Abstract
Ever since the idea arose that melatonin might promote sleep and resynchronize circadian rhythms, many research groups have centered their efforts on obtaining new melatonin receptor ligands whose pharmacophores include an aliphatic chain of variable length united to an N-alkylamide and a methoxy group (or a bioisostere), linked to a central ring. Substitution of the indole ring found in melatonin with a naphthalene or quinoline ring leads to compounds of similar affinity. The next step in this structural approximation is to introduce a quinoxaline ring (a bioisostere of the quinoline and naphthalene rings) as the central nucleus of future melatoninergic ligands.
1. Introduction
The frequent pathology of sleep disorders has diverse origins. The clinical manifestations vary and may include insomnia, hypersomnia, respiratory disorders, complex motor disorders, etc. These disorders can appear alone or associated to each other, thereby making this a very complex pathology. Insomnia is the most frequent disorder within the general population [1]. The complaints from more than 50% of primary care patients are related to insomnia and an estimated 9 to 18% of the adult population suffers from chronic insomnia [2,3].
In the regulation of the awake-sleep system, a complex neural network is involved, in which different areas of the brain are activated and inhibited, following a circadian rhythm that lasts 24 h. The suprachiasmatic nucleus (SCN) is responsible for synchronizing the circadian rhythm as well as for promoting sleep, by means of melatonin (MLT), the main hormone secreted by the pineal gland [4,5]. In fact, it has been observed that administration of MLT results in an increase of sleepiness.
Melatonin regulates sleep and the circadian rhythms by means of its receptors MT1 and MT2 which in recent years have become one of the most interesting pharmacological targets [4,6].The diversity and distribution of MT1 and MT2 in the different tissues indicate that each one of these receptors possibly possesses a different physiological function, although this possibility must be more thoroughly studied. Some studies suggest that the effect of promoting sleep is related to the suppression of activity in the SCN neurons by means of activation of the receptors MT1. Other studies report that the phase changes of the neuronal rhythm are related to the activation of the receptors MT2 at dawn [4,7].
Ever since the idea arose that MLT might promote sleep and resynchronize circadian rhythms, many research groups have centered their efforts on obtaining new melatonin receptor ligands [4,6,8,9,10,11]. As can be observed in Figure 1, several series of MT2 ligands have been described as a competitive agonists or antagonists with varying degrees of selectivity (IIK7 is a selective MT2 receptor agonist and luzindole and 4P-PDOT are a selective MT2 receptor antagonists) as well as compounds with affinity for both melatoninergic receptor subtypes (melatonin, ramelteon, agomelatine, LY156735 and tasimelteon) [12,13,14,15].
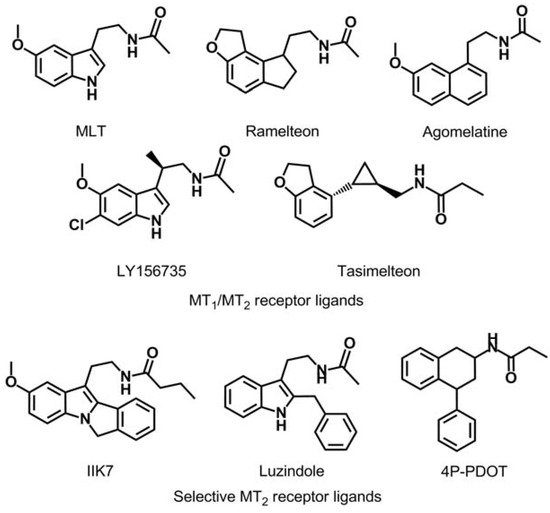
Figure 1.
Chemical structures of MT1/MT2 receptors agonist and MT2-selective ligands.
Figure 1.
Chemical structures of MT1/MT2 receptors agonist and MT2-selective ligands.
Recently MT2 receptor subtype selectivity has registered some important advances that could result in a safer compound with a more favorable pharmacological profile [16,17,18,19]. During the last two decades, numerous research groups have sought to understand how melatonin interacts with its receptors. A number of structure-activity relationships (SAR) have been studied proposing molecular models of the melatonin binding site and 3D pharmacophore models [20,21,22,23,24,25,26,27]. These models have been reviewed and completed in recent reviews [6,28].
A brief description of the general SAR concepts developed in these decades follows. The pharmacophore structure that can be found in almost all MLT receptor agonists is composed by an amide group connected by a linker chain of variable length to an aromatic nucleus (indole or a bioisostere) carrying a methoxy group, or a bioisostere, such as bromine.
Since the indole ring of MLT is not essential for binding to the receptor, several potent ligands have been designed by employing bioisosteric replacement of the indole moiety by other aromatic rings. Substitution of the indole ring with the naphthalene ring system leads to agomelatine, which presents higher affinity for MT1/MT2 than MLT. In addition, replacement of the naphthalene ring with a quinoline scaffold leads to compounds of similar affinity [29,30]. The bioequivalency disclosed between the indole moiety replaced by naphthalene and quinolone rings is a clear example of a structural approach, in which the next logical step was to introduce the quinoxaline ring as shown in Figure 2. The quinoxaline derivatives represent a privileged chemical structure because they possess important biological properties with applications in diverse therapeutic fields as anticancerous [31], anti-inflammatory/antioxidant [32], antibacterial [33] and sedative and anticonvulsant agents [34]. Thus, in this study, we decided to design and synthesize quinoxaline derivatives in order to verify whether the quinoxaline is a valuable heterocyclic bioisostere of the melatonin indole nucleus.
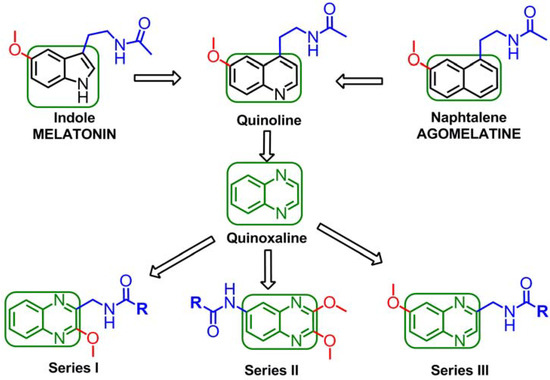
Figure 2.
Structural approach of the indole ring and the naphthalene ring to quinoline ring and to quinoxaline ring.
Figure 2.
Structural approach of the indole ring and the naphthalene ring to quinoline ring and to quinoxaline ring.
In addition, early SAR studies showed that both the 5-methoxy group and the N-acetylamino side chain of melatonin are crucial for high receptor affinity and that the relative spatial distance between these groups is also an important factor [35]. However, based on extended SAR data of a series of tryptamines, it has been concluded that the 5-methoxy group is not an essential requirement for biological activity, although it clearly shows major interactions with a specific binding site in the melatonin receptor for a broad variety of ligands [36] and it is known to increase potency by forming a hydrogen bond with His211 in the putative transmembrane domain 5 (TM5) [37,38].
Moreover, it has been reported that appropriate substituents ortho to the ethylamido side chain in the naphthalenic melatonin analogues modulate the binding affinity to the melatonin receptor [39]. Thus, we were further interested in the effects of switching the methoxy group from position 6 of the quinoxaline scaffold (analogous to position 5 in the melatonin indole ring) to position 2, contiguous to the N-acylamido side chain. Herein, we report a preliminary design, synthesis and biological evaluation of three novel series of quinoxaline derivatives as shown in Figure 2.
2. Results and Discussion
2.1. Chemistry
The synthetic route followed for the synthesis of series I is shown in Scheme 1. First, one of the chlorine atoms of 2,3-dichloroquinoxaline was replaced by a methoxy group using sodium methoxide in tetrahydrofuran (THF) leading to compound 1 [40]. According to the procedure described by Herrman et al. [41,42] a nitrile group was then introduced into position 2 of the quinoxaline by treatment with tetraethylammonium cyanide in the presence of acetonitrile at 50–60 °C to give compound 2.
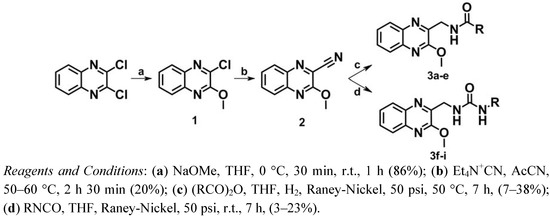
Scheme 1.
Synthesis of series I.
Scheme 1.
Synthesis of series I.
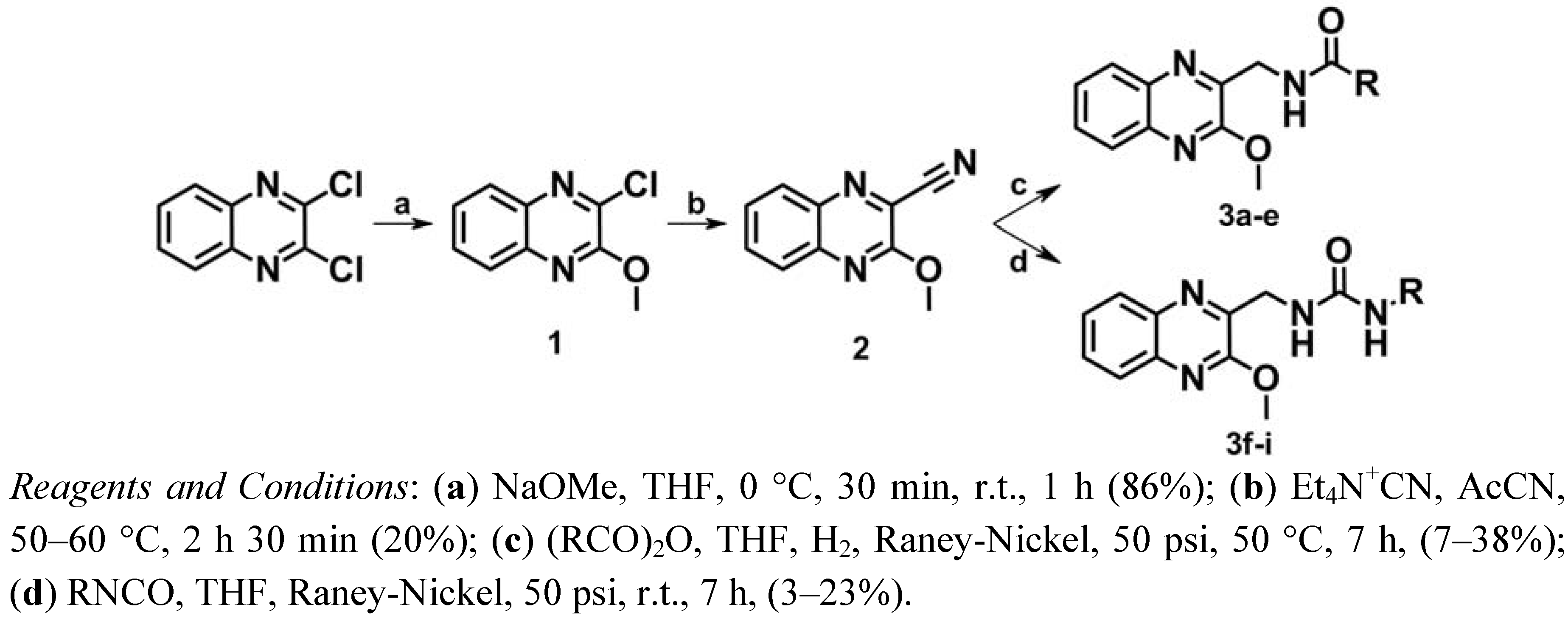
The amide derivatives 3a–e were obtained according to previously described methods [43,44]. Hydrogenation of the nitrile group of 2 over Raney-Nickel in THF and concomitant N-acylation with a suitable anhydride gave the desired amide derivatives. In the case of the urea derivatives 3f–i, several trials and strategies were carried out. At the beginning, a classical two-step strategy was designed in which a catalytic hydrogenation of the nitrile group of 2 over Raney-Nickel in ammonia and ethanol provided the desired amines which were converted to urea by reaction of the primary amine with the corresponding isocyanate [45]. Due to the failure of this strategy, further investigations regarding these reactions led to the development of a novel one-step procedure. Reduction of the nitrile group of 2 by hydrogen over Raney-Nickel in THF and concomitant addition of an appropriate isocyanate gave the desired urea derivatives mixed with the hydrolyzed isocyanates, as expected.
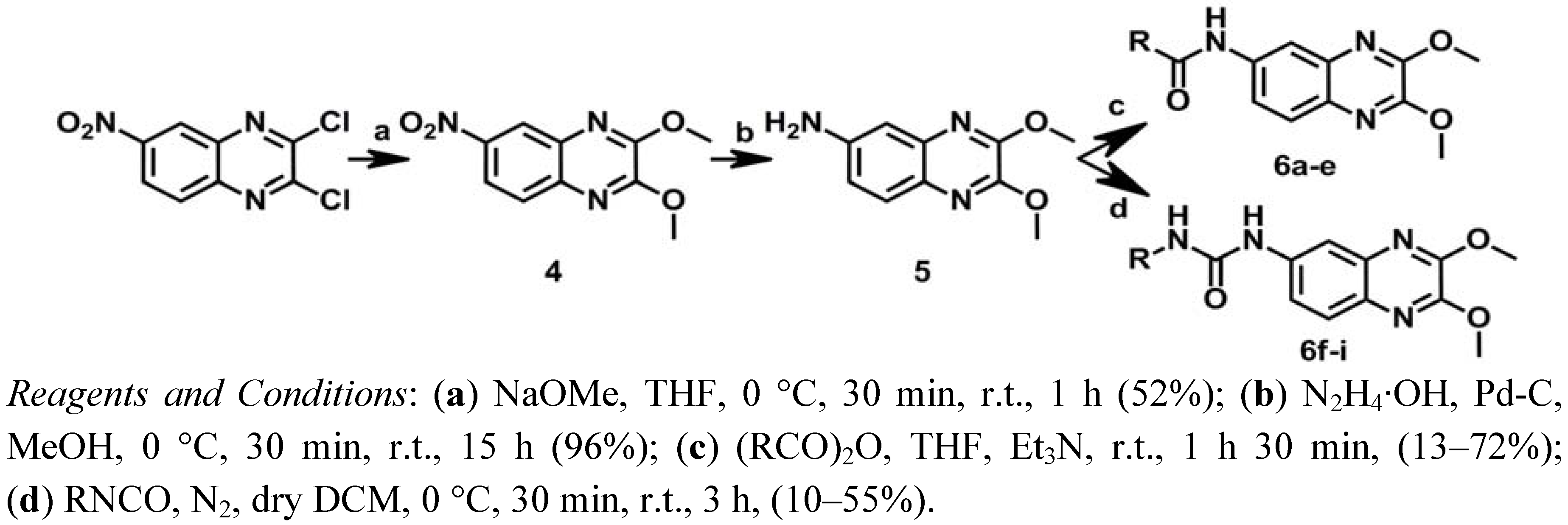
With the aim of obtaining the compounds of series II, a new synthetic route was designed, as shown in Scheme 2. 2,3-Dichloro-6-nitroquinoxaline was first treated with sodium methoxide in THF to give 4 [16]. Subsequently, compound 5 was obtained through the reduction of the nitrile group of 4with hydrazinium hydroxide and palladium on carbon [46]. Finally, N-acylation of compound 5 using a suitable anhydride and triethylamine in THF yielded the corresponding amide analogs 6a–e [47], while treatment of 5 with different isocyanates in dichloromethane (DCM) afforded the urea analogs 6f–i [45].
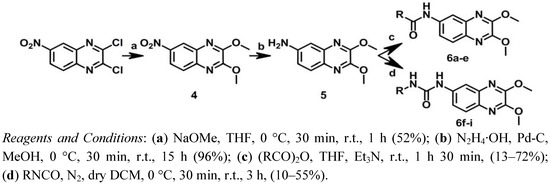
Scheme 2.
Synthesis of series II.
Scheme 2.
Synthesis of series II.
Compounds 10a–e of series III were synthesized as depicted in Scheme 3. A modified Beirut reaction was adopted for the preparation of 7 by condensation of 6-methoxybenzofuroxane and malononitrile.
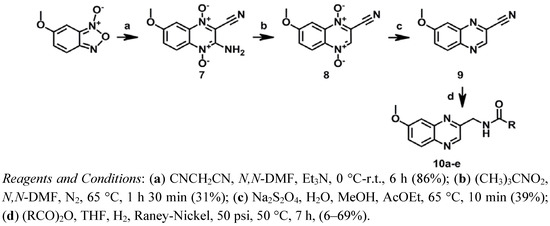
Scheme 3.
Synthesis of series III.
Scheme 3.
Synthesis of series III.
Next, compound 7 was deaminated leading to compound 8 by means of the corresponding diazonium salt intermediate, using tert-butyl nitrite in N,N-dimethylformamide (N,N-DMF) as previously described [48]. Treatment of compound 8 with sodium dithionite in water in the presence of methanol (MeOH) and ethyl acetate (AcOEt) provided compound 9 [32,49]. Finally the amide derivatives, 10a–e, were obtained by the hydrogenation of the nitrile group of 9 over Raney-Nickel in THF and concomitant N-acylation with a suitable anhydride as previously described [43,44].
2.2. Pharmacology and Structure-Activity Relationship
The binding affinity of the newly synthesized quinoxalines for MT1 and MT2 receptors was tested through competitive binding experiments using 2-[125I]Iodomelatonin as radioligand. Biological evaluation was performed according to previously described methods [50] and the obtained results are reported in Table 1.

Table 1.
MT1 and MT2 binding affinities (µM) of new synthesized Quinoxalines.
| Quinoxalines | Comp. | R | MT1 Ki (µM) ± SEM | MT2 Ki (µM) ± SEM |
|---|---|---|---|---|
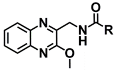 | 3a | CH3 | 2.98 ± 0.36 | 0.88 ± 0.30 |
| 3b | CH2CH3 | 2.60 ± 0.23 | 0.66 ± 0.01 | |
| 3c | CH2CH2CH3 | 0.75 ± 0.36 | 1.10 ± 0.03 | |
| 3d | CH(CH3)2 | 1.23 ± 0.32 | 0.47 ± 0.02 | |
| 3e | Ph | >103 | >103 | |
| 3f | NHCH2CH3 | >103 | 0.40 ± 0.13 | |
| 3g | NHCH2CH2CH3 | >103 | 0.44 ± N.D. | |
| 3h | NHCH(CH3)2 | >103 | >103 | |
| 3i | NHPh | >103 | >103 | |
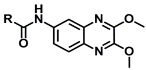 | 6a | CH3 | 20.00 ± 1.82 | 0.08 ± N.D. |
| 6b | CH2CH3 | 17.60 ± 7.81 | 4.36 ± 1.22 | |
| 6c | CH2CH2CH3 | 11.50 ± 2.80 | 1.35 ± 0.31 | |
| 6d | CH(CH3)2 | 3.40 ± 1.37 | 10.50 ± 2.96 | |
| 6e | Ph | >103 | >103 | |
| 6f | NHCH2CH3 | 3.41 ± 1.89 | 28.80 ± N.D | |
| 6g | NHCH2CH2CH3 | 1.63 ± 0.44 | 0.489 ± 0.07 | |
| 6h | NHCH(CH3)2 | >103 | >103 | |
| 6i | NHPh | >103 | >103 | |
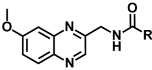 | 10a | CH3 | >103 | >103 |
| 10b | CH2CH3 | >103 | 0.34 ± 0.15 | |
| 10c | CH2CH2CH3 | 0.21 ± 0.11 | 0.10 ± 0.01 | |
| 10d | CH(CH3)2 | 0.32 ± 0.04 | 0.16 ± 0.00 | |
| 10e | Ph | >103 | >103 | |
 | MLT | 0.14·× 10−3 ± 0.03·× 10−3 | 0.41·× 10−3 ± 0.04·× 10−3 |
Binding affinities (μM) are expressed as mean Ki ± SEM of at least three independent experiments. N.D. not determined.
As can be observed in Table 1, compounds 10c and 10d of Series III present the best affinity profile towards MT1/MT2 receptors. Comparing these data with their analogues 3c and 3d of Series I, it can be concluded that the existence of the six-atom distance between the methoxy group and the first nitrogen atom of the side chain is essential for obtaining a good melatoninergic MT1/MT2 binding affinity. Derivative 10c exhibited the best melatoninergic binding affinity with both receptors and therefore, this compound could be selected as a hit in the search for new and more active quinoxalines acting as MT1/MT2 receptor ligands.
As shown by compounds of Series I and II, shifting the methoxy group from position C6 to C2 of the quinoxaline moiety can modulate the affinity and selectivity of these ligands depending on the distance between the methoxy group and the N-acylamino chain. The comparison of the obtained data for compounds of both series and Series III reveals that switching the methoxy group to position 2 of the quinoxaline ring has a much greater effect on binding affinity to the MT2 receptor subtype than it does on affinity to MT1 receptor, displaying a significant selectivity for MT2 receptor in Series II, where the relative spatial distance (six-atoms) between the two key pharmacophoric elements is also the optimal.
Compound 6a showed the most promising profile in terms of affinity and selectivity for the MT2 receptor with MT2/MT1 selectivity ratio of 250 and the highest binding affinity (0.08 μM) for the MT2 receptor. These results indicate that compound 6a is a potent and selective MT2 ligand. Furthermore, the functional activity of two of the best compounds of this series (6c and 6a) has been evaluated only on the MT2 receptor subtype, due to the weak affinity for the MT1 subtype. The results are shown in Table 2. Compound 6c showed full agonist profile with a EC50 = 1.3 μM and Emax = 84%. In the case of compound 6a, due to the fact that this compounds exhibited an EC50 > 10 μM, the compound 6a might considered as an antagonist.

Table 2.
MT2 GTPγS (+) binding affinities of compounds 6a and 6c.
| Compound | MT2 | |
|---|---|---|
| EC50 ± SEM (μM) | Emax ± SEM (%) | |
| MLT | 0.49 ×·10−3 ± 0.05·× 10−3 | 100 |
| 6a | >10 | |
| 6c | 1.3 ± 0.26 | 84 ± 9.5 |
Concentration-response curves were analysed by non-linear regression. Agonist potency was expressed as EC50 ± SEM (μM) while the maximal efficacy, Emax ± SEM was expressed as a Percentage of that observed with melatonin 1 μM (100%).
With regard to the amide/urea substitution, the results are inconclusive, therefore, it cannot be determined which group (amide/urea) leads to better affinity for MT1/MT2 receptors. Comparing the results obtained from Series I, it could be assumed that the amide derivatives 3a–e generally present better affinity for MT1 receptor than their homologous urea derivatives 3f–i. However, the affinity of urea derivatives for MT2 is higher than the affinity of amide derivatives for this same receptor, provided that the aliphatic chain linked to the urea/amide group is a linear aliphatic chain. In contrast, taking into account the results obtained for the compounds from Series II, it could be concluded that the urea derivatives 6a–e present higher affinity for both receptors than their homologous amide derivatives 6f–i, provided that the aliphatic chain linked to the urea/amide group is a linear aliphatic chain. Thus, it can be said that the introduction of a urea function, instead of the amide function existing in the MLT, does not negatively affect the affinity of the ligands for MT1 and MT2 receptors. In addition, since it is already well known that urea derivatives usually present better metabolic stability than amide derivatives, the urea derivatives are preferred.
Finally, the introduction of a benzene ring, replacing the aliphatic chain substituted over the amide/urea group, results in a total loss of affinity. By contrast, substitution with an aliphatic linear chain in general, and particularly with a propyl group, appeared to be the best option in order to obtain high affinity compounds.
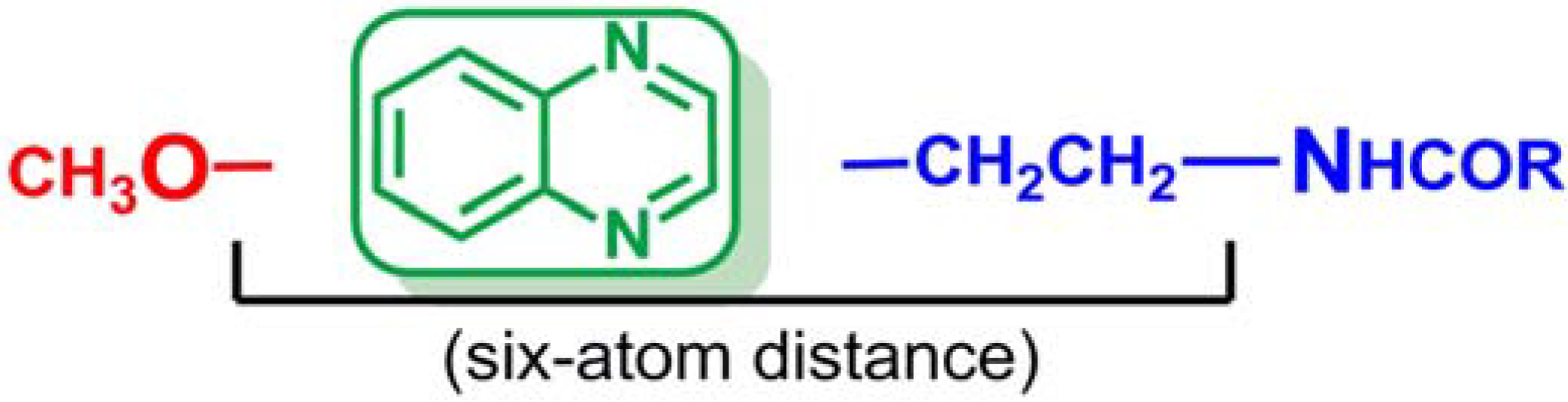
With these results, we were not able to state that the quinoxaline scaffold can act as a useful bioisostere of indole in the search for melatonin receptor ligands. In addition, The SAR study suggested that a six-atom length is the best distance between the methoxy group and the first nitrogen atom of the side chain in order to obtain high MT1/MT2 affinity. Moreover, new bibliographic review has revealed that the optimum distance should be obtained through a two-methylene linker in order to allow compounds to adopt a MLT-like configuration [51]. Thus, the structural requirements initially described have been redefined and the synthesis of a new series of quinoxalines has been proposed in which the aforementioned distance is obtained through a two-methylene linker (Figure 3). These conclusions open a new line of research in order to determine if the quinoxaline ring is suitable as central scaffold of melatoninergic ligands.
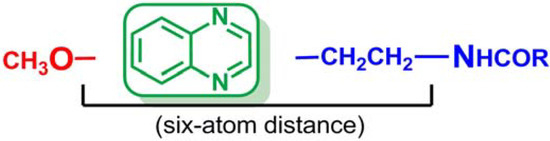
Figure 3.
Structural requirements of future series of quinoxalines as MT1/MT2 receptor ligands.
Figure 3.
Structural requirements of future series of quinoxalines as MT1/MT2 receptor ligands.
3. Experimental
3.1. Chemical Synthesis
3.1.1. General Remarks
All of the synthesized compounds were chemically characterized by thin layer chromatography (TLC), infrared (IR), proton nuclear magnetic resonance (1H-NMR) and elemental microanalyses (CHN). Alugram SIL G/UV254 (Layer: 0.2 mm) (Macherey-Nagel GmbH & Co. KG., Düren, Germany) was used for TLC, and Silica gel 60 (0.040–0.063 mm, Merck, Darmstadt, Germany) was used for flash column chromatography. The 1H-NMR spectra were recorded on a Bruker 400 Ultrashield instrument (Bruker, Rheinstetten, Germany) 400 MHz), using TMS as internal standard and with DMSO-d6 or CDCl3 as solvents; the chemical shifts are reported in ppm (δ) and coupling constant (J) values are given in Hertz (Hz). Signal multiplicities are represented by: s (singlet), bs (broad singlet), d (doublet), dd (doublet of doublets), ddd (doublet of doublet of doublets), t (triplet), dt (doublet of triplets), tt (triplet of triplets); q (quartet); dq (doublet of quartets) and m (multiplet). The IR spectra were recorded on a Nicolet Nexus FTIR (Thermo, Madison, WI, USA) in KBr pellets. Elemental microanalyses were obtained on a CHN 900 Elemental Analyzer (Leco, Tres Cantos, Spain) from vacuum-dried samples. The analytical results for C, H and N, were within ±0.4 of the theoretical values. Chemicals were purchased from Panreac Química S.A. (Montcada i Reixac, Spain), Sigma-Aldrich-Fluka Química S.A. (Alcobendas, Spain), Alfa Aesar-Avocado GmbH & Co. KG (Karlsruhe, Germany), and E. Merck (Darmstadt, Germany).
3.1.2. Synthesis of 2-Chloro-3-methoxyquinoxaline (1)
2,3-Dichloroquinoxaline (90%, 2.30 g, 10.04 mmol) and THF (20.00 mL) were placed in a flask. The mixture was cooled to 0 °C. A solution of sodium methoxide was obtained from the reaction between Na (0.50 g) and methanol (5.00 mL). NaOMe (2.34 mL, 10.04 mmol) was added dropwise to the other suspension. The color of the mixture changed from purple to yellow. The mixture was stirred during 30 min at 0 °C and during 1 h at room temperature. DCM (80.00 mL) was added and the mixture was quenched with brine. The DCM solution was dried over Na2SO4 and filtered. The solvent was removed under reduced pressure to give compound 1 as a yellow solid. Yield: 86%. IR (cm−1): 3032, 2987, 2936. 1H-NMR (CDCl3) δ: 7.96 (ddd, 1H, H8, J8-7 = 8.3 Hz, J8-6 = 1.5 Hz, J8-5 = 0.5 Hz); 7.88 (ddd, 1H, H5, J5-6 = 8.3 Hz, J5-7 = 1.4 Hz, J5-8 = 0.5 Hz); 7.71 (ddd, 1H, H6, J6-5 = 8.4 Hz, J6-7 = 7.1 Hz, J6-8 = 1.5 Hz); 7.60 (ddd, 1H, H7, J7-8 = 8.4 Hz, J7-6 = 7.1 Hz, J7-5 = 1.5 Hz); 4.20 (s, 3H, OCH3) ppm. Anal. Calcd for C9H7N2OCl: C: 55.53%; H: 3.60%; N: 14.40%; Found: C: 55.23%; H: 3.34%; N: 14.41%.
3.1.3. Synthesis of 3-Methoxyquinoxaline-2-carbonitrile (2)
Compound 1 (1.59 g, 8.17 mmol) and tetraethylammonium cyanide (2.55 g, 16.32 mmol) were dissolved in acetonitrile (60.00 mL). The mixture was heated between 50–60 °C during 2 h 30 min. The solution was poured onto ice and a black precipitate was formed which was purified by silica gel column chromatography using DCM as mobile phase. The solvent was removed under reduced pressure to yield compound 2. Yield: 20%. IR (cm−1): 3026, 2943, 2232. 1H-NMR (CDCl3) δ: 8.08 (ddd, 1H, H8, J8-7 = 8.4 Hz, J8-6 = 1.5 Hz, J8-5 = 0.5 Hz); 7.88 (ddd, 1H, H5, J5-6 = 8.3 Hz, J5-7 = 1.4 Hz, J5-8 = 0.5 Hz); 7.71 (ddd, 1H, H6, J6-5 = 8.4 Hz, J6-7 = 6.9 Hz, J6-8 = 1.4 Hz); 7.60 (ddd, 1H, H7, J7-8 = 8.4 Hz, J7-6 = 6.9 Hz, J7-5 = 1.5 Hz); 4.20 (s, 3H, OCH3) ppm. Anal. Calcd for C10H7N3O: C: 64.86%; H: 3.78%; N: 22.70%; Found: C: 65.23%; H: 3.61%; N: 22.70%.
3.1.4. General Procedure for Synthesis of N-(3-Methoxyquinoxalin-2-ylmethyl)-alkylamidea 3a–e
The previously obtained compound 2, THF and the corresponding anhydride in proportion 1:2 were placed in a hydrogenation flask. A spatula of Raney-Nickel was added and the mixture was hydrogenated during 7 h at 50 °C and 50 psi. Next, DCM was added and the mixture was decanted off. Next, the mixture was quenched with water, dried over Na2SO4 and filtered. The solvent was removed under reduced pressure to obtain brown oil. Finally, n-hexane was added to the oil and a white solid precipitated, which was filtered and washed with diethyl ether and water. If necessary, it was purified by silica gel column chromatography using DCM/MeOH as mobile phase. The solvent was removed under reduced pressure and the desired amides 3a–e were thus obtained.
N-(3-Methoxyquinoxalin-2-ylmethyl)-acetamide (3a). Yield: 7%. IR (cm−1): 3314, 3084, 2937, 1648. 1H-NMR (CDCl3) δ: 8.11 (dd, 1H, H8, J8-7 = 8.1 Hz, J8-6 = 1.1 Hz); 7.91 (ddd, 1H, H5, J5-6 = 8.3 Hz, J5-7 = 1.3 Hz); 7.71 (ddd, 1H, H6, J6-5 = 8.3 Hz, J6-7 = 7.1 Hz, J6-8 = 1.3 Hz); 7.62 (ddd, 1H, H7, J7-8 = 8.3 Hz, J7-6 = 7.1 Hz, J7-5 = 1.4 Hz); 4.76 (d, 2H, CH2, JCH2-NH = 4.3 Hz); 4.17 (s, 3H, OCH3); 2.21 (s, 3H, CH3) ppm. Anal. Calcd for C12H13N3O2 · 1/4 H2O: C: 61.14%; H: 5.73%; N: 17.83%; Found: C: 61.26%; H: 5.47%; N: 17.65%. M.p.: 231–235 °C.
N-(3-Methoxyquinoxalin-2-ylmethyl)-propionamide (3b). Yield: 27%. IR (cm−1): 3308, 3078, 2969, 2943, 1638.1H-NMR (DMSO-d6) δ: 8.27 (t, 1H, NH, JNH-CH2 = 5.3 Hz); 7.96 (dd, 1H, H8, J8-7 = 8.2 Hz, J8-6 = 1.2 Hz); 7.85 (dd, 1H, H5, J5-6 = 8.3 Hz, J5-7 = 1.0 Hz); 7.72 (ddd, 1H, H6, J6-5 = 8.3 Hz, J6-7 = 7.0 Hz, J6-8 = 1.5 Hz); 7.63 (ddd, 1H, H7, J7-8 = 8.3 Hz, J7-6 = 7.0 Hz, J7-5 = 1.5 Hz); 4.53 (d, 2H, CH2NH, JCH2-NH = 5.6 Hz); 4.07 (s, 3H, OCH3); 2.23 (q, 2H, CH2CH3, JCH2-CH3 = 7.6 Hz); 1.06 (t, 3H, CH2CH3, JCH3-CH2 = 7.6 Hz) ppm. Anal. Calcd for C13H15N3O2 · 1/10 H2O: C: 63.21%; H: 6.16%; N: 17.02%; Found: C: 63.44%; H: 6.08%; N: 16.65%. M.p.: 164 °C.
N-(3-Methoxyquinoxalin-2-ylmethyl)-butyramide (3c). Yield: 9%. IR (cm−1): 3309, 3065, 2951, 1641. 1H-NMR (DMSO-d6) δ: 8.30 (bs, 1H, NH); 7.93 (d, 1H, H8, J8-7 = 7.9 Hz); 7.84 (d, 1H, H5, J5-6 = 8.2 Hz); 7.70 (dd, 1H, H6, J6-5 = 8.2 Hz, J6-7 = 6.7 Hz); 7.61 (dd, 1H, H7, J7-8 = 8.1 Hz, J7-6 = 6.9 Hz); 4.52 (d, 2H, CH2NH, JCH2-NH = 5.4 Hz); 4.07 (s, 3H, OCH3); 2.18 (t, 2H, CH2CH2CH3, JCH2-CH2 = 7.2 Hz); 1.56 (dd, 2H, CH2CH2CH3, JCH2-CH2 = 7.2 Hz, JCH2-CH3 = 7.2 Hz); 0.91 (t, 3H, CH3, JCH3-CH2 = 7.3 Hz) ppm. Anal. Calcd for C14H17N3O2·1/8 H2O: C: 64.31%; H: 6.60%; N: 16.08%; Found: C: 64.34%; H: 6.65%; N: 15.93%. M.p.: 259 °C.
N-(3-Methoxyquinoxalin-2-ylmethyl)-isobutyramide (3d).Yield: 28%. IR (cm−1): 3312, 3065, 2968, 2937, 1644. 1H-NMR (DMSO-d6) δ: 8.23 (t, 1H, NH, JNH-CH2 = 5.6 Hz); 7.93 (dd, 1H, H8, J8-7 = 8.2 Hz, J8-6 = 1.0 Hz); 7.85 (dd, 1H, H5, J5-6 = 8.2 Hz, J5-7 = 0.9 Hz); 7.72 (ddd, 1H, H6, J6-5 = 8.3 Hz, J6-7 = 7.1 Hz, J6-8 = 1.5 Hz); 7.62 (ddd, 1H, H7, J7-8 = 8.4 Hz, J7-6 = 7.1 Hz, J7-5 = 1.5 Hz); 4.51 (d, 2H, CH2NH, JCH2-NH = 5.6 Hz); 4.07 (s, 3H, OCH3); 2.55 (q, 1H, CH(CH3)2, JCH-(CH3)2 = 6.8 Hz); 1.08 (d, 6H, CH(CH3)2, J(CH3)2-CH = 6.8 Hz) ppm. Anal. Calcd for C14H17N3O2:C: 64.86%; H: 6.56%; N: 16.22%; Found: C: 64.55%; H: 6.79%; N: 15.91%. M.p.: 142–144 °C.
N-(3-Methoxy-quinoxalin-2-ylmethyl)-benzamide (3e). Yield: 38%. IR (cm−1): 3391, 3071, 3026, 2943, 2904, 1659. 1H-NMR (DMSO-d6) δ: 9.00 (t, 1H, NH, JNH-CH2 = 5.4 Hz); 7.94-7.92 (m, 3H, H8+H2'+H6'); 7.86 (d, 1H, H5, J5-6 = 8.3 Hz); 7.71 (dd, 1H, H6, J6-5 = 8.1 Hz, J6-7 = 7.2 Hz); 7.63-7.47 (m, 4H, H7+H3'+H4'+H5'); 4.74 (d, 2H, CH2, JCH2-NH = 5.4 Hz); 4.10 (s, 3H, OCH3) ppm. Anal. Calcd for C17H15N3O2 · 1/8 H2O: C: 69.09%; H: 5.17%; N: 14.23%; Found: C: 69.13%; H: 5.14%; N: 13.99%. M.p.: 137 °C.
3.1.5. General Procedure for Synthesis of 1-Alkyl-3-(3-methoxyquinoxalin-2-ylmethyl)ureas 3f–i
Compound 2 (1 eq.), THF and the corresponding isocyanate (2 eq.) were placed in a hydrogenation tube. A spatula of Raney-Nickel was added and the reaction was carried out during 7 h at room temperature and 50 psi. Next, DCM was added and the mixture was decanted off with the aim of leaving behind the catalyst. Next, the mixture was quenched with water, dried over Na2SO4 and filtered. The solvent was removed under reduced pressure and the orange oil obtained was purified by flash chromatography using DCM/MeOH as mobile phase. The obtained solid was dissolved in water/diethyl ether with vigorous stirring and then quenched. The organic layer was dried over Na2SO4 and filtered. The solvent was removed under reduced pressure and the desired urea derivatives 3f–i, were obtained.
Ethyl-3-(3-methoxyquinoxalin-2-ylmethyl)urea (3f). Yield: 3%. IR (cm−1): 3334, 3061, 2978, 2939, 1629. 1H-NMR (DMSO-d6) δ: 7.96 (dd, 1H, H8, J8-7 = 8.2 Hz, J8-6 = 1.2 Hz); 7.85 (dd, 1H, H5, J5-6 = 8.2 Hz, J5-7 = 1.2 Hz); 7.71 (ddd, 1H, H6, J6-5 = 8.4 Hz, J6-7 = 7.0 Hz, J6-8 = 1.4 Hz); 7.63 (ddd, 1H, H7, J7-8 = 8.4 Hz, J7-6 = 7.1 Hz, J7-5 = 1.4 Hz); 6.41 (t, 1H, ArCH2NH, JNH-CH2 = 5.0 Hz); 6.33 (t, 1H, NHCH2CH3, JNH-CH2 = 5.3 Hz); 4.50 (d, 2H, ArCH2NH, JCH2-NH = 5.4 Hz); 4.07 (s, 3H, OCH3); 3.05 (dq, 2H, CH2CH3, JCH2-CH3 = 7.2 Hz, JCH2-NH = 5.8 Hz); 1.02 (dt, 3H, CH3, JCH3-CH2 = 7.2 Hz, JCH3-NH = 1.3 Hz) ppm. Anal. Calcd for C13H16N4O2: C: 60.00%; H: 6.15%; N: 21.54%; Found: C: 59.91%; H: 6.12%; N: 21.14%. M.p.: 160.5–160.8 °C.
1-(3-Methoxyquinoxalin-2-ylmethyl)-3-propylurea (3g). Yield: 9%. IR (cm−1): 3330, 3061, 2960, 1633. 1H-NMR (DMSO-d6) δ: 7.95 (d, 1H, H8, J8-7 = 8.1 Hz); 7.85 (d, 1H, H5, J5-6 = 8.2 Hz); 7.71 (dd, 1H, H6, J6-5 = 8.0 Hz, J6-7 = 7.3 Hz); 7.63 (dd, 1H, H7, J7-8 = 8.0 Hz, J7-6 = 7.1 Hz); 6.41 (t, 1H, ArCH2NH, JNH-CH2 = 5.5 Hz); 6.37 (t, 1H, NHCH2CH2CH3, JNH-CH2 = 6.0 Hz); 4.50 (d, 2H, ArCH2NH, JCH2-NH = 5.2 Hz); 4.07 (s, 3H, OCH3); 2.98 (dd, 2H, NHCH2CH2CH3, JCH2-NH = 6.4 Hz, JCH2-CH2 = 6.7 Hz); 1.40 (tq, 2H, NHCH2CH2CH3, JCH2-CH2 = 6.8 Hz, JCH2-CH3 = 7.1 Hz); 0.86 (t, 3H, CH3, JCH3-CH2 = 7.4 Hz) ppm. Anal. Calcd for C14H18N4O2: C: 61.31%; H: 6.57%; N: 20.44%; Found: C: 60.99%; H: 6.45%; N: 20.31%. M.p.: 115.3–116 °C.
1-Isopropyl-3-(3-methoxyquinoxalin-2-ylmethyl)urea (3h). Yield: 23%. IR (cm−1): 3363, 3283, 3100, 2971, 1620. 1H-NMR (DMSO-d6) δ: 7.95 (dd, 1H, H8, J8-7 = 8.1 Hz, J8-6 = 1.1 Hz); 7.85 (dd, 1H, H5, J5-6 = 8.2 Hz, J5-7 = 0.9 Hz); 7.71 (ddd, 1H, H6, J6-5 = 8.2 Hz, J6-7 = 7.7 Hz, J6-8 = 1.2 Hz); 7.63 (ddd, 1H, H7, J7-8 = 8.0 Hz, J7-6 = 7.4 Hz, J7-5 = 1.1 Hz); 6.34 (t, 1H, ArCH2NH, JNH-CH2 = 5.1 Hz); 6.26 (d, 1H, NHCH(CH3)2, JNH-CH = 7.6 Hz); 4.49 (d, 2H, ArCH2NH, JCH2-NH = 5.3 Hz); 4.07 (s, 3H, OCH3); 3.71–3.67 (m, 1H, CH(CH3)2); 1.06 (d, 6H, CH-(CH3)2, J(CH3)2-CH = 6.5 Hz) ppm. Anal. Calcd for C14H18N4O2: C: 61.31%; H: 6.57%; N: 20.44%; Found: C: 61.33%; H: 6.58%; N: 20.43%. M.p.: 171.2 °C.
1-(3-Methoxyquinoxalin-2-ylmethyl)-3-phenyl-urea (3i). Yield: 4%. IR (cm−1): 3310, 3177, 3023, 2952, 2907, 1640. 1H-NMR (DMSO-d6) δ: 9.01 (s, 1H, CONH-Ph); 7.99 (dd, 1H, H8, J8-7 = 8.2 Hz, J8-6 = 1.2 Hz); 7.87 (dd, 1H, H5, J5-6 = 8.3 Hz, J5-7 = 1.1 Hz); 7.73 (ddd, 1H, H6, J6-5 = 8.4 Hz, J6-7 = 7.1 Hz, J6-8 = 1.5 Hz); 7.64 (ddd, 1H, H7, J7-8 = 8.4 Hz, J7-6 = 7.1 Hz, J7-5 = 1.5 Hz); 7.43 (dd, 2H, H2'+H6', J2'-3' = J6'-5' = 8.6 Hz, J2'-4' = J6'-4' = 1.1 Hz); 7.24 (dd, 2H, H3'+H5', J3'-2' = J5'-6' = 8.4Hz, J3'-4' = J5'-4' = 7.5 Hz); 6.91 (tt, 1H, H4', J4'-3' = J4'-5' = 7.5 Hz, J4'-2' = J4'-6' = 1.1Hz); 6.83 (t, 1H, ArCH2NH, JNH-CH2 = 5.2 Hz); 4.61 (d, 2H, ArCH2NH, JCH2-NH = 5.2 Hz); 4.10 (s, 3H, OCH3) ppm. Anal. Calcd for C17H16N4O2 · 1/8 H2O: C: 65.75%; H: 5.24%; N: 18.05%; Found: C: 65.76%; H: 5.32%; N: 18.12%. M.p.: 201–201.5 °C.
3.1.6. Synthesis of 2,3-Dimethoxy-6-nitroquinoxaline (4)
2,3-Dichloro-6-nitroquinoxaline (5.12 g, 20.35 mmol) and THF (100.00 mL) were placed in a round bottomed flask. The mixture was cooled to 0 °C. NaOMe (9.6 mL, 41.00 mmol) prepared in situ by means of a reaction between Na (1.20 g) and MeOH (12.00 mL) was added dropwise to the other suspension. The color of the mixture changed from white-yellow to orange. The mixture was stirred during 30 min at 0 °C and during 1 h at room temperature. Then, DCM was added and the mixture was quenched with brine. The DCM solution was dried over Na2SO4 and filtered. The solvent was removed under reduced pressure in order to obtain compound 4 as a yellow solid. Yield: 52%. IR (cm−1): 3116, 3014, 2956. 1H-NMR (DMSO-d6) δ: 8.50 (d, 1H, H5, J5-7 = 2.6 Hz); 8.30 (dd, 1H, H7, J7-8 = 9.0 Hz, J7-5 = 2.6 Hz); 7.93 (d, 1H, H8, J8-7 = 9.0 Hz); 4.11 (s, 3H, OCH3-C2); 4.10 (s, 3H, OCH3-C3) ppm. Anal. Calcd for C10H9N3O4: C: 51.06%; H: 3.83%; N: 17.87%; Found: C: 50.99%; H: 3.79%; N: 17.40%.
3.1.7. Synthesis of 6-Amino-2,3-dimethoxyquinoxaline (5)
Compound 4 (2.50 g, 10.64 mmol) was disolved in MeOH (150.00 mL) and the mixture was cooled to 0 °C. Then a tip of spatula of palladium on carbon and a great excess of hydrazine hydroxide were added. After stirring 30 min at 0 °C the mixture was stirred at room temperature for 15 h. The mixture was filtered to eliminate the catalyst and the solvent was removed under reduced pressure. Then, DCM was added and the mixture was quenched with brine. The DCM solution was dried over Na2SO4 and filtered. The solvent was removed under reduced pressure yielding to compound 5. Yield: 96%. IR (cm−1): 3442, 3347, 3219, 2937. 1H-NMR (DMSO-d6) δ: 7.42 (d, 1H, H8, J8-7 = 8.6 Hz); 6.87 (dd, 1H, H7, J7-8 = 8.6 Hz, J7-5 = 2.4 Hz); 6.78 (d, 1H, H5, J5-7 = 2.5 Hz); 5.43 (s, 2H, NH2); 3.97 (s, 3H, OCH3-C2); 3.94 (s, 3H, OCH3-C3) ppm. Anal. Calcd for C10H11N3O2: C: 58.54%; H: 5.37%; N: 20.49%; Found: C: 58.08%; H: 5.37%; N: 20.41%.
3.1.8. General Procedure for Synthesis of N-(2,3-Dimethoxyquinoxalin-6-yl)alkylamides 6a–e
Compound 5 (1 eq.) was placed in suspension in THF. Next, the corresponding anhydride (3 eq.) and a catalytic amount of triethylamine were added dropwise. The reaction was carried out during 1 h 30 min at room temperature. THF was removed under reduced pressure. The obtained residue was treated with n-hexane and the obtained solid was then filtered. The solid was washed with water to yield the desired compounds 6a–e.
N-(2,3-Dimethoxyquinoxalin-6-yl)acetamide (6a). Yield: 13%. IR (cm−1): 3276, 3116, 2943, 1664. 1H-NMR (DMSO-d6) δ: 10.19 (s, 1H, NH); 8.21 (d, 1H, H5, J5-7 = 2.2 Hz); 7.67 (d, 1H, H8, J8-7 = 8.8 Hz); 7.57 (dd, 1H, H7, J7-8 = 8.9 Hz, J7-5 = 2.2 Hz); 4.03 (s, 3H, OCH3-C2); 4.01 (s, 3H, OCH3-C3); 2.10 (s, 3H, CH3) ppm. Anal. Calcd for C12H13N3O3 · 1/2 H2O: C: 56.25%; H: 5.47%; N: 16.41%; Found: C: 56.23%; H: 5.42%; N: 16.04%. M.p.: 290 °C.
N-(2,3-Dimethoxyquinoxalin-6-yl)propionamide (6b). Yield: 68%. IR (cm−1): 3284, 3175, 3105, 2987, 2943, 1663. 1H-NMR (DMSO-d6) δ: 10.11 (s, 1H, NH); 8.21 (s, 1H, H5); 7.67 (dd, 1H,H8, J8-7 = 8.8 Hz, J8-5 = 1.2 Hz); 7.60 (ddd, 1H, H7, J7-8 = 8.9 Hz, J7-5 = 2.2 Hz, JH7-NH = 1.5 Hz); 4.03 (d, 3H, OCH3-C2, JOCH3-OCH3 = 1.4 Hz); 4.01 (d, 3H, OCH3-C3, JOCH3-OCH3 = 1.4 Hz); 2.38 (dq, 2H, CH2, JCH2-CH3 = 7.5 Hz, JCH2-NH = 1.3 Hz); 2.12 (dt, 3H,CH3, JCH3-CH2 = 7.5 Hz, JCH3-NH = 1.4 Hz) ppm. Anal. Calcd for C13H15N3O3 · 1/2 H2O: C: 57.78%; H: 5.93%; N: 15.56%; Found: C: 58.15%; H: 5.92%; N: 15.61%. M.p.: 182–183 °C.
N-(2,3-Dimethoxyquinoxalin-6-yl)butyramide (6c). Yield: 26%. IR (cm−1): 3289, 2949, 1656. 1H-NMR (DMSO-d6) δ: 10.11 (s, 1H, NH); 8.21 (s, 1H, H5); 7.66 (dd, 1H, H7, J7-8 = 8.4 Hz, J7-5 = 2.9 Hz); 7.59 (d, 1H, H8, J8-7 = 8.7 Hz); 4.03 (d, 3H, OCH3-C2, JOCH3-OCH3 = 2.6 Hz); 4.01 (d, 3H, OCH3-C3, JOCH3-OCH3 = 2.6 Hz); 2.36–2.32 (m, 2H, COCH2CH2CH3); 1.66–1.62 (m, 2H, COCH2CH2CH3); 0.96–0.92 (m, 3H, CH3) ppm. Anal. Calcd for C14H17N3O3: C: 61.09%; H: 6.18%; N: 15.27%; Found: C: 60.78%; H: 6.32%; N: 15.59%. M.p.: 136–140 °C.
N-(2,3-Dimethoxyquinoxalin-6-yl)isobutyramide (6d). Yield: 72%. IR (cm−1): 3253, 3026, 2969, 1653. 1H-NMR (DMSO-d6) δ: 10.07 (s, 1H, NH); 8.21 (d, 1H, H5, J5-7 = 2.2 Hz); 7.67 (d, 1H, H8, J8-7 = 8.9 Hz); 7.62 (dd, 1H,H7, J7-8 = 8.9 Hz, J7-5 = 2.3 Hz); 4.03 (d, 3H, OCH3-C2, JOCH3-OCH3 = 1.2 Hz); 4.01 (d, 3H, OCH3-C3, JOCH3-OCH3 = 1.3 Hz); 2.64 (dq, 1H, CH(CH3)2, JCH-(CH3)2 = 6.7 Hz); 1.14 (d, 6H, CH(CH3)2, J(CH3)2-CH = 6.8 Hz) ppm. Anal. Calcd for C14H17N3O3 · 1/8 H2O: C: 60.60%; H: 6.22%; N: 15.15%; Found: C: 60.60%; H: 6.07%; N: 15.12%. M.p.: 201–203 °C.
N-(2,3-Dimethoxyquinoxalin-6-yl)benzamide (6e). Yield: 42%. IR (cm−1): 3285, 3061, 2984, 2939, 1651. 1H-NMR (DMSO-d6) δ: 10.49 (s, 1H, NH); 8.37 (d, 1H, H5, J5-7 = 2.2 Hz); 8.00–7.98 (m, 2H, H2'+H6'); 7.87 (dd, 1H, H7, J7-8 = 8.9 Hz, J7-5 = 2.4 Hz); 7.73 (d, 1H, H8, J8-7 = 8.8 Hz); 7.65–7.53 (m, 3H, H3'+H4'+H5'); 4.05 (s, 3H, OCH3-C2, JOCH3-OCH3 = 0.5 Hz); 4.03 (s, 3H, OCH3-C3, JOCH3-OCH3 = 0.5 Hz) ppm. Anal. Calcd for C17H15N3O3 · 1/8 H2O: C: 65.54%; H: 4.90%; N: 13.49%; Found: C: 65.52%; H: 4.99%; N: 13.40%. M.p.: 165–170 °C.
3.1.9. General Procedure for Synthesis of 1-(2,3-Dimethoxyquinoxaline-6-yl)-3-alkylureas 6f–i
Compound 5 (1 eq.) was dissolved in dry DCM (15 mL) and placed under N2 atmosphere. The mixture was cooled to 0 °C and the corresponding isocyanate (2 eq.) was added. The reaction was carried out during 30 min at 0 °C and 3 h at room temperature. Next, the obtained precipitate was filtered to achieve the corresponding compounds 6f–i.
1-(2,3-Dimethoxyquinoxalin-6-yl)-3-ethylurea (6f). Yield: 24%. IR (cm−1): 3303, 3103, 2981, 1633. 1H-NMR (DMSO-d6) δ: 8.69 (s, 1H, CONHAr); 7.97 (d, 1H, H5, J5-7 = 2.2 Hz); 7.60 (d, 1H, H8, J8-7 = 8.8 Hz); 7.41 (dd, 1H, H7, J7-8 = 8.8 Hz, J7-5 = 2.5 Hz); 6.20 (t, 1H, CONHCH2, JNH-CH2 = 5.4 Hz); 4.02 (d, 3H, OCH3-C2, JOCH3-OCH3 = 0.9 Hz); 3.99 (s, 3H, OCH3-C3, JOCH3-OCH3 = 0.9 Hz); 3.14 (dq, 2H, CH2, JCH2-NH = 5.9 Hz, JCH2-CH3 = 7.2 Hz); 1.08 (t, 3H, CH3, JCH3-CH2 = 7.3 Hz) ppm. Anal. Calcd for C13H16N4O3 · 1/4 H2OC: 55.61%; H: 5.88%; N: 19.96%; Found: C: 55.57%; H: 5.66%; N: 19.94%. M.p.: >300 °C.
1-(2,3-Dimethoxyquinoxalin-6-yl)-3-propylurea (6g). Yield: 29%. IR (cm−1): 3289, 2949, 1656. 1H-NMR (DMSO-d6) δ: 8.67 (s, 1H, CONHAr); 7.96 (dd, 1H, H5, J5-7 = 2.2 Hz, J5-8 = 0.6 Hz); 7.60 (dd, 1H, H8, J8-7 = 8.8 Hz, J8-5 = 0.6 Hz); 7.40 (ddd, 1H, H7, J7-8 = 8.9 Hz, J7-5 = 2.4 Hz, JH7-NH = 0.8 Hz); 6.24 (t, 1H, CONHCH2, JNH-CH2 = 5.8 Hz); 4.02 (s, 3H, OCH3-C2, JOCH3-OCH3 = 0.9 Hz); 3.99 (s, 3H, OCH3-C3, JOCH3-OCH3 = 0.9 Hz); 3.08 (dd, 2H, NHCH2CH2CH3, JCH2-CH2 = 6.4 Hz, JCH2-NH = 6.3 Hz); 1.47 (tq, 2H, NHCH2CH2CH3, JCH2-CH3 = 7.3 Hz, JCH2-CH2 = 7.2 Hz); 0.89 (t, 3H, CH3, JCH3-CH2 = 7.4 Hz) ppm. Anal. Calcd for C14H18N4O3: C: 57.93%; H: 6.21%; N: 19.31%; Found: C: 57.78%; H: 6.11%; N: 19.31%. M.p.: 122–123 °C.
1-(2,3-Dimethoxyquinoxalin-6-yl)-3-isopropylurea (6h). Yield: 10%. IR (cm−1): 3334, 3297, 3097, 2962, 1637. 1H-NMR (DMSO-d6) δ: 8.55 (s, 1H, CONHAr); 7.95 (dd, 1H, H5, J5-7 = 2.3 Hz, J5-8 = 1.2 Hz); 7.60 (dd, 1H, H8, J8-7 = 8.9 Hz, J8-5 = 1.0 Hz); 7.39 (ddd, 1H,H7, J7-8 = 8.9 Hz, J7-5 = 2.4 Hz, JH7-NH = 1.1 Hz); 6.10 (d, 1H, CONHCH(CH3)2, JNH-CH = 7.7 Hz); 4.02 (s, 3H, OCH3-C2, JOCH3-OCH3 = 1.3 Hz); 3.99 (s, 3H, OCH3-C3, JOCH3-OCH3 = 1.3 Hz); 3.84–3.75 (m, 1H, CH); 1.12 (dd, 6H, CH(CH3)2, J(CH3)2-CH = 6.5 Hz, J(CH3)2-NH = 1.2 Hz) ppm. Anal. Calcd for C14H18N4O3 · 1/4 H2O: C: 57.05%; H: 6.28%; N: 19.02%; Found: C: 57.12%; H: 6.24%; N: 18.88%. M.p.: 250 °C.
1-(2,3-Dimethoxyquinoxalin-6-yl)-3-phenylurea (6i). Yield: 55%. IR (cm−1): 3275; 3068, 3030, 2997, 2939; 1639. 1H-NMR (DMSO-d6) δ: 8.94 (s, 1H, NH); 8.77 (s, 1H, NH); 8.02 (d, 1H, H5, J5-7 = 2.1 Hz); 7.66 (d, 1H, H8, J8-7 = 8.8 Hz); 7.54–7.43 (m, 3H, H7+H2'+H6'); 7.30 (dd, 2H, H3'+H5', J3'-2' = J5'-6' = 8.1Hz, J3'-4' = J5'-4' = 7.7 Hz); 6.99 (t, 1H, H4’, J4'-3' = J4'-5' = 7.4 Hz); 4.03 (s, 3H, OCH3-C2); 4.00 (s, 3H, OCH3-C3) ppm. Anal. Calcd for C17H16N4O3: C: 62.96%; H: 4.94%; N: 17.28%; Found: C: 63.30%; H: 5.23%; N: 17.56%. M.p.: 260 °C.
3.1.10. Synthesis of 3-Amino-7-methoxy-1,4-di-N-oxidequinoxaline-2-carbonitrile (7)
5-Methoxybenzofuroxan (2.50 g, 15.06 mmol), malononitrile (1.80 mL, 18.00 mmol) and N,N-DMF (5.00 mL) were placed in a flask. Next, the mixture was cooled to 0 °C and triethylamine (1.50 mL, 10.77 mmol) was added dropwise. The reaction was carried out during 6 h at room temperature. Diethyl ether and methanol were added and the mixture was filtered. The obtained compound was used without further purification. This compound has already been described by Monge et al. [24].
3.1.11. Synthesis of 7-Methoxy-1,4-di-N-oxidequinoxaline-2-carbonitrile (8)
Compound 7 (1.60 g, 6.88 mmol) and N,N-DMF (30.00 mL) were placed in a flask under a N2 atmosphere. Next, the mixture was heated to 65 °C and tert-butyl nitrite (2.80 mL, 23.57 mmol) was added dropwise. Effervescence could be observed. After stirring 10 min at 65 °C, another portion of tert-butyl nitrite (2.10 mL, 17.68 mmol) was added and the mixture was stirred for 1 h 30 min. The solvent was removed under reduced pressure. Finally, the solid was purified by traditional column chromatography using DCM as mobile phase to yield compound 8. Yield: 31%. IR (cm−1): 3090, 2975, 2238, 1367. 1H-NMR (DMSO-d6) δ: 9.19 (s, 1H, H3); 8.38 (d, 1H, H5, J5-6 = 9.4 Hz); 7.74 (d, 1H, H8, J8-6 = 2.7 Hz); 7.69 (dd, 1H, H6, J6-5 = 9.4 Hz, J6-8 = 2.8 Hz); 4.02 (s, 3H, OCH3) ppm. Anal. Calcd for C10H7N3O3 · 1/8 H2O: C: 54.73%; H: 3.31%; N: 19.16%; Found: C: 54.86%; H: 3.30%; N: 18.85%.
3.1.12. Synthesis of 7-Methoxyquinoxaline-2-carbonitrile (9)
Compound 8 (0.91 g, 0.97 mmol) and AcOEt/MeOH (1:1, 40.00 mL) were placed in a flask and heated to 65 °C. Next, a solution of sodium dithionite in water was prepared in situ (1.01 g of Na2O4S2in 20.00 mL of water) and added over the mixture. The reaction was carried out during 10 min at 65 °C. Next, the mixture was filtered with the aim of removing the sodium dithionite, and the solvent was removed under reduced pressure. Finally, the crude residue was purified by flash column chromatography to yield compound 9. Yield: 39%. IR (cm−1): 3090, 3039, 2975, 2226. 1H-NMR (DMSO-d6) δ: 9.22 (s, 1H, H3); 8.13 (d, 1H, H5, J5-6 = 9.3 Hz); 7.73 (dd, 1H, H6, J6-5 = 9.3 Hz, J6-8 = 2.8 Hz); 7.58 (d, 1H, H8, J8-6 = 2.8 Hz); 4.00 (s, 3H, OCH3) ppm. Anal. Calcd for C10H7N3O · 1/8 H2O: C: 64.09%; H: 3.87%; N: 22.43%; Found: C: 64.36%; H: 3.73%; N: 22.29%.
3.1.13. General Procedure for Synthesis of N-[(7-Methoxyquinoxalin-2-yl)methyl]alkylamides 10a–e
Compound 9 (1 eq.), THF (20 mL) and the corresponding anhydride (2 eq.) were placed in a hydrogenation flask. A spatula of Raney-Nickel was added and the mixture was hydrogenated during 7 h at 50 °C and 50 psi. Next, DCM was added and the mixture was decanted off with the aim of leaving off the catalyst. Next, the mixture was quenched with water, dried over Na2SO4 and filtered. The solvent was removed under reduced pressure and the orange oil obtained was purified through flash chromatography in order to obtain the amide derivatives 10a–e.
N-[(7-Methoxyquinoxalin-2-yl)methyl]acetamide (10a). Yield: 6%. IR (cm−1): 3289, 3068, 2965, 2920, 1648. 1H-NMR (DMSO-d6) δ: 8.71 (s, 1H, H3); 8.62 (t, 1H, NH, JNH-CH2 = 5.3 Hz); 7.97 (d, 1H,H5, J5-6 = 9.1 Hz); 7.62 (dd, 1H, H6, J6-5 = 9.1 Hz, J6-8 = 2.8 Hz); 7.40 (d, 1H,H8, J8-6 = 2.6 Hz); 4.56 (d, 2H, CH2NH, JCH2-NH = 5.8 Hz); 3.95 (s, 3H, OCH3); 1.93 (s, 3H, CH3) ppm. Anal. Calcd for C12H13N3O2 · 1/8 H2O: C: 61.74%; H: 5.68%; N: 18.01%; Found: C: 61.99%; H: 5.69%; N: 17.67%. M.p.: 123.9–126.9 °C.
N-[(7-Methoxyquinoxalin-2-yl)methyl]propionamide (10b). Yield: 24%. IR (cm−1): 3280, 3068, 2971, 2913, 1643. 1H-NMR (DMSO-d6) δ: 8.70 (s, 1H, H3); 8.54 (t, 1H, NH, JCH2-NH = 5.6 Hz); 7.97 (d, 1H, H5, J5-6 = 9.1 Hz); 7.46 (dd, 1H, H6, J6-5 = 9.1 Hz, J6-8 = 2.8 Hz); 7.40 (d, 1H, H8, J8-6 = 2.8 Hz); 4.56 (d, 2H, CH2NH, JCH2-NH = 5.8 Hz); 3.95 (s, 1H, OCH3); 2.21 (q, 2H, CH2CH3, JCH2-CH3 = 7.6 Hz); 1.04 (t, 3H, CH3, JCH3-CH2 = 7.6 Hz) ppm. Anal. Calcd for C13H15N3O2 · 1/4 H2O: C: 62.53%; H: 6.21%; N: 16.83%; Found: C: 62.92%; H: 6.23%; N: 16.85%. M.p.: 143–145 °C.
N-[(7-Methoxyquinoxalin-2-yl)methyl]butyramide (10c). Yield: 24%. IR (cm−1): 3282, 3081, 2965, 1643. 1H-NMR (DMSO-d6) δ: 8.69 (s, 1H, H3); 8.58 (s, 1H, NH); 7.96 (d, 1H, H5, J5-6 = 8.7 Hz); 7.45 (d, 1H, H6, J6-5 = 8.4 Hz); 7.38 (s, 1H, H8); 4.56 (d, 2H, CH2NH, JCH2-NH = 4.8 Hz); 3.94 (s, 3H, OCH3); 2.17 (t, 2H CH2CH2CH3, JCH2-CH2 = 6.5 Hz); 1.60–1.50 (m, 2H, CH2CH2CH3); 0.88 (t, 3H, CH3, JCH3-CH2 = 6.6 Hz) ppm. Anal. Calcd for C14H17N3O2 · 1/2 H2O: C: 62.69%; H: 6.72%; N: 15.67%; Found: C: 62.49%; H: 6.55%; N: 15.53%. M.p.: 128–129 °C.
N-[(7-Methoxyquinoxalin-2-yl)methyl]isobutyramide (10d). Yield: 62%. IR (cm−1): 3280, 3074, 2971, 2926, 1648. 1H-NMR (DMSO-d6) δ: 8.67 (s, 1H, H3); 8.53 (t, 1H, NH, JNH-CH2 = 5.3 Hz); 7.97 (d, 1H, H5, J5-6 = 9.2 Hz); 7.46 (dd, 1H, H6, J6-5 = 9.2 Hz, J6-8 = 2.8 Hz); 7.39 (d, 1H, H8, J8-6 = 2.7 Hz); 4.55 (d, 2H, CH2NH, JCH2-NH = 5.8 Hz); 3.95 (s, 3H, OCH3); 2.52–2.48 (m, 1H, CH); 1.07 (d, 6H, CH(CH3)2, J(CH3)2-CH = 6.9 Hz) ppm. Anal. Calcd for C14H17N3O2: C: 64.86%; H: 6.56%; N: 16.22%; Found: C: 64.76%; H: 6.67%; N: 16.27%. M.p.: 133–133.5 °C.
N-[(7-Methoxyquinoxalin-2-yl)methyl]benzamide (10e). Yield: 29%. IR (cm−1): 3299, 3061, 3010, 2965, 2920, 1640. 1H-NMR (DMSO-d6) δ: 9.28 (t, 1H, NH, JNH-CH2 = 5.4 Hz); 8.79 (s, 1H, H3); 7.98 (d, 1H, H5, J5-6 = 9.1 Hz); 7.94 (dd, 2H, H2'+H6', J2'-3' = J6'-5' = 7.1Hz, J2'-4' = J6'-4' = 1.2 Hz); 7.59-7.44 (m, 4H, H6+H3'+H4'+H5'); 7.41 (d, 1H,H8, J8-6 = 2.7 Hz); 4.79 (d, 2H, CH2NH, JCH2-NH = 5.7 Hz); 3.94 (s, 3H, OCH3) ppm. Anal. Calcd for C17H15N3O2 · 1/4 H2O: C: 68.57%; H: 5.21%; N: 14.12%; Found: C: 68.66%; H: 5.35%; N: 13.95%. M.p.: 142–143.9 °C.
3.2. Pharmacology
3.2.1. Reagents and Chemicals
2-[125I]Iodomelatonin (2200 Ci/mmol) was purchased from NEN (Boston, MA, USA). Other drugs and chemicals were purchased from Sigma-Aldrich (Saint Quentin, France).
3.2.2. Cell Culture
HEK (provided by A.D. Strosberg, Paris, France) and CHO cell lines stably expressing the human melatonin MT1 or MT2 receptors were grown in DMEM medium supplemented with 10% fetal calf serum, 2 mM glutamine, 100 IU/mL penicillin and 100 µg/mL streptomycin. Grown at confluence at 37 °C (95% O2/5% CO2), they were harvested in PBS containing EDTA 2 mM and centrifuged at 1,000 g for 5 min (4 °C). The resulting pellet was suspended in Tris 5 mM (pH 7.5), containing EDTA 2 mM and homogenized using a Kinematica polytron. The homogenate was then centrifuged (95,000 g, 30 min, 4 °C) and the resulting pellet suspended in 75 mM Tris (pH 7.5), 12.5 mM MgCl2 and 2 mM EDTA. Aliquots of membrane preparations were stored at −80 °C until use.
3.2.3. Binding Assays
2-[125I]Iodomelatonin binding assay conditions are essentially as previously described [50]. Briefly, binding is initiated by the addition of membrane preparations from stable transfected HEK or CHO cells diluted in binding buffer (50 mM Tris-HCl buffer, pH 7.4, containing 5 mM MgCl2) to 2-[125I]Iodomelatonin (25 or 200 pM for MT1 and MT2 receptors, respectively, expressed in HEK cells or 20 pM for MT1 and MT2 receptors expressed in CHO cells) and the tested drug. Non-specific binding was defined in the presence of 1 µM melatonin. After 120 min of incubation at 37 °C, the reaction was stopped by rapid filtration through GF/B filters presoaked in 0.5% (v/v) polyethylenimine. Filters were washed three times with 1 mL of ice-cold 50 mM Tris-HCl buffer, pH 7.4. Data from dose-response curves (seven concentrations in duplicate) were analyzed using the program PRISM (Graph Pad Software Inc., San Diego, CA, USA) to yield IC50 (inhibitory concentration 50). Results were expressed as Ki using the Cheng and Prusoff equation: Ki = IC50/[1 + (L/KD)], where [L] is the concentration of radioligand used in the assay and KD, the dissociation constant of the radioligand characterizing the membrane preparation.
The equilibrium binding constant (KD) for the 2-iodomelatonin at hMT1 and hMT2 receptors expressed in CHO and HEK cell membranes were 21 ± 3 pM and 107 ± 11 pM (n = 4), for CHO-hMT1 and CHO-hMT2 membranes, respectively and 10 ± 1 pM and 83 ± 19 pM (n = 4) for HEK-hMT1 and HEK-hMT2, respectively.
[35S]GTPγS binding assay was performed according to published methodology [50]. Briefly, membranes from transfected CHO cells expressing MT1 and MT2 receptor subtypes and compounds were diluted in binding buffer (20 mM HEPES, pH 7.4, 100 mM NaCl, 3 μM GDP, 3 mM MgCl2, and 20 μg/mL saponin). Incubation was started by the addition of 0.2 nM [35S]GTPγS to membranes (20 μg/mL) and drugs, and further followed for 1h at room temperature.
Usual levels of [35S]GTPγS binding (expressed in dpm) were for CHO-MT2 membranes: 2,000 for basal activity, 8,000 in the presence of melatonin 1 μM and 180 in the presence of GTPγS 10 μM which defined the non-specific binding. Data from the dose-response curves (seven concentrations in duplicate) were analyzed by using the program PRISM (Graph Pad Software Inc., San Diego, CA, USA) to yield EC50 (effective concentration 50%) and Emax (maximal effect) for agonists.
4. Conclusions
In conclusion, we were not able to assert whether the quinoxaline moiety can replace the melatonin indole nucleus as a useful bioisostere in the search of new melatoninergic ligands. Series III showed the best binding affinities to MT1/MT2 receptors due to the fact that the distance between the methoxy group and the nitrogen atom of the amide/urea group is comparable to the distance in the MLT molecule, a six-atom distance. Compound 10c showed the best binding affinity to both receptors. We found a large selectivity on the MT2 receptor subtype by switching the methoxy group to position 2 and establishing a six-atom distance between this group and N-acylamido chain (Series II), displaying a very selective MT2 ligand with a MT2/MT1 ratio of 250 and the higher binding affinity to MT2 (0.08 μM) of this study. This may provide a good preliminary starting point taking into consideration the new structural characteristics were set up as essential requirements for obtaining new quinoxalines as potent MT1 and MT2 receptor ligands.
Acknowledgments
Saioa Ancizu is indebted to the Navarre Government for a grant. We would also like to express our gratitude to ADA of the University of Navarra for the grant given to Nerea Castrillo. We want to thank Carmen Elizalde for her help in the identification assays of this study and to Laura Stokes for having edited the English language in this manuscript.
References and Notes
- Gállego, J.; Toledo, J.B.; Urrestarazu, E.; Iriarte, J. Clasificación de los trastornos del sueño. An. Sist. Sanit. Navar. 2007, 30, 19–36. [Google Scholar] [Green Version]
- Roth, T. Insomnia: Definition, prevalence, etiology, and consequences. Clin. Sleep Med. 2007, 3, S7–S10. [Google Scholar]
- Sarrais, F.; De Castro, T. El insomnio. Anales del Sistema Sanitario de Navarra 2007, 30, 121–134. [Google Scholar]
- Spadoni, G.; Bedini, A.; Rivara, S.; Mor, M. Melatonin receptor agonists: New options for insomnia and depression treatment. CNS Neurosci. Ther. 2011, 17, 733–741. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Srinivasan, V.; Spence, D.W.; Cardinali, D.P. Role of the melatonin system in the control of sleep: Therapeutic implications. CNS Drugs 2007, 21, 995–1018. [Google Scholar] [CrossRef]
- Zlotos, D.P. Recent advances in melatonin receptor ligands. Arch. Pharm. 2005, 338, 229–247. [Google Scholar] [CrossRef]
- Dubocovich, M.L. Melatonin receptors: Role on sleep and circadian rhythm regulation. Sleep Med. 2007, 8, S34–S42. [Google Scholar] [CrossRef]
- Alarma-Estrany, P.; Pintor, J. Melatonin receptors in the eye: Location, second messengers and role in ocular physiology. Pharmacol. Ther. 2007, 113, 507–522. [Google Scholar] [CrossRef]
- El Kazzouli, S.; Griffon du Bellay, A.; Berteina-Raboin, S.; Delagrange, P.; Caignard, D.-H.; Guillaumet, G. Design and synthesis of 2-phenylimidazo[1,2-a]pyridines as a novel class of melatonin receptor ligands. Eur. J. Med. Chem. 2011, 46, 4252–4257. [Google Scholar]
- Markl, C.; Attia, M.I.; Julius, J.; Sethi, S.; Witt-Enderby, P.A.; Zlotos, D.P. Synthesis and pharmacological evaluation of 1,2,3,4-tetrahydropyrazino[1,2-a]indole and 2-[(phenylmethylamino)methyl]-1H-indole analogues as novel melatoninergic ligands. Bioorg. Med. Chem. 2009, 17, 4583–4594. [Google Scholar] [CrossRef]
- Ettaoussi, M.; Sabaouni, A.; Rami, M.; Boutin, J.A.; Delagrange, P.; Renard, P.; Spedding, M.; Caignard, D.-H.; Berthelot, P.; Yous, S. Design, synthesis and pharmacological evaluation of new series of naphthalenic analogues as melatoninergic (MT1/MT2) and serotoninergic 5-HT2C dual ligands (I). Eur. J. Med. Chem. 2012, 49, 310–323. [Google Scholar] [CrossRef]
- Fisher, S.P.; Sugden, D. Sleep-promoting action of IIK7, a selective MT2 melatonin receptor agonist in the rat. Neurosci. Lett. 2009, 457, 93–96. [Google Scholar] [CrossRef]
- Leclerc, V.; Ettaoussi, M.; Rami, M.; Farce, A.; Boutin, J.A.; Delagrange, P.; Caignard, D.-H.; Renard, P.; Berthelot, P.; Yous, S. Design and synthesis of naphthalenic derivatives as new ligands at the melatonin binding site MT3. Eur. J. Med. Chem. 2011, 46, 1622–1629. [Google Scholar] [CrossRef]
- Srinivasan, V.; Brzezinski, A.; Pandi-Perumal, S.R.; Spence, D.W.; Cardinali, D.P.; Brown, G.M. Melatonin agonists in primary insomnia and depression-associated insomnia: Are they superior to sedative-hypnotics? Prog. Neuro-Psychoph. 2011, 35, 913–923. [Google Scholar] [CrossRef]
- Koike, T.; Hoashi, Y.; Takai, T.; Nakayama, M.; Yukuhiro, N.; Ishikawa, T.; Hirai, K.; Uchikawa, O. 1,6-Dihydro-2H-indeno 5,4-b furan Derivatives: Design, Synthesis, and Pharmacological Characterization of a Novel Class of Highly Potent MT2-Selective Agonists. J. Med. Chem. 2011, 54, 3436–3444. [Google Scholar] [CrossRef]
- Poissonnier-Durieux, S.; Ettaoussi, M.; Pérès, B.; Boutin, J.A.; Audinot, V.; Bennejean, C.; Delagrange, P.; Caignard, D.H.; Renard, P.; Berthelot, P.; et al. Synthesis of 3-phenylnaphthalenic derivatives as new selective MT2 melatoninergic ligands. Bioorg. Med. Chem. 2008, 16, 8339–8348. [Google Scholar] [CrossRef]
- Durieux, S.; Chanu, A.; Bochu, C.; Audinot, V.; Coirnailleau, S.; Boutin, J.A.; Delagrange, P.; Caignard, D.H.; Bennejean, C.; Renard, P.; et al. Design and synthesis of 3-phenyltetrahydronaphthalenic derivatives as new selective MT2 melatoninergic ligands. Part II. Bioorg. Med. Chem. 2009, 17, 2963–2974. [Google Scholar] [CrossRef]
- Rivara, S.; Lodola, A.; Mor, M.; Bedini, A.; Spadoni, G.; Lucini, V.; Pannacci, M.; Fraschini, F.; Scaglione, F.; Ochoa-Sanchez, R.; et al. N-(Substituted anilinoethyl)amides: Design, synthesis and pharmacological characterization of a new class of melatonin receptor ligands. J. Med. Chem. 2007, 50, 6618–6626. [Google Scholar]
- Hu, Y.; Ho, M.K.; Chan, K.H.; New, D.C.; Wong, Y.H. Synthesis of substituted N-[3-(3-methoxyphenyl)propyl]amides as highly potent MT2-selective melatonin ligands. Bioorg. Med. Chem. Lett. 2010, 20, 2582–2585. [Google Scholar] [CrossRef]
- Navajas, C.; Kokkola, T.; Poso, A.; Honka, N.; Gynther, J.; Laitinen, J.T. A rhodopsin-based model for melatonin recognition at its G protein-coupled receptor. Eur. J. Pharmacol. 1996, 304, 173–183. [Google Scholar] [CrossRef]
- Sugden, D.; Chong, N.W.S.; Lewis, D.F.W. Structural requirements at the melatonin receptor. Br. J. Pharmacol. 1995, 114, 618–623. [Google Scholar] [CrossRef]
- Spadoni, G.; Balsamini, C.; Diamantini, G.; Di Giacomo, B.; Tarzia, G.; Mor, M.; Plazzi, P.V.; Rivara, S.; Lucini, V.; Nonno, R.; et al. Conformationally restrained melatonin analogues: Synthesis, binding affinity for the melatonin receptor, evaluation of the biological activity, and molecular modeling study. J. Med. Chem. 1997, 40, 1990–2002. [Google Scholar]
- Sicsic, S.; Serraz, I.; Andrieux, J.; Bremont, B.; Mathe-Allainmat, M.; Poncet, A.; Shen, S.; Langlois, M. Three-dimensional quantitative structure-activity relationship of melatonin receptor ligands: a comparative molecular field analysis study. J. Med. Chem. 1997, 40, 739–748. [Google Scholar] [CrossRef]
- Grol, C.J.; Jansen, J.M. The high affinity melatonin binding site probed with conformationally restricted ligands. II. Homology modeling of the receptor. Bioorg. Med. Chem. 1996, 4, 1333–1339. [Google Scholar] [CrossRef]
- Mor, M.; Rivara, S.; Silva, C.; Bordi, F.; Plazzi, P.V.; Spadoni, G.; Diamantini, G.; Balsamini, C.; Tarzia, G.; Fraschini, F.; et al. Melatonin receptor ligands: Synthesis of new melatonin derivatives and comprehensive comparative molecular field analysis (CoMFA) study. J. Med. Chem. 1998, 41, 3831–3844. [Google Scholar]
- Jansen, J.M.; Copinga, S.; Gruppen, G.; Molinari, E.J.; Dubocovich, M.L.; Grol, C.J. The high affinity melatonin binding site probed with conformationally restricted ligand. I. POharmacophore and minireceptor models. Bioorg. Med. Chem. 1996, 4, 1321–1332. [Google Scholar] [CrossRef]
- Marot, C.; Chavatte, P.; Morin-Allory, L.; Viaud, M.C.; Guillaumet, G.; Renard, P.; Lesiur, D.; Michel, A. Pharmacophoric search and 3D-QSAR comparative molecular field analysis studies on agonists of melatonin sheep receptors. J. Med. Chem. 1998, 41, 4453–4465. [Google Scholar] [CrossRef]
- Spadoni, G.; Bedini, A.; Rivara, S.; Mor, M. Melatonin Receptor Agonists: New Options for Insomnia and Depression Treatment. CNS Neurosci. Ther. 2011, 17, 733–741. [Google Scholar] [CrossRef]
- Li, P.-K.; Chu, G.-H.; Gillen, M.L.; Witt-Enderby, P.A. Synthesis and receptor binding studies of quinolinic derivatives as melatonin receptor ligands. Bioorg. Med. Chem. Lett. 1997, 7, 2177–2180. [Google Scholar] [CrossRef]
- Yous, S.; Andrieux, J.; Howell, H.E.; Morgan, P.J.; Renard, P.; Pfeiffer, B.; Lesieur, D.; Guardiola-Lemaitre, B. Novel naphthalenic ligands with high affinity for the melatonin receptor. J. Med. Chem. 1992, 35, 1484–1486. [Google Scholar] [CrossRef]
- Waring, M.; Ben-Hadda, T.; Kotchevar, A.; Ramdani, A.; Touzani, R.; Elkadiri, S.; Hakkou, A.; Bouakka, M.; Ellis, T. 2,3-Bifunctionalized quinoxalines: Synthesis, DNA interactions and evaluation of anticancer, anti-tuberculosis and antifungal activity. Molecules 2002, 7, 641–656. [Google Scholar] [CrossRef]
- Burguete, A.; Pontiki, E.; Hadjipavlou-Litina, D.; Ancizu, S.; Villar, R.; Solano, B.; Moreno, E.; Torres, E.; Pérez, S.; Aldana, I.; et al. Synthesis and Biological Evaluation of New Quinoxaline Derivatives as Antioxidant and Anti-Inflammatory Agents. Chem. Biol. Drug Des. 2011, 77, 255–267. [Google Scholar] [CrossRef]
- Ancizu, S.; Moreno, E.; Solano, B.; Villar, R.; Burguete, A.; Torres, E.; Pérez-Silanes, S.; Aldana, I.; Monge, A. New 3-methylquinoxaline-2-carboxamide 1,4-di-N-oxide derivatives as anti-mycobacterium tuberculosis agents. Bioorg. Med. Chem. 2010, 18, 2713–2719. [Google Scholar] [CrossRef]
- Olayiwola, G.; Obafemi, C.A.; Taiwo, F.O. Synthesis and neuropharmacological activity of some quinoxalinone derivatives. Afr. J. Biotechnol. 2007, 6, 777–786. [Google Scholar]
- Mathé-Allainmat, M.; Andrieux, J.; Langlois, M. Recent developments in melatonin receptor ligands. Expert Opin. Ther. Pat. 1997, 7, 1447–1458. [Google Scholar] [CrossRef]
- Garratt, P.J.; Jones, R.; Rowe, S.J.; Sugden, D. Mapping the Melatonin Receptor. 1. The 5-Methoxy group is not an essential requirement for biological activity. Bioorg. Med. Chem. Lett. 1994, 4, 1555–1558. [Google Scholar] [CrossRef]
- Garratt, P.J.; Travard, S.; Vonhoff, S.; Tsotinis, A.; Sugden, D. Mapping the melatonin receptor. 4. Comparison of the binding affinities of a series of substituted phenylalkyl amides. J. Med. Chem. 1996, 39, 1797–1805. [Google Scholar] [CrossRef]
- Davies, D.J.; Faust, R.; Garratt, P.J.; Marivingt-Mounir, C.; Davidson, K.; Teh, M.T.; Sugden, D. Binding affinity and biological activity of oxygen and sulfur isosteres ata melatonin receptors as a function of their hydrogen bonding capability. Bioorg. Chem. 2004, 32, 1–12. [Google Scholar] [CrossRef]
- Langlois, M.; Bremont, B.; Shen, S.; Poncet, A.; Andrieux, J.; Sicsic, S.; Serraz, I.; Mathé-Allainmat, M.; Renard, P.; Delagrange, P. Design and synthesis of new naphthalenic derivatives as ligands for 2-[125I]iodomelatonin binding sites. J. Med. Chem. 1995, 38, 2050–2060. [Google Scholar] [CrossRef]
- Beavers, M. P.; Dudash, J.; Zhang, Y. 3-Oxo-3,4-dihydro-quinoxalin-2-yl-amino)-benzamide Derivatives and Related Compound as Glycogen Phosphorylase Inhibitors for the Treatment of Diabetes and Obesity. WO/2005/067932 2005. [Google Scholar]
- Ellis, G.P.; Romney-Alexander, T.M. Cyanation of aromatic halides. Chem. Rev. 1987, 87, 779–794. [Google Scholar] [CrossRef]
- Hermann, K.; Simchen, G. Synthese cyan-substituierter heterocyclen mit tetraethyl-ammoniumcyanid. Liebigs Ann. Chem. 1981, 1981, 333–341. [Google Scholar] [CrossRef]
- Tarzia, G.; Diamantini, G.; Di Giacomo, B.; Spadoni, G.; Esposti, D.; Nonno, R.; Lucini, V.; Pannacci, M.; Fraschini, F.; Stankov, B.M. 1-(2-Alkanamidoethyl)-6-methoxyindole derivatives: A new class of potent iIndole melatonin analogues. J. Med. Chem. 1997, 40, 2003–2010. [Google Scholar] [CrossRef]
- Mor, M.; Spadoni, G.; Di Giacomo, B.; Diamantini, G.; Bedini, A.; Tarzia, G.; Plazzi, P.V.; Rivara, S.; Nonno, R.; Lucini, V.; et al. Synthesis, pharmacological characterization and QSAR studies on 2-substituted indole melatonin receptor ligands. Bioorg. Med. Chem. 2001, 9, 1045–1057. [Google Scholar] [CrossRef]
- Galiano, S.; Ceras, J.; Cirauqui, N.; Pérez, S.; Juanenea, L.; Rivera, G.; Aldana, I.; Monge, A. Novel series of substituted biphenylmethyl urea derivatives as MCH-R1 antagonists for the treatment of obesity. Bioorg. Med. Chem. 2007, 15, 3896–3911. [Google Scholar] [CrossRef]
- Koyuncu, S.; Zafer, C.; Koyuncu, F.B.; Aydin, B.; Can, M.; Sefer, E.; Ozdemir, E.; Icli, S. A New Donor-acceptor Double-cable Carbazole Polymer with Perylene Bisimide Pendant Group: Synthesis, Electrochemical, and Photovoltaic Properties. J. Polym. Sci. A Polym. Chem. 2009, 47, 6280–6291. [Google Scholar] [CrossRef]
- Wallez, V.; Durieux-Poissonnier, S.; Chavatte, P.; Boutin, J.A.; Audinot, V.; Nicolas, J.-P.; Bennejean, C.; Delagrange, P.; Renard, P.; Lesieur, D. Synthesis and structure-affinity-activity relationships of novel benzofuran derivatives as MT2 melatonin receptor selective ligands. J. Med. Chem. 2002, 45, 2788–2800. [Google Scholar] [CrossRef]
- Monge, A.; Martinez-Crespo, F.J.; Lopez de Cerain, A.; Palop, J.A.; Narro, S.; Senador, V.; Marin, A.; Sainz, Y.; Gonzalez, M. Hypoxia-selective agents derived from 2-quinoxalinecarbonitrile 1,4-di-N-oxides. 2. J. Med. Chem. 1995, 38, 4488–4494. [Google Scholar]
- Solano, B.; Junnotula, V.; Marín, A.; Villar, R.; Burguete, A.; Vicente, E.; Pérez-Silanes, S.; Aldana, I.; Monge, A.; Dutta, S.; et al. Synthesis and biological evaluation of new 2-arylcarbonyl-3-trifluoromethylquinoxaline 1,4-di-N-oxide derivatives and their reduced analogues. J. Med. Chem. 2007, 50, 5485–5492. [Google Scholar] [CrossRef]
- Audinot, V.; Mailliet, F.; Lahaye-Brasseur, C.; Bonnaud, A.; Le Gall, A.; Amossé, C.; Dromaint, S.; Rodriguez, M.; Nagel, N.; Galizzi, J.P.; et al. New selective ligands of human cloned melatonin MT1 and MT2 receptors. Naunyn-Schmiedebergs Arch. Pharmacol. 2003, 367, 553–561. [Google Scholar] [CrossRef]
- Farce, A.; Chugunov, A.O.; Logé, C.; Sabaouni, A.; Yous, S.; Dilly, S.; Renault, N.; Vergoten, G.; Efremov, R.G.; Lesieur, D.; et al. Homology modeling of MT1 and MT2 receptors. Eur. J. Med. Chem. 2008, 43, 1926–1944. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
