Antiprotozoal Activity of Quinonemethide Triterpenes from Maytenus ilicifolia (Celastraceae)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Maytenin (1) and Pristimerin (2): Leishmanicidal, Trypanocidal and Cytotoxic Activities against Extracellular Parasites and Macrophages
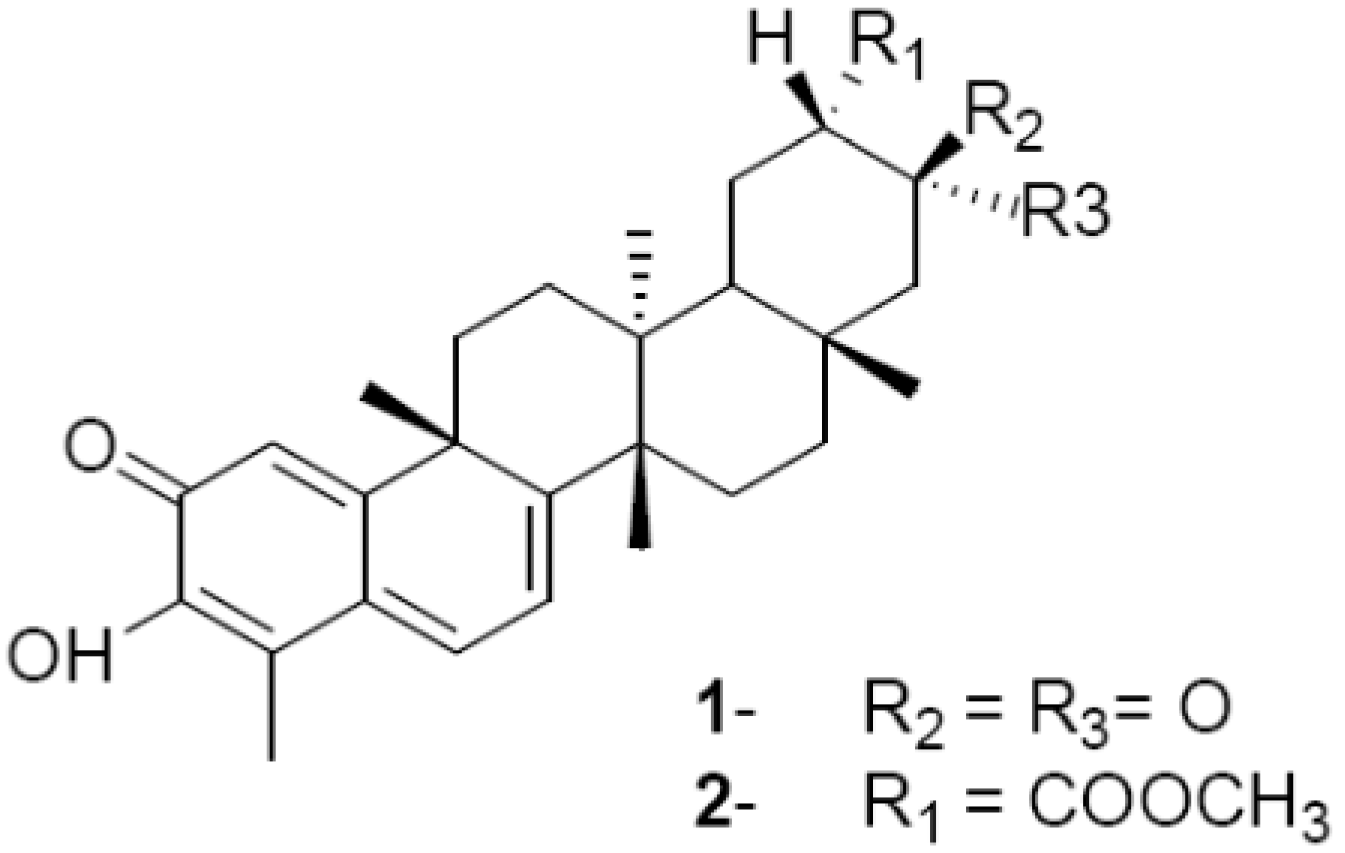
| compound | L. amazonensis (IC50) | L. chagasi (IC50) | T. cruzi (IC50) | BALB/c (LC50) | ||
|---|---|---|---|---|---|---|
| promastigotes | amastigotes | promastigotes | amastigotes | epimastigotes | macrophages | |
| maytenin | 0.09 (243.65) | 0.47 (45.21) | 0.46 (46.41) | 0.25 (85) | 0.25 (85) | 21.25 |
| pristimerin | 0.05 (193.63) | 0.88 (11.03) | 0.41 (23.85) | 0.43 (22.65) | 0.30 (32.37) | 9.71 |
| control | 6.75 a (3.0) | 9.30 (2.18) | 6.75 a | n.d. | 31.20 × 103 | 20.32 a |
2.2. Leishmanicidal Activity of Compounds 1 and 2 on Intracellular Parasites
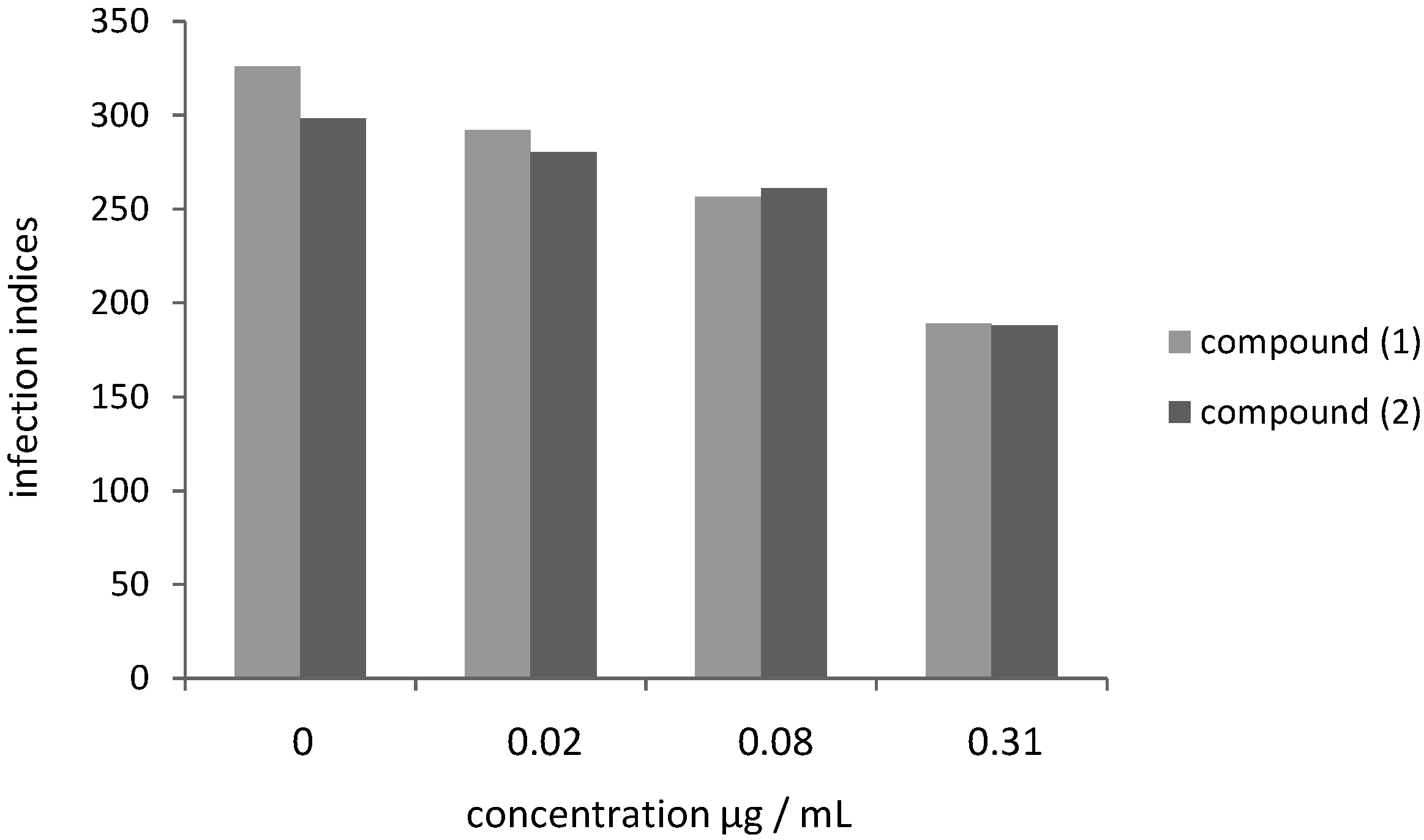
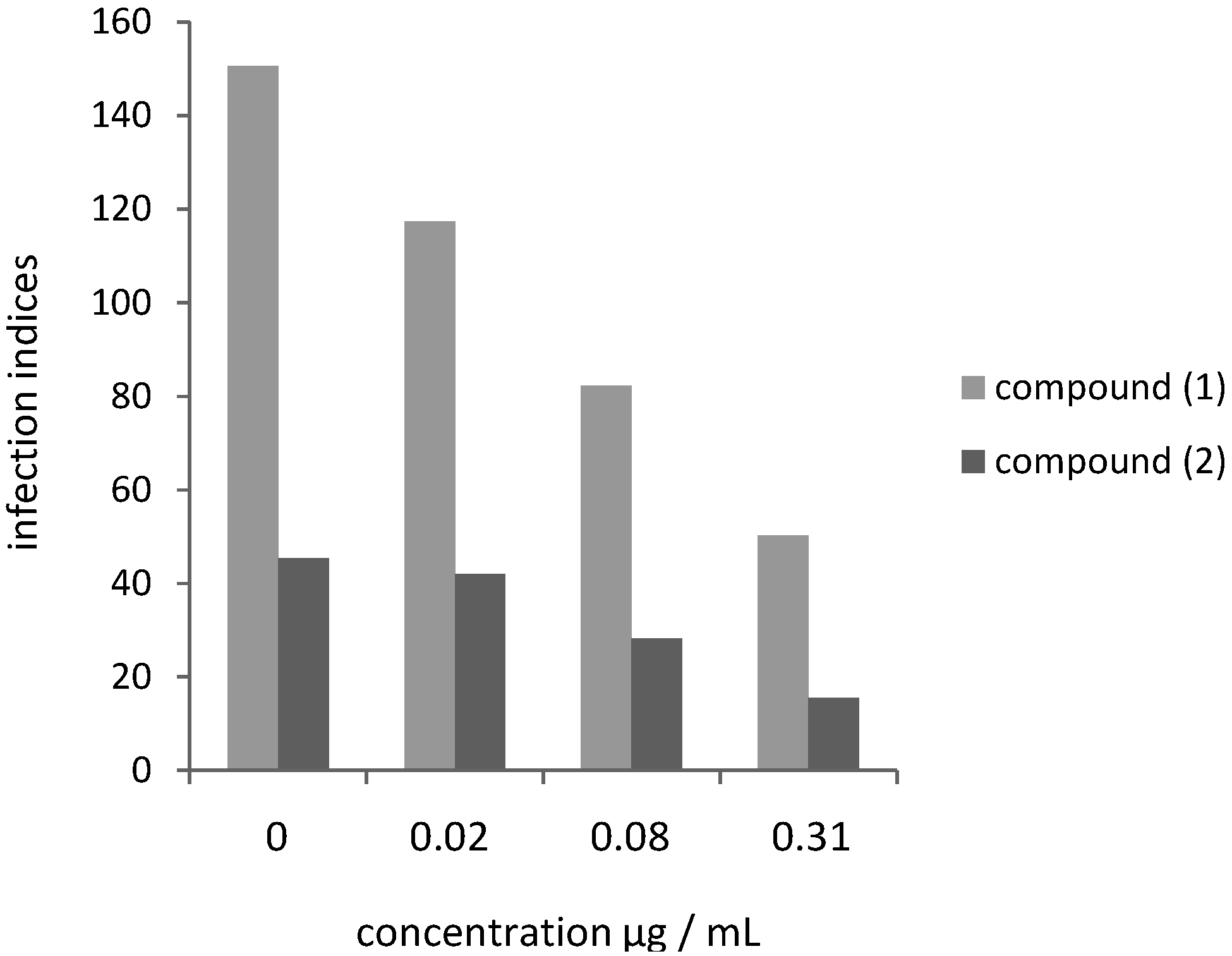
3. Experimental
3.1. General Methods
3.2. Plant Material
3.3. Extraction and Isolation of Quinonemethide Triterpenes from Maytenus ilicifolia
3.4. Parasite Culture
3.5. Evaluation of Leishmanicidal Activity
3.5.1. Promastigotes
3.5.2. Cytotoxicity on Macrophages
3.5.3. Amastigotes
3.6. Evaluation of Trypanocidal Activity
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the compounds 1 and 2 are available from the authors.
References
- Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; den Boer, M.; Team, W.L.C. Leishmaniasis worldwide and global estimates of its incidence. PLoS One 2012, 7, e35671. [Google Scholar]
- Rassi, A.; de Rezende, M. Leishmaniasis worldwide and global estimates of its incidence. Infect. Dis. Clin. N. Am. 2012, 26, 275–291. [Google Scholar] [CrossRef]
- Chawla, B.; Madhubala, R. Drug targets in Leishmania. J. Parasit. Dis. 2010, 34, 1–13. [Google Scholar] [CrossRef]
- Vinhaes, M.C.; Dias, J.C. Chagas disease in Brazil. Cad. Saude Publica 2000, 16, S7–S12. [Google Scholar] [CrossRef]
- Croft, S.L.; Brun, R. In Vitro and in Vivo Models for the Identification and Evaluation of Drugs Active against Trypanosoma and Leishmania. In Drugs against Parasitic Diseases: R&D Methodologies and Issues (Discoveries and Drug Development); Fairlamb, A.H., Ridley, R.G., Vial, H.J., Eds.; World Health Organization, Special Programme for Research and Training in Tropical Diseases: Geneva, Switzerland, 2003; pp. 165–176. [Google Scholar]
- Da Silva Mota, J.; Leite, A.C.; Batista, J.M., Jr.; Noelí López, S.; Luz Ambrósio, D.; Duó Passerini, G.; Kato, M.J.; da Silva Bolzani, V.; Barretto Cicarelli, R.M.; Furlan, M. In vitro trypanocidal activity of phenolic derivatives from Peperomia obtusifolia. Planta Med. 2009, 75, 620–623. [Google Scholar] [CrossRef]
- De Mesquita, M.; Desrivot, J.; Bories, C.; Fournet, A.; de Paula, J.; Grellier, P.; Espindola, L. Antileishmanial and trypanocidal activity of Brazilian Cerrado plants. Mem. Inst. Oswaldo Cruz 2005, 100, 783–787. [Google Scholar] [CrossRef]
- Truiti, M.C.; Ferreira, I.C.; Zamuner, M.L.; Nakamura, C.V.; Sarragiotto, M.H.; Souza, M.C. Antiprotozoal and molluscicidal activities of five Brazilian plants. Braz. J. Med. Biol. Res. 2005, 38, 1873–1878. [Google Scholar] [CrossRef]
- Gunatilaka, A.A.L. Triterpenoid Quinonemethides and Related Compounds (Celastroloids); Springer-Verlag: New York, NY, USA, 1996; Volume 67, pp. 1–123. [Google Scholar]
- Corsino, J.; de Carvalho, P.R.; Kato, M.J.; Latorre, L.R.; Oliveira, O.M.; Araújo, A.R.; Bolzani, V.D.; França, S.C.; Pereira, A.M.; Furlan, M. Biosynthesis of friedelane and quinonemethide triterpenoids is compartmentalized in Maytenus aquifolium and Salacia campestri. Phytochemistry 2000, 55, 741–748. [Google Scholar] [CrossRef]
- Peng, B.; Xu, L.; Cao, F.; Wei, T.; Yang, C.; Uzan, G.; Zhang, D. HSP90 inhibitor, celastrol, arrests human monocytic leukemia cell U937 at G0/G1 in thiol-containing agents reversible way. Mol. Cancer 2010, 9, 79. [Google Scholar]
- Dos Santos, V.A.; Dos Santos, D.P.; Castro-Gamboa, I.; Zanoni, M.V.; Furlan, M. Evaluation of antioxidant capacity and synergistic associations of quinonemethide triterpenes and phenolic substances from Maytenus ilicifolia (Celastraceae). Molecules 2010, 15, 6956–6973. [Google Scholar] [CrossRef]
- Lião, L.M.; Silva, G.A.; Monteiro, M.R.; Albuquerque, S. Trypanocidal activity of quinonemethide triterpenoids from Cheiloclinium cognatum (Hippocrateaceae). Z. Naturforsch. C 2008, 63, 207–210. [Google Scholar]
- Goijman, S.G.; Turrens, J.F.; Marini-Bettolo, G.B.; Stoppani, A.O. Inhibition of growth and macromolecular biosynthesis in Trypanosoma cruzi by natural products. Effects of miconidine and tingenone. Medicina (B Aires) 1984, 44, 361–370. [Google Scholar]
- Muelas-Serrano, S.; Nogal-Ruiz, J.J.; Gómez-Barrio, A. Setting of a colorimetric method to determine the viability of Trypanosoma cruzi epimastigotes. Parasitol. Res. 2000, 86, 999–1002. [Google Scholar] [CrossRef]
- Siqueira-Neto, J.L.; Song, O.R.; Oh, H.; Sohn, J.H.; Yang, G.; Nam, J.; Jang, J.; Cechetto, J.; Lee, C.B.; Moon, S.; et al. Antileishmanial high-throughput drug screening reveals drug candidates with new scaffolds. PLoS Negl. Trop. Dis. 2010, 4, e675. [Google Scholar] [CrossRef]
- Le Fichoux, Y.; Rousseau, D.; Ferrua, B.; Ruette, S.; Lelièvre, A.; Grousson, D.; Kubar, J. Short- and long-term efficacy of hexadecylphosphocholine against established Leishmania infantum infection in BALB/c mice. Antimicrob. Agents Chemother. 1998, 42, 654–658. [Google Scholar]
- Bollini, M.; Casal, J.J.; Alvarez, D.E.; Boiani, L.; González, M.; Cerecetto, H.; Bruno, A.M. New potent imidazoisoquinolinone derivatives as anti-Trypanosoma cruzi agents: Biological evaluation and structure-activity relationships. Bioorg. Med. Chem. 2009, 17, 1437–1444. [Google Scholar] [CrossRef]
- Croft, S.L.; Sundar, S.; Fairlamb, A.H. Drug resistance in leishmaniasis. Clin. Microbiol. Rev. 2006, 19, 111–126. [Google Scholar] [CrossRef]
- Escobar, P.; Matu, S.; Marques, C.; Croft, S.L. Sensitivities of Leishmania species to hexadecylphosphocholine (miltefosine), ET-18-OCH(3) (edelfosine) and amphotericin B. Acta Trop. 2002, 81, 151–157. [Google Scholar] [CrossRef]
- Miguel, D.C.; Yokoyama-Yasunaka, J.K.; Andreoli, W.K.; Mortara, R.A.; Uliana, S.R. Tamoxifen is effective against Leishmania and induces a rapid alkalinization of parasitophorous vacuoles harbouring Leishmania (Leishmania) amazonensis amastigotes. J. Antimicrob. Chemother. 2007, 60, 526–534. [Google Scholar] [CrossRef]
- Gunatilaka, A.A.L.; Fernando, C.H.; Kikuchi, T.; Tezuka, Y. 1H and 13C-NMR analysis of three quinone-methide triterpenoids. Magn. Reson. Chem. 1989, 27, 803–807. [Google Scholar] [CrossRef]
- Santos, V.A.; Regasini, L.O.; Nogueira, C.R.; Passerini, G.D.; Martinez, I.; Bolzani, V.S.; Graminha, M.A.; Cicarelli, R.M.; Furlan, M. Antiprotozoal sesquiterpene pyridine alkaloids from Maytenus ilicifolia. J. Nat. Prod. 2012, 75, 991–995. [Google Scholar] [CrossRef]
- Jeller, A.H.; Silva, D.H.; Lião, L.M.; Bolzani, V.A.S.; Furlan, M. Antioxidant phenolic and quinonemethide triterpenes from Cheiloclinium cognatum. Phytochemistry 2004, 65, 1977–1982. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dos Santos, V.A.F.F.M.; Leite, K.M.; Da Costa Siqueira, M.; Regasini, L.O.; Martinez, I.; Nogueira, C.T.; Galuppo, M.K.; Stolf, B.S.; Pereira, A.M.S.; Cicarelli, R.M.B.; et al. Antiprotozoal Activity of Quinonemethide Triterpenes from Maytenus ilicifolia (Celastraceae). Molecules 2013, 18, 1053-1062. https://doi.org/10.3390/molecules18011053
Dos Santos VAFFM, Leite KM, Da Costa Siqueira M, Regasini LO, Martinez I, Nogueira CT, Galuppo MK, Stolf BS, Pereira AMS, Cicarelli RMB, et al. Antiprotozoal Activity of Quinonemethide Triterpenes from Maytenus ilicifolia (Celastraceae). Molecules. 2013; 18(1):1053-1062. https://doi.org/10.3390/molecules18011053
Chicago/Turabian StyleDos Santos, Vania A. F. F. M., Karoline M. Leite, Mariana Da Costa Siqueira, Luis O. Regasini, Isabel Martinez, Camila T. Nogueira, Mariana Kolos Galuppo, Beatriz S. Stolf, Ana Maria Soares Pereira, Regina M. B. Cicarelli, and et al. 2013. "Antiprotozoal Activity of Quinonemethide Triterpenes from Maytenus ilicifolia (Celastraceae)" Molecules 18, no. 1: 1053-1062. https://doi.org/10.3390/molecules18011053
APA StyleDos Santos, V. A. F. F. M., Leite, K. M., Da Costa Siqueira, M., Regasini, L. O., Martinez, I., Nogueira, C. T., Galuppo, M. K., Stolf, B. S., Pereira, A. M. S., Cicarelli, R. M. B., Furlan, M., & Graminha, M. A. S. (2013). Antiprotozoal Activity of Quinonemethide Triterpenes from Maytenus ilicifolia (Celastraceae). Molecules, 18(1), 1053-1062. https://doi.org/10.3390/molecules18011053




