Bioassay-Guided Isolation of DPP-4 Inhibitory Fractions from Extracts of Submerged Cultured of Inonotus obliquus
Abstract
:1. Introduction
2. Results and Discussion
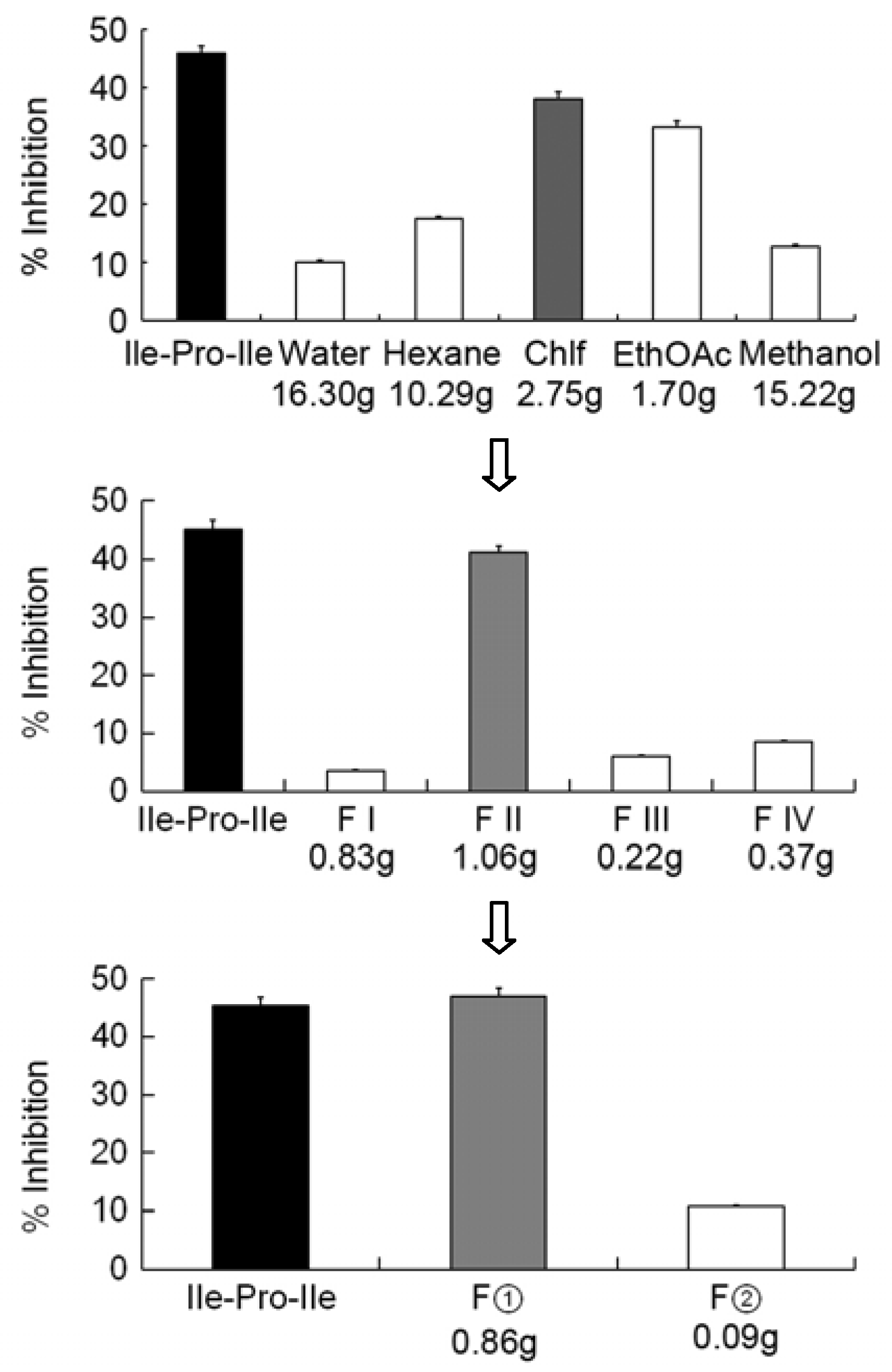
2.1. Isolation of the DPP-4 Inhibitory Components from Mycelium Powders of I. obliquus
2.2. Structural Determination of the Isolated Compounds
2.3. Molecular Docking of the Isolated Compounds with DPP-4
3. Experimental
3.1. Fermentation and Product Preparation
3.2. Extraction and Isolation
3.3. UPLC-Q-TOF-MS Experiments
3.4. DPP-4 Inhibition Assay
3.5. Molecular Docking
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Taylor, S.I. Deconstructing type 2 diabetes. Cell 1999, 97, 9–12. [Google Scholar] [CrossRef]
- Chen, L.; Magliano, D.J.; Zimmet, P.Z. The worldwide epidemiology of type 2 diabetes mellitus-present and future perspectives. Nat. Rev. Endocrinol. 2012, 8, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Cornell, S. Key considerations in pharmacotherapy for type 2 diabetes mellitus: A multiple target organ approach. J. Clin. Pharm. Ther. 2012, 37, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Barnett, A. DPP-4 inhibitors and their potential role in the management of type 2 diabetes. Int. J. Clin. Pract. 2006, 60, 1454–1470. [Google Scholar] [CrossRef] [PubMed]
- Drucker, D.J. Glucagon-like peptides. Diabetes 1998, 47, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Zinman, B. Dipeptidyl peptidase-4 inhibitors and the management of type 2 diabetes mellitus. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Utzschneider, K.M.; Tong, J.; Montgomery, B.; Udayasankar, J.; Gerchman, F.; Marcovina, S.M.; Watson, C.E.; Ligueros-Saylan, M.A.; Foley, J.E.; Holst, J.J.; et al. The dipeptidyl peptidase-4 inhibitor vildagliptin improves beta-cell function and insulin sensitivity in subjects with impaired fasting glucose. Diabetes Care 2008, 31, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Williams-Herman, D.; Round, E.; Swern, A.; Musser, B.; Davies, M.J.; Kaufman, K.D.; Amatruda, J.M. Safety and tolerability of sitagliptin, a selective DPP-4 inhibitor, in patients with type 2 diabetes: Pooled analysis of 6139 patients in clinical trials for tip to 2 years. Diabetologia 2008, 51, S365–S366. [Google Scholar]
- Ahren, B.; Simonsson, E.; Larsson, H.; Landin-Olsson, M.; Torgeirsson, H.; Jansson, P.A.; Sandqvist, M.; Bavenholm, P.; Efendic, S.; Eriksson, J.W.; et al. Inhibition of dipeptidyl peptidase IV improves metabolic control over a 4-week study period in type 2 diabetes. Diabetes Care 2002, 25, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Covington, P.; Christopher, R.; Davenport, M.; Fleck, P.; Mekki, Q.A.; Wann, E.R.; Karim, A. Pharmacokinetic, Pharmacodynamic, And tolerability profiles of the dipeptidyl peptidase-4 inhibitor alogliptin: A randomized, Double-blind, Placebo-controlled, Multiple-dose study in adult patients with type 2 diabetes. Clin. Ther. 2008, 30, 499–512. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.L. Inonotus obliquus. Edible Fungi of China 2002, 21, 7–8. [Google Scholar]
- Sun, J.E.; Ao, Z.H.; Lu, Z.M.; Xu, H.Y.; Zhang, X.M.; Dou, W.F.; Xu, Z.H. Antihyperglycemic and antilipidperoxidative effects of dry matter of culture broth of Inonotus obliquus in submerged culture on normal and alloxan-diabetes mice. J. Ethnopharmacol. 2008, 118, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Shibnev, V.A.; Mishin, D.V.; Garaev, T.M.; Finogenova, N.P.; Botikov, A.G.; Deryabin, P.G. Antiviral activity of Inonotus obliquus fungus extract towards infection caused by hepatitis C virus in cell cultures. Bull. Exp. Biol. Med. 2011, 151, 612–614. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Kim, D.S.; Park, K.C. Antioxidant effect of Inonotus obliquus. J. Ethnopharmacol. 2005, 96, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.O.; Park, H.W.; Kim, J.H.; Lee, J.Y.; Moon, S.H.; Shin, C.S. Anti-cancer effect and structural characterization of endo-polysaccharide from cultivated mycelia of Inonotus obliquus. Life Sci. 2006, 79, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Taji, S.; Yamada, T.; Wada, S.; Tokuda, H.; Sakuma, K.; Tanaka, R. Lanostane-type triterpenoids from the sclerotia of Inonotus obliquus possessing anti-tumor promoting activity. Eur. J. Med. Chem. 2008, 43, 2373–2379. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Park, J.; Min, B.S.; Shim, S.H. Chemical Constituents from the Sclerotia of Inonotus obliquus. J. Korean Soc. Appl Bi. 2011, 54, 287–294. [Google Scholar] [CrossRef]
- Zjawiony, J.K. Biologically active compounds from aphyllophorales (polypore) fungi. J. Nat. Prod. 2004, 67, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Fazenda, M.L.; Seviour, R.; McNeil, B.; Harvey, L.M. Submerged culture fermentation of “higher fungi”: The macrofungi. Adv. Appl. Microbiol. 2008, 63, 33–103. [Google Scholar] [PubMed]
- Mohamed, H.N.; Man, Y.C.; Mustafa, S.; Manap, Y.A. Tentative identification of volatile flavor compounds in commercial Budu, A Malaysian fish sauce, Using GC-MS. Molecules 2012, 17, 5062–5080. [Google Scholar] [CrossRef] [PubMed]
- Bukvicki, D.; Gottardi, D.; Veljic, M.; Marin, P.D.; Vannini, L.; Guerzoni, M.E. Identification of volatile components of liverwort (Porella cordaeana) extracts using GC/MS-SPME and their antimicrobial activity. Molecules 2012, 17, 6982–6995. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Tang, X.; Zhang, Y.; Zhao, W. Determination of the volatile composition in brown millet, milled millet and millet bran by gas chromatography/mass spectrometry. Molecules 2012, 17, 2271–2282. [Google Scholar] [CrossRef] [PubMed]
- Careri, M.; Mangia, A.; Musci, M. Overview of the applications of liquid chromatography–mass spectrometry interfacing systems in food analysis: Naturally occurring substances in food. J. Chromatogr. A 1998, 794, 263–297. [Google Scholar] [CrossRef]
- Xing, J.; Xie, C.; Lou, H. Recent applications of liquid chromatography-mass spectrometry in natural products bioanalysis. J. Pharm. Biomed. Anal. 2007, 44, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Medina, I.C.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Use of high-performance liquid chromatography with diode array detection coupled to electrospray-Qq-time-of-flight mass spectrometry for the direct characterization of the phenolic fraction in organic commercial juices. J. Chromatogr. A 2009, 1216, 4736–4744. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.D.; Zhou, H.C.; Lin, Y.M. Antioxidant Activities of Extract and Fractions from the Hypocotyls of the Mangrove Plant Kandelia candel. Int. J. Mol. Sci. 2010, 11, 4080–4093. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.D.; Zhou, H.C.; Lin, Y.M. Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa. Int. J. Mol. Sci. 2011, 12, 1146–1160. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Tamai, Y.; Terazawa, M. Chemical constituents of Inonotus obliquus II: A new triterpene, 21,24-cyclopentalanosta-3 beta,21,25-triol-8-ene from sclerotium. J. Wood Sci. 2001, 47, 313–316. [Google Scholar] [CrossRef]
- Nakajima, Y.; Sato, Y.; Konishi, T. Antioxidant small phenolic ingredients in Inonotus obliquus (persoon) Pilat (Chaga). Chem. Pharm. Bull. 2007, 55, 1222–1226. [Google Scholar] [CrossRef] [PubMed]
- Zhukovich, E.N.; Semenova, M.Y.; Sharikova, L.A.; Pribytkova, T.F. Tetracyclic triterpenes from Inonotus obliquus (PERS.) PIL. (CHAGA) growing in Russia. Pharm. Chem. J. 2010, 44, 495–496. [Google Scholar] [CrossRef]
- Sun, Y.; Yin, T.; Chen, X.H.; Zhang, G.; Curtis, R.B.; Lu, Z.H.; Jiang, J.H. In Vitro Antitumor Activity and Structure Characterization of Ethanol Extracts from Wild and Cultivated Chaga Medicinal Mushroom, Inonotus obliquus (Pers.:Fr.) Pilat (Aphyllophoromycetideae). Int. J. Med. Mushrooms 2011, 13, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Jin, C.; Lee, S.L. Antioxidative activities of phenolic compounds isolated from Inonotus obliquus. Kor. J. Pharmacogn. 2007, 38, 164–169. [Google Scholar]
- Morris, G.M.; Lim-Wilby, M. Molecular docking. Methods Mol. Biol. 2008, 443, 365–382. [Google Scholar] [PubMed]
- Xu, H.Y.; Sun, J.E.; Lu, Z.M.; Zhang, X.M.; Dou, W.F.; Xu, Z.H. Beneficial effects of the ethanol extract from the dry matter of a culture broth of Inonotus obliquus in submerged culture on the antioxidant defence system and regeneration of pancreatic -cells in experimental diabetes in mice. Nat. Prod. Res. 2010, 24, 542–553. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Pang, C.; Yang, C.J.; Zheng, Y.T.; Xu, H.Y.; Lu, Z.M.; Xu, Z.H. Antihyperglycemic and Antilipidperoxidative Effects of Polysaccharides Extracted from Medicinal Mushroom Chaga, Inonotus obliquus (Pers.: Fr.) Pilat (Aphyllophoromycetideae) on Alloxan-Diabetes Mice. Int. J. Med. Mushrooms 2010, 12, 235–244. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
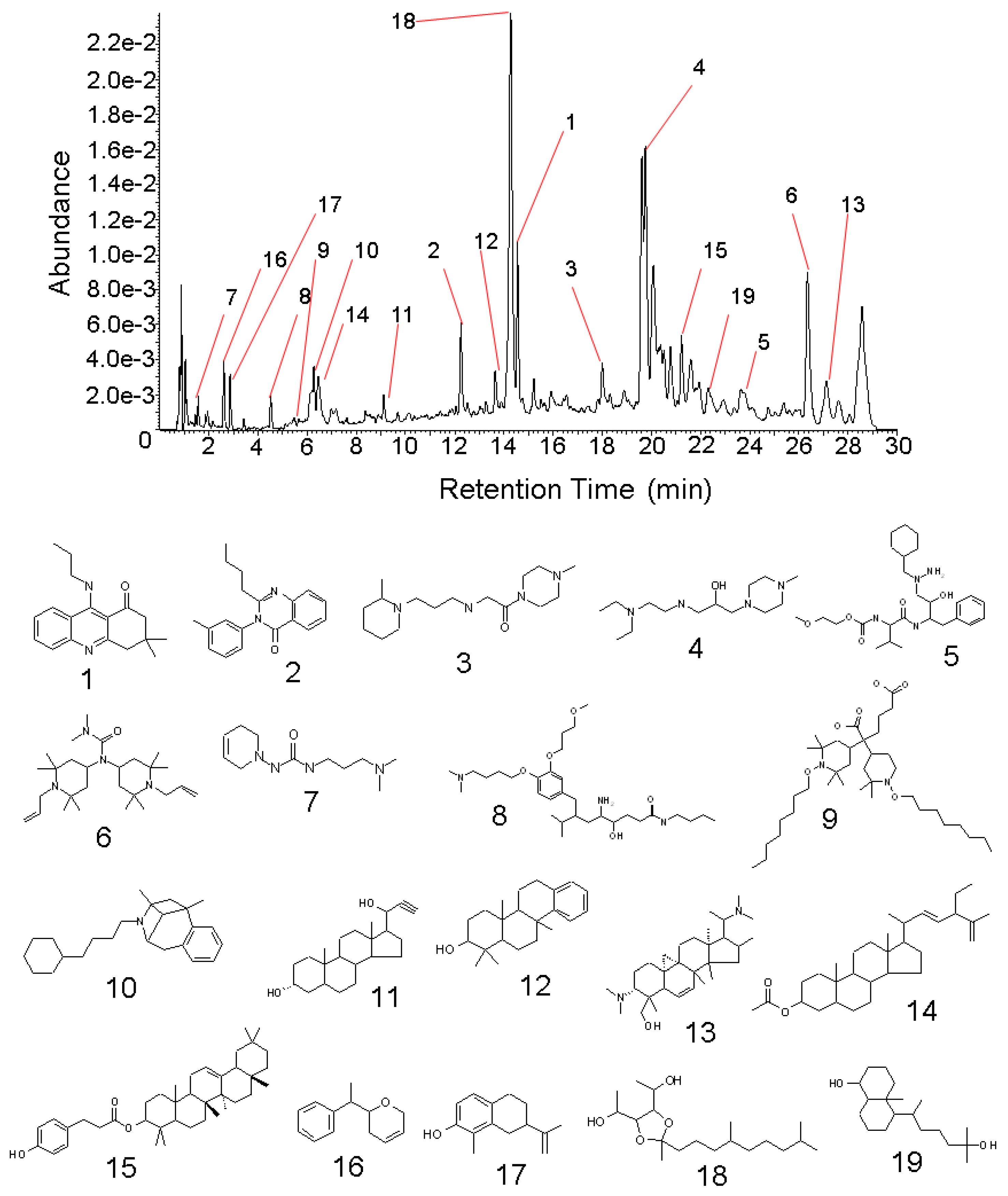
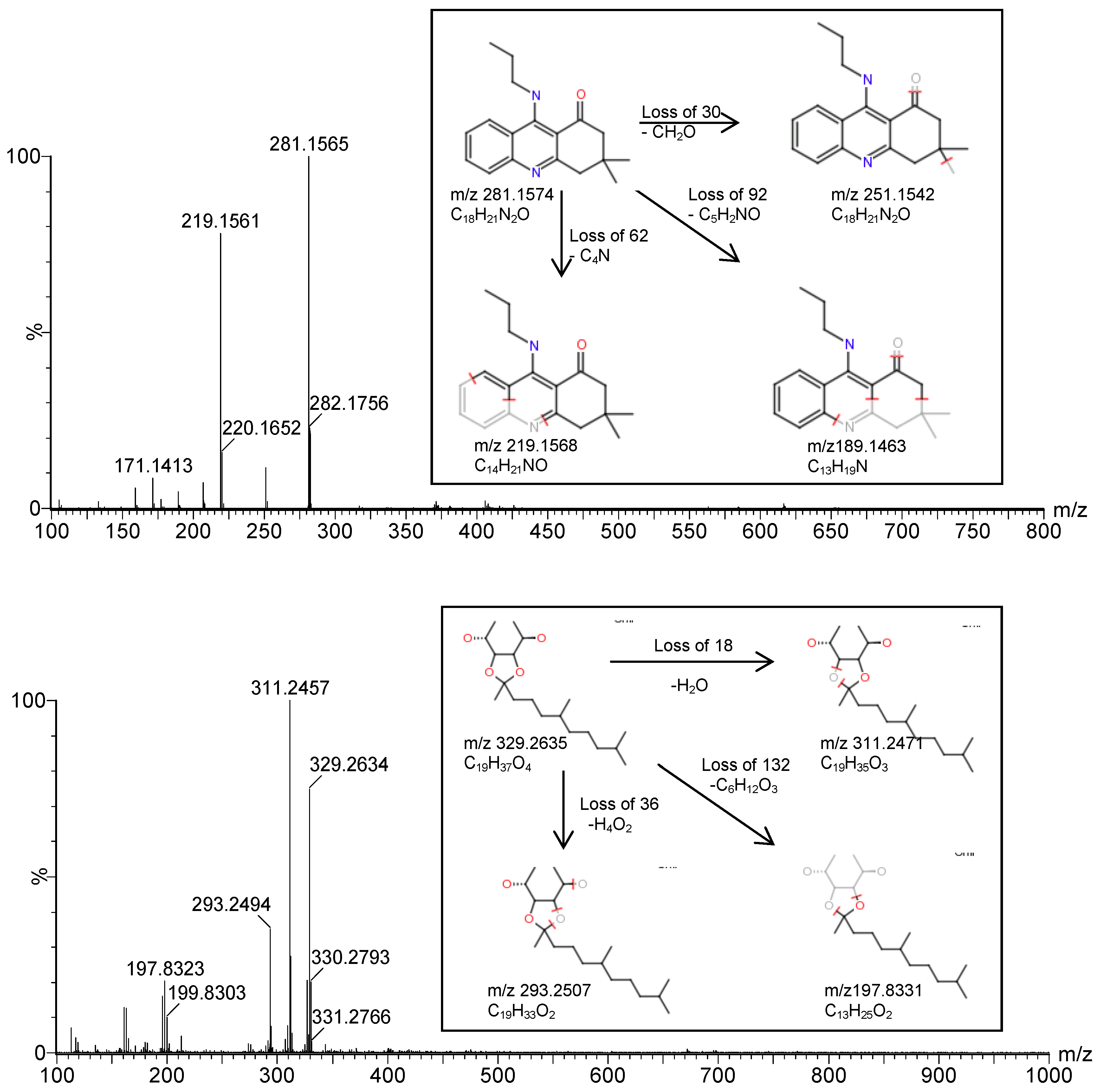
| Compound | Experimental mass (m/z) | Theoretical mass (m/z) | Mass error | DBE | Molecular formula | i-FIT | i-FIT (Norm) | |
|---|---|---|---|---|---|---|---|---|
| mDa ppm | ||||||||
| 1 | 281.1565 | 281.1574 | −0.9 | −3.2 | 9.5 | C18H21N2O | 150.0 | 0.0 |
| 2 | 291.1724 | 291.1737 | −1.3 | −4.4 | 10.5 | C19H19N2O | 46.1 | 0.3 |
| 3 | 295.2505 | 295.2498 | 0.7 | 2.4 | 3.5 | C16H31N4O | 166.9 | 0.0 |
| 4 | 271.2498 | 271.2498 | 0.0 | 0.0 | 0.5 | C14H31N4O | 79.0 | 0.0 |
| 5 | 491.3223 | 491.3233 | −1.0 | −2.0 | 7.5 | C26H43N4O5 | 54.6 | 0.3 |
| 6 | 445.3879 | 445.3893 | −1.4 | −3.1 | 5.5 | C27H49N4O | 69.2 | 1.0 |
| 7 | 225.1876 | 225.1872 | 0.4 | 1.8 | 2.5 | C11H21N4O | 40.3 | 0.4 |
| 8 | 564.4618 | 564.4614 | 0.4 | 0.7 | -0.5 | C32H57N3O5 | 50.4 | 0.0 |
| 9 | 677.5576 | 677.5571 | 0.5 | 0.7 | 6.5 | C40H73N2O6 | 36.9 | 0.0 |
| 10 | 338.2864 | 338.2848 | 1.6 | 4.7 | 7.5 | C24H36N | 127.2 | 0.0 |
| 11 | 329.2453 | 329.2467 | −1.4 | −4.7 | 6.5 | C22H33O2 | 53.0 | 0.7 |
| 12 | 311.2392 | 311.2375 | 1.5 | 4.8 | 7.5 | C22H31O | 173.1 | 1.2 |
| 13 | 443.3663 | 443.3648 | 1.5 | 3.3 | 6.5 | C28H47N2O2 | 100.2 | 0.0 |
| 14 | 456.3778 | 456.3760 | 1.8 | 3.9 | 7.5 | C31H47O2 | 42.9 | 0.0 |
| 15 | 573.4431 | 573.4453 | −2.2 | −3.8 | 10.5 | C39H57O3 | 36.9 | 0.4 |
| 16 | 187.1162 | 187.1156 | 0.6 | 3.5 | 6.5 | C13H15O | 96.8 | 0.4 |
| 17 | 201.1278 | 201.1279 | −0.1 | −0.5 | 6.5 | C14H17O | 41.4 | 0.3 |
| 18 | 329.2634 | 329.2635 | −0.1 | −0.3 | 1.5 | C19H37O4 | 109.7 | 0.1 |
| 19 | 295.2632 | 295.2637 | −0.5 | −1.7 | 2.5 | C19H35O2 | 121.3 | 0.1 |
| Compound | Molecular Weight | Energy (kJ/mol) | RMSD |
|---|---|---|---|
| Sitagliptin | 505.3 | −90.2814 | 1.21861 |
| 1 | 281.2 | −54.5608 | 2.20387 |
| 2 | 291.2 | −63.6279 | 4.57834 |
| 3 | 295.2 | −67.4692 | 2.77483 |
| 4 | 271.2 | −61.6337 | 6.21484 |
| 5 | 491.3 | −105.071 | 1.47814 |
| 6 | 445.4 | −64.0713 | 10.2671 |
| 7 | 225.2 | −67.2371 | 6.84517 |
| 8 | 564.5 | −113.391 | 4.66248 |
| 9 | 677.6 | −86.1653 | 7.27826 |
| 10 | 338.3 | −70.4691 | 3.83635 |
| 11 | 329.2 | −60.2719 | 6.73284 |
| 12 | 311.2 | −51.5762 | 3.88163 |
| 13 | 443.4 | −61.9833 | 4.26127 |
| 14 | 456.4 | −85.7413 | 1.27186 |
| 15 | 573.4 | −85.1947 | 2.18643 |
| 16 | 187.1 | −48.9167 | 3.37182 |
| 17 | 201.1 | −52.3578 | 0.087938 |
| 18 | 329.3 | −73.6836 | 8.26177 |
| 19 | 295.3 | −59.8366 | 4.57368 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Geng, Y.; Lu, Z.-M.; Huang, W.; Xu, H.-Y.; Shi, J.-S.; Xu, Z.-H. Bioassay-Guided Isolation of DPP-4 Inhibitory Fractions from Extracts of Submerged Cultured of Inonotus obliquus. Molecules 2013, 18, 1150-1161. https://doi.org/10.3390/molecules18011150
Geng Y, Lu Z-M, Huang W, Xu H-Y, Shi J-S, Xu Z-H. Bioassay-Guided Isolation of DPP-4 Inhibitory Fractions from Extracts of Submerged Cultured of Inonotus obliquus. Molecules. 2013; 18(1):1150-1161. https://doi.org/10.3390/molecules18011150
Chicago/Turabian StyleGeng, Yan, Zhen-Ming Lu, Wei Huang, Hong-Yu Xu, Jin-Song Shi, and Zheng-Hong Xu. 2013. "Bioassay-Guided Isolation of DPP-4 Inhibitory Fractions from Extracts of Submerged Cultured of Inonotus obliquus" Molecules 18, no. 1: 1150-1161. https://doi.org/10.3390/molecules18011150
APA StyleGeng, Y., Lu, Z.-M., Huang, W., Xu, H.-Y., Shi, J.-S., & Xu, Z.-H. (2013). Bioassay-Guided Isolation of DPP-4 Inhibitory Fractions from Extracts of Submerged Cultured of Inonotus obliquus. Molecules, 18(1), 1150-1161. https://doi.org/10.3390/molecules18011150




