Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers
Abstract
:1. Introduction
2.Results
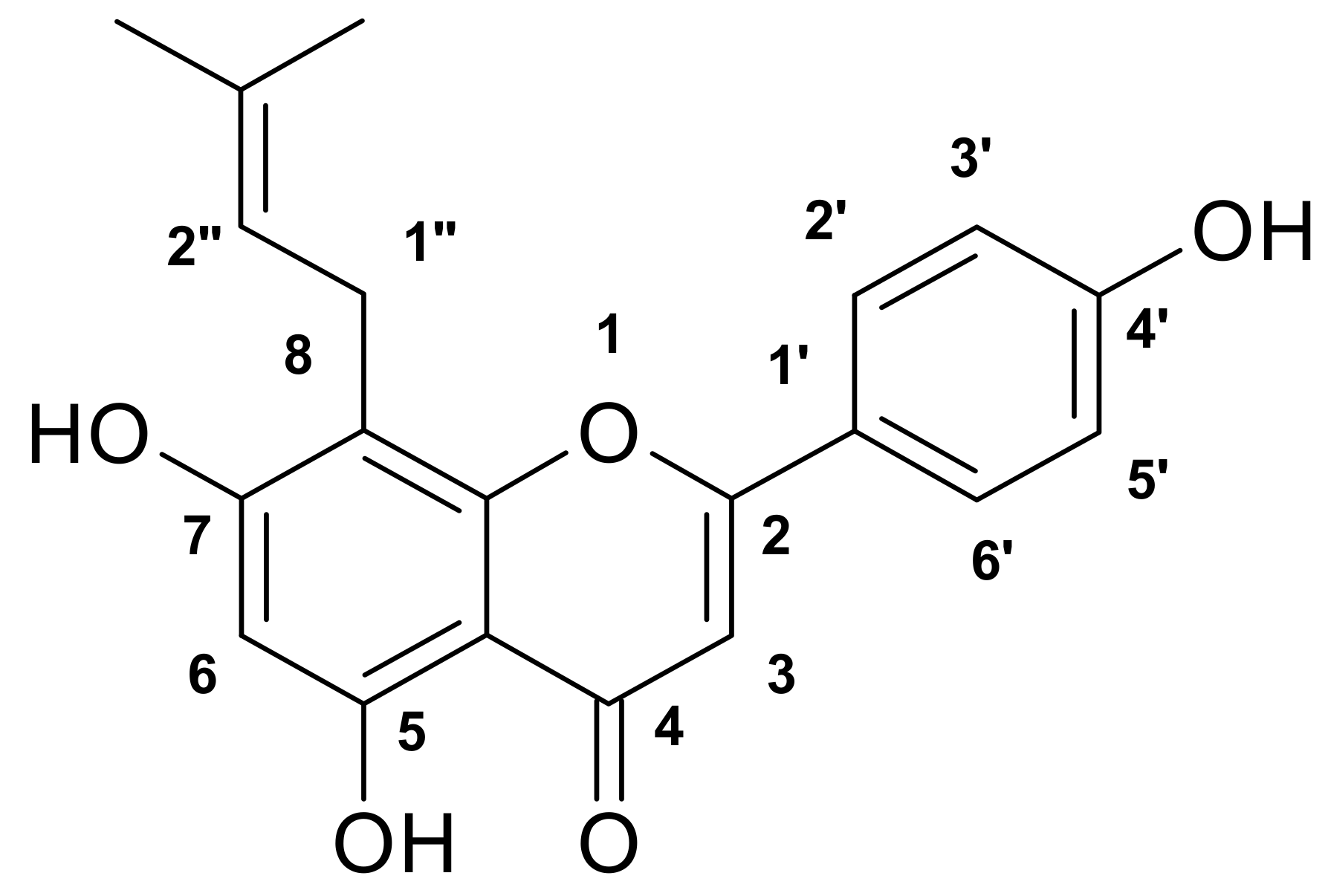
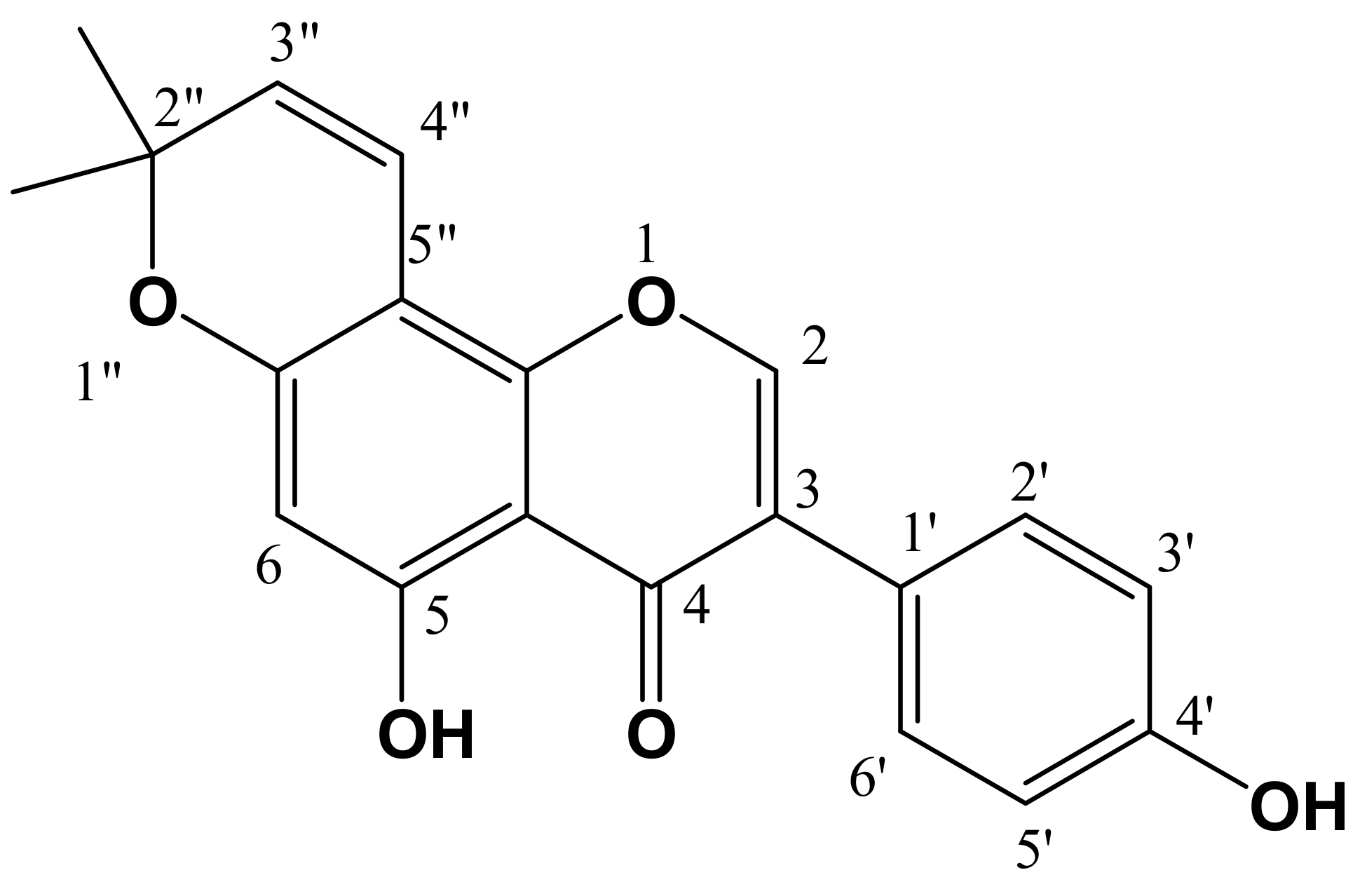
| Microorganisms | AmphotericinB a | GM b | Compound (1) | Compound (2) | ||
|---|---|---|---|---|---|---|
| 25 c | 50 d | 25 | 50 | |||
| Bacteria | ||||||
| Staphylococcus aureus ATCC 25923 | nd | 18.3 ± 0.61 | 11 ± 1.10 | 13 ± 1.23 | 10 ± 2.03 | 12 ± 1.27 |
| Escherichia coli ATCC 25922 | nd | 23 ± 0.02 | 11 ± 1.51 | 22 ± 1.51 | 17 ± 2.12 | 19 ± 0.23 |
| Enterococcus faecalis ATTC 29212 | nd | nd | 16 ± 2.33 | 20 ± 0.98 | 13 ± 1.51 | 17 ± 0.15 |
| Pseudomonas aeruginosa ATCC | nd | 18 ± 0.11 | 14 ± 1.01 | 20 ± 1.8 | 12 ± 2.17 | 16 ± 0.52 |
| Yeasts | ||||||
| Candida glabrata ATCC 90030 | 20 ± 1.04 | nd | 12 ± 0.03 | 16 ± 1.53 | 20 ± 0.52 | 25 ± 1.11 |
| Candida albicans ATCC 90028 | 19 ± 0.25 | nd | 14 ± 0.61 | 19 ± 0.18 | 21 ± 1.25 | 25 ± 1.24 |
| Candida parapsilosis ATCC 22019 | 19 ± 0.51 | nd | 14 ± 1.72 | 19 ± 0.39 | 20 ± 1.87 | 25 ± 0.91 |
| Candida kreusei ATCC 6258 | 19 ± 0.12 | nd | 15 ± 1.92 | 19 ± 0.54 | 21 ± 1.79 | 25 ± 1.63 |
| Microorganism | MIC (µg/mL) | ||||
|---|---|---|---|---|---|
| CP1 a | CP2 b | GM c | OFX d | AP e | |
| Bacteria | |||||
| Staphilococcus aureus ATCC 25923 | 62.5 | 62.5 | nd | 0.25 | nd |
| Escherichia coli ATCC 25922 | 7.81 | 7.81 | nd | 0.12 | nd |
| Enterococcus faecalis ATCC 29212 | 100 | 100 | nd | 1 | nd |
| Pseudomonas aeruginosa ATCC 27950 | 15.62 | 15.62 | 0.5 | 1 | nd |
| Yeast | |||||
| Candida glabrata ATCC 90030 | 15.62 | 7.81 | nd | nd | 0.5 |
| Candida albicans ATCC 90028 | 15.62 | 7.81 | nd | nd | 0.5 |
| Candida parapsilosis ATCC 22019 | 15.62 | 7.81 | nd | nd | 0.5 |
| Candada kreusei ATCC 6258 | 15.62 | 7.81 | nd | nd | 0.5 |
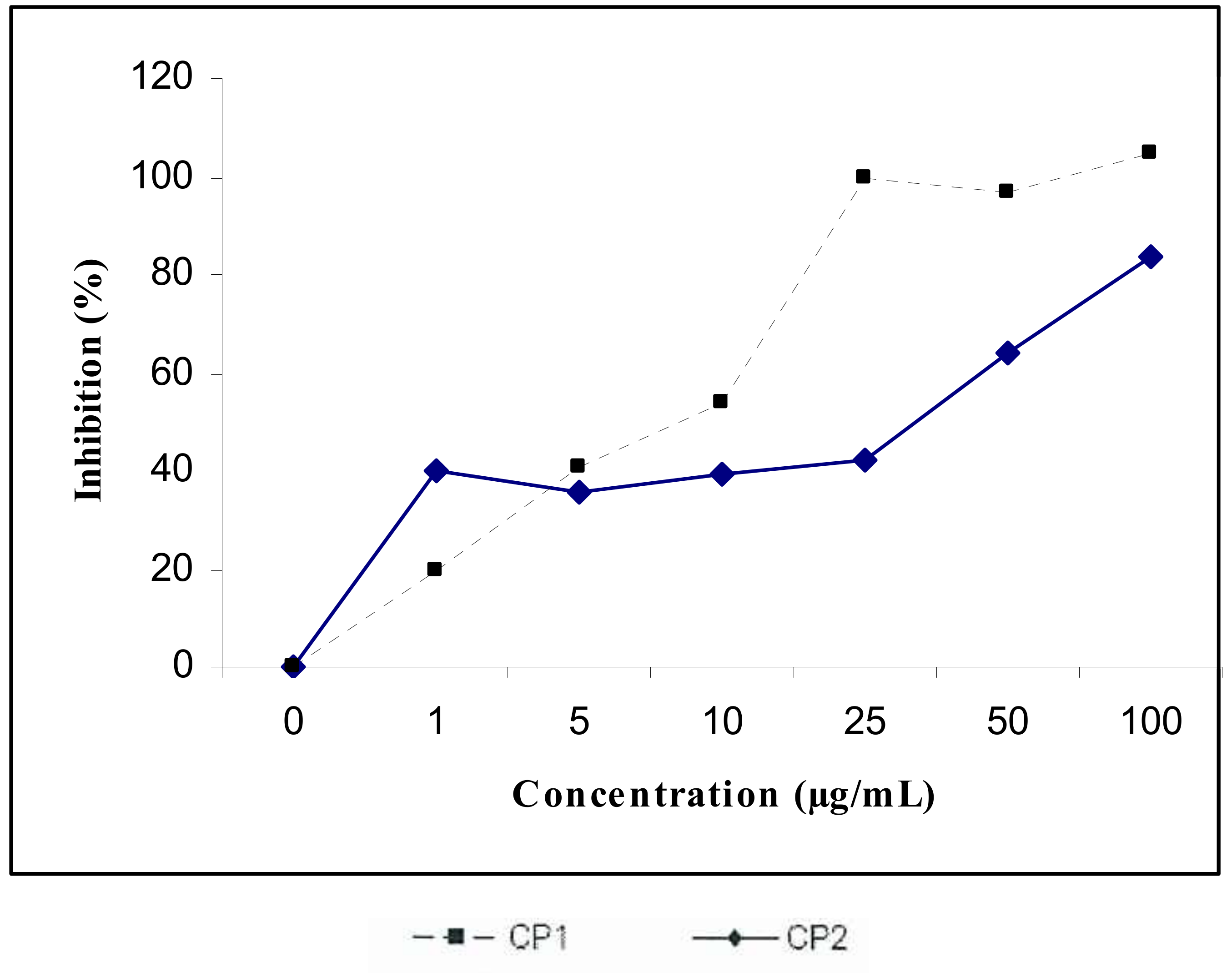
3. Discussion
4. Experimental
4.1. Plant Material
4.2. Extraction and Isolation
4.3. Spectral Analyses
4.4. Structure Identification
4.5. Microrganisms
4.6. Determination of Antibacterial and Antifungal Activities
4.6.1. Disc Diffusion Method
4.6.2. Micro-Well Dilution Assay
4.7. Cytotoxic Activity
5. Conclusions
References and Notes
- Yagi, A.; Fukunaga, M.; Okuzako, N.; Mifuchi, I.; Kawamoto, F. Antifungal substances from Sophora flavescens. Shoyakugaku Zasshi 1989, 43, 343–347. [Google Scholar]
- Yamaki, M.; Kashihara, M.; Takagi, S. Activity of Ku Shen compounds against Staphylococcus aureus and Streptococcus mutans. Phytother. Res. 1990, 4, 235–236. [Google Scholar]
- Bang, K.H.; Kim, Y.K.; Min, B.S.; Na, M.K.; Rhee, Y.H.; Lee, J.P. Antifungal activity of magnolol and honokiol. Arch. Pharm. Res. 2000, 23, 46–49. [Google Scholar]
- Hayes, A.J.; Markovic, B. Toxicity of Australian essential oil Backhousia citrodora (lemon myrtle). Antimicrobila activity and in vitro cytotoxicity. Food Chem. Toxicol. 2002, 40, 535–543. [Google Scholar]
- Pezzuto, J.M. Plant-derived anticancer agents. Biochem. Pharmacol. 1997, 2, 21–133. [Google Scholar]
- Nomura, T. Biological activities of phenolic constituents of mulberry tree and related plants. In Progress in the Chemistry of Organic Natural Products; Hertz, W., Grisebach, H., Kirby, G.W., Tamm, C.H., Eds.; Springer Verlag, Wien: New York, NY, USA, 1988; Volume 3, p. 189. [Google Scholar]
- Kang, S.S.; Kim, J.S.; Son, K.H.; Chang, H.W.; Kim, H.P. A new prenylated flavanone from the roots of Sophora flavescens. Fitoterapia 2000, 71, 511–515. [Google Scholar]
- Chi, Y.S; Jong, H.G.; Son, K.H.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of naturally occurring prenylated flavonoids on enzymes metabolizing arachidonic acid: Cyclooxygenases and lipoxygenases. Biochem. Pharmacol. 2001, 62, 1185–1191. [Google Scholar]
- Son, K.H.; Kwon, S.J.; Chang, H.W.; Kim, H.P.; Kang, S.S. Papyriflavonol A. A new prenylated flavonol, from the root barks of Broussnetia papyrifera. Fitoterapia 2001, 72, 456–458. [Google Scholar]
- Tringali, C. Bioactive Compounds from Natural Sources (Isolation, Characterization and Biological Properties); Taylor & Francis: London, UK, 2001. [Google Scholar]
- Kim, D.W.; Chi, Y.S.; Son, K.H.; Chang, H.W.; Kim, J.S.; Kang, S.S.; Kim, H.P. Effect of sophoraflavanone G, a prenylated flavonoids from Sophora flavescens, on cyclooxygenase-2 and in vivo inflammatory response . Arch. Pharm. Res. 2002, 20, 125–135. [Google Scholar]
- Mittle, R.; Merquiol, E.; Hallak-Herr, E.; Rachmilevitch, S.; Kaplan, A.; Cohen, M. Living under a dormant canopy: A molecular acclimation mechanism of the desert plant Retama raetam. Plant J. 2001, 25, 407–416. [Google Scholar]
- El Bahri, L.; Djegham, M.; Bellil, H. Retama raetam W. A poisonous plant of North Africa. Vet. Hum. Toxicol. 2000, 41, 33–35. [Google Scholar]
- Halim, A.O.B.; Abdel Fattah, H.; Halim, A.F.; Murakoshi, I. Comparative chemical and biological studies of the alkaloidal content of Lygos species and varieties growing in Egypt. Acta Pharm. Hung. 1997, 67, 241–247. [Google Scholar]
- Nawwar, M.A.M.; El Sherbeiny, A.E.A.; El Ansari, M.A. Plant constitutents of Tamarix aphylla flowers (Tamaricaceae). Cell. Mol. Life Sci. 1975, 31, 1118. [Google Scholar] [CrossRef]
- Kassem, M.; Mosharrafa, S.; Saleh, N. Two new flavonoids from Retama raetam. Fitoterapia 2000, 71, 649–654. [Google Scholar]
- Han, A.R.; Kang, Y.J.; Windono, T.; Lee, S.K.; Seo, E.K. Prenylated flavonoids from the heartwood of Artocarpus communis with inhibitory activity of lipopolysaccharide-induced nitric oxide production. J. Nat. Prod. 2006, 69, 719–721. [Google Scholar]
- Chibber, S.S.; Sharma, R.P. Derrone, a new pyranoisoflavone from Derris robusta seeds. Phytochemistry 1980, 19, 1857–1858. [Google Scholar]
- Polydoro, M.; de Souza, K.C.B.; Andrades, M.E.; da silva, E.G.; Bonatto, F.; Heydrich, J.; Dal-Pizzol, F.; Schapoval, E.E.S.; Bassani, V.L.; Moreira, J.C.F. Antioxidant, a pro-oxidant and cytotoxic effect of Achrrocline satureioides extracts. Life Sci. 2004, 74, 2815–2826. [Google Scholar]
- Jarvis, W.R.; Martone, W.J. Predominant pathogens in hospital infections. J. Antimicrob.Chemother. 1992, 2, 19–24. [Google Scholar]
- Carmeli, Y.; Troillet, N.; Eliopoulos, G.M.; Samore, M.H. Emergence of Antibiotic-resistant Pseudomonas aeruginosa: Comparison of risks associated with different antipseudomonal agents. Antimicrob. Agents Chemother. 1999, 43, 1379–1382. [Google Scholar]
- Pereira, J.A.; Pereira, A.P.G.; Ferreira, I.C.F.R.; Valentão, P.; Andrade, P.B.; Seabra, R.; Estevinho, L.; Bento, A. Table olives from Portugal: Compounds. antioxidant potential, and antimicrobial activity. J. Agric. Food Chem. 2006, 54, 8425–8431. [Google Scholar]
- Cowan, M.M. Plant product as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar]
- Tsuchia, H.; Sato, M.; Miyazaki, S.; Fujiwara, S.; Tanaka, T.; Lumina, M. Comparative study on the antibacterial activity of phytochemical flavones against methicillin-resistant Staphylococcus aureus. J. Ethnopharmacol. 1999, 50, 7–34. [Google Scholar]
- Sahin, F.; Gulluce, M.; Daferera, D.; Soken, A.; Sokmen, M.; Polissiou, M. Biological activities of the essential oils and methanol extract of Origanum vulgare ssp. vulgare in the Eastern Anatolia region of Turkey . Food Control 2004, 15, 549–557. [Google Scholar]
- Samples Availability: Samples of the compounds 1 and 2 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Edziri, H.; Mastouri, M.; Mahjoub, M.A.; Mighri, Z.; Mahjoub, A.; Verschaeve, L. Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers. Molecules 2012, 17, 7284-7293. https://doi.org/10.3390/molecules17067284
Edziri H, Mastouri M, Mahjoub MA, Mighri Z, Mahjoub A, Verschaeve L. Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers. Molecules. 2012; 17(6):7284-7293. https://doi.org/10.3390/molecules17067284
Chicago/Turabian StyleEdziri, Hayet, Maha Mastouri, Mohamed Ali Mahjoub, Zine Mighri, Aouni Mahjoub, and Luc Verschaeve. 2012. "Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers" Molecules 17, no. 6: 7284-7293. https://doi.org/10.3390/molecules17067284
APA StyleEdziri, H., Mastouri, M., Mahjoub, M. A., Mighri, Z., Mahjoub, A., & Verschaeve, L. (2012). Antibacterial, Antifungal and Cytotoxic Activities of Two Flavonoids from Retama raetam Flowers. Molecules, 17(6), 7284-7293. https://doi.org/10.3390/molecules17067284





