FG020326 Sensitized Multidrug Resistant Cancer Cells to Docetaxel-Mediated Apoptosis via Enhancement of Caspases Activation
Abstract
:Abbreviations
| MDR | multidrug resistance |
| P-gp | P-glycoprotein |
| P-gp−ve | P-gp negative |
| P-gp+ve | P-gp positive |
| PARP | poly-(ADP-ribose) polymerase |
| PBS | phosphate-buffered saline |
| fmk | fluoromethylketone |
| GB | granzyme B |
| pfp | perforin |
1. Introduction
2. Results and Discussion
2.1. KBv200 Cells with Overexpression of P-gp Are Resistant to Docetaxel-Mediated Death
2.2. Effect of FG020326 on the Reversal of MDR
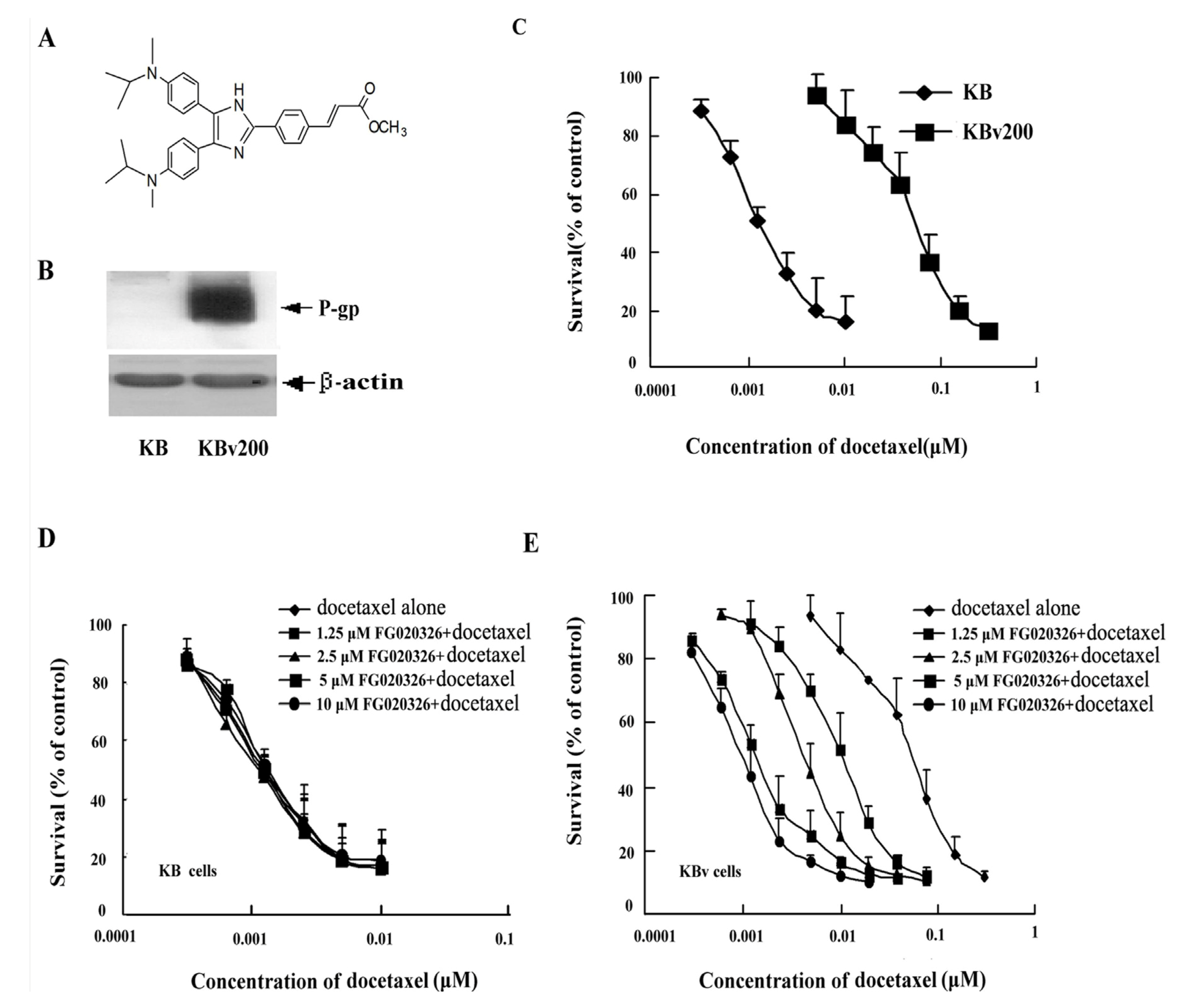
2.3. P-gp+ve KBv200 Cells Are Resistant to Docetaxel-Mediated Apoptosis
| Concertration of FG020326 (μM) | IC50 of docetaxel (μM)) ± SD | Fold-reversal of MDR | ||
|---|---|---|---|---|
| KB | KBV200 | KB | KBV200 | |
| 0 | 1.3 ± 0.2 | 69.8 ± 7.3 | - | - |
| 10 | 1.4 ± 0.5 | 1.5 ± 0.4 ** | 0.87 | 46.53 |
| 5 | 1.3 ± 0.4 | 2.4 ± 1.8 ** | 1.00 | 29.07 |
| 2.5 | 1.3 ± 0.2 | 6.5 ± 5.6 ** | 0.99 | 10.70 |
| 1.25 | 1.4 ± 0.3 | 17.3 ± 11.4 ** | 0.87 | 4.03 |

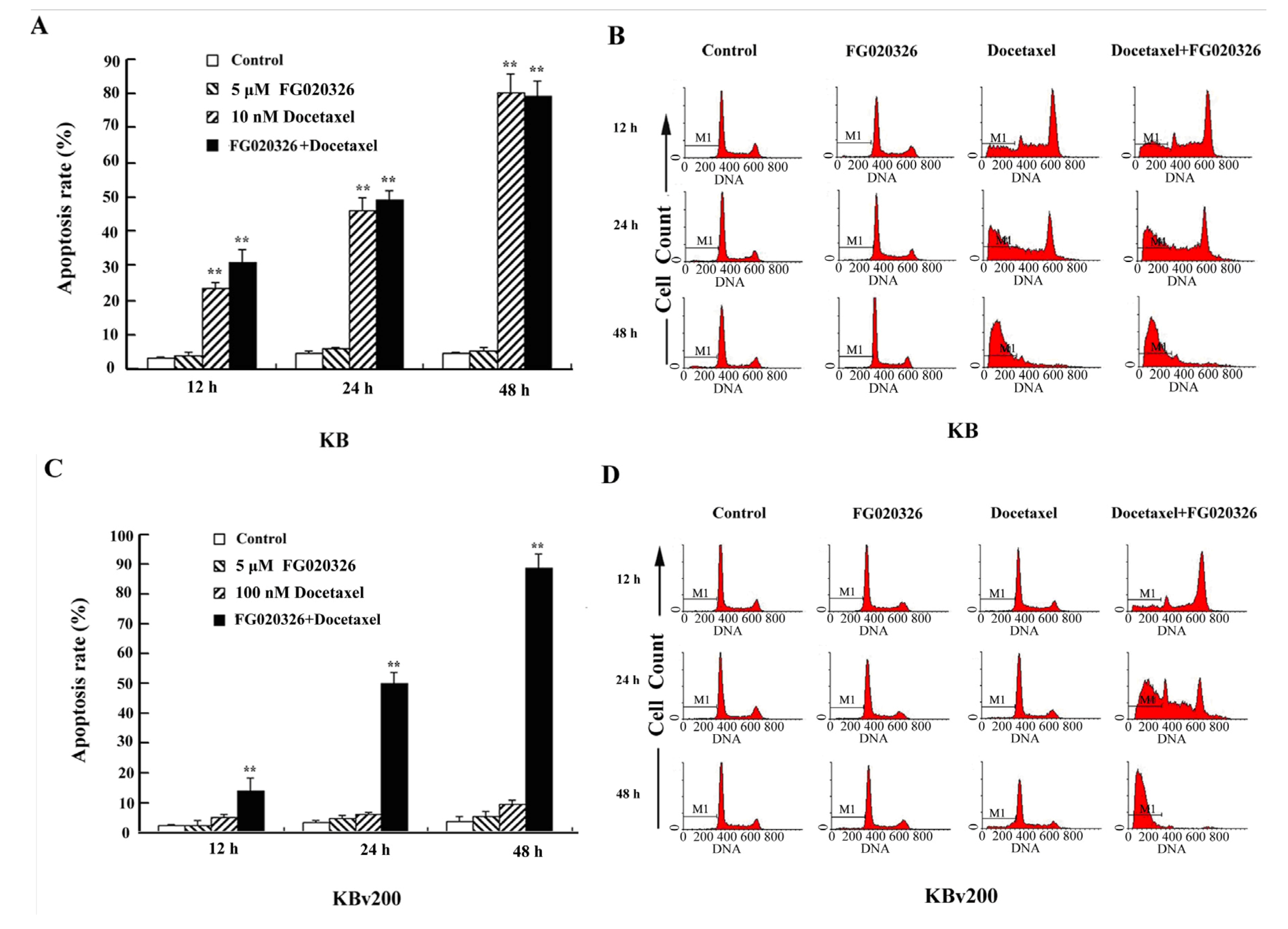
2.4. The Activation of Caspase-8, -9 and -3 Are Involved in Cell Apoptosis Induced by Docetaxel
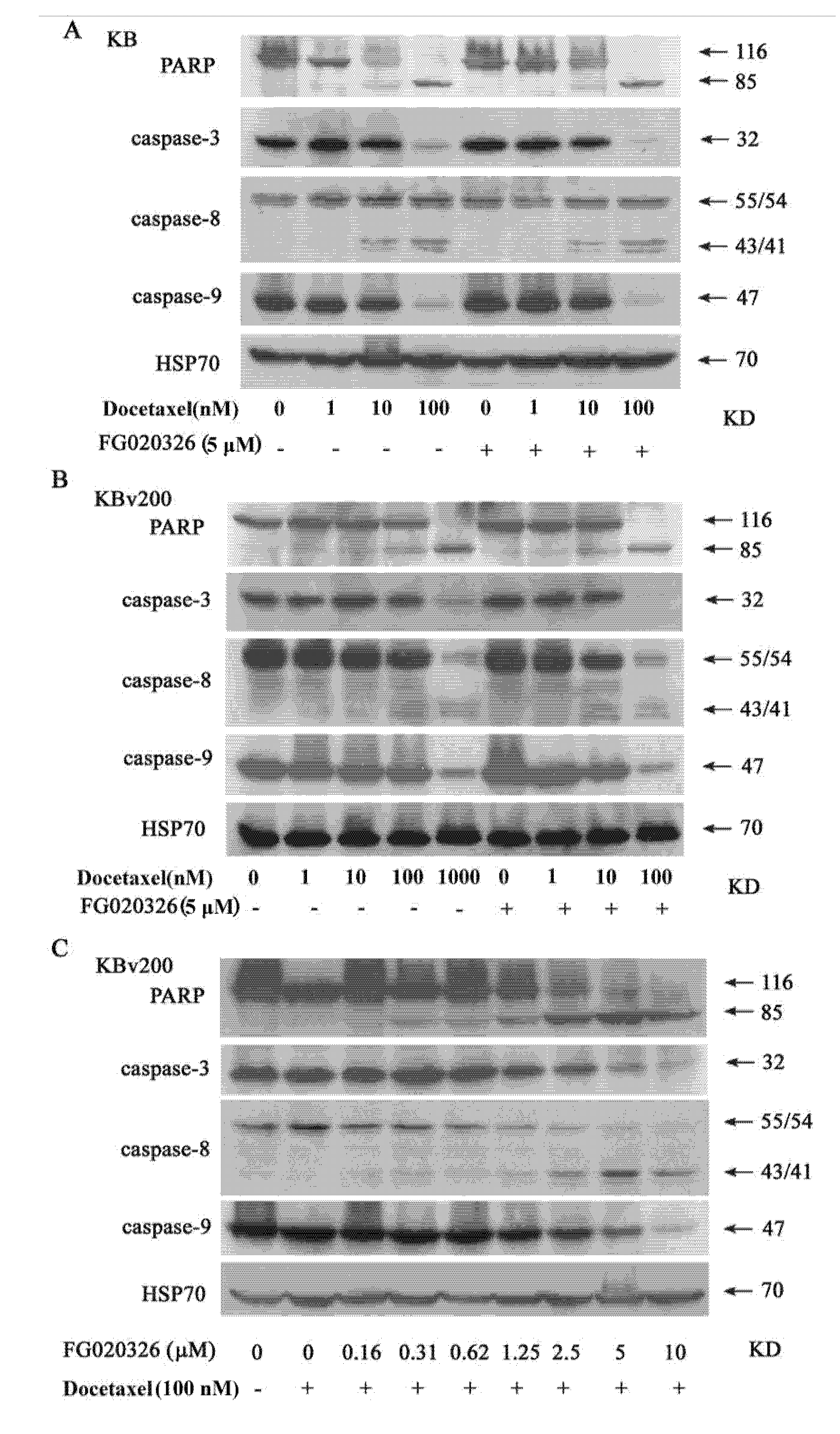
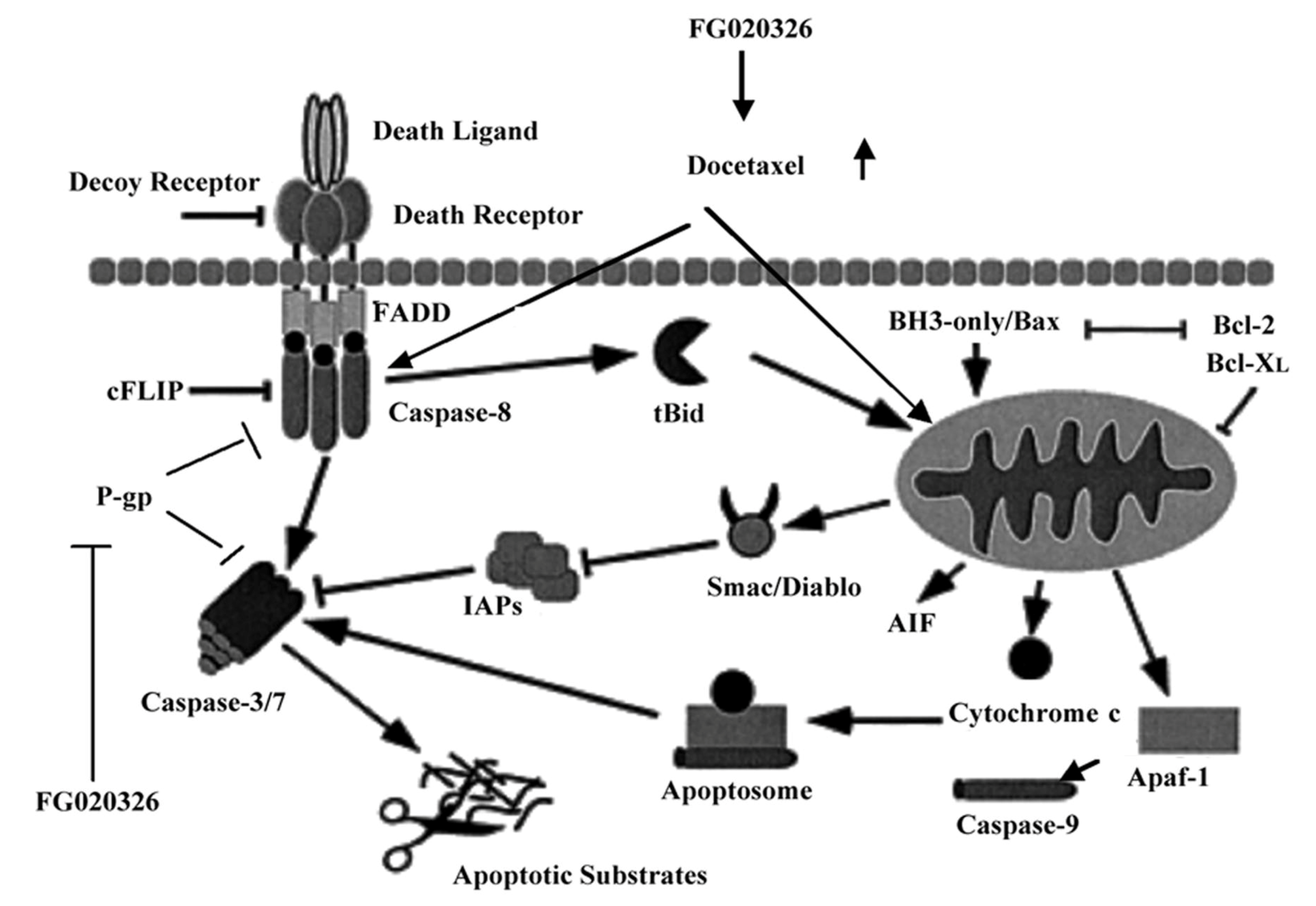
2.5. FG020326 Could Overcome Resistance to Docetaxel-Mediated Apoptosis via the Pathway of Sensitizing Activation Of Caspase-8, -9 and -3 in P-gp+ve KBv200 Cells
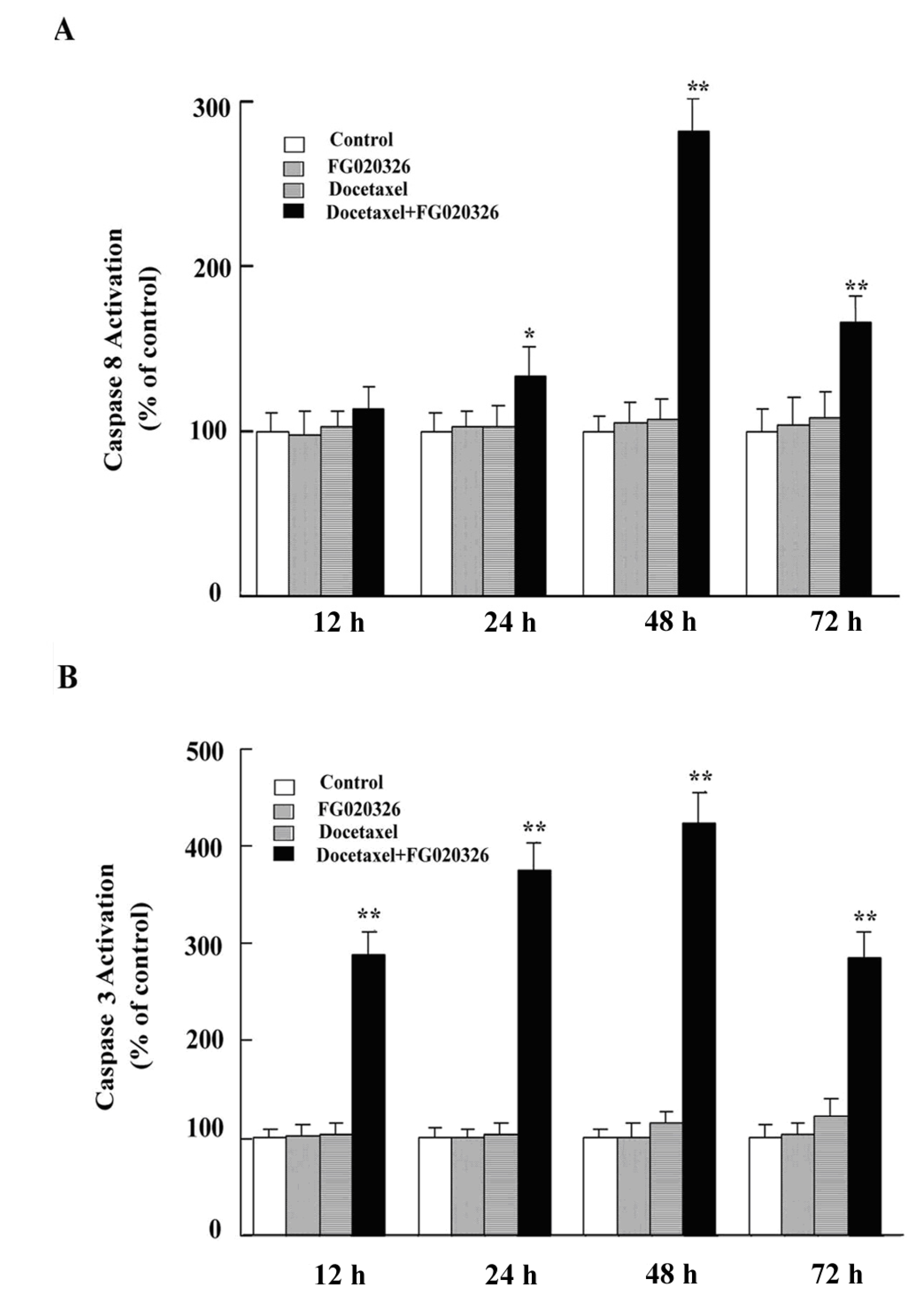
2.6. Discussion
3. Experimental
3.1. Materials
3.2. Cell Lines and Cell Culture
3.3. MTT Cytotoxicity Assay
3.4. Cell Apoptosis Examined by DNA Fragmentation Analysis
3.5. Analysis of Cell Apoptosis by Flow Cytometry
3.6. Apoptotic Cells Detected by HOECHST 33258 Dye
3.7. Immunoblot Analysis
3.8. Caspase-8 Activity Assay
3.10. Statistical Analysis
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compound FG020326 are available from the authors.
References and Notes
- Wilson, C.R.; Sauer, J.; Hooser, S.B. Taxines: A review of the mechanism and toxicity of yew (Taxus spp.) alkaloids. Toxicon 2001, 39, 175–185. [Google Scholar]
- Blagosklonny, M.V.; Fojo, T. Molecular effects of paclitaxel: Myths and reality (a critical review). Int. J. Cancer 1999, 83, 151–156. [Google Scholar] [CrossRef]
- Pucci, B.; Bellincampi, L.; Tafani, M.; Masciullo, V.; Melino, G.; Giordano, A. Paclitaxel induces apoptosis in Saos-2 cells with CD95L upregulation and Bcl-2 phosphorylation. Exp. Cell Res. 1999, 252, 134–143. [Google Scholar] [CrossRef]
- Goncalves, A.; Braguer, D.; Carles, G.; Andre, N.; Prevot, C.; Briand, C. Caspase-8 activation independent of CD95/CD95-L interaction during paclitaxel-induced apoptosis in human colon cancer cells (HT29-D4). Biochem. Pharmacol. 2000, 60, 1579–1584. [Google Scholar] [CrossRef]
- Wang, T.H.; Wang, H.S.; Soong, Y.K. Paclitaxel-induced cell death: Where the cell cycle and apoptosis come together. Cancer 2000, 88, 2619–2628. [Google Scholar] [CrossRef]
- Takahashi, M.; Kigawa, J.; Minagawa, Y.; Itamochi, H.; Shimada, M.; Kamazawa, S.; Sato, S.; Akeshima, R.; Terakawa, N. Sensitivity to paclitaxel is not related to p53-dependent apoptosis in ovarian cancer cells. Eur. J. Cancer 2000, 36, 1863–1868. [Google Scholar] [CrossRef]
- Tan, M.; Jing, T.; Lan, K.H.; Neal, C.L.; Li, P.; Lee, S.; Fang, D.; Nagata, Y.; Liu, J.; Arlinghaus, R.; et al. Phosphorylation on tyrosine-15 of p34Cdc2 by ErbB2 inhibits p34Cdc2 activation and is involved in resistance to taxol-induced apoptosis. Mol. Cell 2002, 9, 993–1004. [Google Scholar] [CrossRef]
- Taniguchi, T.; Takahashi, M.; Shinohara, F.; Sato, T.; Echigo, S.; Rikiishi, H. Involvement of NF-kappaB and mitochondrial pathways in docetaxel-induced apoptosis of human oral squamous cell carcinoma. Int. J. Mol. Med. 2005, 15, 667–673. [Google Scholar]
- Ling, V. Multidrug resistance: Molecular mechanisms and clinical relevance. Cancer Chemother. Pharmacol. 1997, 40, S3–S8. [Google Scholar] [CrossRef]
- Chen, L.M.; Wu, X.P.; Ruan, J.W.; Liang, Y.J.; Ding, Y.; Shi, Z.; Wang, X.W.; Gu, L.Q.; Fu, L.W. Screening novel, potent multidrug-resistant modulators from imidazole derivatives. Oncol. Res. 2004, 14, 355–362. [Google Scholar]
- Fu, L.W.; Deng, Z.A.; Pan, Q.C.; Fan, W. Screening and discovery of novel MDR modifiers from naturally occurring bisbenzylisoquinoline alkaloids. Anticancer Res. 2001, 21, 2273–2280. [Google Scholar]
- Krishna, R.; Mayer, L.D. Multidrug resistance (MDR) in cancer. Mechanisms, reversal using modulators of MDR and the role of MDR modulators in influencing the pharmacokinetics of anticancer drugs. Eur. J. Pharm. Sci. 2000, 11, 265–283. [Google Scholar]
- Sikic, B.I.; Fisher, G.A.; Lum, B.L.; Halsey, J.; Beketic-Oreskovic, L.; Chen, G. Modulation and prevention of multidrug resistance by inhibitors of P-glycoprotein. Cancer Chemother. Pharmacol. 1997, 40, S13–S19. [Google Scholar] [CrossRef]
- Garnier-Suillerot, A.; Marbeuf-Gueye, C.; Salerno, M.; Loetchutinat, C.; Fokt, I.; Krawczyk, M.; Kowalczyk, T.; Priebe, W. Analysis of drug transport kinetics in multidrug-resistant cells: Implications for drug action. Curr. Med. Chem. 2001, 8, 51–64. [Google Scholar]
- Smyth, M.J.; Krasovskis, E.; Sutton, V.R.; Johnstone, R.W. The drug efflux protein, P-glycoprotein, additionally protects drug-resistant tumor cells from multiple forms of caspase-dependent apoptosis. Proc. Natl. Acad. Sci. USA 1998, 95, 7024–7029. [Google Scholar]
- Johnstone, R.W.; Cretney, E.; Smyth, M.J. P-glycoprotein protects leukemia cells against caspase-dependent, but not caspase-independent, cell death. Blood 1999, 93, 1075–1085. [Google Scholar]
- Robinson, L.J.; Roberts, W.K.; Ling, T.T.; Lamming, D.; Sternberg, S.S.; Roepe, P.D. Human MDR 1 protein overexpression delays the apoptotic cascade in Chinese hamster ovary fibroblasts. Biochemistry 1997, 36, 11169–11178. [Google Scholar] [CrossRef]
- Berger, W.; Micksche, M.; Elbling, L. Effects of multidrug resistance-related ATP-binding-cassette transporter proteins on the cytoskeletal activity of cytochalasins. Exp. Cell Res. 1997, 237, 307–317. [Google Scholar] [CrossRef]
- Kang, C.D.; Ahn, B.K.; Jeong, C.S.; Kim, K.W.; Lee, H.J.; Yoo, S.D.; Chung, B.S.; Kim, S.H. Downregulation of JNK/SAPK activity is associated with the cross-resistance to P-glycoprotein-unrelated drugs in multidrug-resistant FM3A/M cells overexpressing P-glycoprotein. Exp. Cell Res. 2000, 256, 300–307. [Google Scholar] [CrossRef]
- Coultas, L.; Strasser, A. The molecular control of DNA damage-induced cell death. Apoptosis 2000, 5, 491–507. [Google Scholar] [CrossRef]
- Kim, R.; Emi, M.; Tanabe, K. Caspase-dependent and -independent cell death pathways after DNA damage. Oncol. Rep. 2005, 14, 595–599. [Google Scholar]
- Ruefli, A.A.; Smyth, M.J.; Johnstone, R.W. HMBA induces activation of a caspase-independent cell death pathway to overcome P-glycoprotein-mediated multidrug resistance. Blood 2000, 95, 2378–2385. [Google Scholar]
- Reed, J.C. Cytochrome c: Can’t live with it—Can’t live without it. Cell 1997, 91, 559–562. [Google Scholar] [CrossRef]
- Orrenius, S. Mitochondrial regulation of apoptotic cell death. Toxicol. Lett. 2004, 149, 19–23. [Google Scholar] [CrossRef]
- Kluck, R.M.; Bossy-Wetzel, E.; Green, D.R.; Newmeyer, D.D. The release of cytochrome c from mitochondria: A primary site for Bcl-2 regulation of apoptosis. Science 1997, 275, 1132–1136. [Google Scholar]
- Susin, S.A.; Lorenzo, H.K.; Zamzami, N.; Marzo, I.; Snow, B.E.; Brothers, G.M.; Mangion, J.; Jacotot, E.; Costantini, P.; Loeffler, M.; et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 1999, 397, 441–446. [Google Scholar] [CrossRef]
- Nutt, L.K.; Gogvadze, V.; Uthaisang, W.; Mirnikjoo, B.; McConkey, D.J.; Orrenius, S. Indirect effects of Bax and Bak initiate the mitochondrial alterations that lead to cytochrome c release during arsenic trioxide-induced apoptosis. Cancer Biol. Ther. 2005, 4, 459–467. [Google Scholar] [CrossRef]
- Zou, H.; Henzel, W.J.; Liu, X.; Lutschg, A.; Wang, X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 1997, 90, 405–413. [Google Scholar]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Luo, X.; Budihardjo, I.; Zou, H.; Slaughter, C.; Wang, X. Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 1998, 94, 481–490. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.; Xu, C.J.; Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 1998, 94, 491–501. [Google Scholar] [CrossRef]
- Konopleva, M.; Zhao, S.; Xie, Z.; Segall, H.; Younes, A.; Claxton, D.F.; Estrov, Z.; Kornblau, S.M.; Andreeff, M. Apoptosis. Molecules and mechanisms. Adv. Exp. Med. Biol. 1999, 457, 217–236. [Google Scholar]
- Houghton, J.A. Apoptosis and drug response. Curr. Opin. Oncol. 1999, 11, 475–481. [Google Scholar] [CrossRef]
- Li, J.; Sasaki, H.; Sheng, Y.L.; Schneiderman, D.; Xiao, C.W.; Kotsuji, F.; Tsang, B.K. Apoptosis and chemoresistance in human ovarian cancer: Is Xiap a determinant? Biol. Signals Recept. 2000, 9, 122–130. [Google Scholar] [CrossRef]
- Costantini, P.; Jacotot, E.; Decaudin, D.; Kroemer, G. Mitochondrion as a novel target of anticancer chemotherapy. J. Natl. Cancer Inst. 2000, 92, 1042–1053. [Google Scholar] [CrossRef]
- Kidd, V.J.; Lahti, J.M.; Teitz, T. Proteolytic regulation of apoptosis. Semin. Cell Dev. Biol. 2000, 11, 191–201. [Google Scholar]
- Cao, D.X.; Qiao, B.; Ge, Z.Q.; Yuan, Y.J. Comparison of burst of reactive oxygen species and activation of caspase-3 in apoptosis of K562 and HL-60 cells induced by docetaxel. Cancer Lett. 2004, 214, 103–113. [Google Scholar] [CrossRef]
- Yin, X.M.; Wang, K.; Gross, A.; Zhao, Y.; Zinkel, S.; Klocke, B.; Roth, K.A.; Korsmeyer, S.J. Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature 1999, 400, 886–891. [Google Scholar] [CrossRef]
- Varfolomeev, E.E.; Schuchmann, M.; Luria, V.; Chiannilkulchai, N.; Beckmann, J.S.; Mett, I.L.; Rebrikov, D.; Brodianski, V.M.; Kemper, O.C.; Kollet, O.; et al. argeted disruption of the mouse Caspase 8 gene ablates cell death induction by the TNF receptors, Fas/Apo1, and DR3 and is lethal prenatally. Immunity 1998, 9, 267–276. [Google Scholar] [CrossRef]
- Herr, I.; Debatin, K.M. Cellular stress response and apoptosis in cancer therapy. Blood 2001, 98, 2603–2614. [Google Scholar] [CrossRef]
- Ruan, J.W.; Fu, L.W.; Huang, Z.S.; Chen, L.M.; Ma, L.; Gu, L.Q. Studies on synthesis of multiaryl-substituted imidazoles and reversal activity on the multidrug resistance. Chem. J. Chin. Univ. 2004, 25, 870–873. [Google Scholar]
- Zhang, X.H.; Zhang, F.Y.; Ji, X.J.; Li, Z.Y. Vincristine-resistant human KB cell line and mechanism of multidrug resistance. Yao Xue Xue Bao 1994, 29, 246–251. [Google Scholar]
- Fu, L.W.; Pan, Q.C.; Huang, W.L.; Yang, X.P. Reversal of tumor multidrug resistance by 2-phenyl-3-(3',5'-dimorpholinomethyl-4'-hydroxy)-benzoyl-indole (HWL-12). Zhongguo Yao Li Xue Bao 1998, 19, 77–80. [Google Scholar]
- Herr, I.; Wilhelm, D.; Bohler, T.; Angel, P.; Debatin, K.M. Activation of CD95 (APO-1/Fas) signaling by ceramide mediates cancer therapy-induced apoptosis. EMBO J. 1997, 16, 6200–6208. [Google Scholar] [CrossRef]
- Hishikawa, K.; Oemar, B.S.; Tanner, F.C.; Nakaki, T.; Luscher, T.F.; Fujii, T. Connective tissue growth factor induces apoptosis in human breast cancer cell line MCF-7. J. Biol. Chem. 1999, 274, 37461–37466. [Google Scholar]
- Jiang, X.H.; Wong, B.C.; Yuen, S.T.; Jiang, S.H.; Cho, C.H.; Lai, K.C.; Lin, M.C.; Kung, H.F.; Lam, S.K. Arsenic trioxide induces apoptosis in human gastric cancer cells through up-regulation of p53 and activation of caspase-3. Int. J. Cancer 2001, 91, 173–179. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.-W.; Wang, X.-K.; Zhang, X.; Liang, Y.-J.; Shi, Z.; Chen, L.-M.; Fu, L.-W. FG020326 Sensitized Multidrug Resistant Cancer Cells to Docetaxel-Mediated Apoptosis via Enhancement of Caspases Activation. Molecules 2012, 17, 5442-5458. https://doi.org/10.3390/molecules17055442
Wang X-W, Wang X-K, Zhang X, Liang Y-J, Shi Z, Chen L-M, Fu L-W. FG020326 Sensitized Multidrug Resistant Cancer Cells to Docetaxel-Mediated Apoptosis via Enhancement of Caspases Activation. Molecules. 2012; 17(5):5442-5458. https://doi.org/10.3390/molecules17055442
Chicago/Turabian StyleWang, Xiu-Wen, Xiao-Kun Wang, Xu Zhang, Yong-Ju Liang, Zhi Shi, Li-Ming Chen, and Li-Wu Fu. 2012. "FG020326 Sensitized Multidrug Resistant Cancer Cells to Docetaxel-Mediated Apoptosis via Enhancement of Caspases Activation" Molecules 17, no. 5: 5442-5458. https://doi.org/10.3390/molecules17055442
APA StyleWang, X.-W., Wang, X.-K., Zhang, X., Liang, Y.-J., Shi, Z., Chen, L.-M., & Fu, L.-W. (2012). FG020326 Sensitized Multidrug Resistant Cancer Cells to Docetaxel-Mediated Apoptosis via Enhancement of Caspases Activation. Molecules, 17(5), 5442-5458. https://doi.org/10.3390/molecules17055442





