Characterization of the Antibacterial Activity and the Chemical Components of the Volatile Oil of the Leaves of Rubus parvifolius L.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Uniform Distribution Experimental Design of Extraction
| No. | Soak time X1 (h) | Grinding degree X2 (mesh) | Distillation time X3 (h) | Ratio of solvent to leaves X4 ( v/w) | Weight of volatile oil (g) |
|---|---|---|---|---|---|
| 1 | 0 | < 20 | 5 | 20 | 0.15 |
| 2 | 2 | 20–40 | 9 | 16 | 0.85 |
| 3 | 4 | > 40 | 4 | 12 | 0.29 |
| 4 | 6 | < 20 | 8 | 8 | 0.16 |
| 5 | 8 | 20–40 | 3 | 22 | 0.21 |
| 6 | 10 | > 40 | 7 | 18 | 0.65 |
| 7 | 12 | < 20 | 2 | 14 | 0.39 |
| 8 | 14 | 20–40 | 6 | 10 | 0.28 |
| 9 | 16 | > 40 | 10 | 24 | 0.85 |
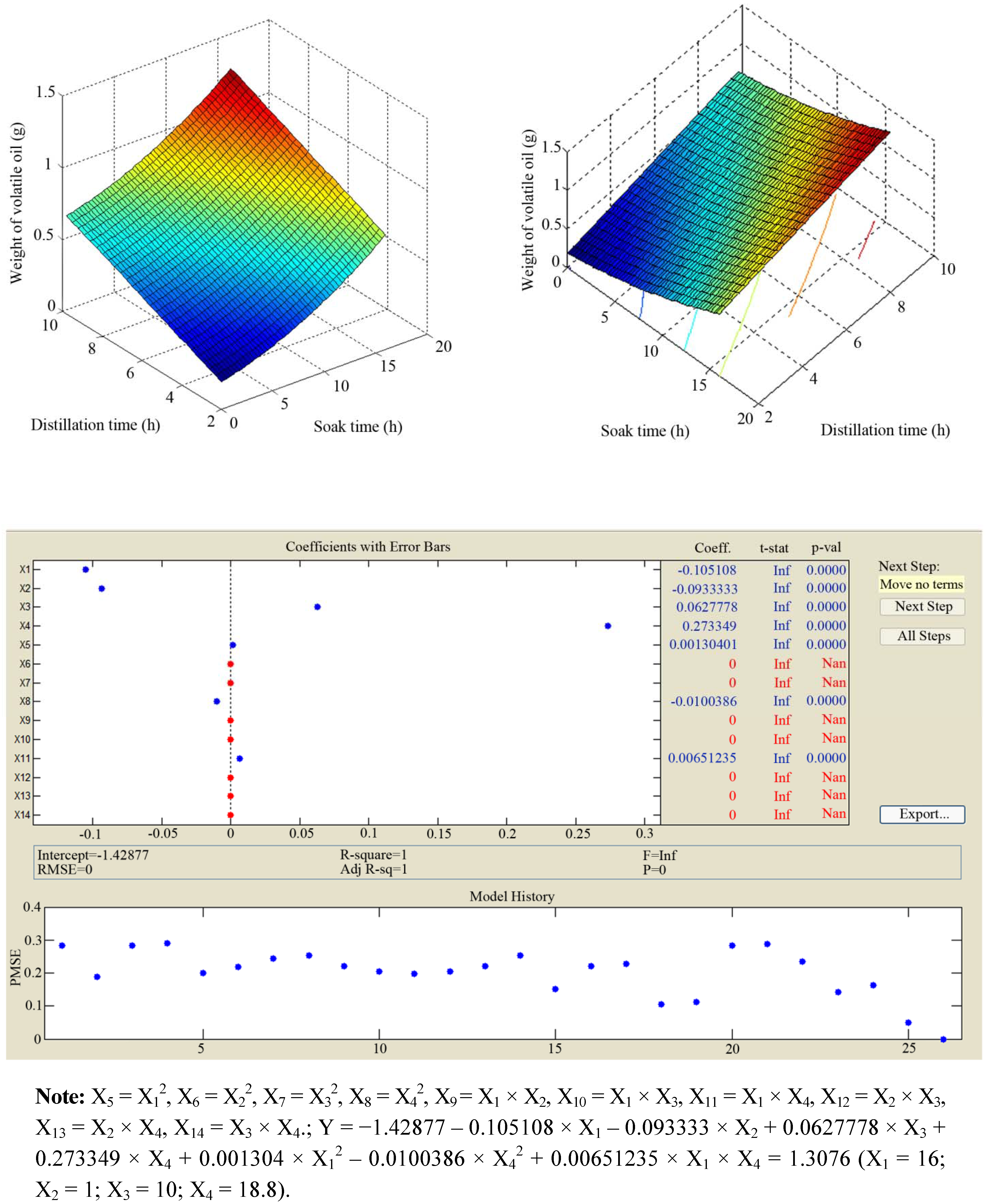
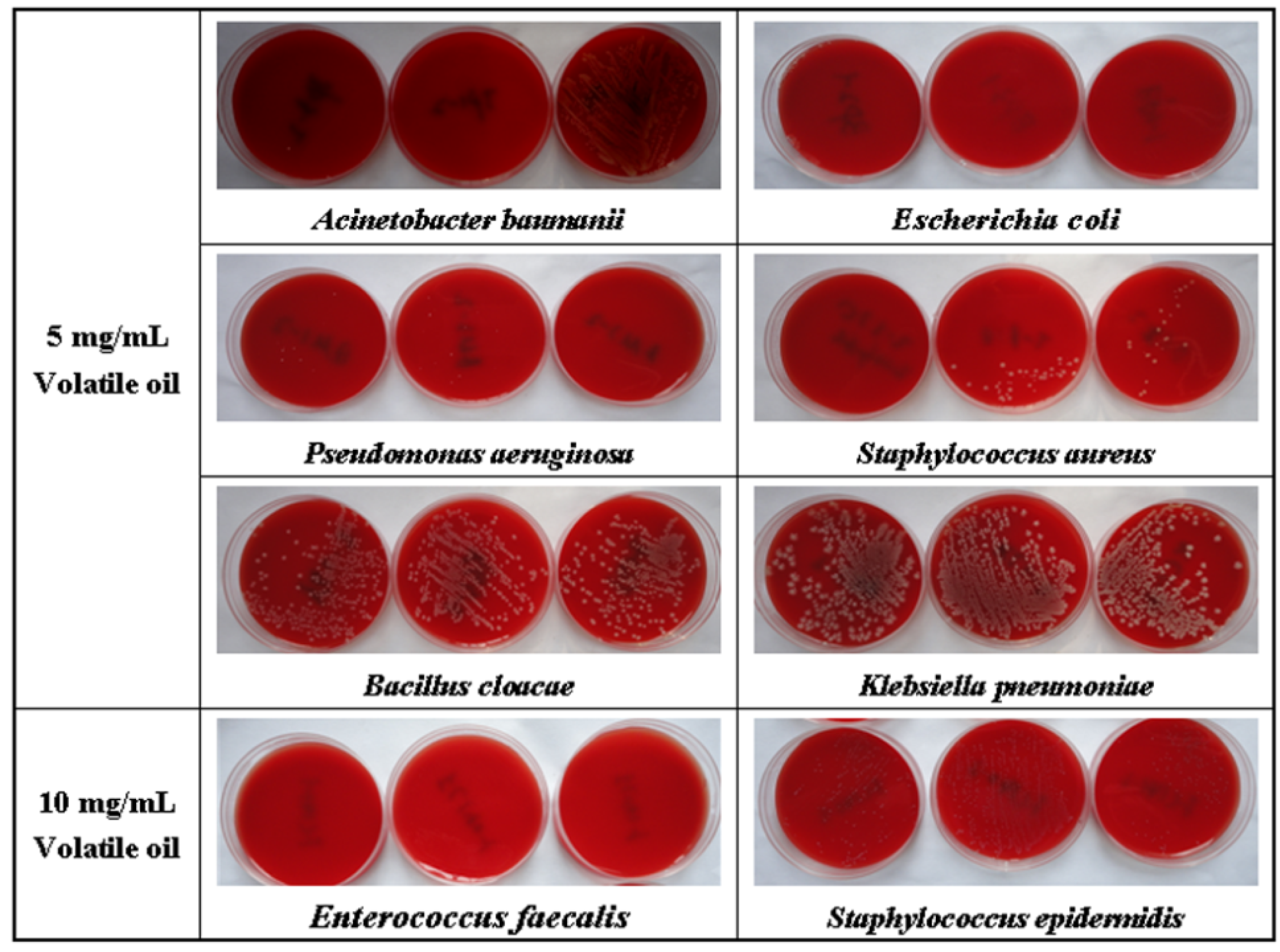
2.2. Antibacterial Activity of Rp Volatile Oil
| Bacterial species | Source | MIC (mg/mL) | MBC (mg/mL) |
|---|---|---|---|
| Gram-positive bacteria | |||
| Staphylococcus aureus | CIS a | 5 | > 5 |
| Staphylococcus aureus | ATCC b29213 | 5 | 10 |
| Staphylococcus epidermidis | CIS | 10 | 10 |
| Enterococcus faecalis | CIS | 10 | 10 |
| Gram-negative bacteria | |||
| Escherichia coli | CIS | 5 | 5 |
| Escherichia coli | ATCC 25922 | 5 | 5 |
| Pseudomonas aeruginosa | CIS | > 2.5 | 5 |
| Pseudomonas aeruginosa | ATCC 27853 | 2.5 | 5 |
| Acinetobacter baumanii | CIS | > 2.5 | 5 |
| Bacillus cloacae | CIS | 5 | 10 |
| Klebsiella pneumoniae | CIS | 5 | > 10 |
2.3. Components of the Rp Volatile Oil
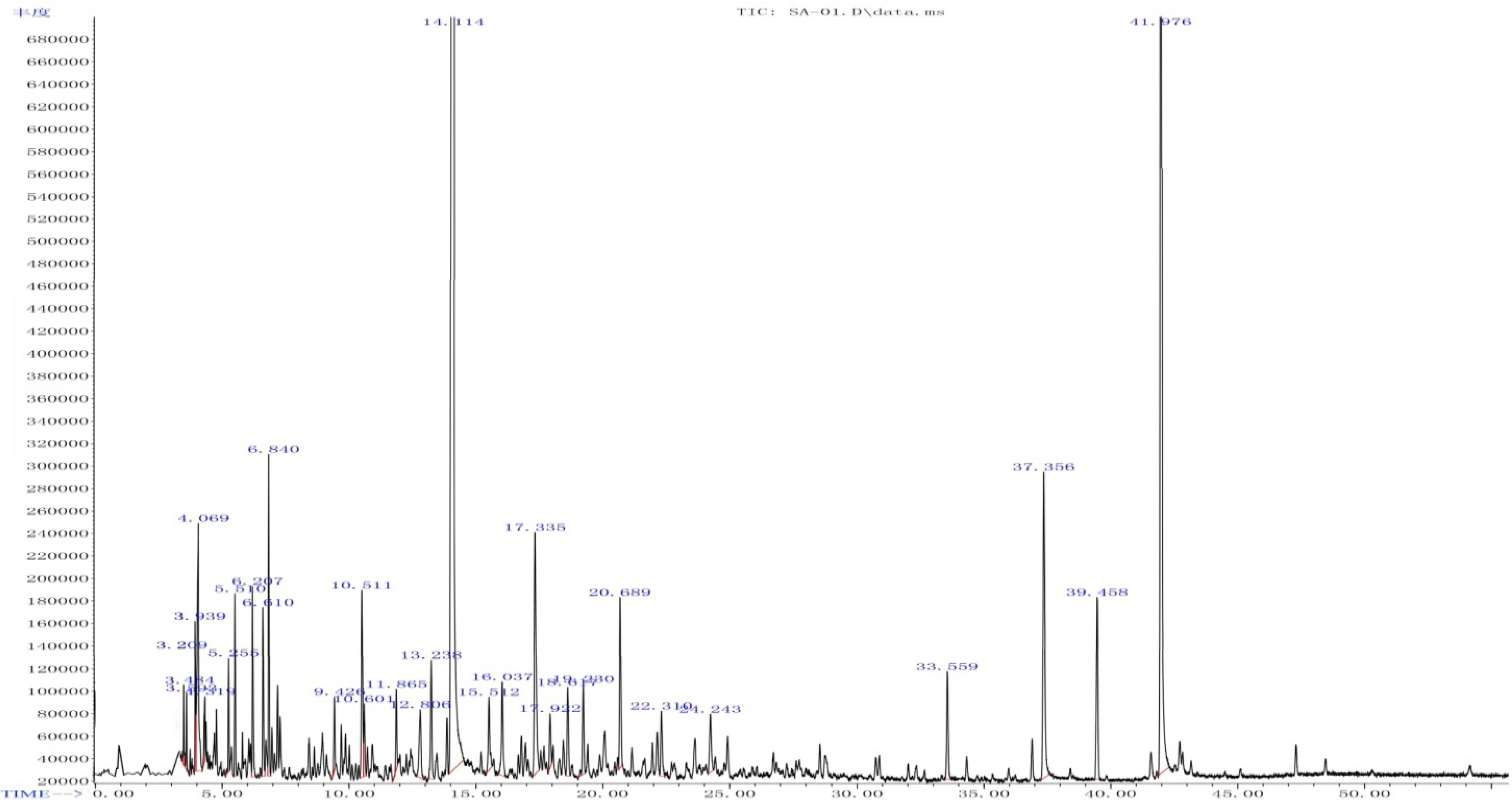
| No | RT (min) | Compound ID | Structural Formula | Formula | Content% |
|---|---|---|---|---|---|
| 1 | 3.209 | 2-Acetylfuran |  | C6H6O2 | 0.28 |
| 2 | 3.484 | Butyl formate |  | C5H10O2 | 0.46 |
| 3 | 3.939 | 5-Methyl furfural |  | C6H6O2 | 0.55 |
| 4 | 4.069 | Hexanoic acid |  | C6H12O2 | 2.03 |
| 5 | 4.319 | trans-3-Hexenoic acid |  | C6H10O2 | 0.32 |
| 6 | 5.255 | Benzyl alcohol |  | C7H8O | 0.45 |
| 7 | 5.510 | Phenyl acetaldehyde |  | C8H8O | 0.77 |
| 8 | 6.207 | cis-α,α,5-Trimethyl-5-vinyl-tetrahydrofuran-2-methanol | 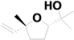 | C10H18O2 | 0.88 |
| 9 | 6.610 | α-Methyl-α-[4-methyl-3-pentenyl]oxiranemethanol | 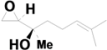 | C10H18O2 | 0.78 |
| 10 | 6.840 | Linalool | 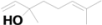 | C10H18O | 1.39 |
| 11 | 9.426 | Naphthalene | 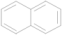 | C10H8 | 0.37 |
| 12 | 10.511 | 2,3-Dihydrobenzofuran |  | C8H8O | 1.21 |
| 13 | 10.601 | α,4-Dimethyl-3-cyclohexene-1-acetaldehyde |  | C10H16O | 0.41 |
| 14 | 11.865 | Nerol |  | C10H18O | 0.42 |
| 15 | 12.806 | Phenylephrine | 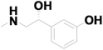 | C9H13NO2 | 0.69 |
| 16 | 13.238 | 2-Methylnaphthalene |  | C11H10 | 0.81 |
| 17 | 14.114 | 4-Hydroxy-3-methoxystyrene | 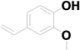 | C9H10O2 | 66.05 |
| 18 | 15.512 | 1,1,6-Trimethyl-1,2-dihydronaphthalene | 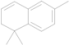 | C13H16 | 0.57 |
| 19 | 16.037 | Methyl 4-formylbenzoate |  | C9H8O3 | 0.77 |
| 20 | 17.335 | 4-tert-Butylbenzoic acid |  | C11H14O2 | 2.22 |
| 21 | 17.922 | Dimethylnaphthalene |  | C12H12 | 0.37 |
| 22 | 18.617 | α,2,6-Trimethyl-benzeneethanamine |  | C11H17N | 0.65 |
| 23 | 19.230 | 2-Methoxy-4-(prop-1-enyl)phenol |  | C10H12O2 | 0.64 |
| 24 | 20.689 | Irisone | 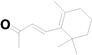 | C13H20O | 1.17 |
| 25 | 22.310 | Dihydroactinidiolide | 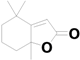 | C11H16O2 | 0.52 |
| 26 | 24.243 | 2-(1,3-Butadienyl)mesitylene |  | C13H16 | 0.52 |
| 27 | 33.559 | Hexahydrofarnesylacetone |  | C18H36O | 0.8 |
| 28 | 39.458 | Kaur-16-ene |  | C20H32 | 1.36 |
| 29 | 41.976 | 3,7,11,15-Tetramethyl-2-hexadecen-1-ol |  | C20H40O | 9.79 |
| Compound types | Content% | Amount |
|---|---|---|
| Alcohol-phenol | 81.09 | 9 |
| Carboxylic acid | 4.57 | 3 |
| Ester | 1.52 | 3 |
| Ketone | 2.25 | 3 |
| Aldehyde | 1.73 | 3 |
| Other | 5.86 | 8 |
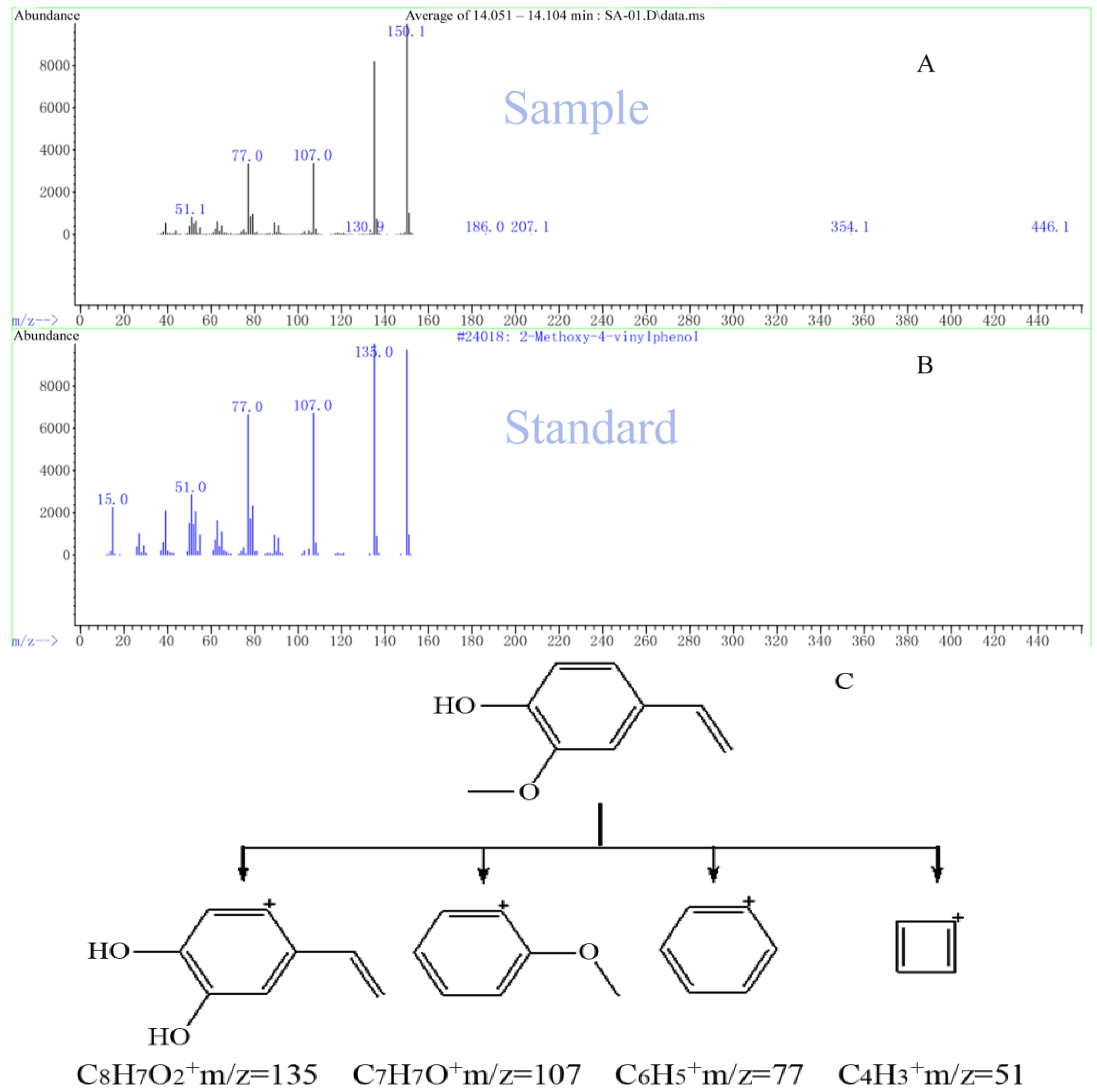
3. Experimental
3.1. Plant Materials
3.2. Other Reagents
3.3. Volatile Rp Oil Extraction
3.4. Bacterial Strains
3.5. Antibacterial Tests
3.6. Analysis of Components
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Wang, Y.; He, J.M.; Chen, H.; Zhang, D.S.; Cai, H.; Shao, H.B. Analysis of flavones in Rubus parvifolius Linn by high performance liquid chromatography combined with electrospray ionization-mass spectrometry and thin-layer chromatography combined with Fourier transform surface enhanced Raman spectroscopy. Fen Xi Hua Xue 2006, 34, 1073–1077. [Google Scholar]
- Gao, J.; Sun, C.R.; Yang, J.H.; Shi, J.M.; Du, Y.G.; Zhang, Y.Y.; Li, J.H.; Wan, H.T. Evaluation of the hepatoprotective and antioxidant activities of Rubus parvifolius L. J. Zhejiang Univ. Sci. B 2011, 12, 135–142. [Google Scholar] [CrossRef]
- Zhu, Z.H.; Zhang, H.Q.; Yuan, M.J. Pharmacological study of Rubus parvifolius L. Zhongguo Zhong Yao Za Zhi 1990, 15, 427–447. [Google Scholar]
- Wang, J.S.; Qiu, Z.Y.; Xia, Y.P.; Li, H.Z.; Ren, L.Y.; Zhang, L. The protective effects of total glycosides Rubus parviflolius on cerebral ischemia in rat. Zhongguo Zhong Yao Za Zhi 2006, 2, 138–141. [Google Scholar]
- Wang, J.S.; Li, H.Z.; Qiu, Z.Y.; Xia, Y.P.; Ren, L.Y.; Zhou, C.L. Protective effects of aqueous extract of Rubus parviflolius on middle cerebral artery occlusion and reperfusion injury in rats. Chin. J. New Drugs Clin. Remed. s 2006, 12, 920–923. [Google Scholar]
- Deighton, N.; Brennan, R.; Finn, C.; Davies, H.V. Antioxidant properties of domesticated and wild Rubus species. J. Sci. Food Agric. 2000, 80, 1307–1313. [Google Scholar] [CrossRef]
- Hsiao, G.; Shen, M.Y.; Lin, K.H.; Lan, M.H.; Wu, L.Y.; Chou, D.S.; Lin, C.H.; Su, C.H.; Sheu, J.R. Antioxidative and hepatoprotective effects of Antrodia camphorata extract. J. Agric. Food Chem. 2003, 51, 3302–3308. [Google Scholar] [CrossRef]
- Gowri Shankar, N.L.; Manavalan, R.; Venkappayya, D.; David Raj, C. Hepatoprotective and antioxidant effects of Commiphora berryi (Arn) Engl bark extract against CCl4-induced oxidative damage in rats. Food Chem. Toxicol. 2008, 46, 3182–3185. [Google Scholar] [CrossRef]
- Zeashan, H.; Amresh, G.; Singh, S.; Rao, C.V. Hepatoprotective and antioxidant activity of Amaranthus spinosus against CCl4 induced toxicity. J. Ethnopharmarcol. 2009, 125, 364–366. [Google Scholar] [CrossRef]
- Ho, C.L.; Tseng, Y.H.; Wang, E.I.; Liao, P.C.; Chou, J.C.; Lin, C.N.; Su, Y.C. Composition, antioxidant and antibacterial activities of the seed essential oil of Calocedrus formosana from Taiwan. Nat. Prod. Commun. 2011, 6, 133–136. [Google Scholar]
- Hamill, F.A.; Apio, S.; Mubiru, N.K.; Mosango, M.; Bukenya-Ziraba, R.; Maganyi, O.W.; Soejarto, D.D. Traditional herbal drugs of southern Uganda. Part III: Isolation and methods for physical characterization of bioactive alkanols from Rubus apetalus. J. Ethnopharmarcol. 2003, 87, 15–19. [Google Scholar] [CrossRef]
- Tan, M.X.; Wang, H.S.; Li, S.; Yang, Y. Studies on the chemical constituents of the volatile oil from the leaves of Rubus parviflolius. Tianran Chanwu Yanjiu Yu Kaifa 2003, 1, 32–33. [Google Scholar]
- Thiem, B.; Goslinska, O. Antimicrobial activity of Rubus chamaemorus leaves. Fitoterapia 2004, 75, 93–95. [Google Scholar] [CrossRef]
- Nissen, L.; Zatta, A.; Stefanini, I.; Grandi, S.; Sgorbati, B.; Biavati, B.; Monti, A. Characterization and antimicrobial activity of essential oils of industrial hemp varieties (Cannabis sativa L.). Fitoterapia 2010, 81, 413–419. [Google Scholar] [CrossRef]
- Coghe, S.; Benoot, K.; Delvaux, F.; Vanderhaegen, B.; Delvaux, F.R. Ferulic acid release and 4-vinylguaiacol formation during brewing and fermentation: indications for feruloyl esterase activity in Saccharomyces cerevisiae. J. Agric. Food Chem. 2004, 52, 602–608. [Google Scholar] [CrossRef]
- Dugelay, I.; Gunata, Z.; Sapis, J.C.; Baumes, R.; Bayonove, C. Role of cinnamoyl esterase activities from enzyme preparations on the formation of volatile phenols during winemaking. J. Agric. Food Chem. 1993, 41, 2092–2096. [Google Scholar] [CrossRef]
- Pongprayoon, U.; Bohlin, L.; Soonthornsaratune, P.; Wasuwat, S. Anti-inflammatory activity of Ipomoea pes-caprae (L.) R. Br. Phytother. Res. 1991, 5, 63–66. [Google Scholar] [CrossRef]
- Morton, C.E.G.; Holt, H.A. A problem encountered using staphylococcus/streptococcus supplement. Med. Lab. Sci. 1989, 46, 72–73. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI), Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically: Approved Guideline, 7th edCLSI: Wayne, NE, USA, 2006; Approved Standard M7–A7.
- Fang, K.T.; Ma, C.X. Orthogonal and Uniform Experimental Design, 1st ed; Science press: Beijing, China, 2001. [Google Scholar]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cai, Y.; Hu, X.; Huang, M.; Sun, F.; Yang, B.; He, J.; Wang, X.; Xia, P.; Chen, J. Characterization of the Antibacterial Activity and the Chemical Components of the Volatile Oil of the Leaves of Rubus parvifolius L. Molecules 2012, 17, 7758-7768. https://doi.org/10.3390/molecules17077758
Cai Y, Hu X, Huang M, Sun F, Yang B, He J, Wang X, Xia P, Chen J. Characterization of the Antibacterial Activity and the Chemical Components of the Volatile Oil of the Leaves of Rubus parvifolius L. Molecules. 2012; 17(7):7758-7768. https://doi.org/10.3390/molecules17077758
Chicago/Turabian StyleCai, Yongqing, Xiaogang Hu, Mingchun Huang, Fengjun Sun, Bo Yang, Juying He, Xianfeng Wang, Peiyuan Xia, and Jianhong Chen. 2012. "Characterization of the Antibacterial Activity and the Chemical Components of the Volatile Oil of the Leaves of Rubus parvifolius L." Molecules 17, no. 7: 7758-7768. https://doi.org/10.3390/molecules17077758
APA StyleCai, Y., Hu, X., Huang, M., Sun, F., Yang, B., He, J., Wang, X., Xia, P., & Chen, J. (2012). Characterization of the Antibacterial Activity and the Chemical Components of the Volatile Oil of the Leaves of Rubus parvifolius L. Molecules, 17(7), 7758-7768. https://doi.org/10.3390/molecules17077758




