Coupling of Nanoporous Chromium, Aluminium-Containing Silicates with an Ionic Liquid for the Transformation of Glucose into 5-(Hydroxymethyl)-2-furaldehyde
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterisation of the Catalysts
| Sample | Si/Al | Si/Cr | SBET (m2·g−1) | Vp (cm3·g−1) | PSD (pore width, nm) |
|---|---|---|---|---|---|
| Al-TUD-1 | 20 | - | 726 (789) | 0.64 (0.69) | 2.5–7 (2.5–7) |
| CrAl-TUD-1 | 27 | 108 | 763 (995) | 0.78 (0.95) | 2.5–7 (2.5–7) |
| Cr-TUD-1 | - | 150 | 484 (515) | 1.02 (1.14) | 3–17 (3–17) |
| BEA | 12 | - | 634 | 0.74 | - |
| Cr-BEA | 13 | 47 | 702 (793) | 0.87 (0.75) | - |
| BEA/TUD-1 | 31 | - | 685 (722) | 0.78 (0.82) | 2.5–10 (2.5–10) |
| Cr-BEA/TUD-1 | 41 | 236 | 802 (717) | 0.92 (0.80) | 2.5–10 (2.5–10) |
 ), CrAl-TUD-1 (
), CrAl-TUD-1 (  ), Cr-TUD-1 (○), and (b) BEA (
), Cr-TUD-1 (○), and (b) BEA (  ), Cr-BEA (
), Cr-BEA (  ), BEA/TUD-1 (○), Cr-BEA/TUD-1 (
), BEA/TUD-1 (○), Cr-BEA/TUD-1 ( 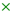 ). The insets show the PSD curves (dp is the pore width) for the respective materials (with matching symbols).
). The insets show the PSD curves (dp is the pore width) for the respective materials (with matching symbols).
 ), CrAl-TUD-1 (
), CrAl-TUD-1 (  ), Cr-TUD-1 (○), and (b) BEA (
), Cr-TUD-1 (○), and (b) BEA (  ), Cr-BEA (
), Cr-BEA (  ), BEA/TUD-1 (○), Cr-BEA/TUD-1 (
), BEA/TUD-1 (○), Cr-BEA/TUD-1 (  ). The insets show the PSD curves (dp is the pore width) for the respective materials (with matching symbols).
). The insets show the PSD curves (dp is the pore width) for the respective materials (with matching symbols).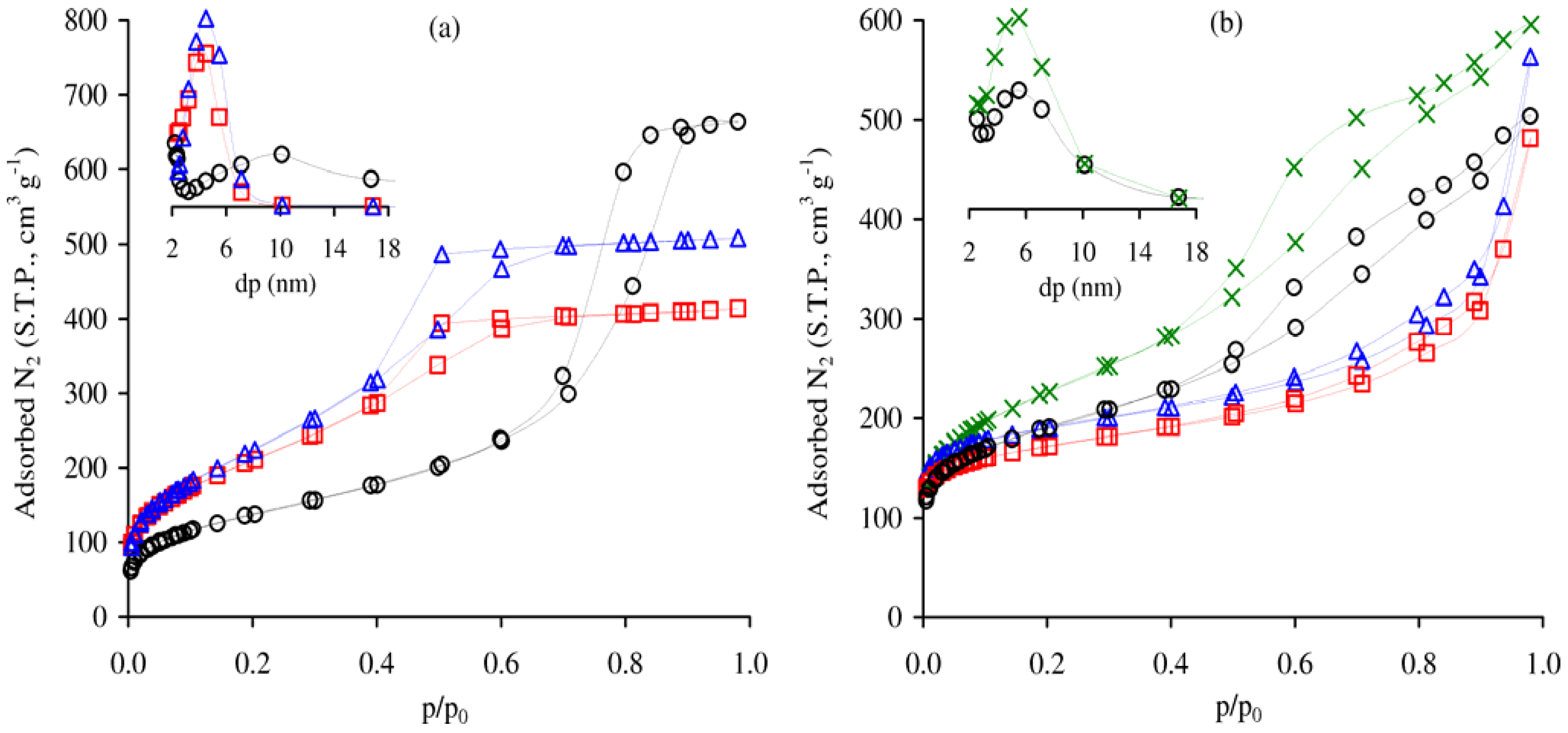

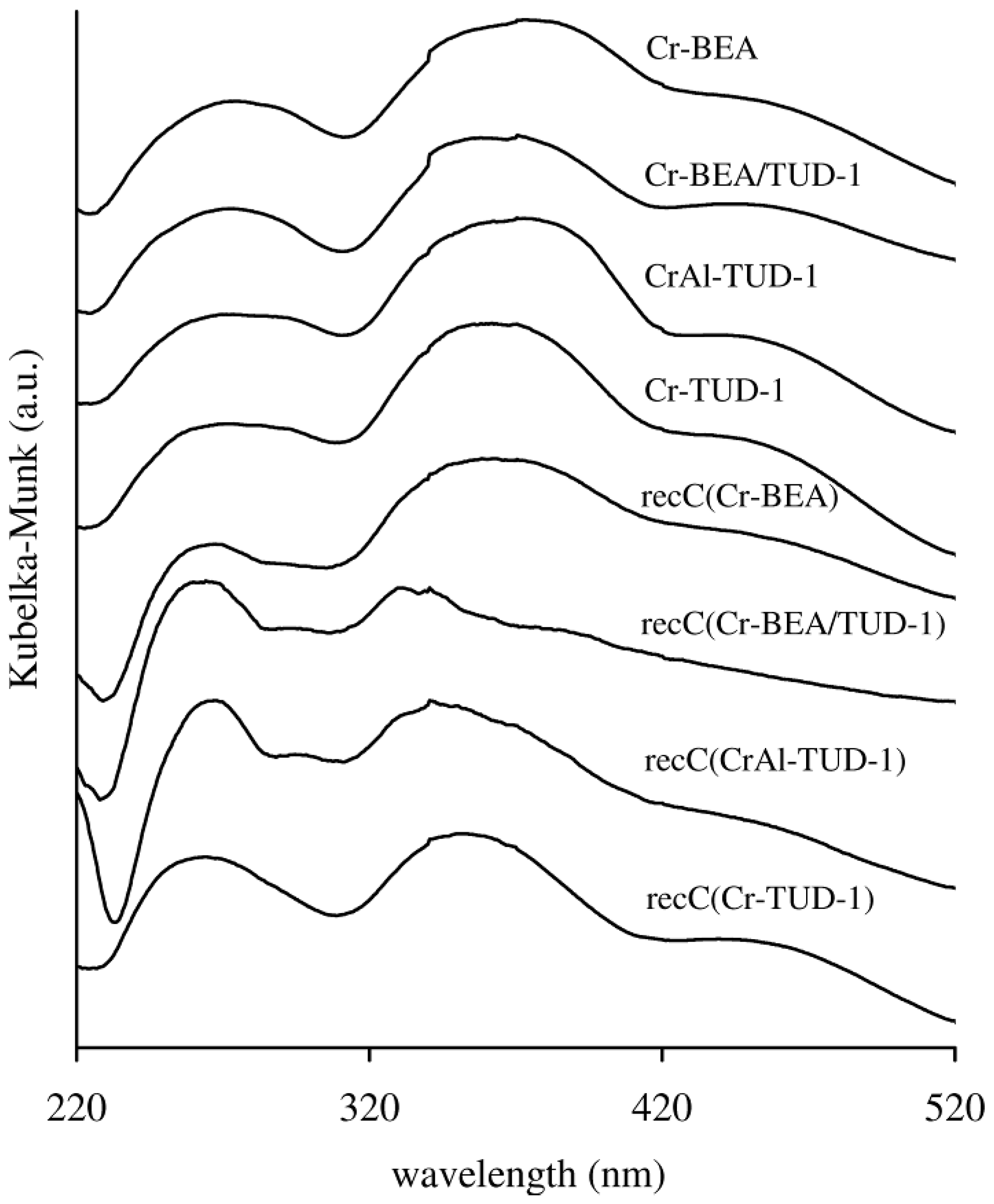
2.2. Catalysis
2.2.1. General Considerations
| Sample | Catalytic BR | Experiment (i) | Experiment (ii) | Experiment (iii) | ||||
|---|---|---|---|---|---|---|---|---|
| Conv. b (%) | Yield c (%) | Conv. b (%) | Yield c (%) | Conv. b (%) | Yield c (%) | Conv. b (%) | Yield c (%) | |
| Al-TUD-1 | 65 | 9 | 23 | 1 | 64 | 9 | 61 | 9 |
| CrAl-TUD-1 | 82 | 54 | 37 | 9 | 70 | 12 | 79 | 59 |
| Cr-TUD-1 | 42 | 39 | 18 | 5 | 42 | 24 | 46 | 42 |
| BEA | 85 | 13 | 11 | 4 | 80 | 17 | 75 | 15 |
| Cr-BEA | 96 | 58 | 39 | 10 | 84 | 28 | 96 | 60 |
| BEA/TUD-1 | 75 | 11 | 34 | 4 | 69 | 15 | 70 | 7 |
| Cr-BEA/TUD-1 | 65 | 36 | 19 | 2 | 62 | 13 | 66 | 58 |
2.2.2. Catalyst Stability
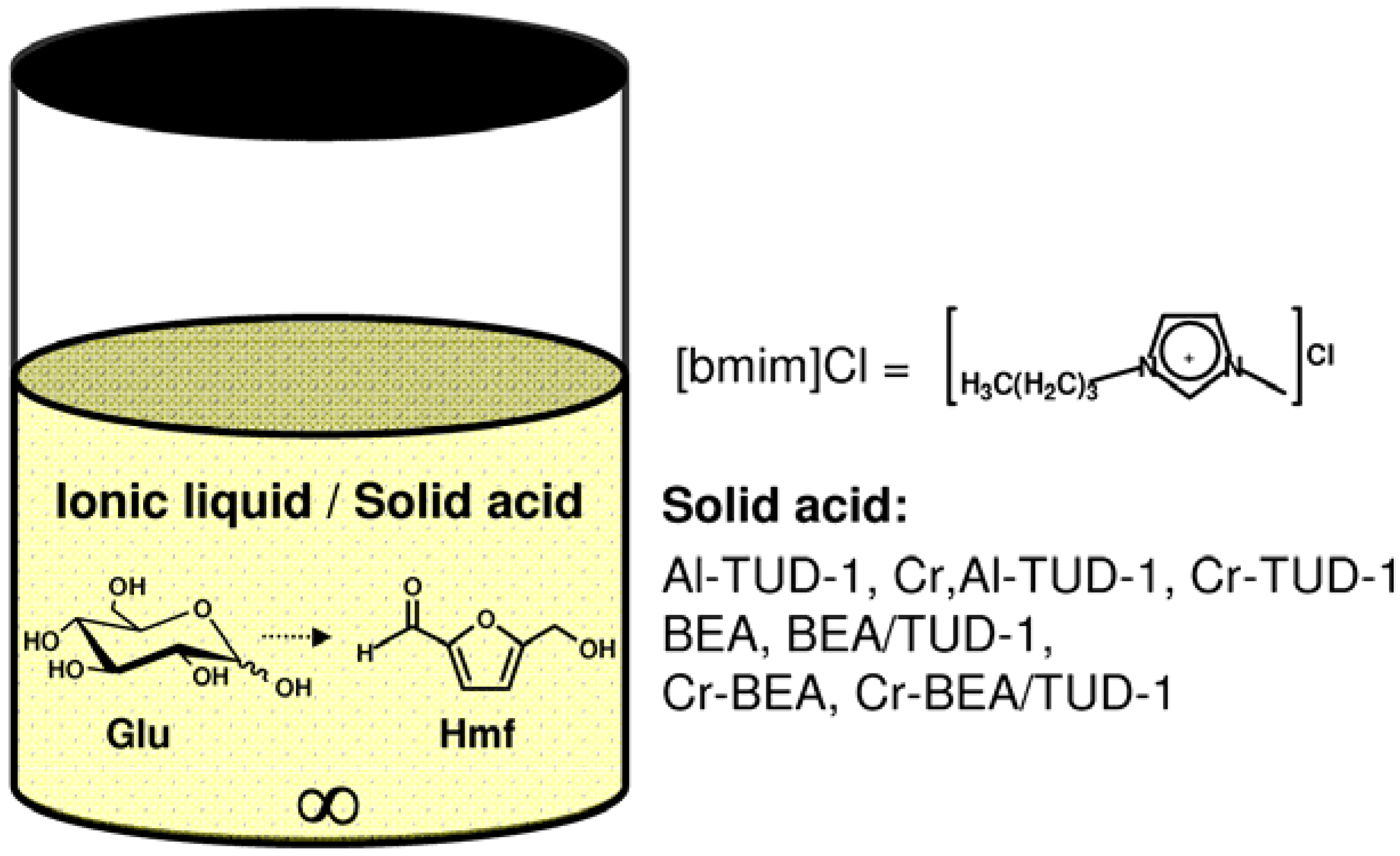
- (i) The washed and dried solid, referred to as recW(solid acid), was tested in the reaction of Glu, using fresh IL as solvent, at 120 °C for 3 h;
- (ii) The recW solid was calcined (450 °C, 3 h, heating rate of 1 °C·min−1) to give recC(solid acid), which was tested in the reaction of Glu, using fresh IL as solvent, at 120 °C for 3 h;
- (iii) The recovered IL, referred to as recIL(“name of solid”), was used as solvent in a 3 h-batch run of the reaction of Glu, at 120 °C, without adding a solid catalyst.

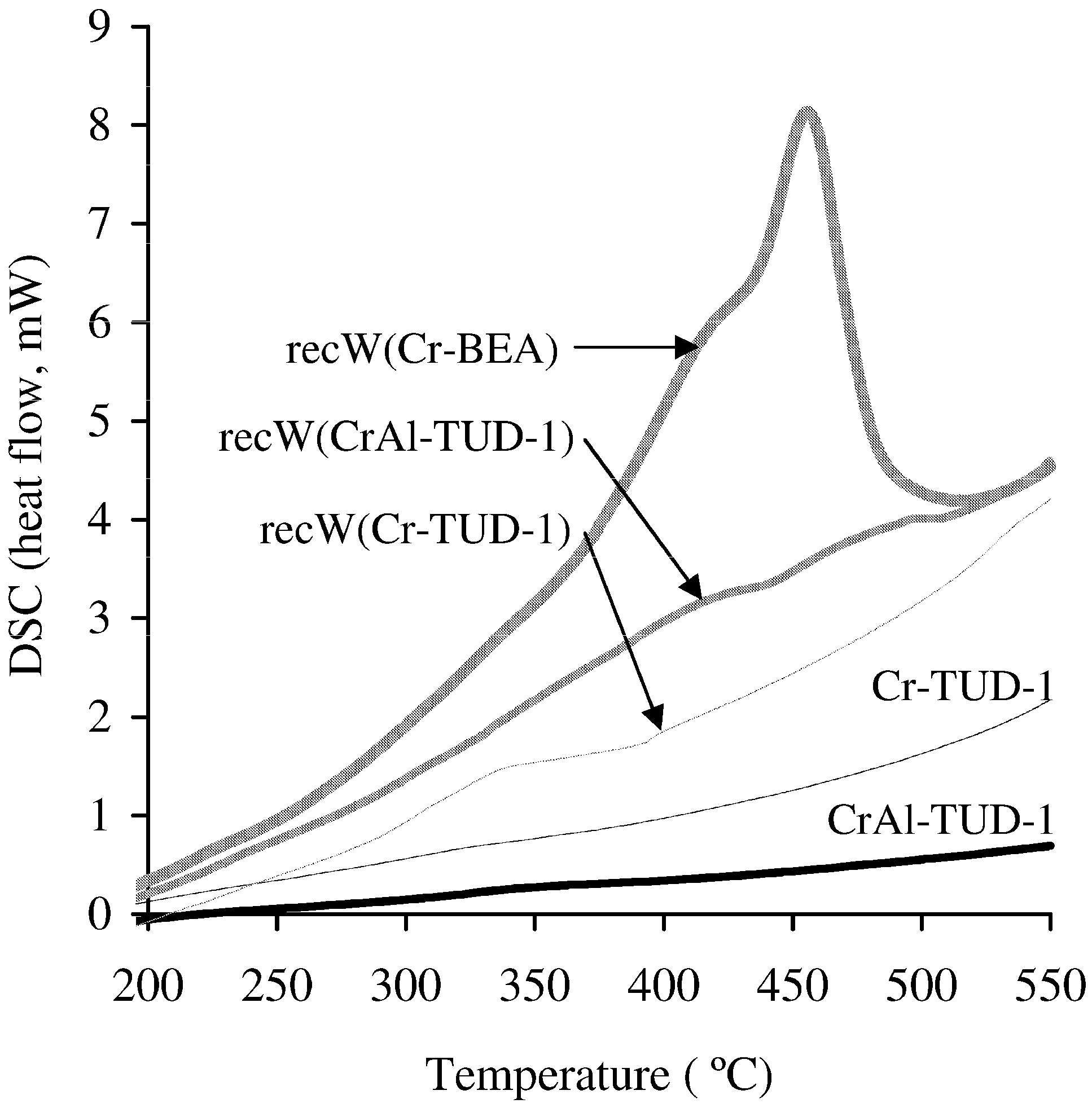
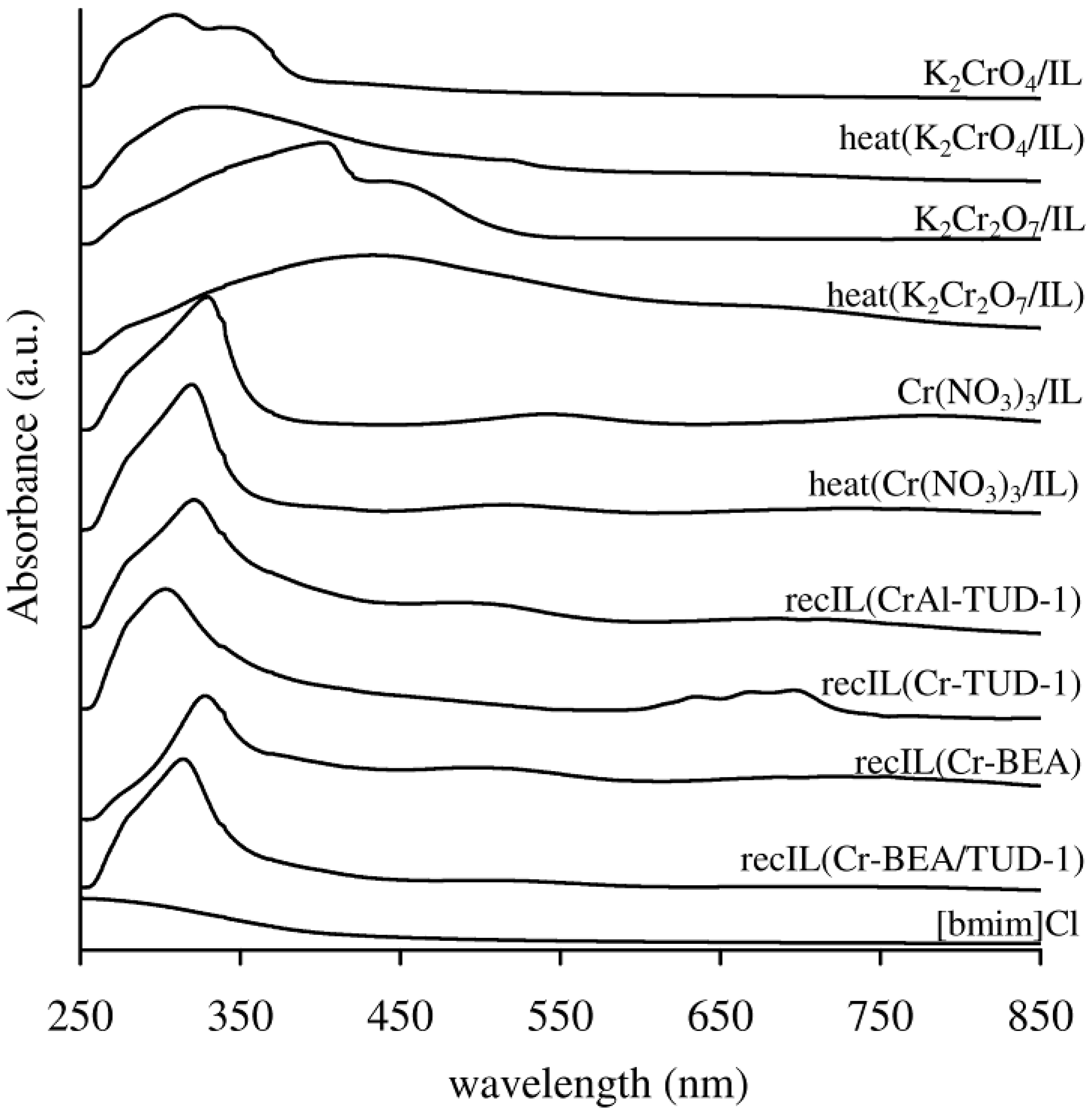
3. Experimental
3.1. General
3.2. Preparation of Al-TUD-1, CrAl-TUD-1 and Cr-TUD-1
3.3. Preparation of BEA, BEA//TUD-1, Cr-BEA and Cr-BEA/TUD-1
3.4. Catalytic Experiments
4. Conclusions
Acknowledgements
References and Notes
- Ragauskas, J.; Williams, C.K.; Davison, B.H.; Britovsek, G.; Cairney, J.; Eckert, C.A.; Frederick, W.J., Jr.; Hallett, J.P.; Leak, D.J.; Liotta, C.L.; et al. The path forward for biofuels and biomaterials. Science 2006, 311, 484–489. [Google Scholar]
- Sannigrahi, P.; Pu, Y.; Ragauskas, A. Cellulosic biorefineries-unleashing lignin opportunities. Curr. Opin. Environ. Sustain. 2010, 2, 383–393. [Google Scholar]
- Karinen, R.; Vilonen, K.; Niemel, M. Biorefining: Heterogeneously catalyzed reactions of carbohydrates for the production of furfural and hydroxymethyl furfural. ChemSusChem 2011, 4, 1002–1016. [Google Scholar]
- Zhao, H.; Holladay, J.E.; Brown, H.; Zhang, Z.C. Metal chlorides in ionic liquid solvents convert sugars to 5-hydroxymethylfurfural. Science 2007, 316, 1597–1600. [Google Scholar]
- Zhao, H.; Holladay, J.E.; Zhang, Z.C. Method for conversion of carbohydrates in ionic liquids to hydroxymethylfurfural. WO Pat. Appl. 2008, 019219A1. [Google Scholar]
- Zhao, H.; Holladay, J.E.; Zhang, Z.C. Methods for conversion of carbohydrates in ionic liquids to value-added chemicals. U.S. Patent 7,939,681 B2, 2011. [Google Scholar]
- Lima, S.; Antunes, M.M.; Pillinger, M.; Valente, A.A. Ionic liquids as tools for the acid-catalyzed hydrolysis/dehydration of saccharides to furanic aldehydes. ChemCatChem 2011, 3, 1686–1706. [Google Scholar]
- Zakrzewska, E.; Bogel-Lukasic, E.; Boguel-Lukasic, R. Ionic liquid-mediated formation of 5-hydroxymethylfurfural-A promising biomass-derived building block. Chem. Rev. 2011, 111, 397–417. [Google Scholar]
- Stählberg, T.; Fu, W.; Woodley, J.M.; Riisager, A. Synthesis of 5-(hydroxymethyl)furfural in ionic liquids: Paving the way to renewable chemicals. ChemSusChem 2011, 4, 451–458. [Google Scholar]
- Rasrendra, C.B.; Makertihartha, I.G.B.N.; Adisasmito, S.; Heeres, H.J. Green chemicals from D-glucose: Systematic studies on catalytic effects of inorganic salts on the chemoselectivity and yield in aqueous solutions. Top. Catal. 2010, 53, 1241–1247. [Google Scholar]
- Peng, L.; Lin, L.; Zhang, J.; Zhuang, J.; Zhang, B.; Gong, Y. Catalytic conversion of cellulose to levulinic acid by metal chlorides. Molecules 2010, 15, 5258–5272. [Google Scholar]
- Patil, S.K.R.; Lund, C.R.F. Formation and growth of humans via aldol addition and condensation during acid-catalyzed conversion of 5-hydroxymethylfurfural. Energ. Fuel. 2011, 25, 4745–4755. [Google Scholar]
- Swatloski, P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellulose with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar]
- Remsing, R.C.; Swatloski, R.P.; Rogers, R.D.; Moyan, G. Mechanism of cellulose dissolution in the ionic liquid 1-n-butyl-3-methylimidazolium chloride: A 13C and 35/37Cl-NMR relaxation study on model systems. Chem. Commun. 2006, 1271–1273. [Google Scholar]
- Liebert, T.; Heinze, T. Interaction of ionic liquids with polysaccharides 5. Solvents and reaction media for the modification of cellulose. Bioresources 2008, 3, 576–601. [Google Scholar]
- Pinkert, A.; Marsh, K.N.; Pang, S.; Staiger, M.P. Ionic liquids and their interaction with cellulose. Chem. Rev. 2009, 109, 6712–6728. [Google Scholar]
- Zakrzewska, E.; Bogel-Lukasik, E.; Bogel-Lukasik, R. Solubility of carbohydrates in ionic liquids. Energ. Fuel. 2010, 24, 737–745. [Google Scholar]
- Guo, J.; Zhang, D.; Liu, C. A theoretical investigation of the interactions between cellulose and 1-butyl-3-methylimidazolium chloride. J. Theor. Comput. Chem. 2010, 9, 611–624. [Google Scholar]
- Gupta, K.M.; Hu, Z.; Jiang, J. Mechanistic understanding of interactions between celulose and ionic liquids: A molecular simulation study. Polymer 2011, 52, 5904–5911. [Google Scholar]
- Imperato, G.; König, B.; Chiappe, C. Ionic green solvents from renewable resources. Eur. J. Org. Chem. 2007, 1049–1058. [Google Scholar]
- Tan, M.; Zhao, L.; Zhang, Y. Production of 5-hydroxymethyl furfural from cellulose in CrCl2/Zeolites/BmimCl system. Biomass Bioenerg. 2011, 35, 1367–1370. [Google Scholar]
- Francisco, M.; Mlinar, A.N.; Yoo, B.; Bell, A.T.; Prausnitz, J.M. Recovery of glucose from an aqueous ionic liquid by adsorption onto a zeolite-based solid. Chem. Eng. J. 2011, 172, 184–190. [Google Scholar]
- Zhang, Z.; Zhao, Z.K. Production of 5-hydroxymethylfurfural from glucose catalyzed by hydroxyapatite supported chromium chloride. Bioresource Technol. 2011, 102, 3970–3972. [Google Scholar]
- Gläser, R. Novel process options for the application of zeolites in supercritical fluids and ionic liquids. Chem. Eng. Technol. 2007, 30, 557–568. [Google Scholar]
- Zhang, Y.; Degirmenci, V.; Li, C.; Hensen, E.J.M. Phosphotungstic acid encapsulated in metal-organic framework as catalysts for carbohydrate dehydration to 5-hydroxymethylfurfural. ChemSusChem 2011, 4, 59–64. [Google Scholar]
- Rinaldi, R.; Meine, N.; vom Stein, J.; Palkovits, R.; Schüth, F. Which controls the depolymerization of cellulose in ionic liquids: The solid acid catalyst or cellulose? ChemSusChem 2010, 3, 266–276. [Google Scholar] [CrossRef]
- Telalović, S.; Ramanathan, A.; Mul, G.; Hanefeld, U. TUD-1: Synthesis and application of a versatile catalyst, carrier, material. J. Mater. Chem. 2010, 20, 642–658. [Google Scholar]
- Shan, Z.; Jansen, J.C.; Zhou, W.; Maschmeyer, Th. AlTUD-1, stable mesoporous aluminas with high surface areas. Appl. Catal. A Gen. 2003, 254, 339–343. [Google Scholar] [CrossRef]
- Anand, R.; Maheswari, R.; Hanefeld, U. Catalytic properties of the novel mesoporous aluminosilicate AlTUD-1. J. Catal. 2006, 242, 82–91. [Google Scholar]
- Hamdy, M.S.; Berg, O.; Jansen, J.C.; Maschemeyer, T.; Arafat, A.; Moulijn, J.A.; Mul, G. Chromium-incorporated TUD-1 as a new visible light-sensitive photo-catalyst for selective oxidation of propane. Catal. Today 2006, 117, 337–342. [Google Scholar]
- Zhang, Z.-X.; Bai, P.; Xu, B.; Yan, Z.F. Synthesis of mesoporous alumina TUD-1 with high thermostability. J. Porous Mater. 2006, 13, 245–250. [Google Scholar]
- Lima, S.; Antunes, M.M.; Fernandes, A.; Pillinger, M.; Ribeiro, M.F.; Valente, A.A. Acid-catalysed conversion of saccharides into furanic aldehydes in the presence of three-dimensional mesoporous Al-TUD-1. Molecules 2010, 15, 3863–3877. [Google Scholar]
- Telalović, S.; Hanefeld, U. Noncovalent immobilization of chiral cyclopropanation catalysts on mesoporous TUD-1: Comparison of liquid-phase and gas-phase ion-exchange. Appl. Catal. A Gen. 2010, 372, 217–223. [Google Scholar]
- Janses, J.C.; Shan, Z.; Marchese, L.; Zhou, W.; Puil, N.V.D.; Maschmeyer, Th. A new templating method for three-dimensional mesopore networks. Chem. Commun. 2001, 713–714. [Google Scholar]
- Sing, K.S.W. Characterization of Adsorbents. In Adsorption, Science and Technology; Nato ASI Series; Rodrigues, A.E., LeVan, M.D., Tondeur, D., Eds.; Kluwer Academic Publishers: Boston, MA, USA, 1989; pp. 3–14. [Google Scholar]
- Sangwichien, C.; Aranovich, G.L.; Donohue, M.D. Density functional theory predictions of adsorption isotherms with hysteresis loops. Colloids Surf. A 2002, 206, 313–320. [Google Scholar]
- Blin, J.L.; Léonard, A.; Su, B.L. Well-ordered spherical mesoporous materials CMI-1 synthesized via an assembly of decaoxyethylene cetyl ether and TMOS. Chem. Mater. 2001, 13, 3542–3553. [Google Scholar]
- Waller, P.; Shan, Z.; Marchese, L.; Tartaglione, G.; Zhou, W.; Jansen, J.C.; Maschmeyer, T. Zeolite nanocrystals inside mesoporous TUD-1: A high-performance catalytic composite. Chem. Eur. J. 2004, 10, 4970–4976. [Google Scholar]
- Lima, S.; Antunes, M.M.; Fernandes, A.; Pillinger, M.; Ribeiro, M.F.; Valente, A.A. Catalytic cyclodehydration of xylose to furfural in the presence of zeolite H-Beta and a micro/mesoporous Beta/TUD-1 composite material. Appl. Catal. A Gen. 2010, 388, 144–148. [Google Scholar]
- Weckhuysen, B.M.; Schoonheydt, R.A.; Jehng, J.-M.; Wachs, I.E.; Cho, S.J.; Ryoo, R.; Kijistra, S.; Poels, E. Combined DRS-RS-EXAFS-XANES-TPR Study of Supported Chromium Catalysts. J. Chem. Soc. Faraday Trans. 1995, 91, 3245–3253. [Google Scholar]
- Weckhuysen, B.M.; Wachs, I.E.; Schoonheydt, R.A. Surface chemistry and spectroscopy of chromium in inorganic oxides. Chem. Rev. 1996, 96, 3327–3349. [Google Scholar]
- Yuvaraj, S.; Palanichamy, M.; Krishnasamy, V. Chromium substitution in a large-pore high-silica zeolite BEA: Synthesis, characterisation and catalytic activity. Chem. Commun. 1996, 2707–2708. [Google Scholar]
- Dzwigaj, S.; Shishido, T. State of chromium in CrSiBEA zeolite prepared by the two-step postsynthesis method: XRD, FTIR, UV-Vis, EPR, TPR, and XAS studies. J. Phys. Chem. C 2008, 112, 5803–5809. [Google Scholar] [CrossRef]
- Binder, J.B.; Raines, R.T. Simple chemical transformation of lignocellulosic biomass into furans for fuels and chemicals. J. Am Chem. Soc. 2009, 131, 1979–1985. [Google Scholar]
- Lima, S.; Antunes, M.M.; Pillinger, M.; Ignatyev, N.; Valente, A.A. Conversion of mono/di/ polysaccharides into furan compounds using 1-alkyl-3-methylimidazolium ionic liquids. Appl. Catal. A Gen. 2009, 363, 93–99. [Google Scholar]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L., Jr. Fast transformation of glucose and di-polysaccharides into 5-hydroxymethylfurfural by microwave heating in an ionic liquid/catalyst system. ChemSusChem 2010, 3, 1071–1077. [Google Scholar]
- Pidko, E.A.; Degirmenci, V.; Santen, R.A.V.; Hensen, E.J.M. Glucose activation by transient Cr2+ dimers. Angew. Chem. Int. Ed. Engl. 2010, 49, 2530–2534. [Google Scholar]
- Sample Availability: Small quantities of the solid acid catalysts are available from the authors on request.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Antunes, M.M.; Lima, S.; Pillinger, M.; Valente, A.A. Coupling of Nanoporous Chromium, Aluminium-Containing Silicates with an Ionic Liquid for the Transformation of Glucose into 5-(Hydroxymethyl)-2-furaldehyde. Molecules 2012, 17, 3690-3707. https://doi.org/10.3390/molecules17043690
Antunes MM, Lima S, Pillinger M, Valente AA. Coupling of Nanoporous Chromium, Aluminium-Containing Silicates with an Ionic Liquid for the Transformation of Glucose into 5-(Hydroxymethyl)-2-furaldehyde. Molecules. 2012; 17(4):3690-3707. https://doi.org/10.3390/molecules17043690
Chicago/Turabian StyleAntunes, Margarida M., Sérgio Lima, Martyn Pillinger, and Anabela A. Valente. 2012. "Coupling of Nanoporous Chromium, Aluminium-Containing Silicates with an Ionic Liquid for the Transformation of Glucose into 5-(Hydroxymethyl)-2-furaldehyde" Molecules 17, no. 4: 3690-3707. https://doi.org/10.3390/molecules17043690
APA StyleAntunes, M. M., Lima, S., Pillinger, M., & Valente, A. A. (2012). Coupling of Nanoporous Chromium, Aluminium-Containing Silicates with an Ionic Liquid for the Transformation of Glucose into 5-(Hydroxymethyl)-2-furaldehyde. Molecules, 17(4), 3690-3707. https://doi.org/10.3390/molecules17043690