New Methodology for the Synthesis of Thiobarbiturates Mediated by Manganese(III) Acetate
Abstract
:1. Introduction
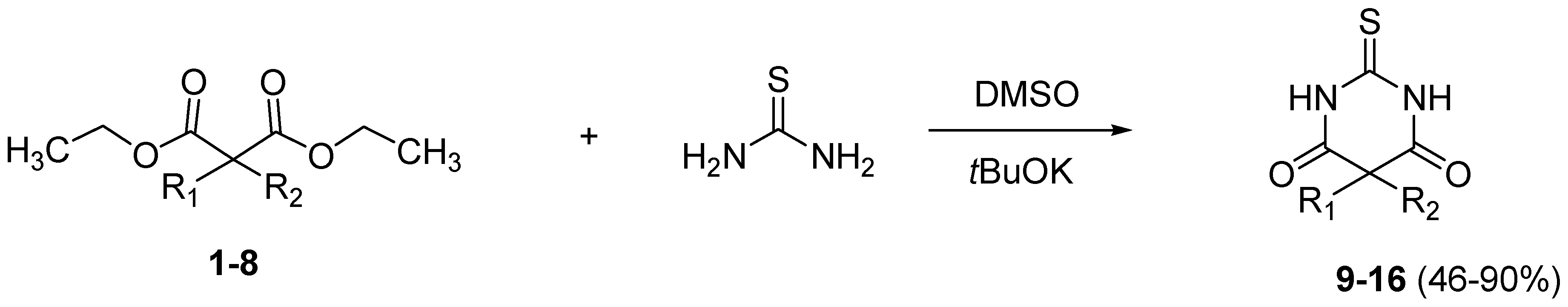
2. Results and Discussion
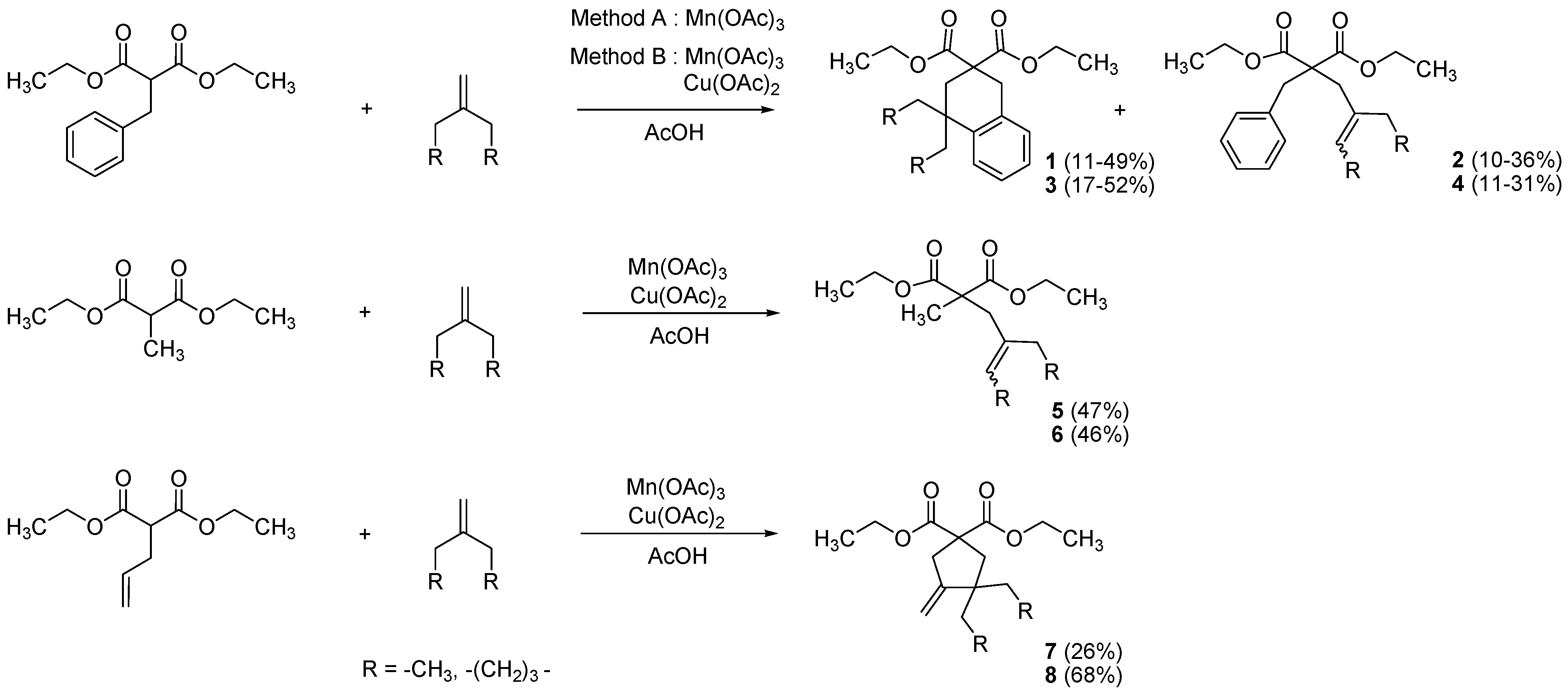
| Entry | R1,R2 (malonate) | Product | Yields |
|---|---|---|---|
| 1 | 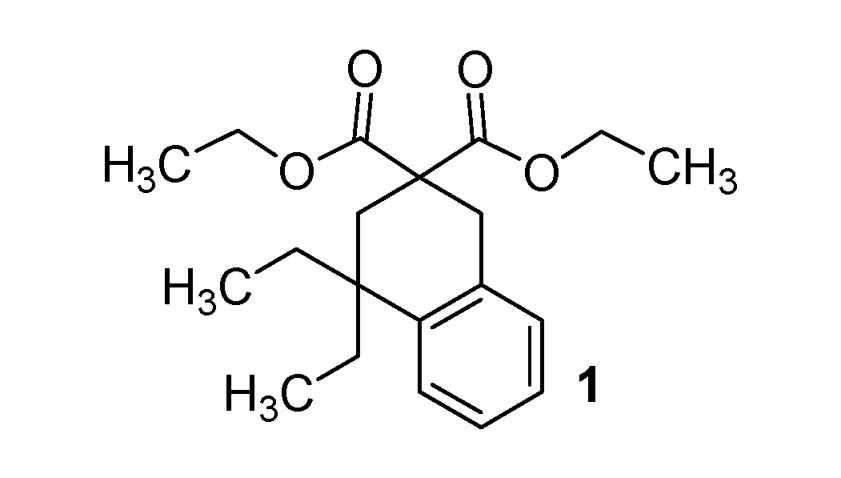 | 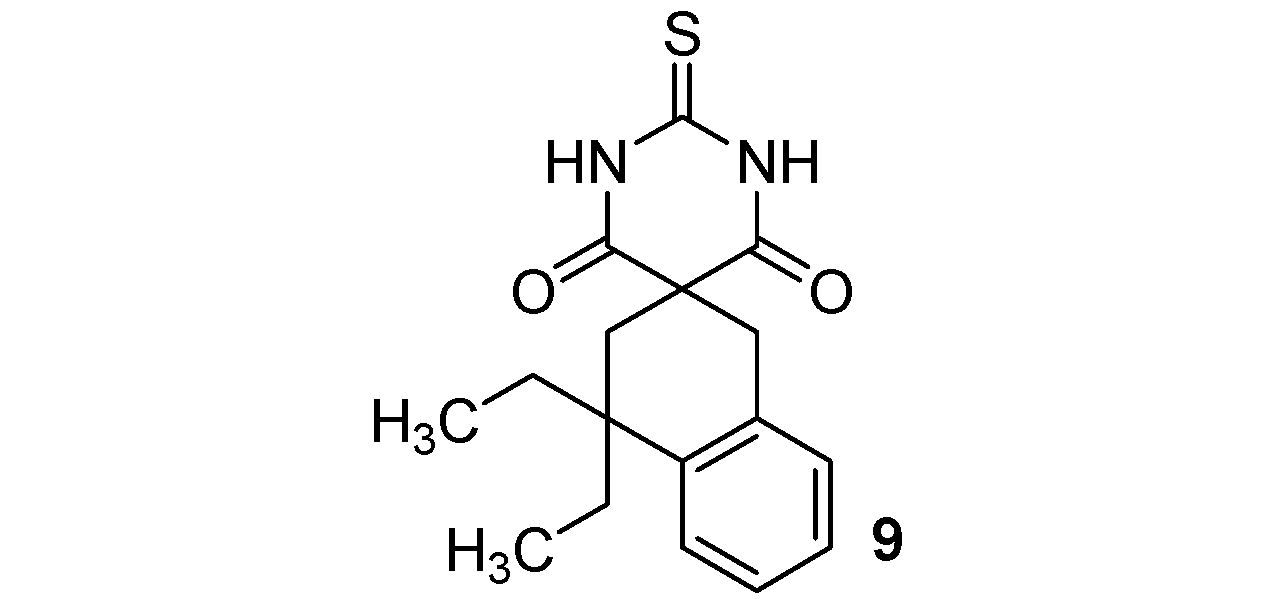 | 53% |
| 2 | 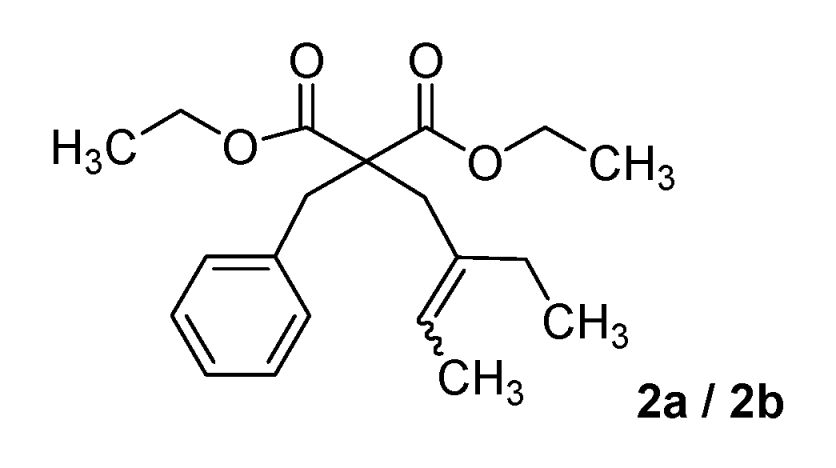 | 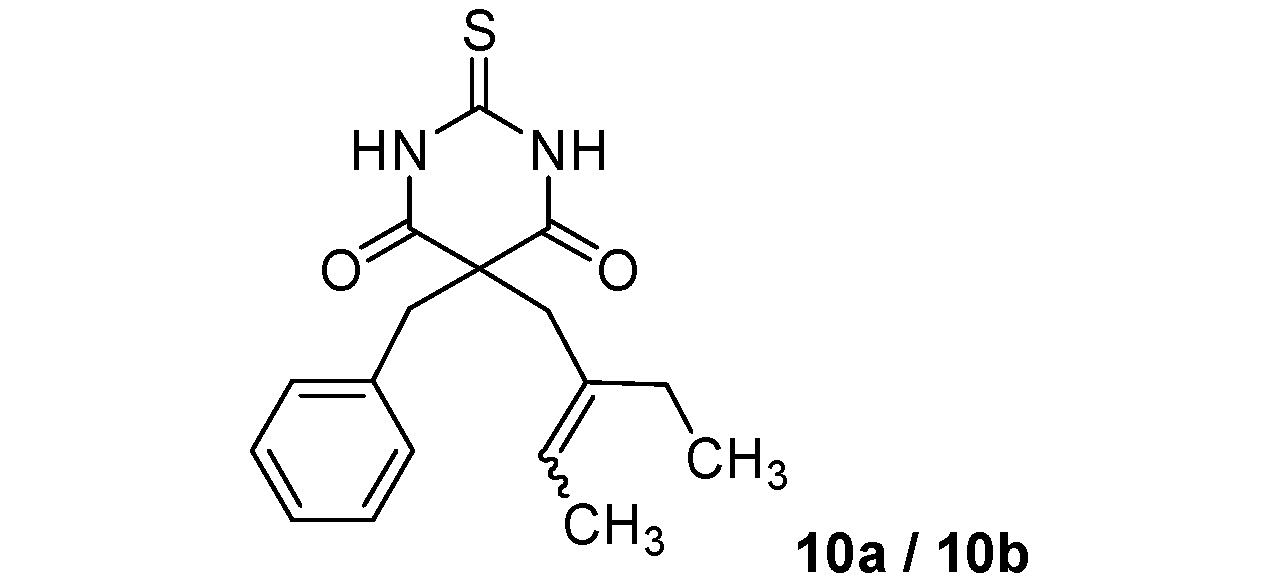 | 46% |
| 3 | 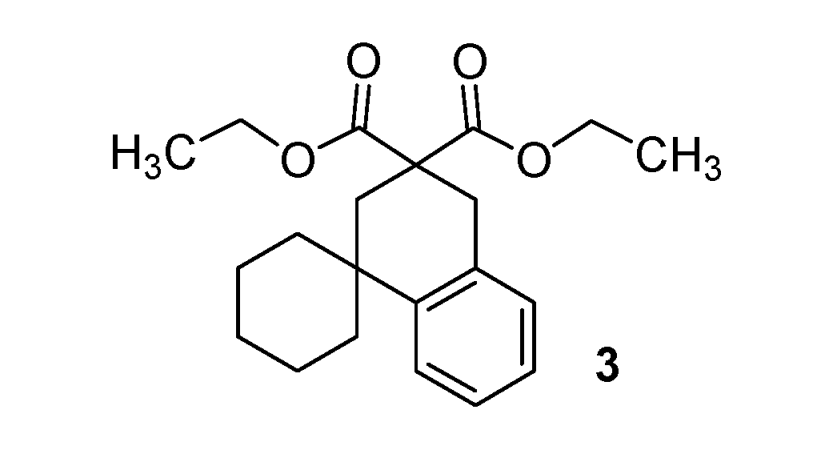 | 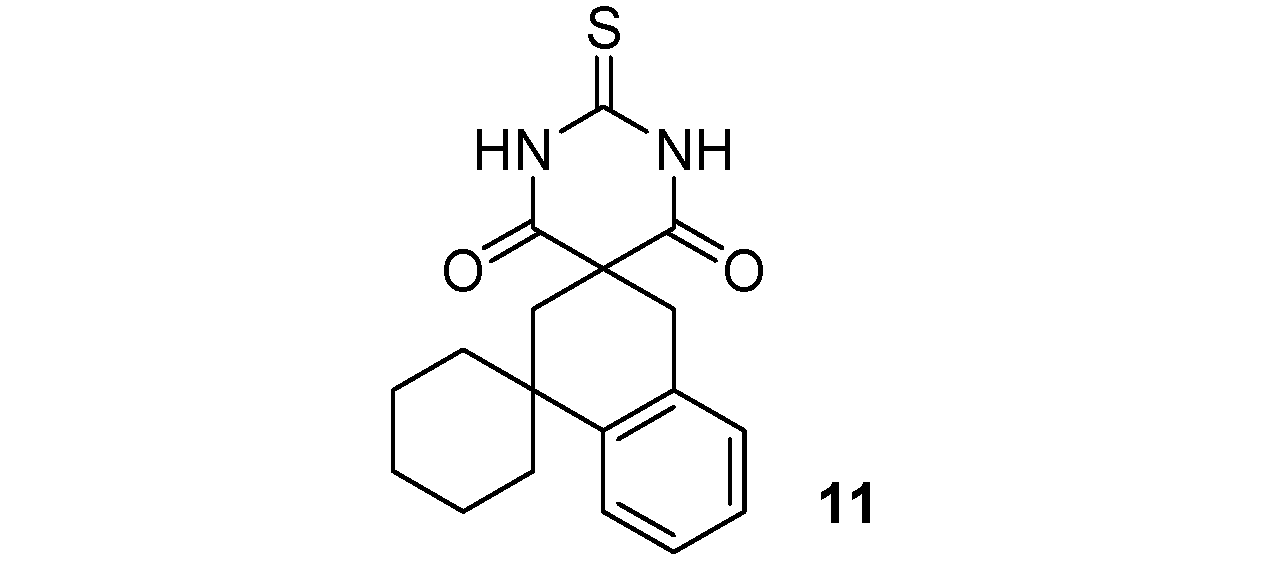 | 64% |
| 4 |  | 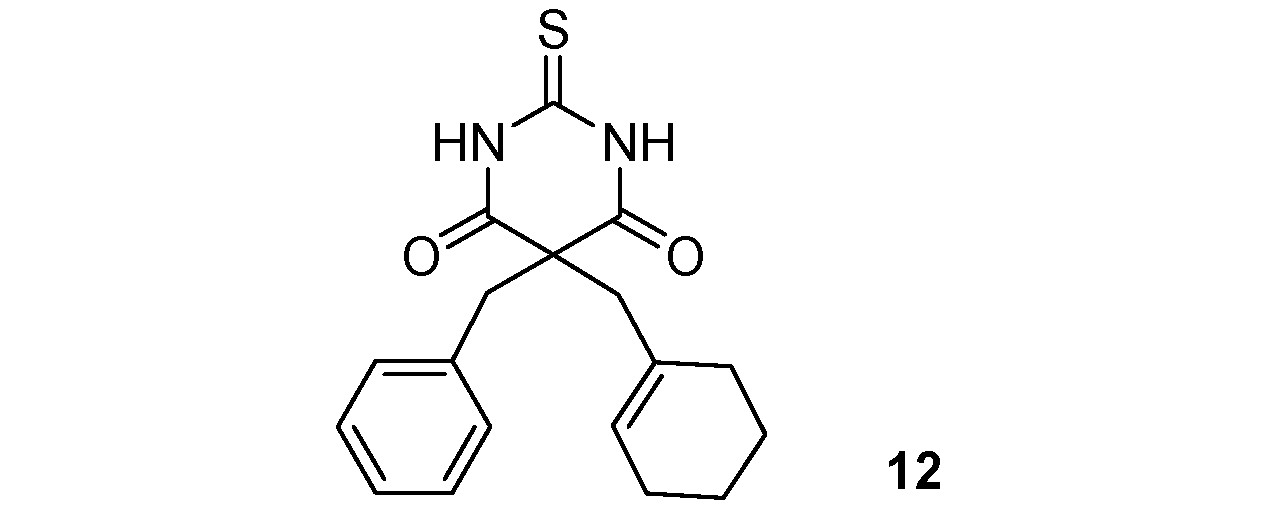 | 88% |
| 5 | 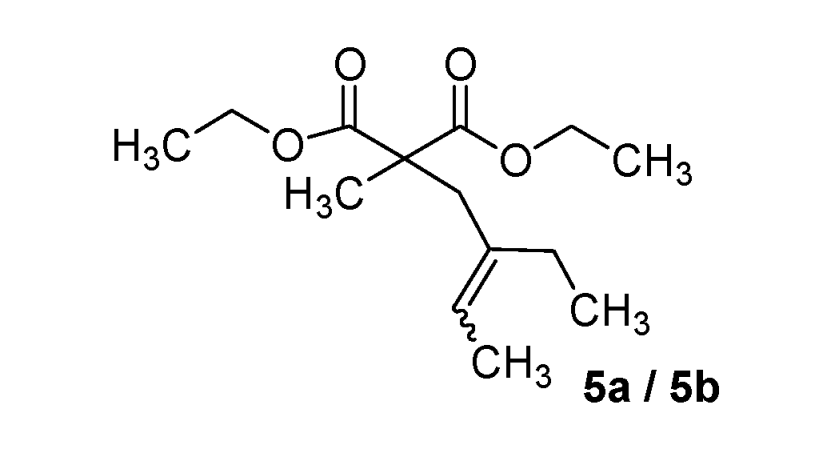 | 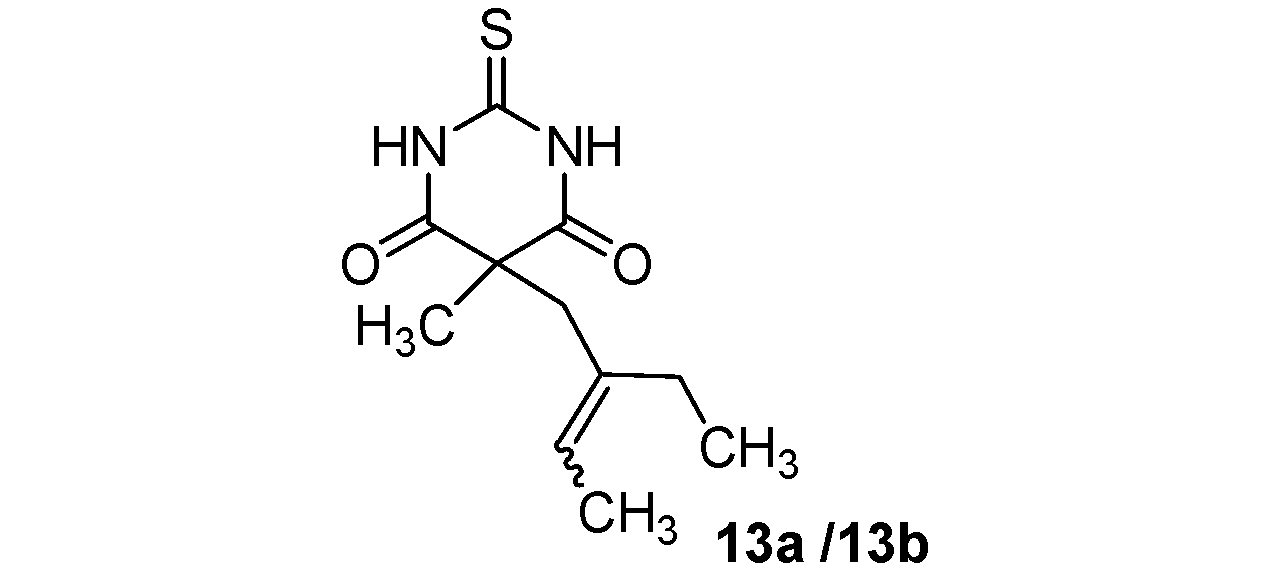 | 75% |
| 6 | 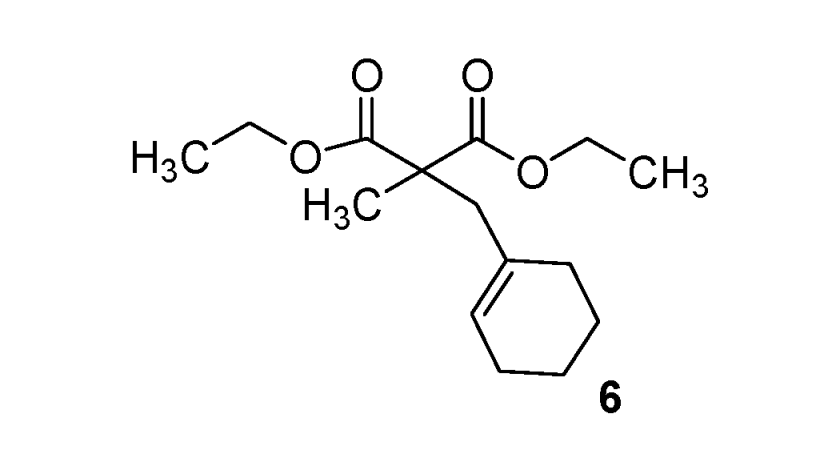 | 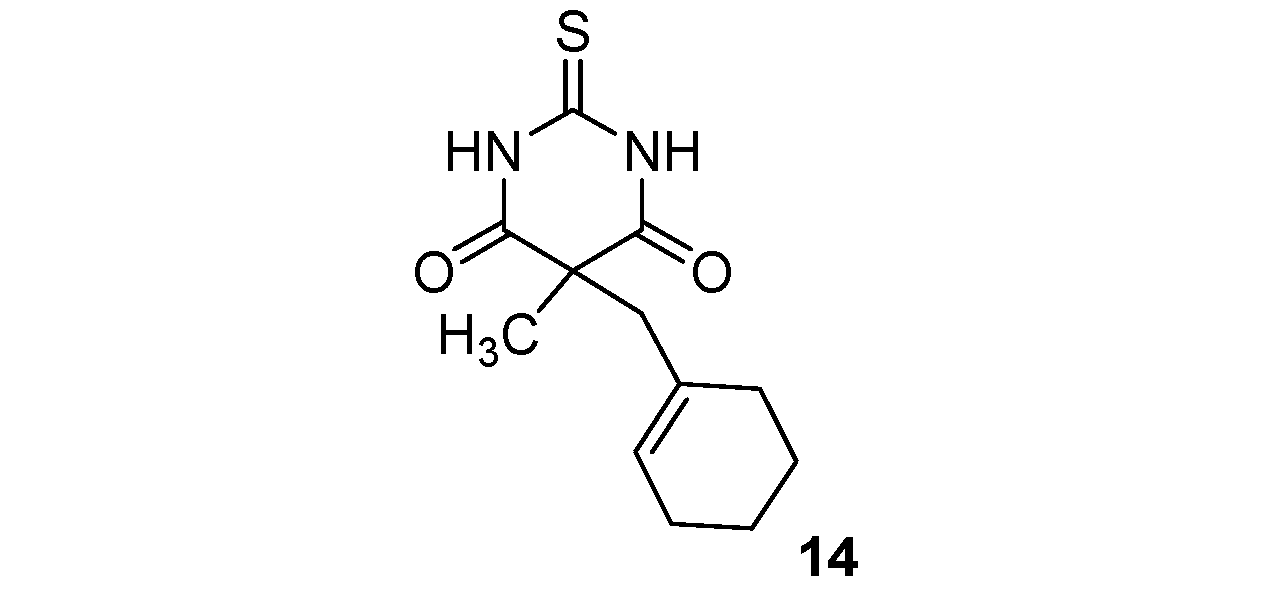 | 90% |
| 7 | 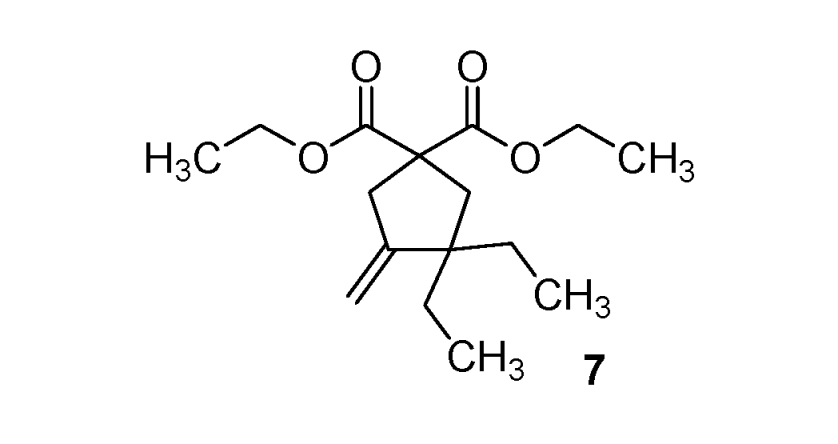 | 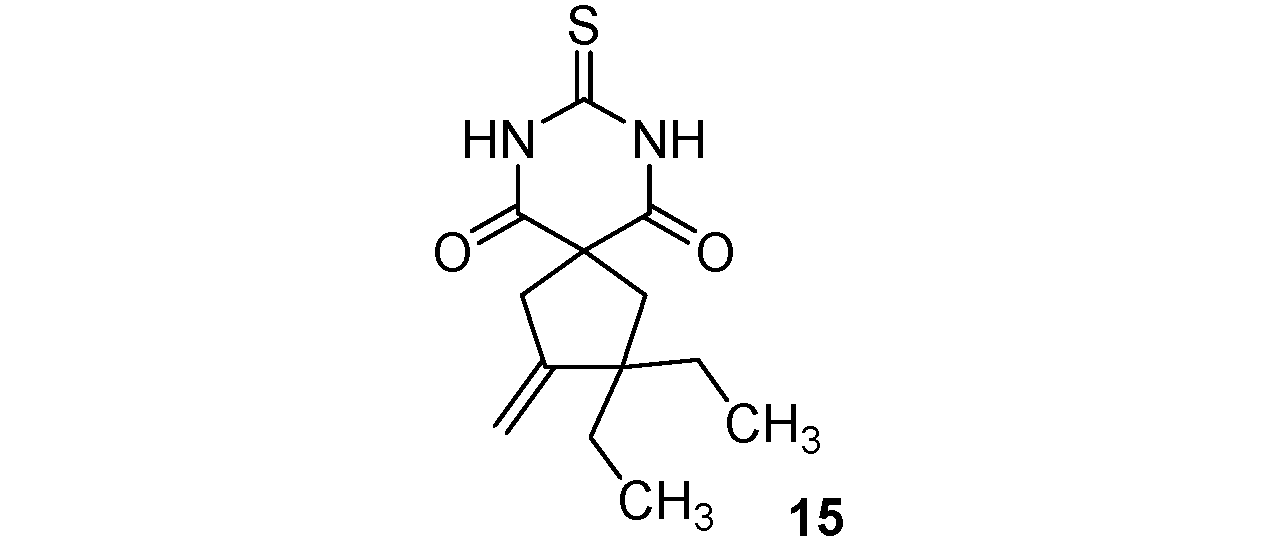 | 70% |
| 8 |  | 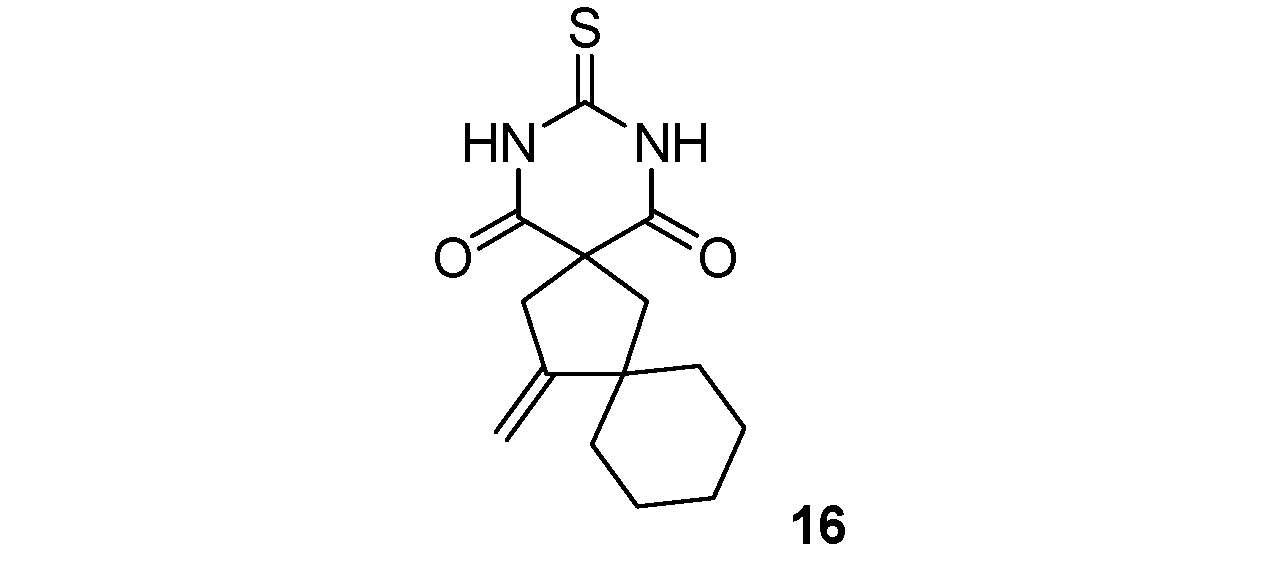 | 54% |
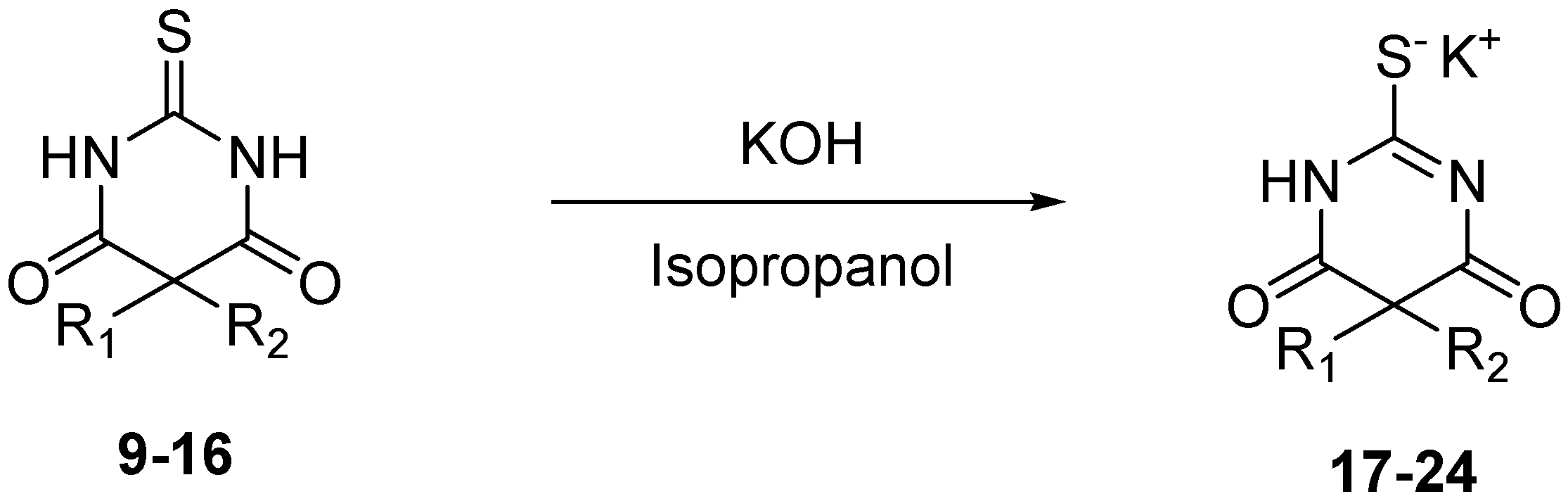
3. Experimental
3.1. General
3.2. General Procedure for the Synthesis of Substituted Malonates 1–8
3.3. General Procedure for the Synthesis of Thiobarbituric Acids 9–16
3.4. General Procedure for Salification of Barbituric Acids to Barbiturate Potassium Salts 17–24
4. Conclusions
Acknowledgements
References and Notes
- Snider, B.B. Manganese(III)-based oxidative free-radical cyclizations. Chem. Rev. 1996, 96, 339–363. [Google Scholar]
- Demir, A.S.; Emrullahoglu, M. Manganese(III) acetate: A versatile reagent in organic chemistry. Curr. Org. Synth. 2007, 4, 321–350. [Google Scholar]
- Dombroski, M.A.; Snider, B.B. Manganese(III)-Based oxidative free-radical. Cyclizations of γ,γ-bis(allylic) acetoacetates. Tetrahedron 1992, 48, 1417–1426. [Google Scholar] [CrossRef]
- Kates, S.A.; Dombroski, M.A.; Snider, B.B. Manganese(III)-based oxidative free-radical cyclization of unsaturated beta-keto esters, 1,3-diketones, and malonate diesters. J. Org. Chem. 1990, 55, 2427–2436. [Google Scholar] [CrossRef]
- Chuang, C.-P.; Tsai, A.-I. A novel oxidative free radical reaction between 2-amino-1,4-benzoquinones and benzoylacetonitriles. Tetrahedron 2007, 63, 9712–9717. [Google Scholar]
- Logoglu, E.; Yilmaz, M.; Katircioglu, H.; Yakut, M.; Mercan, S. Synthesis and biological activity studies of furan derivatives. Med. Chem. Res. 2010, 19, 490–497. [Google Scholar]
- Curti, C.; Crozet, M.D.; Vanelle, P. Microwave-assisted manganese(III) acetate based oxidative cyclizations of alkenes with β-ketosulfones. Tetrahedron 2009, 65, 200–205. [Google Scholar]
- Bouhlel, A.; Curti, C.; Dumètre, A.; Laget, M.; Crozet, M.D.; Azas, N.; Vanelle, P. Synthesis and evaluation of original amidoximes as antileishmanial agents. Bioorg. Med. Chem. 2010, 18, 7310–7320. [Google Scholar]
- Paloque, L.; Bouhlel, A.; Curti, C.; Dumètre, A.; Verhaeghe, P.; Azas, N.; Vanelle, P. Synthesis and evaluation of monoamidoxime derivatives: Toward new antileishmanial compounds. Eur. J. Med. Chem. 2011, 46, 2984–2991. [Google Scholar]
- Biltz, H.; Witteck, H. Alkylated and acylated barbituric acids. Ber. Dtsch. Chem. Ges. 1921, 54, 1035–1059. [Google Scholar]
- Clark-Lewis, J.W.; Thompson, M.J. Preparation of 1,3-dimethylbarbituric acid and formation of 5-ethoxycarbonylacetyl-1,3-dimethylbarbituric acid. J. Chem. Soc. 1959, 1628–1629. [Google Scholar]
- Nguyen, V.; Nishino, H. Novel synthesis of dihydropyrans and 2,8-dioxabicylo[3.3.0]oct-3-enes using Mn(III)-based oxidative cyclization. Tetrahedron Lett. 2004, 45, 3373–3377. [Google Scholar] [CrossRef]
- Tsubusaki, T.; Nishino, H. Manganese(III)-mediated facile synthesis of 3,4-dihydro-2(1H)-quinolinones: Selectivity of the 6-endo and 5-exo cyclization. Tetrahedron 2009, 65, 9448–9459. [Google Scholar]
- Delmas, F.; Gasquet, M.; Timon-David, P.; Madadi, N.; Vanelle, P.; Vaille, A.; Maldonado, J. Synthesis and in vitro anti-protozoan activity of new 5-nitrothiophene oxime ether derivatives. Eur. J. Med. Chem. 1993, 28, 23–27. [Google Scholar] [CrossRef]
- Baraldi, P.G.; El-Kashef, H.; Farghaly, A.R.; Vanelle, P.; Fruttarolo, F. Synthesis of new pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidines and related heterocycles. Tetrahedron 2004, 60, 5093–5104. [Google Scholar]
- Boufatah, N.; Gellis, A.; Maldonado, J.; Vanelle, P. Efficient microwave-assisted synthesis of new sulfonylbenzimidazole-4,7-diones: Heterocyclic quinones with potential antitumor activity. Tetrahedron 2004, 60, 9131–9137. [Google Scholar]
- Verhaeghe, P.; Azas, N.; Gasquet, M.; Hutter, S.; Ducros, C.; Laget, M.; Rault, S.; Rathelot, P.; Vanelle, P. Synthesis and antiplasmodial activity of new 4-aryl-2-trichloromethylquinazolines. Bioorg. Med. Chem. Lett. 2008, 18, 396–401. [Google Scholar]
- Kabri, Y.; Gellis, A.; Vanelle, P. Microwave-assisted synthesis in aqueous medium of new quinazoline derivatives as anticancer agent precursors. Green Chem. 2009, 11, 201–208. [Google Scholar]
- Smith, M.C.; Riskin, B.J. The clinical use of barbiturates in neurological disorders. Drugs 1991, 42, 365–378. [Google Scholar]
- Nagel, S.; von Heinemann, P.; Heiland, S.; Koziol, J.; Gardner, H.; Wagner, S. Selective MMP-inhibition with Ro 28-2653 in acute experimental stroke—A magnetic resonance imaging efficacy study. Brain Res. 2011, 1368, 264–270. [Google Scholar] [CrossRef]
- Wang, J.; Medina, C.; Radomski, M.W.; Gilmer, J.F. N-substituted homopiperazine barbiturates as gelatinase inhibitors. Bioorg. Med. Chem. 2011, 19, 4985–4999. [Google Scholar]
- Vijaya, L.S.; Thirupathi, R.Y.; Suresh, K.B.; Narsimha, R.P.; Crooks, P.A.; Rajitha, B. Synthesis and evaluation of chromenyl barbiturates and thiobarbiturates as potential antitubercular agents. Bioorg. Med. Chem. Lett. 2011, 21, 4329–4331. [Google Scholar]
- Al-Najjar, B.O.; Wahab, H.A.; Muhammad, T.S.T.; Shu-Chien, A.C.; Noruddin, N.A.A.; Taha, M.O. Discovery of new nanomolar peroxisome proliferator-activated receptor γ activators via elaborate ligand-based modeling. Eur. J. Med. Chem. 2011, 46, 2513–2529. [Google Scholar]
- Ma, L.; Li, S.; Zheng, H.; Chen, J.; Lin, L.; Ye, X.; Chen, Z.; Xu, Q.; Chen, T.; Yang, J.; et al. Synthesis and biological activity of novel barbituric and thiobarbituric acid derivatives against non-alcoholic fatty liver disease. Eur. J. Med. Chem. 2011, 46, 2003–2010. [Google Scholar]
- Zheng, H.; Li, S.; Ma, L.; Cheng, L.; Deng, C.; Chen, Z.; Xie, C.; Xiang, M.; Jiang, W.; Chen, L. A novel agonist of PPAR-γ based on barbituric acid alleviates the development of non-alcoholic fatty liver disease by regulating adipocytokine expression and preventing insulin resistance. Eur. J. Pharmacol. 2011, 659, 244–251. [Google Scholar] [CrossRef]
- Gruber, P.; Rechfeld, F.; Kirchmair, J.; Hauser, N.; Boehler, M.; Garczarczyk, D.; Langer, T.; Hofmann, J. Barbituric acid derivative BAS 02104951 inhibits PKCε, PKCη, PKCε/RACK2 interaction, Elk-1 phosphorylation in HeLa and PKCε and η translocation in PC3 cells following TPA-induction. J. Biochem. 2011, 149, 331–336. [Google Scholar]
- Soine, W. Sedative-Hypnotics. In Foye’s Principles of Medicinal Chemistry, 6th; Foye, W.O., Lemke, T.L., Williams, D.A., Eds.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2007; pp. 504–520. [Google Scholar]
- Kepczyńska, E.; Obłoza, E.; Stasiewicz-Urban, A.; Bojarski, J.; Pyka, A. Lipophilicity of thiobarbiturates determined by TLC. Acta Pol. Pharm. 2007, 64, 295–302. [Google Scholar]
- Toon, S.; Rowland, M. Structure-pharmacokinetic relationships among the barbiturates in the rat. J. Pharmacol. Exp. Ther. 1983, 225, 752–763. [Google Scholar]
- Snider, B.B.; Patricia, J.J.; Kates, S.A. Mechanism of manganese(III)-based. Oxidation of β-keto esters. J. Org. Chem. 1988, 53, 2137–2143. [Google Scholar] [CrossRef]
- Citterio, A.; Sebastiano, R.; Marion, A. Synthesis of substituted tetrahydronaphthalenes by manganese(III), cerium(IV), and iron(III) oxidation of substituted diethyl alpha-benzylmalonates in the presence of olefins. J. Org. Chem. 1991, 56, 5328–5335. [Google Scholar]
- Santi, R.; Bergamini, F.; Citterio, A.; Sebastiano, R.; Nicolini, M. Reactivity of malonyl radicals. Synthesis of substituted dihydronaphthalenes by manganese(III) oxidation of diethyl alpha-benzylmalonate in the presence of alkynes. J. Org. Chem. 1992, 57, 4250–4255. [Google Scholar]
- Bergamini, F.; Citterio, A.; Gatti, N.; Nicolini, M.; Santi, R.; Sebastiano, R. Metal-induced electrochemical oxidation of diethyl benzylmalonates in the presence of alkenes and alkynes. Synthesis of substituted tetrahydro- and dihydronaphthalenes. J. Chem. Res. (S) 1993, 9, 364–365. [Google Scholar]
- Bouhlel, A.; Curti, C.; Vanelle, P. Access to original spirocyclic derivatives via inter- or intramolecular reaction mediated by manganese(III) acetate. Tetrahedron 2012, 68, 3596–3604. [Google Scholar]
- Snider, B.B.; Buckman, B.O. Manganese(III) based oxidative free-radical annulations. Tetrahedron 1989, 45, 6969–6978. [Google Scholar]
- Jagodzinska, M.; Huguenot, F.; Candiani, G.; Zanda, M. Assessing the bioisosterism of the trifluoromethyl group with a protease probe. Chem. Med. Chem. 2009, 4, 49–51. [Google Scholar]
- Sun, T.; Watson, S.; Manchanda, R. Disclosed are new phenobarbital salts, methods of preparation, and uses thereof. U.S. Patent 0035904, 11 February 2010. Chem. Abstr. 2010, 152, 223581. [Google Scholar]
- Sample Availability: Samples of the compounds 6, 8, 10, 15, 17–24 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bouhlel, A.; Curti, C.; Vanelle, P. New Methodology for the Synthesis of Thiobarbiturates Mediated by Manganese(III) Acetate. Molecules 2012, 17, 4313-4325. https://doi.org/10.3390/molecules17044313
Bouhlel A, Curti C, Vanelle P. New Methodology for the Synthesis of Thiobarbiturates Mediated by Manganese(III) Acetate. Molecules. 2012; 17(4):4313-4325. https://doi.org/10.3390/molecules17044313
Chicago/Turabian StyleBouhlel, Ahlem, Christophe Curti, and Patrice Vanelle. 2012. "New Methodology for the Synthesis of Thiobarbiturates Mediated by Manganese(III) Acetate" Molecules 17, no. 4: 4313-4325. https://doi.org/10.3390/molecules17044313
APA StyleBouhlel, A., Curti, C., & Vanelle, P. (2012). New Methodology for the Synthesis of Thiobarbiturates Mediated by Manganese(III) Acetate. Molecules, 17(4), 4313-4325. https://doi.org/10.3390/molecules17044313




