Preparation of Quinolinium Salts Differing in the Length of the Alkyl Side Chain
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and HPLC Analysis
2.2. In-Vitro Antimicrobial Activity Assessment
3. Experimental
3.1. General
3.1.1. Synthesis
3.1.2. HPLC Analysis
3.2. In-Vitro Antimicrobial Testing
3.2.1. Antifungal Activity
3.2.2. Antibacterial Activity
4. Conclusions
Acknowledgements
References and Notes
- Kuca, K.; Dohnal, V.; Bielavska, M.; Cabal, J. Determination of benzalkonium bromide homologues in disinfection products using high-performance liquid chromatography. Anal. Lett. 2005, 38, 673–682. [Google Scholar] [CrossRef]
- Semmler, A.; Kohler, H.H. Surface Properties of Alkylpyridinium Chlorides and the Applicability of the Pendant Drop Technique. J. Coll. Interf. Sci. 1999, 218, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Akbas, H.; Kartal, C.I. Reactive Orange 16-dodecylpyridinium chloride interactions in electrolytic solutions. Spectrochim. Acta A 2006, 65, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Faraj, J.A.; Dorati, R.; Schoubben, A.; Worthen, D.; Selmin, F.; Capan, Y.; Leung, K.; Deluca, P.P. Development of a peptide-containing chewing gum as a sustained release antiplaque antimicrobial delivery system. AAPS Pharm.Sci. Tech. 2007, 8, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Tanzer, J.M.; Slee, A.M.; Kamay, B.; Scheer, E.R. In vitro evaluation of seven cationic detergents as antiplaque agents. Antimicrob. Agents Chemother. 1979, 15, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Kuca, K.; Cabal, J.; Patocka, J.; Dohnal, V. Quaternary Heteroarenium Salts as the Competitive Inhibitors of the Brain Acetylcholinesterase. Lett. Drug Des. Discov. 2004, 4, 97–100. [Google Scholar] [CrossRef]
- Grinevich, V.P.; Crooks, P.A.; Sumithran, S.P.; Haubner, A.J.; Ayers, J.T.; Dwoskin, L.P. N-nalkylpyridinium analogs, a novel class of nicotinic receptor antagonists: Selective inhibition of nicotine-evoked [3H] dopamine overflow from superfused rat striatal slices. J. Pharmacol. Exp. Ther. 2003, 306, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, S.; Kontturi, A.K.; Kontturi, K. Improving membrane activity of oligonucleotides by cetylpyridinium chloride: An electrochemical study. Eur. J. Pharm. Sci. 2006, 29, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Karsa, D.R. Industrial Applications of Surfactants, II., 2nd ed.; Royal Society of Chemistry: Cambridge, UK, 1989; pp. 409–412. [Google Scholar]
- Epstein, J.; Kaminski, J.J.; Bodor, N.; Enver, R.; Sowa, J.; Higuchi, T. Micellar Acceleration of Organophosphate Hydrolysis by Hydroximinomethylpyridinium Type Surfactants. J. Org. Chem. 1978, 43, 2816–2821. [Google Scholar] [CrossRef]
- Cabal, J.; Kuca, K.; Micova, J. Kinetics of decomposition of organophosphate Fenitrothion by decontamination foam-making blends. J. Appl. Biomed. 2007, 167–170. [Google Scholar]
- Tiwari, S.; Ghosh, K.K.; Marek, J.; Kuca, K. Cationic Micellar-Catalyzed Hydrolysis of Pesticide Fenitrothion Using alpha-Nucleophiles. Lett. Drug Des. Discov. 2010, 7, 194–199. [Google Scholar] [CrossRef]
- Kotoucova, H.; Cibulka, R.; Hampl, F.; Liska, F. Amphiphilic quaternary pyridinium ketoximes as functional hydrolytic micellar catalysts—Does the nucleophilic function position influence their reactivity? J. Mol. Catal. A Chem. 2001, 174, 59–62. [Google Scholar] [CrossRef]
- Budka, J.; Hampl, F.; Liska, F.; Scrimin, P.; Tecilla, P.; Tonellato, U.J. Micellar nickel(II)-2-pyridineketoxime complexes as powerful catalysts of the cleavage of carboxylic acid esters in weakly acidic conditions. J. Mol. Catal. A Chem. 1996, 104, L201–L204. [Google Scholar] [CrossRef]
- Madaan, P.; Tyagi, V.K. Quaternary Pyridinium Salts: A Review. J. Oleo Sci. 2008, 57, 197–215. [Google Scholar] [CrossRef] [PubMed]
- Bharate, S.B.; Thompson, C.M. Antimicrobial, Antimalarial, and Antileishmanial Activities of Mono- and Bis-quaternary Pyridinium Compounds. Chem. Biol. Drug Des. 2010, 76, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Morales, S.; Compadre, R.L.; Castillo, R.; Breen, P.J.; Compadre, C.M. 3D-QSAR, synthesis, and antimicrobial activity of 1-alkylpyridinium compounds as potential agents to improve food safety. Eur. J. Med. Chem. 2005, 40, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Chanawanno, K.; Chantrapromma, S.; Anantapong, T.; Kanjana-Opas, A.; Fun, H.K. Synthesis, structure and in vitro antibacterial activities of new hybrid disinfectants quaternary ammonium compounds: Pyridinium and quinolinium stilbene benzenesulfonates. Eur. J. Med. Chem. 2010, 45, 4199–4208. [Google Scholar] [CrossRef] [PubMed]
- Quagliotto, P.; Barbero, N.; Barolo, C.; Barni, E.; Compari, C.; Fisicaro, E.; Viscardi, G. Synthesis and properties of cationic surfactants with tuned hydrophylicity. J. Colloid Interf. Sci. 2009, 340, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Kuca, K.; Marek, J.; Stodulka, P.; Musilek, K.; Hanusova, P.; Hrabinova, M.; Jun, D. Preparation of benzalkonium salts differing in the length of a side alkyl chain. Molecules 2007, 12, 2341–2347. [Google Scholar] [CrossRef] [PubMed]
- Marek, J.; Stodulka, P.; Cabal, J.; Soukup, O.; Pohanka, M.; Korabecny, J.; Musilek, K.; Kuca, K. Preparation of the Pyridinium Salts Differing in the Length of the N-Alkyl Substituent. Molecules 2010, 15, 1967–1972. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts. Approved Standard, Third Edition. CSLI document M27-A3, 3rd ed.; CLSI: Wayne, PA, USA, 2008; Volume 28. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi. Approved Standard, Second Edition. CSLI document M38-A2, 2nd ed.; CLSI: Wayne, PA, USA, 2008; Volume 28. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, Seventh Edition, CSLI document M07-A7, 7th ed.; CLSI: Wayne, PA, USA, 2006. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
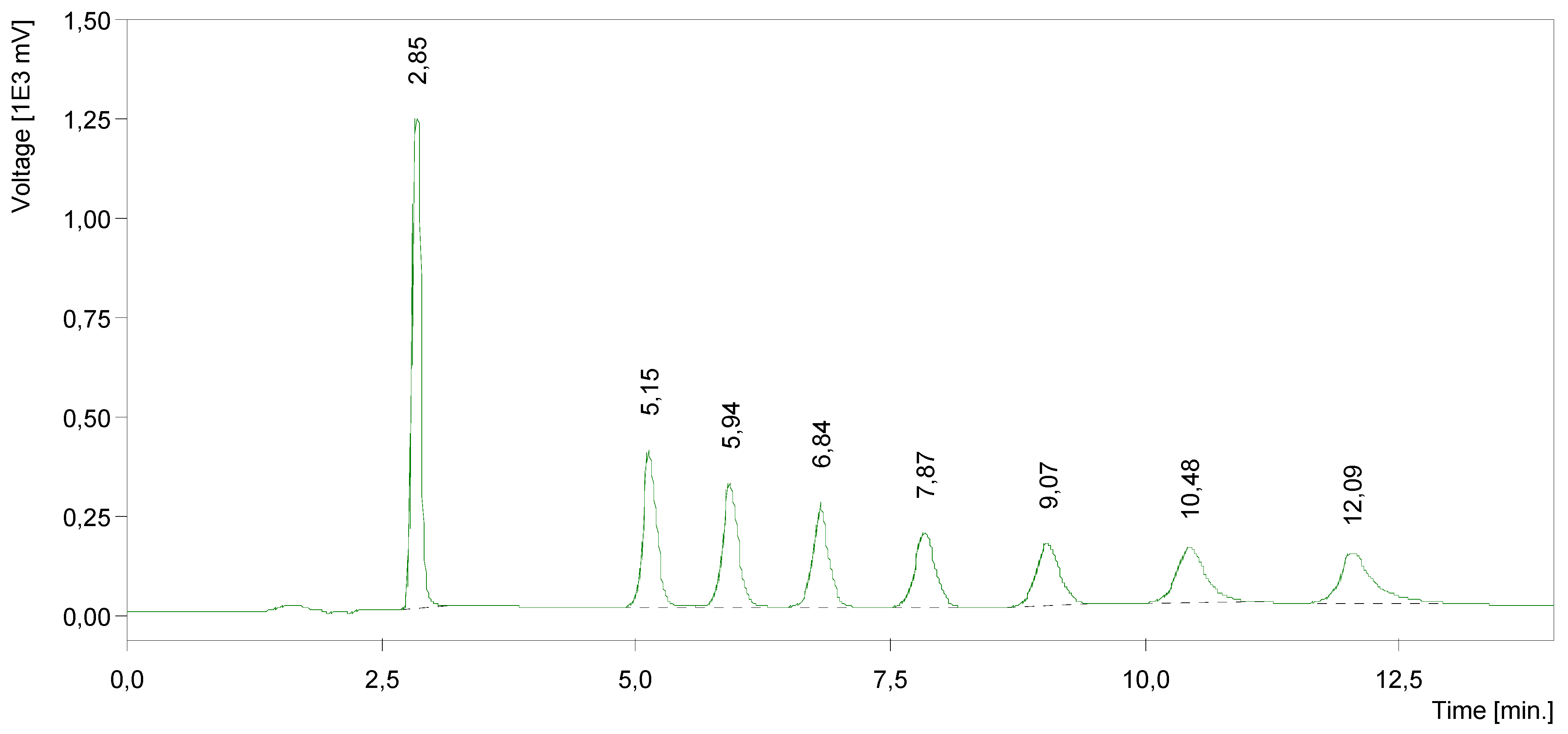
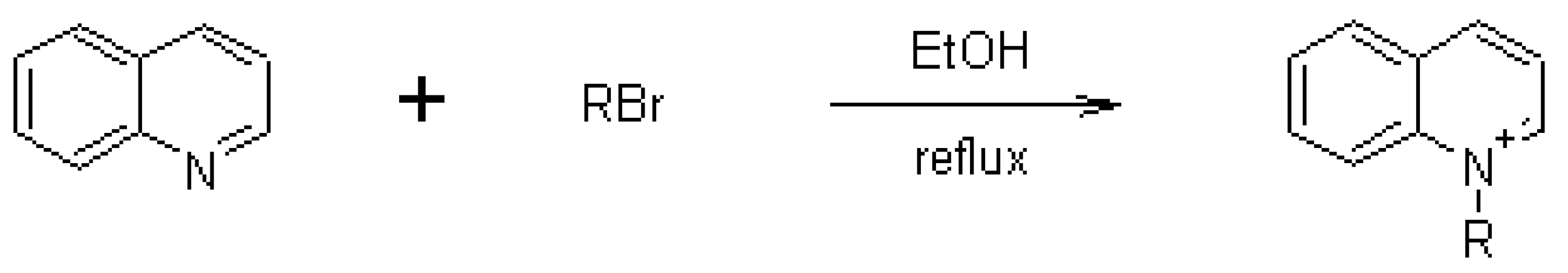
| Compound | Side alkylating chain (R) | Yield (%) | m.p. (°C) | HPLC Rt (min) |
|---|---|---|---|---|
| 3 | C8 | 40.64 | 66–68 | 5.15 |
| 4 | C10 | 34.32 | 48–50 | 5.94 |
| 5 | C12 | 33.71 | 73–75 | 6.84 |
| 6 | C14 | 34.99 | 98–100 | 7.87 |
| 7 | C16 | 22.32 | 90–92 | 9.07 |
| 8 | C18 | 22.16 | 92–94 | 10.48 |
| 9 | C20 | 7,35 | 98–99 | 12,09 |
| Bacterial strains | MIC (μM/L) | MBC (μM/L) | |
|---|---|---|---|
| 24 hours | 48 hours | 48 hours | |
| Staphylococcus aureus ATCC6538 | 15.62 | 15.62 | 15.62 |
| Staphylococcus aureus MRSA HK5996/08 | 15.62 | 15.62 | 15.62 |
| Staphylococcus epidermidis HK6966/08 | 7.81 | 15.62 | 15.62 |
| Enterococcus sp. HK14365/08 | 31.25 | 62.50 | 62.50 |
| Escherichia coli ATCC8739 | 125.00 | 125.00 | 125.00 |
| Klebsiella pneumoniae HK11750/08 | 125.00 | 125.00 | 125.00 |
| Klebsiella pneumoniae ESBL HK14368/08 | 125.00 | 125.00 | 125.00 |
| Pseudomonas aeruginosa ATCC9027 | 250.00 | 250.00 | 250.00 |
| Fungal strains | MIC (μM/L) | MFC (μM/L) | |
|---|---|---|---|
| 24 hours | 48 hours | 48 hours | |
| Candida albicans ATCC 44859 | 15.62 | 15.62 | 15.62 |
| Candida krusei E28 | 15.62 | 15.62 | 15.62 |
| Candida tropicalis 156 | 15.62 | 15.62 | 15.62 |
| Candida glabrata 20/I | 7.81 | 15.62 | 15.62 |
| Trichosporon beigelii 1188 | 15.62 | 62.50 | 62.50 |
| Aspergillus fumigatus 231 | 62.50 | 125.00 | 250.00 |
| Aspergillus corymbifera 272 | 500.00 | 500.00 | 500.00 |
| Trichophyton mentagrophytes 445 | 31.25 | 31.25 | 62.50 |
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Marek, J.; Buchta, V.; Soukup, O.; Stodulka, P.; Cabal, J.; Ghosh, K.K.; Musilek, K.; Kuca, K. Preparation of Quinolinium Salts Differing in the Length of the Alkyl Side Chain. Molecules 2012, 17, 6386-6394. https://doi.org/10.3390/molecules17066386
Marek J, Buchta V, Soukup O, Stodulka P, Cabal J, Ghosh KK, Musilek K, Kuca K. Preparation of Quinolinium Salts Differing in the Length of the Alkyl Side Chain. Molecules. 2012; 17(6):6386-6394. https://doi.org/10.3390/molecules17066386
Chicago/Turabian StyleMarek, Jan, Vladimir Buchta, Ondrej Soukup, Petr Stodulka, Jiri Cabal, Kallol K. Ghosh, Kamil Musilek, and Kamil Kuca. 2012. "Preparation of Quinolinium Salts Differing in the Length of the Alkyl Side Chain" Molecules 17, no. 6: 6386-6394. https://doi.org/10.3390/molecules17066386
APA StyleMarek, J., Buchta, V., Soukup, O., Stodulka, P., Cabal, J., Ghosh, K. K., Musilek, K., & Kuca, K. (2012). Preparation of Quinolinium Salts Differing in the Length of the Alkyl Side Chain. Molecules, 17(6), 6386-6394. https://doi.org/10.3390/molecules17066386




