Corn Silk (Stigma Maydis) in Healthcare: A Phytochemical and Pharmacological Review
Abstract
:1. Introduction
2. Botanical Description
3. Phytochemical Composition
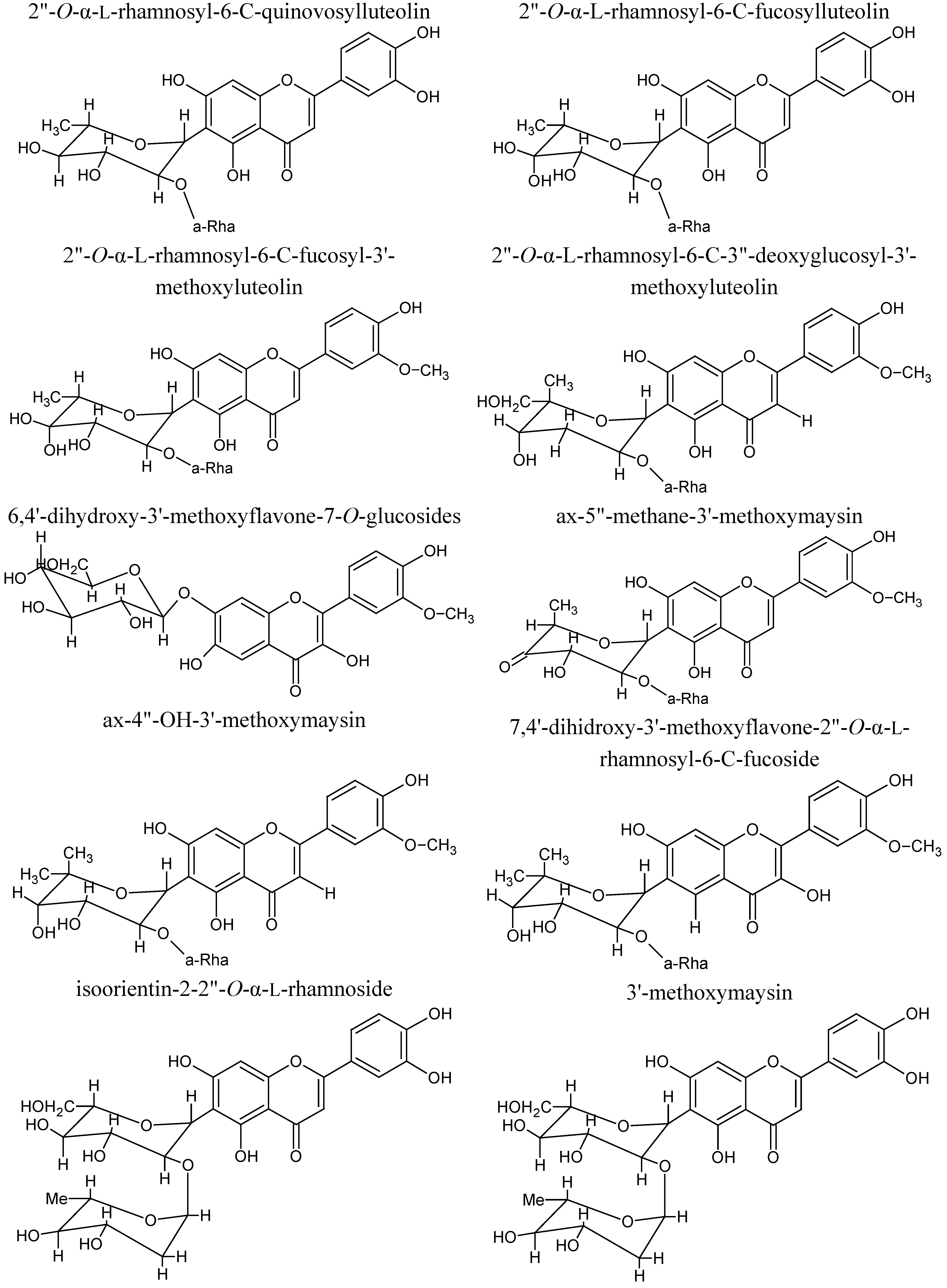
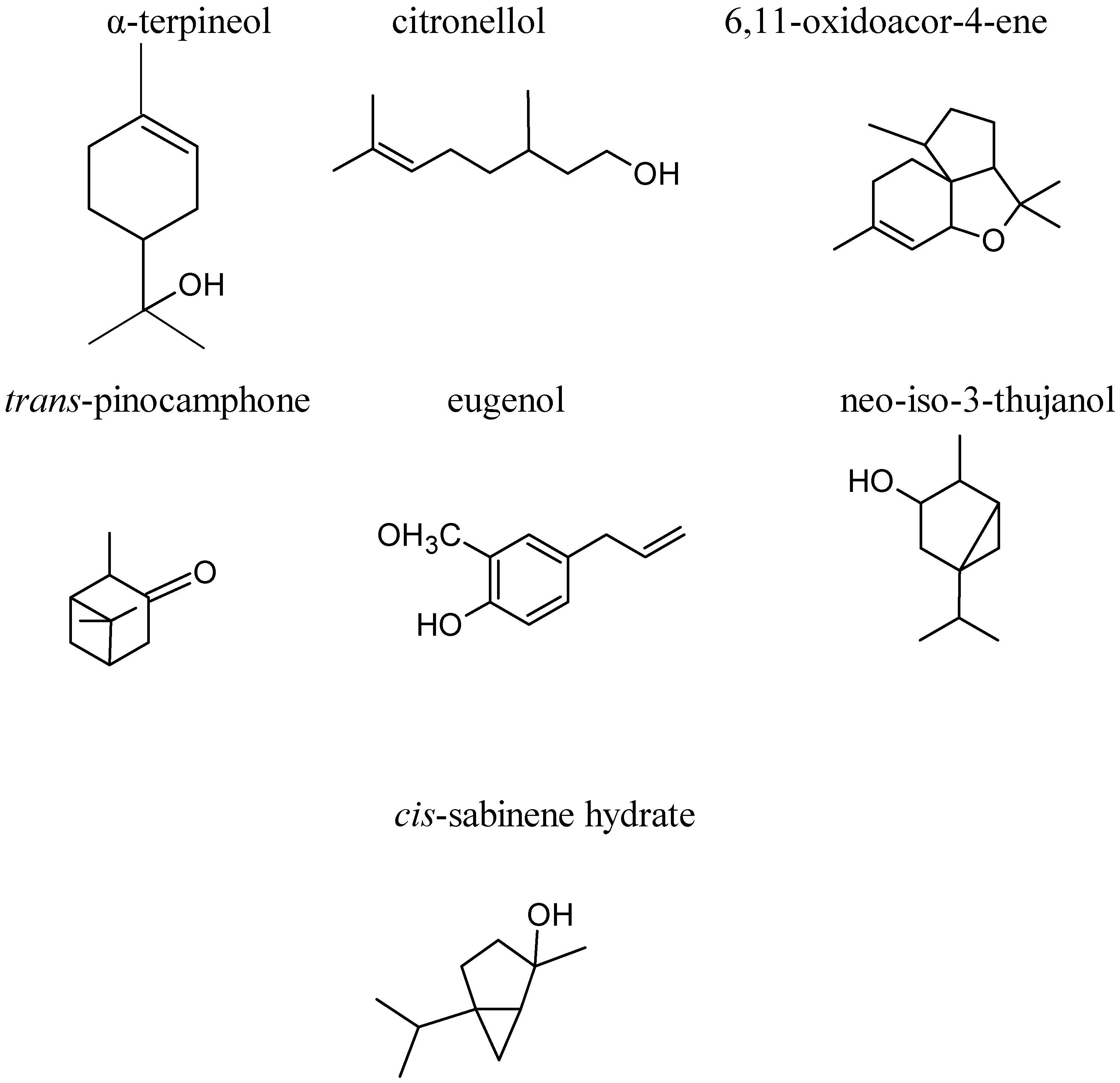
4. Pharmacological Studies for Potential Healthcare
4.1. Antioxidant Activity
4.2. Diuresis and Kaliuresis Effect
4.3. Hyperglycemia Reduction
4.4. Anti-depressant Activity
4.5. Anti-fatigue Activity
4.6. Anti-hyperlipidemic Effects
4.7. Anti-diabetic Effects
4.8. Nephrotoxicity Reduction
4.9. Anti-inflammatory Activity
4.10. Neuroprotective Effects
5. Toxicity
| IN VIVO STUDY | |||
|---|---|---|---|
| Pharmacological activity | Method | Results | References |
| Antioxidant activity | γ-Radiation induced oxidative stress in mice treated for 10 days. | Antioxidant activity against γ-radiation. | [15] |
| Exercise induced oxidative stress in mice treated for 28 days. | Antioxidant activity against oxidative stress during acute exercise. | [39] | |
| Diuresis and kaliuresis effect | Wistar rats were administered with CS extract by orogastric catherer and continuous urine collection for 3 and 5 h. | Exhibition of diuresis and kaliuresis effect. | [12] |
| Wistar rats were treated intragastrically with CS extract for 90 min and urine collection and urinary flow were measured by cannulated to the urinary bladder. | Shows a diuresis effect. | [41] | |
| Hyperglycemia reduction | Adrenaline-induced hyperglycemic mice treated orally with CS extract for 45 and 14 days. | Reduction of blood glucose levels. | [43] |
| Nephrotoxicity reduction | GM-induced nephrotoxicity mice administered with CS extract for 8 days. | Ameliorate nephropathy. | [16] |
| Anti-fatigue activity | Swimming exercise carried out by 10 mice after administration of flavonoid CS for 14 days and loaded with 5% of its body wt. of galvanized wire. | Strong anti-fatigue activity. | [4] |
| Anti-depressant activity | FST and TST carried out on 10 male Swiss mice for 6 and 5 min, respectively, 1h after treated with CS extract. | Strong anti-depressant activity. | [11] |
| Activity times of CS treated mice (normal and diabetic mice) in a black box were observed. | Good anti-depressant activity. | [46] | |
| Anti-hyperlipidemic effect | Hyperlipidemic rats were treated with CS extract for 20 days. | Shows anti-hyperlipidemic effect. | [47] |
| Anti-diabetic effect | Streptozotocin-induced diabetic rats were treated intragastrically with polysaccharides from CS for 4 weeks. | Shows anti-diabetic effect. | [46] |
| Anti-inflammatory effects | Carragenin-induced pleurisy rats were administered orally with CS for 6 h. | Inhibit inflammatory response. | [52] |
| Antioxidant activity | Total antioxidant capacity, DPPH radical scavenging activity, reducing power, and iron-chelating capacity were evaluated in ethanol extract (EF), petroleum ether (PF), acetic ether (AF), n-butanol (BF), and water (WF). | BF exhibited the strongest antioxidant activity. | [2] |
| Total antioxidant capacity by DPPH radical scavenging activity was evaluated in CS ethanolic extract. | Upper parts of CS showed higher antioxidant activity than the lower parts of CS. | [17] | |
| 50% ethanolic extract were tested in DPPH radical scavenging activity, metal chelating activity, nitric oxide-scavenging activity, reducing power determination and ferric thiocyanate (FTC) method. | Ethanol extract showed a comparable antioxidant activity to the standard compounds (BHA, BHT, Vitami C, quercetin, EDTA). | [14] | |
| Dichloromethane extract, petroleum ether extract, 95% ethanol extract, water extract were evaluated for their antioxidant activity in DPPH and β-carotene bleaching assay. | Ethanol extract exhibited the strongest antioxidant activity. | [31] | |
| 70% aqueous acetone extract were tested for ferric reducing antioxidant power (FRAP) assay using different type of hybrid. | The acetone extract of NS 640 hybrid showed a highest antioxidant activity. | [3] | |
| Metaholic extract of CS were evaluated for antioxidant capacity by lipid peroxidation inhibition in liposomes induced by Fe2+/ascorbate system. | Antioxidant activity from matured CS is higher than immature CS. | [13] | |
| DPPH radical scavenging activity, superoxide (SO) scavenging activity, iron chelating capacity, ferric reducing antioxidant power (FRAP) assay were carried out in ethyl acetate extract and ethanol extract. | All extracts exhibited low DPPH radical scavenging activity. | [37] | |
| Anti-glycation effect | Inhibition of AGE formation assay in 80% methanolic extract. | Inhibit non-enzymatic glycation. | [45] |
| Anti-inflammatory effect | Endothelial-monocyte adhesion assay, molecule expression, treatment of TNF-mediated cytotoxicity, LPS-induced TNF released were evaluated in chloroform, ethyl acetate, butanol and water extract. | Ethanol extract inhibits the expression of ICAM-1 and adhesiveness of endothelial cells. | [51] |
| COX-2 determination was conducted on macrophages treated with CS and PGE2 production was measured with PGE2 enzyme immunoassay kit. | CS stimulated COX-2 and secretion of PGE2. | [55] | |
| Neuroprotective effect | Acetylcholinesterase (AChE) and butrylcholinesterase (BChE) inhibitions assay were carried out in ethyl acetate extract and ethanol extract. | Ethyl acetate extract of Z. mays var. intendata strongly inhibit AChE and ethyl acetate extract of Z. mays var. everta strongly inhibit BChE. | [37] |
6. Conclusions
Acknowledgments
References
- Wan Rosli, W.I.; Nurhana, A.R.; Farid, C.G.; Mohsin, S.S.J. Effect of sodium hydroxide (NaOH) and sodium hypochlorite (NaHClO) on morphology and mineral concentrations of Zea mays hairs (cornsilk). Ann. Microsc. 2010, 10, 4–10. [Google Scholar]
- Liu, J.; Wang, C.; Wang, Z.; Zhang, C.; Lu, S.; Liu, J. The antioxidant and free-radical scavenging activities of extract and fractions from corn silk (Zea mays L.) and related flavone glycosides. Food Chem. 2011, 126, 261–269. [Google Scholar]
- Maksimović, Z.; Malenčić, Đ.; Kovačević, N. Polyphenol contents and antioxidant activity of Maydis stigma extracts. Bioresource Technol. 2005, 96, 873–877. [Google Scholar] [CrossRef]
- Hu, Q.L.; Zhang, L.J.; Li, Y.N.; Ding, Y.J.; Li, F.L. Purification and anti-fatigue activity of flavonoids from corn silk. Int. J. Phys. Sci. 2010, 5, 321–326. [Google Scholar]
- Bastien, J.W. Pharmacopeia of qollahuayaandeans. J. Ethnopharmacol. 1982, 8, 97–111. [Google Scholar] [CrossRef]
- Caceres, A.; Giron, L.M.; Martinez, A.M. Diuretic activity of plants used for the treatment of urinary ailments in Guatemala. J. Ethnopharmacol. 1987, 19, 233–245. [Google Scholar] [CrossRef]
- Dat, D.D.; Ham, N.N.; Khac, D.H.; Lam, N.T.; Son, P.T.; Dau, N.V.; Grabe, M.; Johansson, R.; Lindgren, G.; Stjernstrom, N.E. Studies on the individual and combined diuretics effects of four Vietnamese traditional herbal remedies (Zea mays, Imperatacylindrica, Plantago major and Orthosiphonstamineus). J. Ethnopharmacol. 1992, 36, 225–231. [Google Scholar] [CrossRef]
- Grases, F.; March, J.G.; Ramis, M.; Costa-Bauzá, A. The influence of Zea mays on urinary risk factors for kidney stones in rats. Phytother. Res. 1993, 7, 146–149. [Google Scholar] [CrossRef]
- Yesilada, E.; Honda, G.; Sevik, E.; Tabata, M.; Fujita, T.; Tanaka, T.; Takeda, Y.; Takaishi, Y. Traditional medicine in Turkey. V. Folk medicine in the inner Taurus Mountains. J. Ethnopharmacol. 1995, 46, 133–152. [Google Scholar]
- Steenkamp, V. Phytomedicines for the prostate. Fitoterapia 2003, 74, 545–552. [Google Scholar]
- Ebrahimzadeh, M.A.; Mahmoudi, M.; Ahangar, N.; Ehteshami, S.; Ansaroudi, F.; Nabavi, S.F.; Nabavi, S.M. Antidepressant activity of corn silk. Pharmacologyonline 2009, 3, 647–652. [Google Scholar]
- Velazquez, D.V.O.; Xavier, H.S.; Batista, J.E.M.; Castro-Chaves, C.D. Zea mays L. extracts modify glomerular function and potassium urinary excretion in conscious rats. Phytomedicine 2005, 12, 363–369. [Google Scholar]
- Maksimovic, Z.A.; Kovačević, N. Preliminary assay on the antioxidative activity of Maydis stigma extracts. Fitoterapia 2003, 74, 144–147. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Pourmorad, F.; Hafe, S. Antioxidant Activities of Iranian Corn Silk. Turk. J. Biol. 2008, 32, 43–49. [Google Scholar]
- Bai, H.; Hai, C.; Xi, M.; Liang, X.; Liu, R. Protective Effect of Maize Silks (Maydis stigma) Ethanol Extract on Radiation-Induced Oxidative Stress in Mice. Plant Food Hum. Nutr. 2010, 65, 271–276. [Google Scholar]
- Sepehri, G.; Derakhshanfar, A.; Zade, F.Y. Protective effects of corn silk extract administration on gentamicin-induced nephrotoxicity in rat. Comp. Clin. Pathol. 2011, 20, 89–94. [Google Scholar] [CrossRef]
- Alam, E.A. Evaluation of antioxidant and antibacterial activities of Egyptian Maydis stigma (Zea mays hairs) rich in some bioactive constituents. J. Am. Sci. 2011, 7, 726–729. [Google Scholar]
- Anonymous. Corn Silk Tablet. Available online: http://www.nutritional-supplement-bible.com/cornsilk-tablets.html (accessed on 13 April 2011).
- Anonymous. Cornsilk Tea Common Uses AND Benefits. Available online: http://www.cornsilktea.net (accessed on 13 April 2011).
- Wan Rosli, W.I.; Nurhanan, A.R.; Mohsin, S.S.J.; Farid, C.G. Aqueous, alcoholic treated and proximate analysis of maydis stigma (Zea mays hairs). Ann. Microsc. 2008, 8, 66–72. [Google Scholar]
- Liu, J.; Lin, S.; Wang, Z.; Wang, C.; Wang, E.; Zhang, Y.; Liu, J. Supercritical fluid extraction of flavonoids from Maydis stigma and its nitrite-scavenging ability. Food Bioprod. Process. 2011, 89, 333–339. [Google Scholar]
- Anonymous. Maize. Available online: http://www.newworldencyclopedia.org/entry/Maize (accessed on 29 June 2011).
- Leon, N.D.; Coors, J.G. Genetic Improvement of Corn for Lingo-Cellulosic Biofeedstock Production. In Genetic Improvement of Bioenergy Crops; Vermerris, W., Ed.; Springer Science: New York, NY, USA, 2008; pp. 185–210. [Google Scholar]
- Inglett, G.E. Corn: Culture, Processing, Products; The AVI Publishing Company: Westport, CT, USA, 1970. [Google Scholar]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Snook, M.E.; Widstrom, N.W.; Wiseman, B.R.; Byrne, P.F.; Harwood, J.S.; Costello, C.E. New C-4"-hyroxy derivatives of maysin and 3'-methoxymaysin isolated from corn silks (Zea mays). J. Agric. Food Chem. 1995, 43, 2740–2745. [Google Scholar]
- Ren, S.C.; Liu, Z.L.; Ding, X.L. Isolation and identification of two novel flavone glycosides from corn silk (Stigma maydis). J. Med. Plants Res. 2009, 32, 1009–1015. [Google Scholar]
- Elliger, C.A.; Chan, B.G.; Waiss, A.C., Jr.; Lundin, R.E.; Haddon, W.F. C-Glycosylflavones from Zea mays that inhibit insect development. Phytochemistry 1980, 19, 293–297. [Google Scholar]
- Snook, M.E.; Widstrom, N.W.; Gueldner, R.C. Reversed-phase high-performance liquid chromatographic procedure for the determination of maysin in corn silks. J. Chromatogr. A 1989, 477, 439–447. [Google Scholar] [CrossRef]
- Waiss, A.C.; Chan, B.G.; Elliger, C.A.; Wiseman, B.R.; McMillian, W.W.; Widstrom, N.W.; Zuber, M.S.; Keaster, A.J. Maysin, a flavone glycoside from corn silks with antibiotic activity toward corn earworm. J. Economic Entomol. 1979, 72, 256–258. [Google Scholar]
- El-Ghorab, A.; El-Massry, K.F.; Shibamoto, T. Chemical Composition of the Volatile Extract and Antioxidant Activities of the Volatile and Nonvolatile Extracts of Egyptian Corn Silk (Zea mays L.). J. Agric. Food Chem. 2007, 55, 9124–9127. [Google Scholar]
- Wang, C.; Zhang, T.; Liu, J.; Lu, S.; Zhang, C.; Wang, E.; Wang, Z.; Zhang, Y.; Liu, J. Subchronic toxicity study of cornsilk with rats. J. Ethnopharmacol. 2011, 137, 36–43. [Google Scholar]
- Hashim, P. Centella asiatica in food and beverage applications and its potential antioxidant and neuroprotective effect. J. Int. Food Res. 2011, 18, 2217–2222. [Google Scholar]
- Vladimir-Knežević, S.; Blažeković, B.; Štefan, M.B.; Alegro, A.; Kőszegi, T.; Petrik, J. Antioxidant activities and polyphenolic contents of three selected micromeria species from croatia. Molecules 2011, 16, 1454–1470. [Google Scholar]
- Zhu, K.X.; Lian, C.X.; Guo, X.N.; Peng, W.; Zhou, H.M. Antioxidant activities and total phenolic contents of various extracts from defatted wheat germ. Food Chem. 2011, 126, 1122–1126. [Google Scholar] [CrossRef]
- Hassas-Roudsari, M.; Chang, P.R.; Pegg, R.B.; Tyler, R.T. Antioxidant capacity of bioactive extracted from canola meal by subcritical water, ethanolic and hot water extraction. Food Chem. 2009, 114, 717–726. [Google Scholar] [CrossRef]
- Kan, A.; Orhan, I.; Coksari, G.; Sener, B. In-vitro neuroprotective properties of the maydis stigma extracts from four corn varieties. Int. J. Food Sci. Nutr. 2011, 63, 1–4. [Google Scholar]
- Bump, E.A.; Brown, J.M. Role of glutathione in the radiation response of mammalian cells in vitro and in vivo. Pharmacol. Ther. 1990, 47, 117–136. [Google Scholar]
- Hu, Q.I.; Deng, Z.I. Protective effects of flavonoids from corn silk on oxidative stress induced by exhaustive exercise in mice. Afr. J. Biot. 2011, 10, 3163–3167. [Google Scholar]
- Nguyen, T.; Sherratt, P.J.; Pickett, C.B. Regulatory mechanisms controlling gene expression mediated by the antioxidant response element. Annu. Rev. Pharm. Toxicol. 2003, 43, 233–260. [Google Scholar]
- Pinheiro, A.C.S.; Pais, A.A.; Tardivo, A.C.B.; Alves, M.J.Q.F. Effect of aqueous extract of corn silks (Zea mays L.) on the renal excretion of water and electrolytes and arterial pressure in anesthetized wistar rats. Revista Brasileira de Plantas Medicinais 2011, 13, 375–381. [Google Scholar]
- Preedy, V.R.; Watson, R.R. Glossary. In Handbook of Disease Burdens and Quality of Life Measures; Springer: New York, NY, USA, 2010; p. 4230. [Google Scholar]
- Guo, J.; Liu, T.; Han, L.; Liu, Y. The effects of corn silk on glycaemic metabolism. Nutr. Metab. 2009, 6, 47. [Google Scholar]
- Ramasamy, R.; Vannucci, S.J.; Yan, S.S.D.; Herold, K.; Yan, S.F.; Schmidt, A.M. Advanced glycation end products and RAGE: A common threa in aging, diabetes, neurodegeneration, and inflammation. Glycobiology 2005, 15, 16–28. [Google Scholar]
- Farsi, D.A.; Harris, C.S.; Reid, L.; Bennett, S.A.L.; Haddad, P.S.; Martineau, L.C.; Arnason, J.T. Inhibition of non-enzymatic glycation by silk extracts from a mexican land race and modern inbred lines of maize (Zea mays). Phytother. Res. 2008, 22, 108–112. [Google Scholar] [CrossRef]
- Zhao, W.; Yin, Y.; Yu, Z.; Liu, J.; Chen, F. Comparison of anti-diabetic effects of polysaccharides from corn silk on normal and hyperglycemia rats. Int. J. Biol. Macromol. 2012, 50, 1133–1137. [Google Scholar]
- Kaup, S.R.; Arunkumar, N.; Bernhardt, L.K.; Vasari, R.G.; Shetty, S.S.; Pai, S.R.; Arunkumar, B. Antihyperlipedemic activity of Cynodondactylon extract in high-cholestrol diet fed Wistar rats. Genomic Med. Biomark. Health Sci. 2011, 3, 98–102. [Google Scholar] [CrossRef]
- Schreiner, G.E. Toxic nephropathy: Adverse renal effects caused by drugs and chemicals. JAMA 1965, 191, 849–850. [Google Scholar] [CrossRef]
- Nguemfo, E.L.; Dimo, T.; Azebaze, A.G.B.; Asongalem, E.A.; Alaoui, K.; Dongmo, A.B.; Cherrah, Y.; Kamtchouing, P. Anti-inflammatory and anti-nociceptive activities of the stem bark extracts from Allanblackia monticola STANER L.C. (Guttiferae). J. Ethnopharmacol. 2007, 114, 417–424. [Google Scholar] [CrossRef]
- Stoecklin, G.; Lu, M.; Rattenbacher, B.; Moroni, C. A constitutive decay element promotes tumor necrosis factor alpha mRNA degradation via an AU-rich element-independent pathway. Mol. Cell. Biol. 2003, 23, 3506–3515. [Google Scholar] [CrossRef]
- Habtemariam, S. Extract of Corn silk (Stigma of Zea mays) Inhibit the Tumour Necrosis Factor-α- and Bacterial Lipopolysaccharide-Induced Cell Adhesion and ICAM-1 Expression. Planta Med. 1998, 64, 314–318. [Google Scholar] [CrossRef]
- Wang, G.Q.; Xu, T.; Bu, X.M.; Liu, B.Y. Anti-inflammation effects of corn silk in a rat model of carrageenin-induced pleurisy. Inflammation 2011, 35, 822–827. [Google Scholar]
- Miguel, M.G. Antioxidant and Anti-inflammatory Activities of Essential oil: A Short Review. Molecules 2010, 15, 9252–9287. [Google Scholar] [CrossRef]
- Mao, Y.W.; Tseng, H.W.; Liang, W.L.; Chen, I.S.; Chen, S.T.; Lee, M.H. Anti-Inflammatory and Free Radical Scavenging Activities of The Constituents Isolated from Machilus zuihoensis. Molecules 2011, 16, 9451–9455. [Google Scholar] [CrossRef]
- Kim, K.; Shin, H.H.; Choi, S.K.; Choi, H.S. Corn Silk Induced Cyclooxygenase-2 in Murine Macrophages. Biosci. Biotechnol. Biochem. 2005, 69, 1848–1853. [Google Scholar] [CrossRef]
- Senol, F.S.; Orhan, I.; Yilmaz, G.; Cicek, M.; Sener, B. Acetylcholinesterase, butyrylcholinesterase, and tyrosinase inhibition studies and antioxidant activities of 33 Scutellaria L. taxa from Turkey. Food Chem. Toxicol. 2009, 48, 781–788. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hasanudin, K.; Hashim, P.; Mustafa, S. Corn Silk (Stigma Maydis) in Healthcare: A Phytochemical and Pharmacological Review. Molecules 2012, 17, 9697-9715. https://doi.org/10.3390/molecules17089697
Hasanudin K, Hashim P, Mustafa S. Corn Silk (Stigma Maydis) in Healthcare: A Phytochemical and Pharmacological Review. Molecules. 2012; 17(8):9697-9715. https://doi.org/10.3390/molecules17089697
Chicago/Turabian StyleHasanudin, Khairunnisa, Puziah Hashim, and Shuhaimi Mustafa. 2012. "Corn Silk (Stigma Maydis) in Healthcare: A Phytochemical and Pharmacological Review" Molecules 17, no. 8: 9697-9715. https://doi.org/10.3390/molecules17089697
APA StyleHasanudin, K., Hashim, P., & Mustafa, S. (2012). Corn Silk (Stigma Maydis) in Healthcare: A Phytochemical and Pharmacological Review. Molecules, 17(8), 9697-9715. https://doi.org/10.3390/molecules17089697




