Antioxidant and Antimicrobial Activities of 7-Hydroxy-calamenene-Rich Essential Oils from Croton cajucara Benth.
Abstract
:1. Introduction
2. Results and Discussion
| Components | Samples (in%) | ||||
|---|---|---|---|---|---|
| SV001 | SV002 | SV003 | SV004 | SV005 | |
| a-Pinene | 7.5 | 24.7 | 0.1 | 0.5 | t |
| Linalool | 6.3 | 11.6 | 11.0 | 9.9 | 13.2 |
| 7-Hydroxycalamenene | 37.5 | n.d. | 28.4 | 30.9 | 32.9 |
| b-Caryophyllene | 4.1 | 5.7 | 2.8 | 4.0 | 2.6 |
| Microorganisms | SV001 | SV002 | SV003 | SV004 | SV005 |
|---|---|---|---|---|---|
| A. fumigatus | 4.8 | 3.2 | 4.5 | 4.6 | 5 |
| A. niger | 5.7 | 3.4 | 5.6 | 5.9 | 6 |
| A. ochraceus | 4.5 | 3.1 | 4.5 | 4.7 | 5 |
| F. solani | 3.1 | 2.2 | 3.4 | 3.5 | 3.5 |
| M. gypseum | 14 | 3 | 13 | 14 | 14 |
| M. circinelloides | 8 | 15 | 6 | 10 | 10 |
| R. oryzae | 6 | 12 | 5 | 8 | 10 |
| T. rubrum | 5.8 | 0 | 5.5 | 5.7 | 6 |
| M. smegmatis | 10 | 24 | 12 | 18 | 18 |
| E. faecalis | 10 | 6 | 8 | 6 | 9 |
| S. epidermidis | 6 | 9 | 7 | 8 | 20 |
| S. aureus MRSA | 7 | 5 | 7 | 13 | 38 |
| L. casei | 8 | 7 | 35 | 11 | 14 |
| C. albicans | 6 | 10 | 7 | 8 | 8 |
| Microorganisms | SV001 | SV002 | SV003 | SV004 | SV005 | 7-OH |
|---|---|---|---|---|---|---|
| M. smegmatis | 39.06 | 5000 | 78.12 | 156.25 | 156.25 | 39.06 |
| M. tuberculosis | 4.88 | 4.88 | 4.88 | 4.88 | 4.88 | 312.5 |
| MRSA | 0.019 | na | 0.019 | 0.004 | 0.001 | 39.06 |
| C. albicans | 1.22 | 1250 | 156.25 | 0.001 | 0.038 | 78.125 |
| M. circinelloides | nd | nd | nd | nd | 3.63 × 10−8 | 19.53 |
| R. oryzae | nd | nd | nd | nd | 0.152 | 39.06 |
 ) and EC50 (
) and EC50 (  ) of standards, essential oils and 7-hydroxycalamenene.
) of standards, essential oils and 7-hydroxycalamenene.
 ) and EC50 (
) and EC50 (  ) of standards, essential oils and 7-hydroxycalamenene.
) of standards, essential oils and 7-hydroxycalamenene.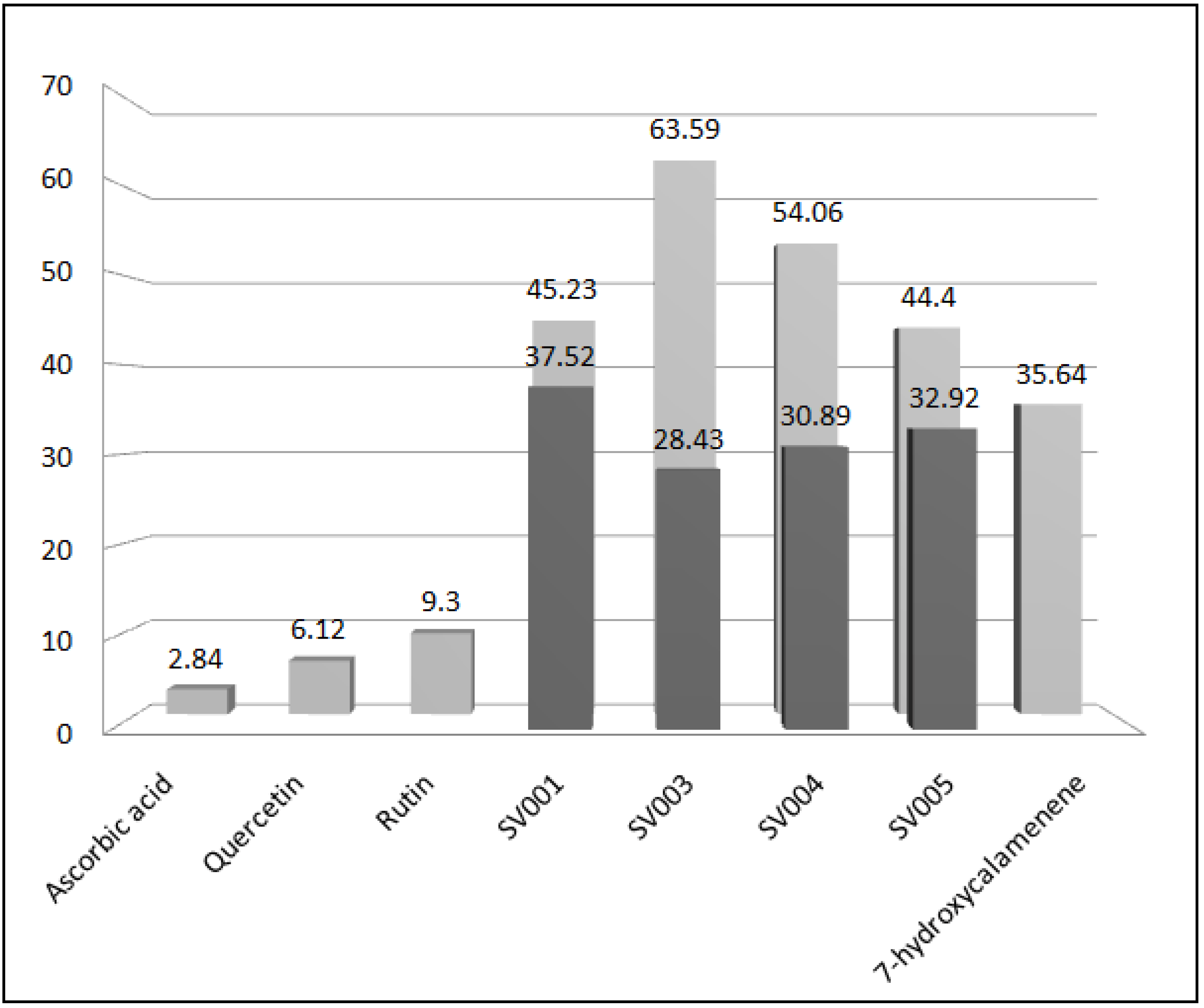
3. Experimental
3.1. Plant Material
3.2. Chemicals
3.3. Extraction and Analysis of the Essential Oils
3.4. Microorganisms
3.5. Antimicrobial Analysis of the Essential Oils
3.6. Thin-layer Chromatography (TLC) and Bioautography
3.7. Minimal Inhibitory Concentration (MIC) and Minimum Microbicidal/Microbiostatic Concentration Assays
3.8. Antioxidant Activity
4. Conclusions
Acknowledgments
References
- Hiruma-Lima, C.A.; Gracioso, J.S.; Bighetti, E.J.B.; Grassi-Kassisse, D.M.; Nunes, D.S.; Souza Brito, A.R.M. Effect of essential oil obtained from Croton cajucara Benth. on gastric ulcer healing and protective factors of the gastric mucosa. Phytomedicine 2002, 9, 523–529. [Google Scholar] [CrossRef]
- Perazzo, F.F.; Carvalho, J.C.T.; Rodrigues, M.; Morais, E.K.L.; Maciel, M.A.M. Comparative anti-inflammatory and antinociceptive effects of terpenoids and an aqueous extract obtained from Croton cajucara Benth. Rev. Bras. Farmacogn. 2007, 17, 521–528. [Google Scholar]
- Chaves, F.C.M.; Bizzo, H.R.; Angelo, P.M.C.; Xavier, J.J.B.N.; Sá-Sobrinho, A.F. Rendimento e Composição Química do Óleo Essencial de Folha de Dois Morfotipos de Sacaca (Croton cajucara Benth.). Rev. Bras. Pl. Med. 2006, 8, 117–119. [Google Scholar]
- Pereira, A.Q.; Chaves, F.C.M.; Pinto, S.C.; Leitão, S.G.; Bizzo, H.R. Isolation and Identification of cis-7-Hydroxycalamenene from the Essential Oil of Croton cajucara Benth. J. Essent. Oil Res. 2011, 23, 20–23. [Google Scholar]
- Rosa, M.S.C.S.; Mendonça-Filho, R.R.; Bizzo, H.R.; Rodrigues, I.A.; Soares, R.M.A.; Souto-Padrón, T.; Alviano, C.S.; Lopes, A.H.C.S. Antileishmanial activity of linalool-rich essential oil from Croton cajucara Benth. Antimicrob. Agents Chemother. 2003, 47, 1895–1901. [Google Scholar] [CrossRef]
- Alviano, W.S.; Mendonça-Filho, R.R.; Alviano, D.S.; Bizzo, H.R.; Souto-Padrón, T.; Rodrigues, M.L.; Bolognese, A.M.; Alviano, C.S.; Souza, M.M.G. Antimicrobial activity of Croton cajucara Benth. linalool-rich essential oil on artificial biofilms and planktonic microorganisms. Oral Microbiol. Immunol. 2005, 20, 101–105. [Google Scholar] [CrossRef]
- Tullio, V.; Nostro, A.; Mandras, N.; Dugo, P.; Banche, G.; Cannatelli, M.A.; Cuffini, A.M.; Alonzo, V.; Carlone, N.A. Antifungal activity of essential oils against filamentous fungi determined by broth microdilution and vapour contact methods. J. Appl. Microbiol. 2006, 102, 1544–1550. [Google Scholar]
- Sun, Q.N.; Najvar, L.K.; Bocanegra, R.; Loebenberg, D.; Graybill, J.R. In vivo activity of posaconazole against Mucor spp. in an immunosuppressed-mouse model. Antimicrob. Agents Chemother. 2002, 46, 2310–2312. [Google Scholar] [CrossRef]
- Dannaoui, E.; Meis, J.F.G.M.; Loebenberg, D.; Verweij, P.E. Activity of posaconazole in treatment of experimental disseminated zygomycosis. Antimicrob. Agents Chemother. 2003, 47, 3647–3650. [Google Scholar] [CrossRef]
- Azevedo, M.M.B.; Pereira, A.Q.; Chaves, F.C.M.; Bizzo, H.R.; Alviano, C.S.; Alviano, D.S. Antimicrobial activity of the essential oils from the leaves of two morphotypes of Croton cajucara Benth. J. Essent. Oil Res. 2012, 24, 351–357. [Google Scholar] [CrossRef]
- Stephan, J.; Mailaender, C.; Etienne, G.; Daffe, M.; Niederweis, M. Multidrug resistance of a porin deletion mutant of Mycobacterium smegmatis. Antimicrob. Agents Chemother. 2004, 48, 4163–4170. [Google Scholar] [CrossRef]
- Wiid, I.; Van Helden, E.H.; Hon, D.; Lombard, C.; Van Helden, P. Potentiation of isoniazid activity against Mycobacterium tuberculosis by melatonin. Antimicrob. Agents Chemother. 1999, 43, 975–977. [Google Scholar]
- Maus, C.E.; Plikaytis, B.B.; Shinnick, T.M. Molecular analysis of cross-resistance to capreomycin, kanamycin, amikacin, and viomycin in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2005, 49, 3192–3197. [Google Scholar] [CrossRef]
- Shandil, R.K.; Jayaram, R.; Kaur, P.; Gaonkar, S.; Suresh, B.L.; Mahesh, B.N.; Jayashree, R.; Nandi, V.; Bharath, S.; Balasubramanian, V. Moxifloxacin, ofloxacin, sparfloxacin, and ciprofloxacin against Mycobacterium tuberculosis: evaluation of in vitro and pharmacodynamic indices that best predict in vivo efficacy. Antimicrob. Agents Chemother. 2007, 51, 576–582. [Google Scholar] [CrossRef]
- Sartoratto, A.; Machado, A.L.M.; Delarmelina, C.; Figueira, G.M.; Duarte, C.T.; Rehder, V.L. Composition and antimicrobial activity of essential oils from aromatic plants used in Brazil. Braz. J. Microbiol. 2004, 35, 275–280. [Google Scholar] [CrossRef]
- van Den Dool, H.; Kratz, P.D.J.A. Generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed; Allured Business Media: Carol Stream, IL, USA, 2007. [Google Scholar]
- Cruz, M.C.S.; Santos, P.O.; Barbosa, A.M., Jr.; Melo, D.L.F.M.; Alviano, C.S.; Antoniolli, A.R.; Alviano, D.S.; Trindade, R.C. Antifungal activity of Brazilian medicinal plants involved in popular treatment of mycoses. J. Ethnopharmacol. 2007, 111, 409–412. [Google Scholar] [CrossRef]
- Subha, T.S.; Gnanamani, A. Effect of active fraction of methanolic extract of Acorus calamus on sterol metabolism of Candida albicans. J. Appl. Biosci. 2008, 8, 243–250. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI), Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Approved standard, M27-A2, CLSI: Wayne, PA, USA, 2002.
- Clinical and Laboratory Standards Institute (CLSI), Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; approved standard M38-A, CLSI: Wayne, PA, USA, 2002.
- Clinical and Laboratory Standards Institute (CLSI), Susceptibility Testing of Mycobacteria, Nocardiae, and Other Aerobic Actinomycetes; approved standard M24-A, CLSI: Wayne, PA, USA, 2011.
- Clinical and Laboratory Standards Institute (CLSI), Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; approved standard M7-A6, CLSI: Wayne, PA, USA, 2006.
- Khan, R.; Islam, B.; Akram, M.; Shakil, S.; Ahmad, A.; Ali, S.M.; Siddiqui, M.; Khan, A.U. Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules 2009, 14, 586–597. [Google Scholar] [CrossRef]
- Calvin, A.; Potterat, O.; Wolfender, J.L.; Hostettmann, K.; Dyatmyko, W. Use of On-flow LC/1H NMR for the study of an antioxidant fraction from Orophea enneandra and isolation of a polyacetylene, lignans, and a tocopherol derivative. J. Nat. Prod. 1998, 61, 1497–1501. [Google Scholar] [CrossRef]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; Santos, T.C.; Coube, C.S.; Leitão, S.G. Screening of brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Azevedo, M.M.B.; Chaves, F.C.M.; Almeida, C.A.; Bizzo, H.R.; Duarte, R.S.; Campos-Takaki, G.M.; Alviano, C.S.; Alviano, D.S. Antioxidant and Antimicrobial Activities of 7-Hydroxy-calamenene-Rich Essential Oils from Croton cajucara Benth. Molecules 2013, 18, 1128-1137. https://doi.org/10.3390/molecules18011128
Azevedo MMB, Chaves FCM, Almeida CA, Bizzo HR, Duarte RS, Campos-Takaki GM, Alviano CS, Alviano DS. Antioxidant and Antimicrobial Activities of 7-Hydroxy-calamenene-Rich Essential Oils from Croton cajucara Benth. Molecules. 2013; 18(1):1128-1137. https://doi.org/10.3390/molecules18011128
Chicago/Turabian StyleAzevedo, Mariana M. B., Francisco C. M. Chaves, Catia A. Almeida, Humberto R. Bizzo, Rafael S. Duarte, Galba M. Campos-Takaki, Celuta S. Alviano, and Daniela S. Alviano. 2013. "Antioxidant and Antimicrobial Activities of 7-Hydroxy-calamenene-Rich Essential Oils from Croton cajucara Benth." Molecules 18, no. 1: 1128-1137. https://doi.org/10.3390/molecules18011128
APA StyleAzevedo, M. M. B., Chaves, F. C. M., Almeida, C. A., Bizzo, H. R., Duarte, R. S., Campos-Takaki, G. M., Alviano, C. S., & Alviano, D. S. (2013). Antioxidant and Antimicrobial Activities of 7-Hydroxy-calamenene-Rich Essential Oils from Croton cajucara Benth. Molecules, 18(1), 1128-1137. https://doi.org/10.3390/molecules18011128




