu/newcrop/morton/capulin.html/ (accessed on 15 July 2012).
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierce, B.; Kader, A.A. Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. J. Agric. Food Chem. 2002, 50, 4976–4982. [Google Scholar] [CrossRef]
- McCune, L.M.; Kubota, C.; Stendell-Hollis, N.R.; Thomson, C.A. Cherries and health: A review. Crit. Rev. Food Sci. Nutr. 2011, 51, 1–12. [Google Scholar]
- Karaman, S.; Tütem, E.; Baskan, S.K.; Apak, R. Comparison of antioxidant capacity and phenolic composition of peel and flesh of some apple varieties. J. Sci. Food Agric. 2013, 93, 867–875. [Google Scholar] [CrossRef]
- Lago-Vanzela, E.S.; Da-Silva, R.; Gomes, E.; García-Romero, E.; Hermosín-Gutiérrez, I. Phenolic composition of the edible parts (flesh and skin) of Bordô grape (Vitis labrusca) using HPLC-DAD-ESI-MS/MS. J. Agric. Food Chem. 2011, 59, 13136–13146. [Google Scholar] [CrossRef]
- Sun, J.; Chu, Y.F.; Wu, X.; Liu, R.H. Antioxidant and antiproliferative activities of common fruits. J. Agric. Food Chem. 2002, 50, 7449–7454. [Google Scholar] [CrossRef]
- Scalzo, J.; Politi, A.; Pellegrini, N.; Mezzetti, B.; Battino, M. Plant genotype affects total antioxidant capacity and phenolic contents in fruit. Nutrition 2005, 21, 207–213. [Google Scholar] [CrossRef]
- Ferretti, G.; Bacchetti, T.; Belleggia, A.; Neri, D. Cherry antioxidants: From farm to table. Molecules 2010, 15, 6993–7005. [Google Scholar] [CrossRef]
- Capanoglu, E.; Boyacioglu, D.; de Vos, R.C.H.; Hall, R.D.; Beekwilder, J. Procyanidins in fruit from Sour cherry (Prunus cerasus) differ strongly in chainlength from those in Laurel cherry (Prunus lauracerasus) and Cornelian cherry (Cornus mas). J. Berry Res. 2011, 1, 137–146. [Google Scholar]
- Liu, Y.; Liu, X.; Zhong, F.; Tian, R.; Zhang, K.; Zhang, X.; Li, T. Comparative study of phenolic compounds and antioxidant activity in different species of cherries. J. Food Sci. 2011, 76, C633–C638. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Peng, C.; Jiao, R.; Wong, Y.M.; Yang, N.; Huang, Y. Anti-hypertensive nutraceuticals and functional foods. J. Agric. Food Chem. 2009, 57, 4485–4499. [Google Scholar] [CrossRef]
- Fan, Y.; Chen, W.; Guo, Y.; Wang, Q.; Song, B. Cellular mechanisms underlying Hyperin-induced relaxation of rat basilar artery. Fitoterapia 2011, 82, 626–631. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Ibarra, M.; Cogolludo, A.L.; Duarte, J.; Zaragoza-Arnaez, F.; Moreno, L.; Lopez-Lopez, G.; Tamargo, J. Endothelium dependent vasodilator effects of the flavonoid quercetin and its methylated metabolites in rat conductance and resistance arteries. J. Pharmacol. Exp. Ther. 2002, 302, 66–72. [Google Scholar] [CrossRef]
- Ibarrola, D.A.; Ibarrola, M.H.; Vera, C.; Montalbetti, Y.; Ferro, E.A. Hypotensive effect of crude root extract of Solanum sisymbriifolium (Solanaceae) in normo- and hypertensive rats. J. Ethnopharmacol. 1996, 54, 7–12. [Google Scholar] [CrossRef]
- Suzuki, A.; Kagawa, D.; Ochiai, R.; Tokimitsu, I.; Saito, I. Green coffee bean extract and its metabolites have a hypotensive effect in spontaneously hypertensive rats. Hypertens. Res. 2002, 25, 99–107. [Google Scholar] [CrossRef]
- Koçyildiz, Z.C.; Birman, H.; Olgac, V.; Akgün-Dar, K.; Melikoğlu, G.; Meriçli, A.H. Crataegus tanacetifolia leaf extract prevents L-NAME-induced hypertension in rats: A morphological study. Phytother. Res. 2006, 20, 66–70. [Google Scholar] [CrossRef]
- Reagan–Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2007, 22, 659–661. [Google Scholar] [CrossRef]
- Taubert, D.; Berkels, R.; Roesen, R.; Klaus, W. Chocolate and blood pressure in elderly individuals with isolated systolic hypertension. JAMA 2003, 290, 1029–1030. [Google Scholar] [CrossRef]
- Endemann, D.H.; Schiffrin, E.L. Endothelial dysfunction. J. Am. Soc. Nephrol. 2004, 15, 1983–1992. [Google Scholar] [CrossRef]
- Rivera-Pastrana, D.M.; Yahia, E.M.; González-Aguilar, G.A. Phenolic and carotenoid profiles of papaya fruit (Carica papaya L.) and their contents under low temperature storage. J. Sci. Food Agric. 2010, 90, 2358–2365. [Google Scholar] [CrossRef]
- García-Solís, P.; Yahia, E.M.; Morales-Tlalpan, V.; Díaz-Muñoz, M. Screening of antiproliferative effect of aqueous extracts of plant foods consumed in Mexico on the breast cancer cell line MCF-7. Int. J. Food Sci. Nutr. 2009, 60, 32–46. [Google Scholar]
- Cantín, C.M.; Moreno, M.A.; Gogocerna, Y. Evaluation of the antioxidant capacity, phenolic compounds, and vitamin C content of different peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. J. Agric. Food Chem. 2009, 56, 4586–4592. [Google Scholar]
- Corral-Aguayo, R.D.; Yahia, E.M.; Carrillo-López, A.; González-Aguilar, G. Correlation between some nutritional components and the total antioxidant capacity measured with six different assays in eight horticultural crops. J. Agric. Food Chem. 2008, 56, 10498–10504. [Google Scholar] [CrossRef]
- Belmokhtar, M.; Bouanani, N.E.; Ziyyat, A.; Mekhfi, H.; Bnouham, M.; Aziz, M.; Mateo, P.; Fischmeister, R.; Legssyer, A. Antihypertensive and endothelium-dependent vasodilator effects of aqueous extract of Cistus ladaniferus. Biochem. Biophys. Res. Commun. 2009, 389, 45–49. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luna-Vázquez, F.J.; Ibarra-Alvarado, C.; Rojas-Molina, A.; Rojas-Molina, J.I.; Yahia, E.M.; Rivera-Pastrana, D.M.; Rojas-Molina, A.; Zavala-Sánchez, Á.M. Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties. Molecules 2013, 18, 14597-14612. https://doi.org/10.3390/molecules181214597
Luna-Vázquez FJ, Ibarra-Alvarado C, Rojas-Molina A, Rojas-Molina JI, Yahia EM, Rivera-Pastrana DM, Rojas-Molina A, Zavala-Sánchez ÁM. Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties. Molecules. 2013; 18(12):14597-14612. https://doi.org/10.3390/molecules181214597
Chicago/Turabian StyleLuna-Vázquez, Francisco J., César Ibarra-Alvarado, Alejandra Rojas-Molina, Juana I. Rojas-Molina, Elhadi M. Yahia, Dulce M. Rivera-Pastrana, Adriana Rojas-Molina, and Ángel Miguel Zavala-Sánchez. 2013. "Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties" Molecules 18, no. 12: 14597-14612. https://doi.org/10.3390/molecules181214597
APA StyleLuna-Vázquez, F. J., Ibarra-Alvarado, C., Rojas-Molina, A., Rojas-Molina, J. I., Yahia, E. M., Rivera-Pastrana, D. M., Rojas-Molina, A., & Zavala-Sánchez, Á. M. (2013). Nutraceutical Value of Black Cherry Prunus serotina Ehrh. Fruits: Antioxidant and Antihypertensive Properties. Molecules, 18(12), 14597-14612. https://doi.org/10.3390/molecules181214597