Abstract
An environmentally-friendly, enzyme-promoted procedure for the Henry reaction was first studied using water-in-[Bmim][PF6] microemulsions as reaction medium. The Amano acylase from Aspergillus oryzae showed better catalytic activity for the addition reactions of nitromethane with a series of aromatic aldehydes, and a highest yield of 90% was obtained.
1. Introduction
The Henry (nitroaldol) reaction is considered one of the most powerful and atom economical C–C bond-formation reactions and is widely employed in organic chemistry. The resulting products, β-nitro alcohols, are often used as key intermediates in the synthesis of numerous biologically active compounds, including natural products, insecticides, fungicides and antibiotics [1,2,3,4,5]. Therefore, a variety of catalysts have been developed for this reaction, and basic catalysts are the most common ones [1,4,6,7,8]. However, this type of reaction is often complicated under strong alkaline conditions because of the unwanted side reactions, such as aldol, Michael and elimination reactions [6,9,10]. On the other hand, it's very gratifying to see that some Henry reactions have been performed under mild reaction conditions more recently, e.g., solvent free [11,12,13,14], in aqueous media [9,15,16,17] or using enzymes as catalysts [1,18,19,20].
Biocatalysis has become a powerful and useful tool for organic synthesis because of its high selectivity, mild reaction conditions and green features. Moreover, enzyme promiscuity has given a new push to its applications [21,22]. In 2006, the Griengl group reported the first example of a biocatalytic asymmetric Henry reaction catalyzed by hydroxynitrile lyase [18]. After that, aminoacylase [10], transglutaminase [19] and lipase [23] have also proved to exhibit nitroaldol activity. However, to the best of our knowledge, these enzyme-catalyzed Henry reactions were carried out in organic media, and the harm from organic solvents was unavoidable. Therefore, it is significant to develop new enzymatic methods for the Henry reaction in eco-friendly media.
Thus, as one part of our continuing interest in enzyme promiscuity and green chemistry, we wish to report the first examples of biocatalytic Henry reactions in ionic liquid (IL)-based microemulsions (Scheme 1). ILs are usually considered as “green” solvents. Compared with ILs, water-in-IL (w/IL) microemulsions are more suitable for an enzyme-catalyzed conversion [24]. In microemulsions, the enzyme is located in the so-called “water pool” where it is exposed to a living environment similar to the natural one, thereby exhibiting good stability and activity [25,26,27]. At the same time, enzyme can be dispersed in the medium at a molecular level [24], which increases the odds of interaction between enzyme and substrate molecules. Here, 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim][PF6], a hydrophobic ionic liquid, was selected as oil phase, and the acylase-catalyzed Henry reaction in w/IL microemulsions was explored (Scheme 1). We were gratified to observe that excellent results were obtained in our preliminary study.
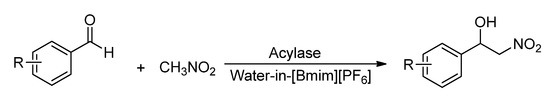
Scheme 1.
The acylase-catalyzed Henry reaction in water-in-[Bmim][PF6] microemulsions.
Scheme 1.
The acylase-catalyzed Henry reaction in water-in-[Bmim][PF6] microemulsions.
2. Results and Discussion
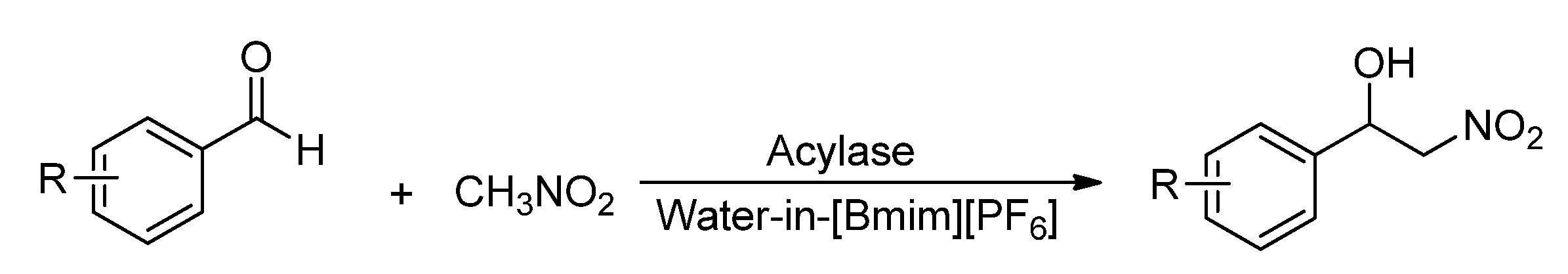
TX-100/H2O/[Bmim]PF6 microemulsions was first prepared according to the phase diagrams in literature [28]. Then the Henry reaction in microemulsions was studied selecting 4-nitrobenzaldehyde and nitromethane as a model reaction. Nine commercial enzymes were investigated to find the most suitable catalyst to catalyze the Henry reaction in w/IL microemulsions. As shown in Table 1, the best result of 88% yield was achieved by using Amano acylase from Aspergillus oryzae (AOA, entry 1), although other tested enzymes, even the non-enzyme protein bovine serum albumin (entry 10), also showed good catalytic activities. Next, some control experiments were performed to demonstrate the specific catalytic effect of the enzymes on the model reaction. The results showed that the model reaction could be performed smoothly in the microemulsions in absence of any enzyme (entry 11), although the yield was lower, but only a small amount of product was obtained under solvent-free conditions in the absence of enzyme (entry 12) and almost no product was produced in [Bmim][PF6] alone using AOA as a catalyst (entry 13). All these showed that this Henry reaction could be freely carried out in water-in-[Bmim][PF6] microemulsions, and AOA could effectively promote this reaction.

Table 1.
Henry reaction catalyzed by different enzymes in IL-based microemulsions a.
 | ||
|---|---|---|
| Entry | Enzyme | Yield (%) b |
| 1 | Amano acylase from Aspergillus oryzae | 88 |
| 2 | Lipase from Rhizopus niveus | 72 |
| 3 | Amano lipase PS from Burkholderia cepacia | 71 |
| 4 | Amano lipase from Pseudomonas fluorescens | 70 |
| 5 | Lipase from Candida rugosa | 68 |
| 6 | Lipase from bovine pancreas | 68 |
| 7 | Amano lipase M from Mucor javanicus | 67 |
| 8 | Amano lipase A from Aspergillus niger | 66 |
| 9 | Acylase I from Aspergillus melleus | 65 |
| 10 | Bovine serum albumin | 64 |
| 11 | Control test c | 62 |
| 12 | Control test d | 24 |
| 13 | Control test e | 1 |
a Reaction conditions: Enzyme (30 mg), 4-nitrobenzaldehyde (1 mmol), nitromethane (4 mmol) and TX-100/H2O/[Bmim]PF6 microemulsions (1 mL, water 16.7%, TX-100 60%, [Bmim]PF6 23.3%, in weight), was shaken at 30 °C for 48 h; b Determined by HPLC; c No enzyme; d Under solvent-free and enzyme-free conditions; e In 1 mL [Bmim]PF6 (with enzyme).
The influence of water content (ω0, where ω0 = [H2O]/[TX-100]) on the activity of AOA encapsulated in the “water pool” was investigated. W/IL microemulsions show a spherical droplet structure for which the droplet radius is directly proportional to the ω0 value and thus the microenvironment around the enzyme can be tuned by simply changing the ω0 value. Therefore ω0 is a key parameter and it plays a significant role in the enzyme-catalyzed reactions in w/IL microemulsions. It was found from the data listed in Table 2 that the ω0 value had an impact on this enzymatic reaction and the best result was obtained at ω0 = 10 (Table 2, entry 3).

Table 2.
Effect of ω0 value on the Henry reaction a.
| Entry | ω0 | Water (%) | TX-100 (%) | [Bmim]PF6 (%) | Yield b (%) |
|---|---|---|---|---|---|
| 1 | 4 | 6.7 | 60 | 33.3 | 84 |
| 2 | 8 | 13.3 | 60 | 26.7 | 85 |
| 3 | 10 | 16.7 | 60 | 23.3 | 88 |
| 4 | 12 | 20.0 | 60 | 20.0 | 82 |
| 5 | 14 | 23.4 | 60 | 16.6 | 80 |
| 6 | 16 | 26.7 | 60 | 13.3 | 75 |
a Reaction conditions: AOA (30 mg), 4-nitrobenzaldehyde (1 mmol), nitromethane (4 mmol) and TX-100/H2O/[Bmim]PF6 microemulsions (1 mL), was shaken at 30 °C for 48 h; b Determined by HPLC.
Next, the influences of enzyme loading, temperature and molar ratio were studied respectively, and the optimal values of these factors were determined to be 30 mg/mL, 40 °C and 1:5, respectively. As shown in Figure 1, the effect of enzyme loading on the yield was not obvious, and the satisfying result could be obtained when 30 mg/mL AOA was adopted. However, temperature and molar ratio had significant effects on this reaction (Figure 2 and Figure 3).
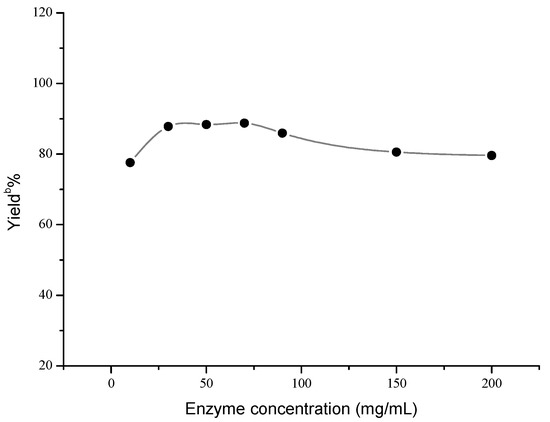
Figure 1.
Influence of enzyme concentration on the Henry reaction a.
Figure 1.
Influence of enzyme concentration on the Henry reaction a.
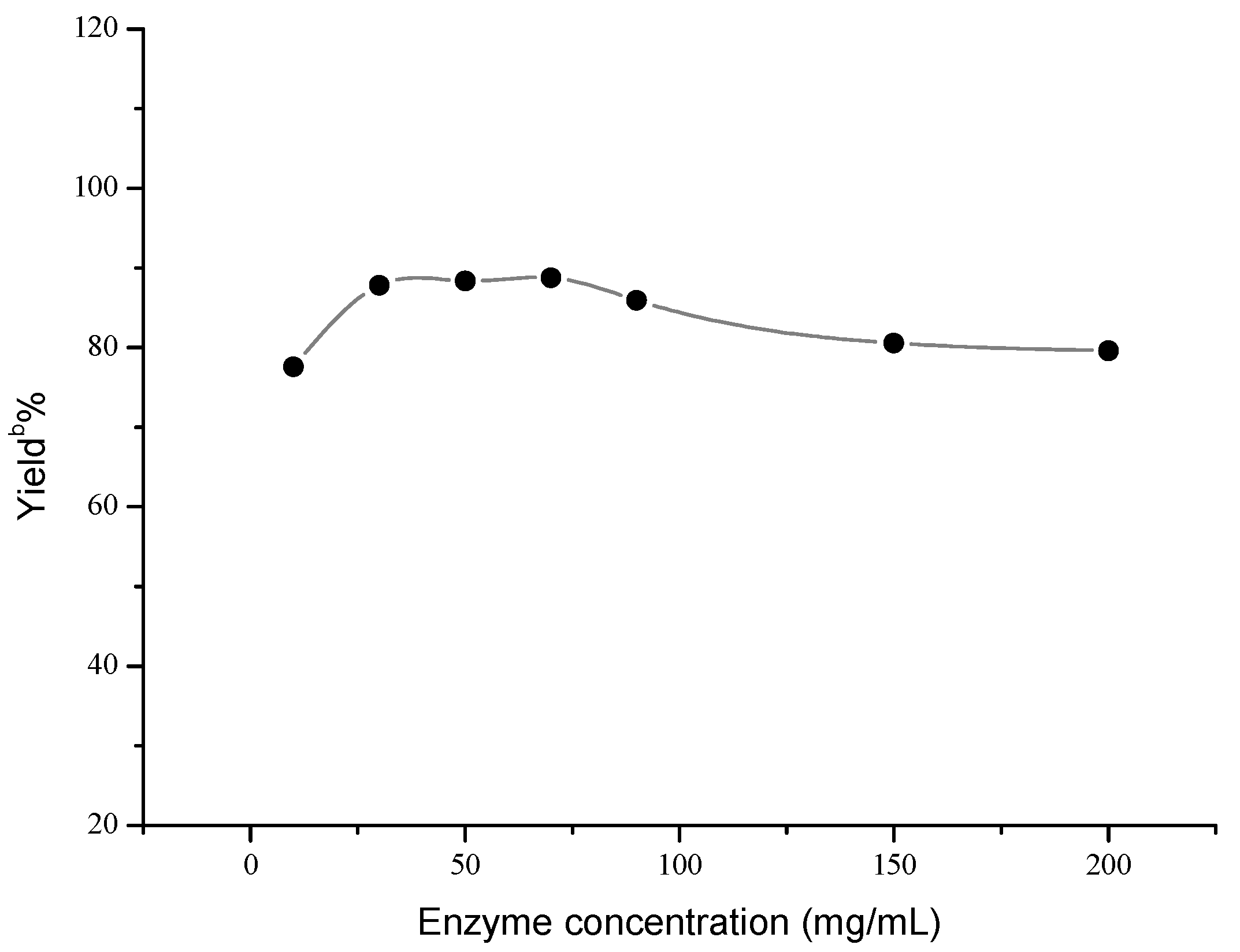
a Reaction conditions: AOA (10–200 mg), 4-nitrobenzaldehyde (1 mmol), nitromethane (4 mmol) and microemulsions (1 mL), was shaken at 30 °C for 48 h; b Yields were determined by HPLC.
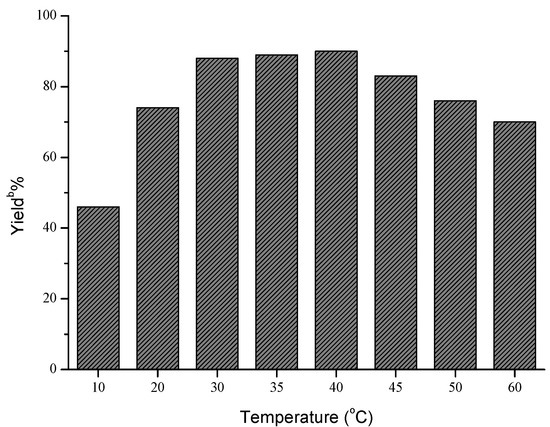
Figure 2.
Influence of temperature on the Henry reaction a.
Figure 2.
Influence of temperature on the Henry reaction a.
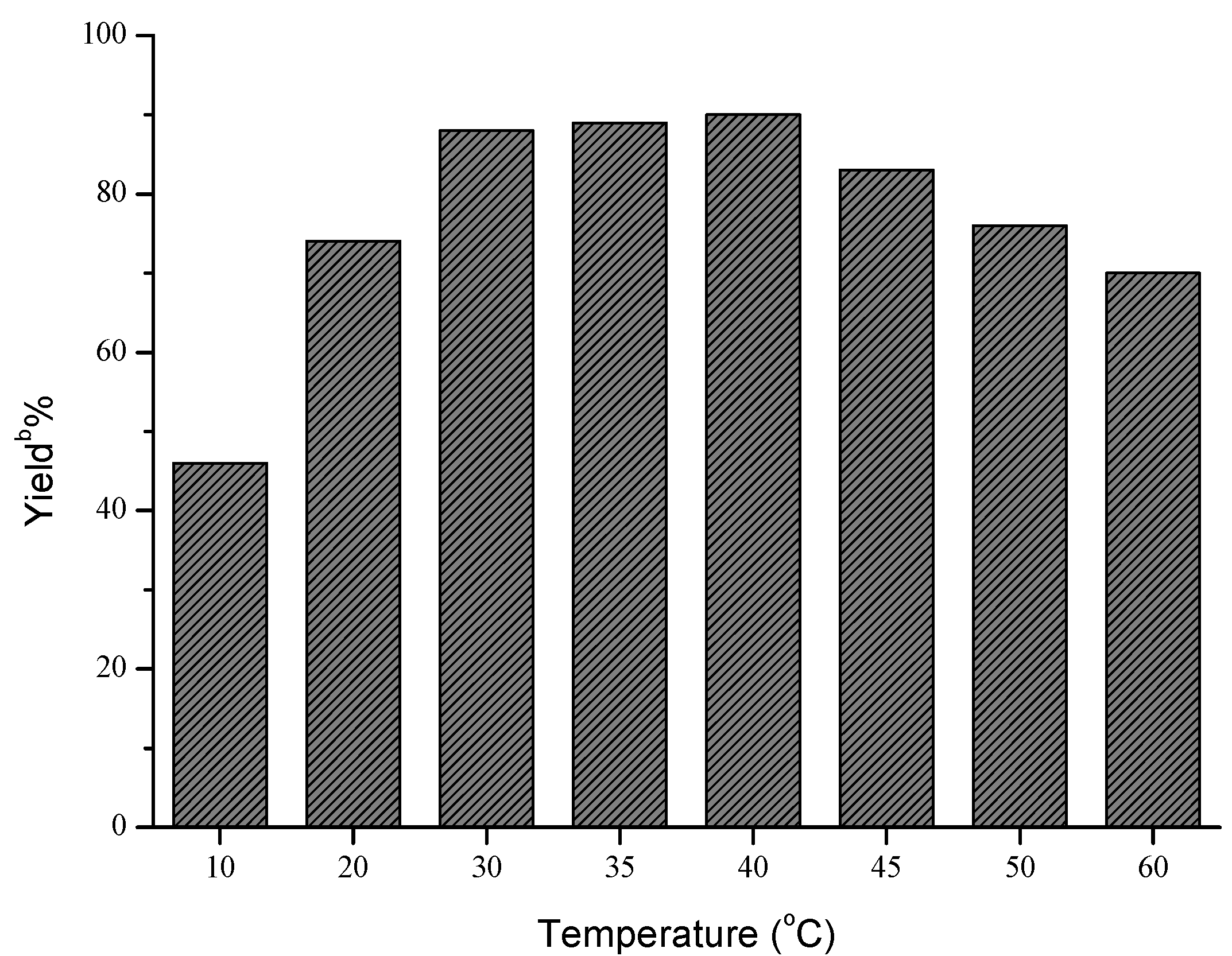
a Reaction conditions: AOA (30 mg), 4-nitrobenzaldehyde (1 mmol), nitromethane (4 mmol) and microemulsions (1 mL), was shaken at certain temperatures for 48 h; b Yields were determined by HPLC.
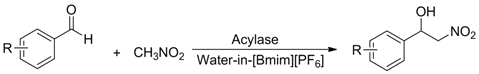
With the optimized conditions in hand, more aromatic aldehydes were evaluated to show the generality and scope of this new enzymatic promiscuity and the results are summarized in Table 3. It can be seen that a wide range of aromatic aldehydes can effectively react with nitromethane in water-in-[Bmim][PF6] microemulsions in the presence of AOA. In general, benzaldehyde and its derivatives bearing an electron-donating group gave the products in relatively lower yields (entries 1–3 and 7–10). In contrast, aromatic aldehydes containing an electron-withdrawing substituent provided β-nitro alcohols in higher yields. Especially, 2-nitrobenzaldehyde and 4-nitrobenzaldehyde (entries 4 and 6) gave the corresponding product in yields of 87% and 90%, respectively. This may be due to the fact that electron-withdrawing groups can enhance the electrophilicity of the carbonyl carbon in the aldehydes, which facilitates the reaction.
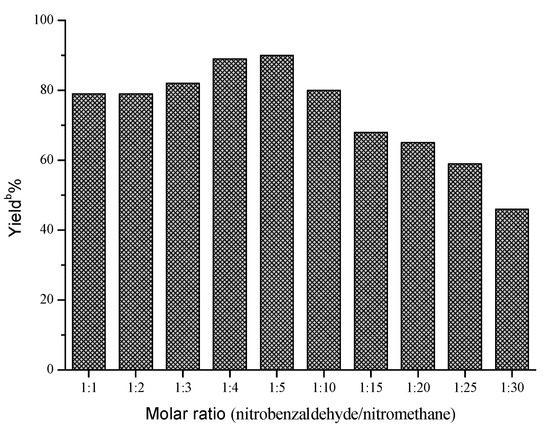
Figure 3.
Influence of molar ratio on the Henry reaction a.
Figure 3.
Influence of molar ratio on the Henry reaction a.
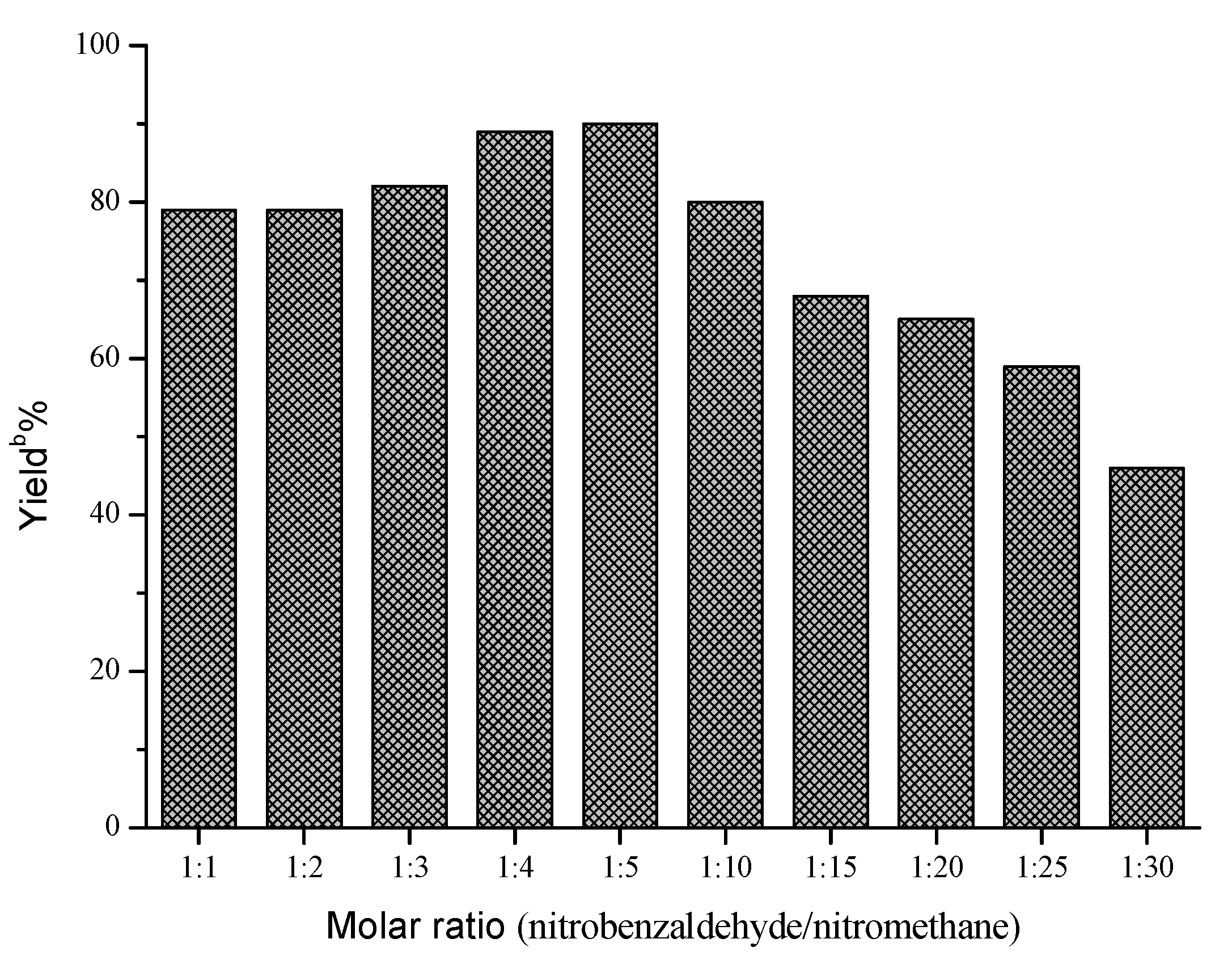
a Reaction conditions: AOA (30 mg), 4-nitrobenzaldehyde (1 mmol), nitromethane (1–30 mmol) and microemulsions (1 mL), was shaken at 40 °C for 48 h; b Yields were determined by HPLC.

Table 3.
Investigation of the reactant scope of the Henry reaction in IL-based microemulsions a.
 | |||
|---|---|---|---|
| Entry | R | Products | Yield b(%) |
| 1 | H | a | 44 |
| 2 | 3-CH3 | b | 28 |
| 3 | 4-CH3 | c | 30 |
| 4 | 2-NO2 | d | 87 |
| 5 | 3-NO2 | e | 79 |
| 6 | 4-NO2 | f | 90 |
| 7 | 2-OH | g | 22 |
| 8 | 3-OH | h | 48 |
| 9 | 4-OH | i | 55 |
| 10 | 4-OCH3 | j | 40 |
| 11 | 2-Cl | k | 77 |
| 12 | 4-Cl | l | 66 |
a Reaction conditions: AOA (30 mg), aldehydes (1 mmol), nitromethane (5 mmol) and microemulsions (1 mL), was shaken at 40 °C for 48 h; b Isolated yield after silica gel column chromatography.
3. Experimental
3.1. Materials and Analytical Methods
Amano acylase from Aspergillus oryzae, lipase from Rhizopus niveus, Amano lipase PS from Burkholderia cepacia, Amano lipase from Pseudomonas fluorescens, lipase from Candida rugosa, Amano lipase M from Mucor javanicus, Amano lipase A from Aspergillus niger and acylase I from Aspergillus melleus were purchased from Sigma-Aldrich Co. LLC (Shanghai, China). Lipase from bovine pancreas and bovine serum albumin were purchased from Aladdin Co., Ltd. (Shanghai, China). Other reagents were obtained from commercial suppliers and used without further purification unless otherwise noted. The reactions were monitored by thin-layer chromatography and visualized using UV light. The 1H-NMR spectra were recorded on a Bruker 400 MHz instrument using CDCl3 as solvent. Chemical shifts (δ) were expressed in ppm with tetramethylsilane (TMS) as internal standard, and coupling constants (J) were reported in Hz. High performance liquid chromatography (HPLC) was carried out on Waters instrument (Waters 2489, 1525) using a C18 column (250 mm × 46 mm). All products are known compounds and characterization data is only given below for a selection of products. Column chromatography was performed on silica gel using ethyl acetate-petroleum ether as mobile phase.
3.2. General Procedure for Henry reaction
Aromatic aldehyde (1 mmol), nitromethane (4 mmol), acylase (30 mg) and microemulsion (1 mL, water 16.7%, TX-100 60%, [Bmim]PF6 23.3%) were added in a 10 mL test tube, then shaken at 260 rpm and 40 °C. After completion of the reaction, enzyme was filtered off to stop the reaction, then the filtrate was extracted with ethyl acetate and the resulting crude product was purified by column chromatography (petroleum ether-ethyl acetate) to give the pure product.
3.3. Physical and 1H-NMR Data of Some Representative Henry Products
2-Nitro-1-phenylethanol (a)
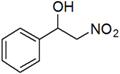
Yellow oil; 1H-NMR: δ = 7.39–7.33 (m, 5H), 5.40–5.36 (m, 1H), 4.58–4.51 (m, 2H), 2.86 (br, 1H).
2-Nitro-1-p-tolylethanol (c)
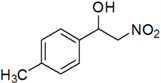
Yellow oil; 1H-NMR: δ = 7.27 (d, J = 7.94 Hz, 2H), 7.19 (d, J = 7.96 Hz, 2H), 5.44–5.38 (m, 2H), 4.62–4.48 (m, 1H), 3.65 (br, 1H), 2.37 (s, 3H).
2-Nitro-1-(2-nitrophenyl)ethanol (d)
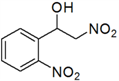
Yellow oil; 1H-NMR: δ = 8.07 (d, J = 8.0 Hz, 1H), 7.95 (d, J = 7.6 Hz, 1H), 7.75 (t, J = 7.4Hz, 1H), 7.55 (t, J = 7.6Hz, 1H), 6.04 (d, J = 8.8 Hz, 1H), 4.57–4.52 (m, 2H), 3.36 (br, 1H).
2-Nitro-1-(3-nitrophenyl)ethanol (e)
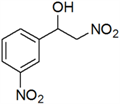
Pale yellow solid; 1H-NMR: δ = 8.26 (d, J = 8.8 Hz, 1H), 8.22 (d, J = 8.0 Hz, 1H), 7.77 (d, J = 7.6 Hz, 1H), 7.61 (d, J = 7.6, 8.0 Hz, 1H), 5.62–5.32 (m, 1H), 4.65–4.57 (m, 2H), 3.43 (br, 1H).
2-Nitro-1-(4-nitrophenyl)ethanol (f)
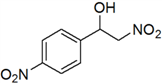
Yellow solid; 1H-NMR: δ = 8.26 (d, J = 8.8 Hz, 2H), 7.63 (d, J = 8.5 Hz, 2H), 5.24–4.76 (m, 1H), 4.52–4.22 (m, 2H), 3.65 (br, 1H).
2-(1-Hydroxy-2-nitroethyl)phenol (g)
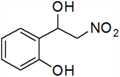
Colorless oil; 1H-NMR: δ = 7.25–6.98 (m, 4H), 5.66 (s, 1H), 4.59–4.51(m, 2H), 4.41–4.36 (m, 1H), 3.25 (br, 1H).
4-(1-Hydroxy-2-nitroethyl)phenol (i)
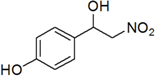
Yellow oil; 1H-NMR: δ = 7.56 (d, J = 9.4 Hz, 2H), 7.16 (d, J = 9.4 Hz, 2H), 5.31 (s, 1H), 4.68–4.61 (m, 2H), 4.58–4.34 (m, 1H), 3.82 (br, 1H).
1-(3-Chlorophenyl)-2-nitroethanol (k)
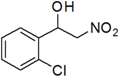
Pale yellow oil; 1H-NMR: δ = 7.66 (d, J = 7.6 Hz, 1H), 7.39–7.26 (m, 3H), 5.85–5.81 (m, 1H), 4.68–4.65 (m, 2H), 3.18 (br, 1H).
1-(4-Chlorophenyl)-2-nitroethanol (l)
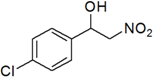
Yellow oil; 1H-NMR: δ = 7.38–7.27 (m, 4H), 5.43–5.39 (m, 1H), 4.57–4.54 (m, 2H), 3.11 (br, 1H).
4. Conclusions
In summary, an efficient and environmentally-friendly method has been developed for the Henry reaction, which is another example of biocatalytic promiscuity. Here, water/[Bmim]PF6 reverse microemulsion was used for the Henry reaction for the first time and AOA could effectively promote this reaction. As a green approach, it not only expands the applications of IL microemulsions, but might push forward the development of other reactions taking advantage of enzymatic promiscuity.
Acknowledgments
We gratefully acknowledge the financial support of the National Natural Science Foundation of China (No. 21262002), Natural Science Foundation of Jiangxi (No. 20114BAB213003, 20122BAB203006, 21262002), Science and Technology Innovation Team of Jiangxi (No. 20113BCB24011) and Graduate Student Innovation Foundation of East China Institute of Technology (No. DYCA12004).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Milner, S.E.; Moody, T.S.; Maguire, A.R. Biocatalytic Approaches to the Henry (Nitroaldol) Reaction. Eur. J. Org. Chem. 2012, 2012, 3059–3067. [Google Scholar] [CrossRef]
- Kisanga, P.B.; Verkade, J.G. P(RNCH2CH2)3N: An efficient promoter for the nitroaldol (Henry) reaction. J. Org. Chem. 1999, 64, 4298–4303. [Google Scholar] [CrossRef]
- Heffner, R.J.; Jiang, J.; Joullie, M.M. Total synthesis of (−)-nummularine F. J. Am. Chem. Soc. 1992, 114, 10181–10189. [Google Scholar] [CrossRef]
- Saraswat, A.; Sharma, L.; Singh, S.; Siddiqui, I.; Singh, R. Chemoselective Henry reaction catalyzed by electro-generated base. Res. Chem. Intermed. 2013, 1–7. [Google Scholar]
- Jiang, T.; Gao, H.; Han, B.; Zhao, G.; Chang, Y.; Wu, W.; Gao, L.; Yang, G. Ionic liquid catalyzed Henry reactions. Tetrahedron Lett. 2004, 45, 2699–2701. [Google Scholar] [CrossRef]
- Rokhum, L.; Bez, G. Ethyl acrylate conjugated polystyryl-diphenylphosphine—An extremely efficient catalyst for Henry reaction under solvent-free conditions (SolFC). Can. J. Chem. 2012, 91, 300–306. [Google Scholar] [CrossRef]
- Khan, F.A.; Dash, J.; Satapathy, R.; Upadhyay, S.K. Hydrotalcite catalysis in ionic liquid medium: A recyclable reaction system for heterogeneous Knoevenagel and nitroaldol condensation. Tetrahedron Lett. 2004, 45, 3055–3058. [Google Scholar] [CrossRef]
- Simoni, D.; Rondanin, R.; Morini, M.; Baruchello, R.; Invidiata, F.P. 1,5,7-Triazabicyclo[4.4.0]dec-1-ene (TBD), 7-methyl-TBD (MTBD) and the polymer-supported TBD (P-TBD): Three efficient catalysts for the nitroaldol (Henry) reaction and for the addition of dialkyl phosphites to unsaturated systems. Tetrahedron Lett. 2000, 41, 1607–1610. [Google Scholar] [CrossRef]
- Bora, P.P.; Bez, G. Henry reaction in aqueous media at neutral pH. Eur. J. Org. Chem. 2013, 2013, 2922–2929. [Google Scholar] [CrossRef]
- Wang, J.-L.; Li, X.; Xie, H.-Y.; Liu, B.-K.; Lin, X.-F. Hydrolase-catalyzed fast Henry reaction of nitroalkanes and aldehydes in organic media. J. Biotechnol. 2010, 145, 240–243. [Google Scholar] [CrossRef]
- Majhi, A.; Kadam, S.T.; Kim, S.S. TMEDA catalyzed Henry (nitroaldol) reaction under metal and solvent-free conditions. Bull. Korean Chem. Soc. 2009, 30, 1767–1770. [Google Scholar] [CrossRef]
- Ballini, R.; Bosica, G.; Parrini, M. A one pot, solvent-free synthesis of acyclic α-nitro ketones through the nitroaldol reaction. Tetrahedron Lett. 1998, 39, 7963–7964. [Google Scholar] [CrossRef]
- Changsheng, R.M.-A.H.R.; Gan, X.C.; Lai, G.; Wang, Z. Rapid microwave-assisted henry reaction in solvent-free processes. Synlett 2006, 2006, 387–390. [Google Scholar]
- Tanaka, K.; Hachiken, S. Enantioselective Henry reaction catalyzed by trianglamine–Cu (OAc)2 complex under solvent-free conditions. Tetrahedron Lett. 2008, 49, 2533–2536. [Google Scholar] [CrossRef]
- Reddy, K.R.; Rajasekhar, C.V.; Krishna, G.G. Zinc-proline complex: An efficient, reusable catalyst for direct nitroaldol reaction in aqueous media. Synth. Commun. 2007, 37, 1971–1976. [Google Scholar] [CrossRef]
- Ren, Y.; Cai, C. Iodine catalysis in aqueous medium: An improved reaction system for Knoevenagel and Nitroaldol condensation. Catalysis Lett. 2007, 118, 134–138. [Google Scholar] [CrossRef]
- Busto, E.; Gotor-Fernández, V.; Gotor, V. Protein-mediated nitroaldol addition in aqueous media. Catalytic promiscuity or unspecific catalysis? Org. Process Res. Dev. 2010, 15, 236–240. [Google Scholar]
- Purkarthofer, T.; Gruber, K.; Gruber-Khadjawi, M.; Waich, K.; Skranc, W.; Mink, D.; Griengl, H. A biocatalytic Henry reaction—The hydroxynitrile lyase from Hevea brasiliensis also catalyzes nitroaldol reactions. Angew. Chem. Int. Ed. 2006, 45, 3454–3456. [Google Scholar] [CrossRef]
- Tang, R.-C.; Guan, Z.; He, Y.-H.; Zhu, W. Enzyme-catalyzed Henry (nitroaldol) reaction. J. Mol. Catal. B: Enzym. 2010, 63, 62–67. [Google Scholar] [CrossRef]
- Gruber-Khadjawi, M.; Purkarthofer, T.; Skranc, W.; Griengl, H. Hydroxynitrile lyase-catalyzed enzymatic nitroaldol (Henry) reaction. Adv. Synth. Catal. 2007, 349, 1445–1450. [Google Scholar] [CrossRef]
- Humble, M.S.; Berglund, P. Biocatalytic promiscuity. Eur. J. Org. Chem. 2011, 2011, 3391–3401. [Google Scholar] [CrossRef]
- Kapoor, M.; Gupta, M.N. Lipase promiscuity and its biochemical applications. Process Biochem. 2012, 47, 555–569. [Google Scholar] [CrossRef]
- Le, Z.-G.; Guo, L.-T.; Jiang, G.-F.; Yang, X.-B.; Liu, H.-Q. Henry reaction catalyzed by Lipase A from Aspergillus niger. Green Chem. Lett. Rev. 2013, 6, 277–281. [Google Scholar] [CrossRef]
- Xue, L.; Qiu, H.; Li, Y.; Lu, L.; Huang, X.; Qu, Y. A novel water-in-ionic liquid microemulsion and its interfacial effect on the activity of laccase. Colloids Surf. B 2011, 82, 432–437. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Kamiya, N.; Goto, M. Biocatalysis in water-in-ionic liquid microemulsions: A case study with horseradish peroxidase. Langmuir 2008, 25, 977–982. [Google Scholar] [CrossRef]
- Pavlidis, I.V.; Gournis, D.; Papadopoulos, G.K.; Stamatis, H. Lipases in water-in-ionic liquid microemulsions: Structural and activity studies. J. Mol. Catal. B: Enzym. 2009, 60, 50–56. [Google Scholar] [CrossRef]
- Xue, L.; Li, Y.; Zou, F.; Lu, L.; Zhao, Y.; Huang, X.; Qu, Y. The catalytic efficiency of lipase in a novel water-in-[Bmim][PF6] microemulsion stabilized by both AOT and Triton X-100. Colloids Surf. B 2012, 92, 360–366. [Google Scholar] [CrossRef]
- Gao, Y.; Han, S.; Han, B.; Li, G.; Shen, D.; Li, Z.; Du, J.; Hou, W.; Zhang, G. TX-100/water/1-butyl-3-methylimidazolium hexafluorophosphate microemulsions. Langmuir 2005, 21, 5681–5684. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
