Antioxidant Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction Efficiency

| Extracts | Ethanol | SFE | SFE | SFE | |
|---|---|---|---|---|---|
| (+10 min) | (−10 min) | (wash) | |||
| Extraction yield | buds | 6.85% | 3.34% | 0.13% | 0.46% |
| barks | 12.73% | 0.90% | 0.16% | 0.22% | |
| leaves | 23.02% | 0.68% | 0.36% | 1.74% | |
| TPC | buds | 6.313 ± 0.15 | 0.151 ± 0.01 | 0.812 ± 0.01 | 1.712 ± 0.01 |
| (g GAE/ | barks | 9.534 ± 0.26 | 0.398 ± 0.01 | 2.018 ± 0.04 | 1.763 ± 0.01 |
| 100 g DW) | leaves | 8.854 ± 0.35 | 0.986 ± 0.02 | 1.248 ± 0.02 | 1.338 ± 0.03 |
| TFC | buds | 2.697 ± 0.17 | 0.395 ± 0.01 | 1.322 ± 0.03 | 2.504 ± 0.05 |
| (g Quercetin/ | barks | 2.030 ± 0.10 | 0.031 ± 0.01 | 1.266 ± 0.10 | 1.373 ± 0.09 |
| 100 g DW) | leaves | 3.348 ± 0.29 | 1.288 ± 0.02 | 1.086 ± 0.05 | 1.916 ± 0.05 |
2.2. Total Phenolic Content
2.3. Total Flavonoid Content
2.4. Antioxidant Activities
2.4.1. DPPH Free Radical Scavenging Ability
| Extracts | Ethanol | SFE | SFE | SFE | |
|---|---|---|---|---|---|
| (+10 min) | (−10 min) | (wash) | |||
| DPPH | buds | 0.073 ± 0.000 | 3.881 ± 0.366 | 3.144 ± 0.228 | 1.457 ± 0.082 |
| IC50 (mg/mL) | barks | 0.072 ± 0.003 | 10.090 ± 0.847 | 1.772 ± 0.221 | 2.446 ± 0.430 |
| leaves | 0.208 ± 0.022 | 0.661 ± 0.039 | 0.562 ± 0.039 | 0.593 ± 0.038 | |
| TEAC | buds | 133.04 ± 11.32 | 6.79 ± 1.8 | 29.10 ± 0.76 | 58.34 ± 0.64 |
| (mmole trolox/g) | barks | 335.78 ± 77.15 | 10.06 ± 4.3 | 51.89 ± 13.57 | 55.67 ± 14.85 |
| leaves | 297.34 ± 65.23 | 39.74 ± 9.13 | 43.98 ± 7.06 | 45.82 ± 8.80 | |
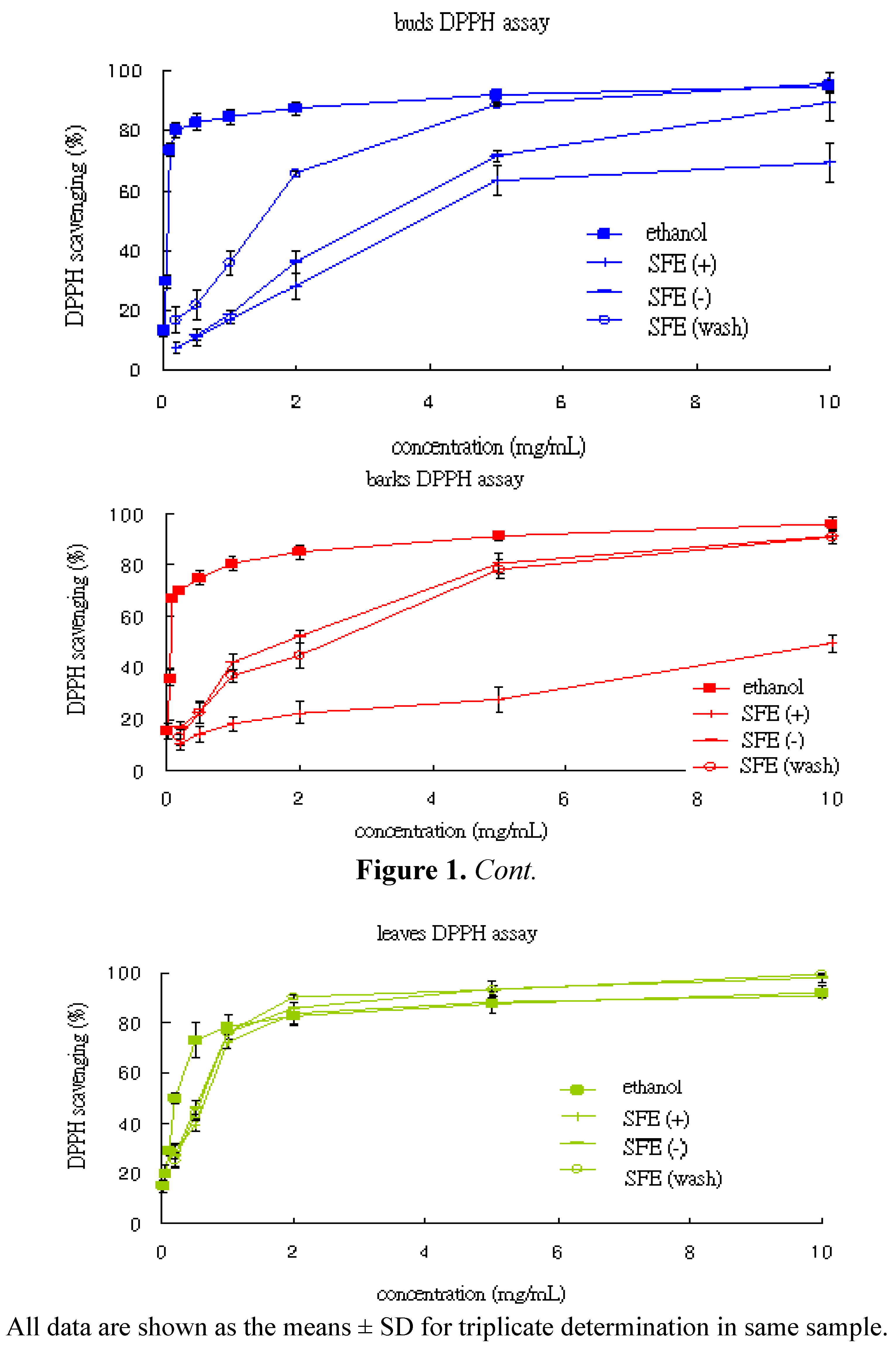
2.4.2. Trolox Equivalent Antioxidant Capacity (TEAC)
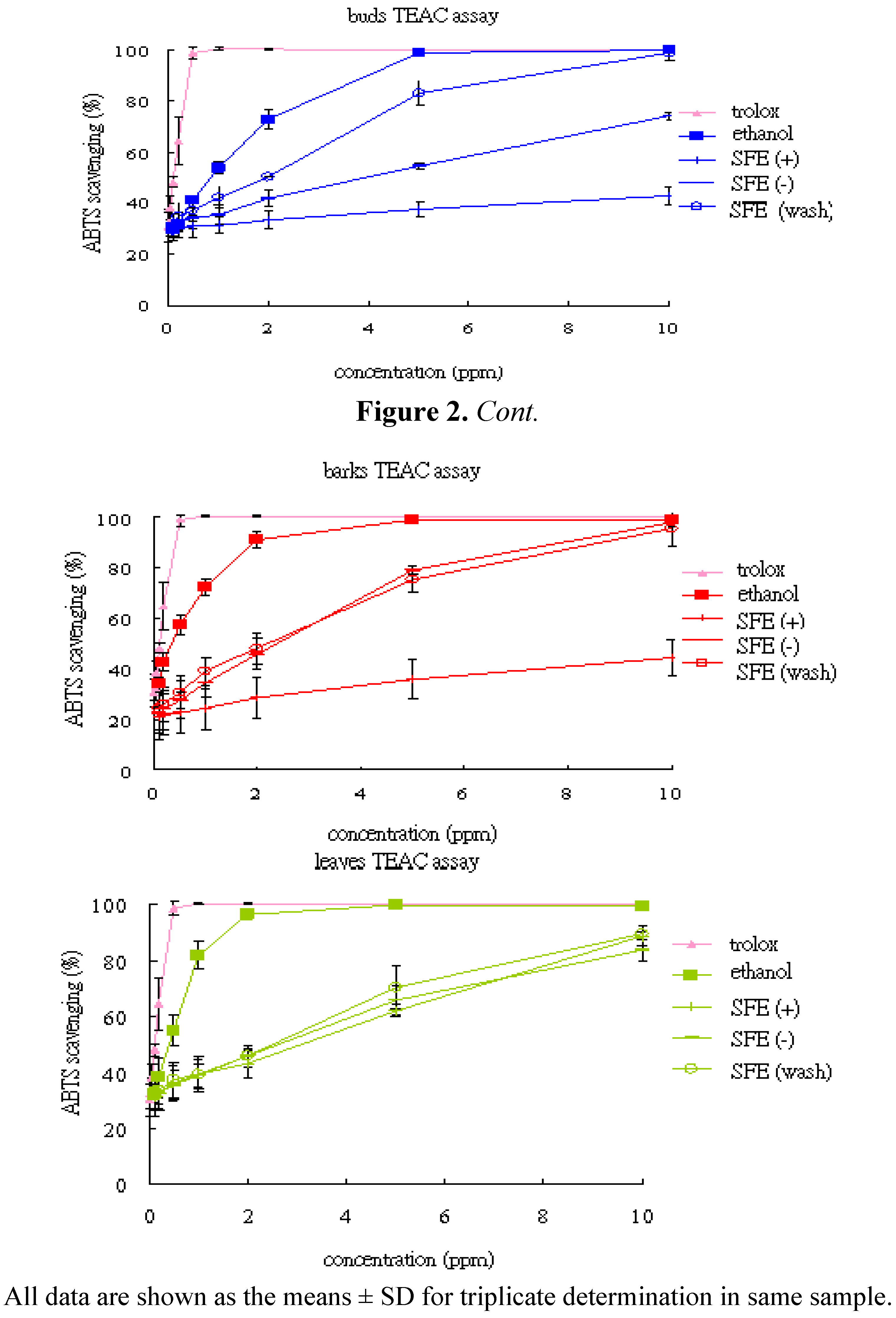
3. Experimental
3.1. Collection of Plant Materials
3.2. Preparation of the Extracts
3.3. Antioxidant Activity Analysis by the DPPH Free Radical Scavenging Method
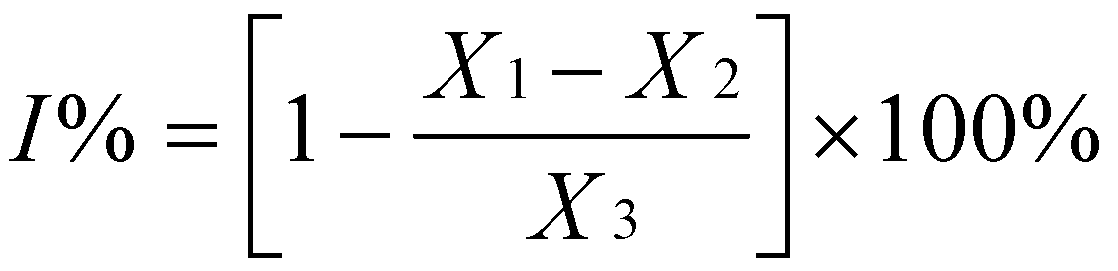
3.4. Trolox Equivalent Antioxidant Capacity (TEAC)
3.5. Antioxidant Activity Analysis by the Total Phenolic Content Determination
3.6. Total Flavonoid Content Determination
4. Conclusions
Acknowledgements
References and Notes
- Yang, D.; Wang, Q.; Ke, L.; Jiang, J.; Ying, T. Antioxidant activities of various extracts of lotus (Nelumbo nuficera Gaertn) rhizome. Asia Pac. J. Clin. Nutr. 2007, 16 (Suppl. 1), 158–163. [Google Scholar]
- Liang, M.T.; Yang, C.H.; Li, S.T.; Yang, C.S.; Chang, H.W.; Liu, C.S.; Cham, T.M.; Chuang, L.Y. Antibacterial and antioxidant properties of Ramulus Cinnamomi using supercritical CO2 extraction. Eur. Food Res. Technol. 2008, 227, 1387–1396. [Google Scholar] [CrossRef]
- Saleem, M.; Nazir, M.; Ali, M.S.; Hussain, H.; Lee, Y.S.; Riaz, N.; Jabbar, A. Antimicrobial natural products: An update on future antibiotic drug candidates. Nat. Prod. Rep. 2009, 27, 238–254. [Google Scholar]
- Liu, C.S.; Yang, C.H.; Chang, C.W.; Chen, C.H.; Chuang, L.Y. Antibacterial properties of Chinese herbal medicines against nosocomial antibiotic rsistan strains of Pseudomonas aerginosa in Taiwan. Am. J. Chin. Med. 2007, 35, 1047–1060. [Google Scholar] [CrossRef]
- Kim, S.H.; Hyun, S.H.; Choung, S.Y. Anti-diabetic effect of cinnamon extract on blood glucose in db/db mice. J. Ethnopharmacol. 2006, 104, 119–123. [Google Scholar] [CrossRef]
- Singh, G. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- Tung, Y.T.; Chua, M.T.; Wang, S.Y.; Chang, S.T. Anti-inflammation activities of essential oil and its constituents from indigenous cinnamon (Cinnamomum osmophloeum) twigs. Bioresour. Technol. 2008, 99, 3908–3913. [Google Scholar] [CrossRef]
- Chang, S.T.; Chen, P.F.; Chang, S.C. Antibacterial activity of leaf essential oils and their constituents from Cinnamomum osmophloeum . J. Ethnopharmacol. 2001, 77, 123–127. [Google Scholar] [CrossRef]
- Tsai, T.H.; Tai, T.H.; Chien, Y.C.; Lee, C.W.; Tsai, O.J. In vitro antimicrobial activities against Cariogenic streptococci and their antioxidant capacities: A comparative study of green tea versus different herbs. Food Chem. 2008, 110, 859–864. [Google Scholar] [CrossRef]
- Prasad, K.N.; Yang, B.; Dong, X.; Jiang, G.; Zhang, H.; Xie, H.; Jiang, Y. Flavonoid contents and antioxidant activities from Cinnamomum species. Food Sci. Emer. Technol. 2009, 10, 627–632. [Google Scholar] [CrossRef]
- Lin, C.C.; Wu, S.J.; Chang, C.H.; Ng, L.K. Antioxidant activity of Cinnamomum cassia . Phytother. Res. 2003, 17, 726–730. [Google Scholar] [CrossRef]
- Lee, H.S.; Kim, B.S.; Kim, M.K. Suppression effect of Cinnamomum barks-derived component on nitric oxide synthase. J. Agric. Food Chem. 2002, 50, 7700–7703. [Google Scholar] [CrossRef]
- Bensebia, O.; Barth, D.; Dahmani, A. Supercritical carbon dioxide extraction of rosemary comparison with steam distillation and hydrodistillation. ISASF Home page. Available online: http://www.isasf.net/fileadmin/files/Docs/Colmar/Paper/N14.pdf (accessed on 31 December 2005).
- Yang, C.H.; Yang, C.S.; Hwang, M.L.; Chang, C.C.; Li, R.X.; Chuang, L.Y. Antimicrobial activity of various parts of Cinnamomum cassia extracted with different extraction methods. J. Food Biochem. 2012. [Google Scholar] [CrossRef]
- Al-Numair, K.S.; Ahmad, D.; Ahmed, S.B.; Al-Assaf, A.H. Nutritive value, level of polyphenols and anti-nutritional factore in Sri Lankan cinnamon (Cinnamomum zeyalnicum) and Chinese Cinnamon (Cinnamomum cassia) . Res. Bult. King Saud. Univ. 2007, 154, 5–21. [Google Scholar]
- Duh, P.D.; Yen, G.C.; Yen, W.J.; Wang, B.S.; Chang, L.E. Effects of pu-erh tea on oxidative damage and nitric oxide scavenging. J. Agric. Food. Chem. 2004, 52, 8169–8176. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.; Davies, M.J.; Gopinathan, V.; Milner, A. A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar]
- Li, H.B.; Wong, C.C.; Cheng, K.W.; Chen, F. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT-Food Sci. Technol. 2008, 41, 385–390. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the author, Li-Yeh Chuang.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, C.-H.; Li, R.-X.; Chuang, L.-Y. Antioxidant Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods. Molecules 2012, 17, 7294-7304. https://doi.org/10.3390/molecules17067294
Yang C-H, Li R-X, Chuang L-Y. Antioxidant Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods. Molecules. 2012; 17(6):7294-7304. https://doi.org/10.3390/molecules17067294
Chicago/Turabian StyleYang, Cheng-Hong, Rong-Xian Li, and Li-Yeh Chuang. 2012. "Antioxidant Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods" Molecules 17, no. 6: 7294-7304. https://doi.org/10.3390/molecules17067294
APA StyleYang, C.-H., Li, R.-X., & Chuang, L.-Y. (2012). Antioxidant Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods. Molecules, 17(6), 7294-7304. https://doi.org/10.3390/molecules17067294




