Antinociceptive and Anti-inflammatory Effects of a Lectin-Like Substance from Clitoria fairchildiana R. Howard Seeds
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification and Chemical Characterization
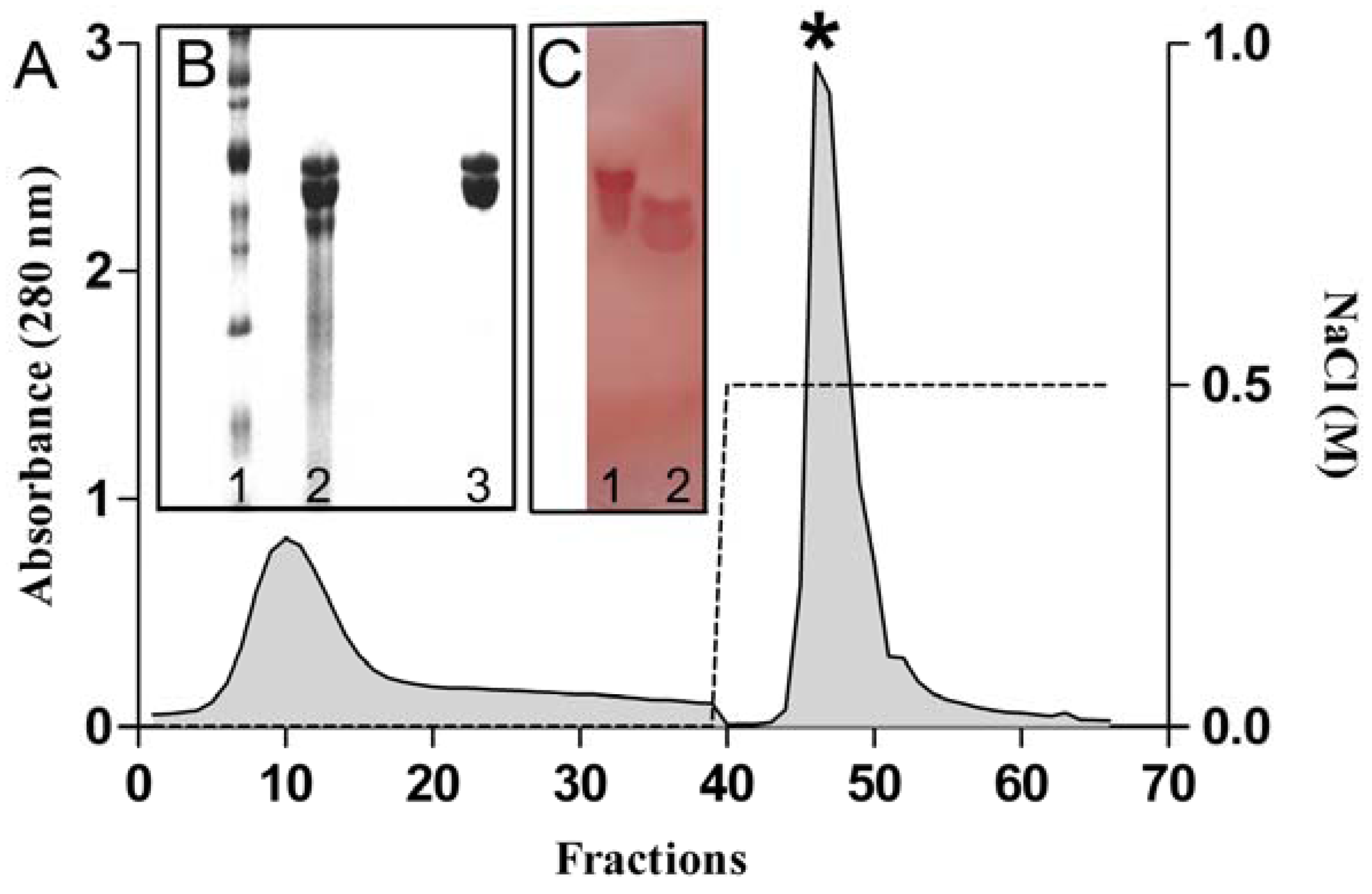
2.2. Cytotoxic Activity against Human Erythrocytes
2.3. Antinociceptive Activity
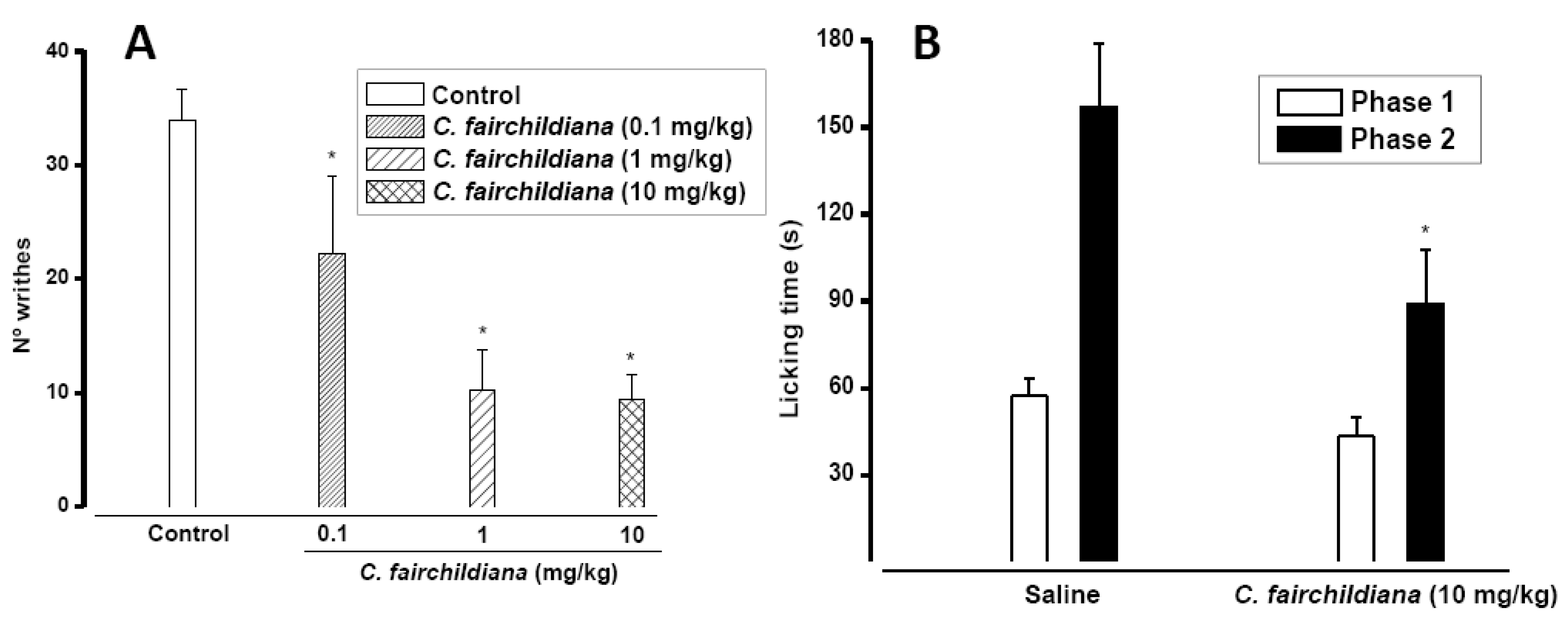
2.4. Anti-inflammatory Activity
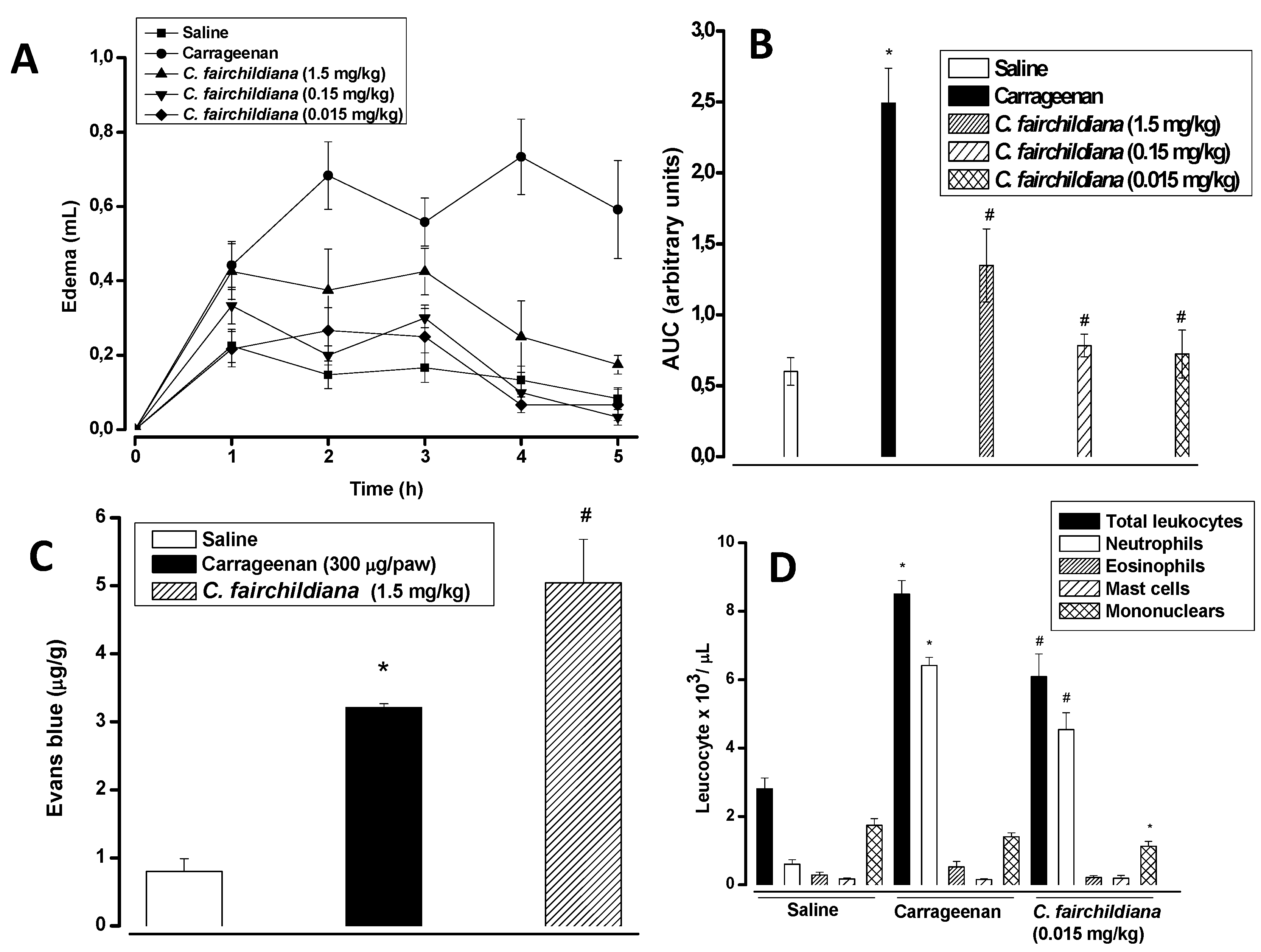
3. Experimental
3.1. Plant Material
3.2. Animals
3.3. Erythrocytes
3.4. Purification of C. fairchildiana Lectin
3.6. Hemagglutinating Activity
3.7. Sugar Specificity
3.8. Electrophoresis Polyacrylamide Gel-SDS (SDS-PAGE)
3.9. Action of Sodium Metaperiodate Oxidizing Agent
3.10. Nociceptive Models in Mice
3.10.1. Writing Test
3.10.2. Formalin Test
3.11. Inflammatory Models in Rats
3.11.1. Paw Edema
3.11.2. Peritonitis
3.12. Assessment of the Potential Cytotoxicity Assay of Lectin from C. fairchildiana in Human Erythrocytes
3.13. Statistical Analysis
4. Conclusions
Conflict of Interest
Acknowledgements
References and Notes
- Beuth, J.; Ko, H.L.; Pulver, G.; Uhlenbruck, G. Importance of lectins for the prevention of bacterial infections and cancer metastases. Glycoconjugate 1995, 12, 1–6. [Google Scholar] [CrossRef]
- Peumans, W.J.; Van Damme, E.J. Lectins as plant defense proteins. Plant Physiol. 1995, 109, 347–352. [Google Scholar]
- Rangel, T.B.A.; Assreuy, A.M.S.; Pires, A.F.; Carvalho, A.U.; Benevides, R.G.; Simões, R.C.; Silva, H.C.; Bezerra, M.J.B.; Nascimento, A.S.F.; Nascimento, K.S.; et al. Crystallization and characterization of an inflammatory lectin purified from the seeds of Dioclea wilsonii. Molecules 2011, 16, 5087–5103. [Google Scholar]
- Oliveira, T.M.; Delatorre, P.; Rocha, B.A.M.; Souza, E.P.; Nascimento, K.S.; Bezerra, G.A.; Moura, T.R.; Benevides, R.G.; Bezerra, E.H.S.; Moreno, F.B.M.B.; et al. Crystal structure of Dioclea rostrata: Insights into understanding the pH-dependent dimer-tetramer equilibrium and thestructural basis for carbohydrate recognition in Diocleinae lectins. J. Struct. Biol. 2008, 164, 177–182. [Google Scholar] [CrossRef]
- Bento, C.A.; Cavada, B.S.; Oliveira, J.T.; Moreira, R.A.; Barja-Fidalgo, C. Rat paw edema and leukocyte immigration induced by plant lectins. Agents Actions 1993, 38, 48–54. [Google Scholar] [CrossRef]
- Rodriguez, D.; Cavada, B.S.; Abreu-de-Oliveira, J.T.; de-Azevedo-Moreira, R.; Russo, M. Differences in macrophage stimulation and leukocyte accumulation in response to intraperitoneal administration of glucose/mannose-binding plant lectins. Braz. J. Med. Biol. Res. 1992, 25, 823–826. [Google Scholar]
- Gomes, J.C.; Ferreira, R.R.; Cavada, B.S.; Moreira, R.A.; Oliveira, J.T. Histamine release induced by glucose (mannose) specific lectins isolated from Brazilian beans. Comparison with concanavalin A. Agents Actions 1994, 41, 132–135. [Google Scholar] [CrossRef]
- Brabosa, T.; Arruda, S.; Cavada, B.S.; Grangeiro, T.B.; Freitas, L.A.; Barral-Netto, M. In vivo lymphocyte activation and apoptosis by lectins of the Diocleinae subtribe. Mem. Inst. Oswaldo Cruz 2001, 96, 673–678. [Google Scholar] [CrossRef]
- Andrade, J.L.; Arruda, S.; Barbosa, T.; Paim, L.; Ramos, M.V.; Cavada, B.S.; Barral-Neto, M. Lectin-induced nitric oxide production. Cell Immunol. 1999, 194, 98–102. [Google Scholar] [CrossRef]
- Barral-Netto, M.; Santos, S.B.; Barral, A.; Moreira, L.I.; Santos, C.F.; Moreira, R.A. Human lymphocyte stimulation by legume lectins from the Diocleae tribe. Immunol. Invest. 1992, 21, 297–303. [Google Scholar] [CrossRef]
- Alencar, N.M.; Assereuy, A.M.; Alencar, V.B.; Melo, S.C.; Ramos, M.V.; Cavada, B.S.; Cunha, F.Q.; Ribeiro, R.A. The galactose-binding lectin from Vatairea macrocarpa seeds induces in vivo neutrophil migration by indirect mechanism. Int. J. Biochem. Cell Biol. 2003, 35, 1674–1674. [Google Scholar] [CrossRef]
- Alencar, N.M.; Assreuy, A.M.; Criddle, D.N.; Souza, E.P.; Soares, P.M.; Havt, A.; Aragão, K.S. Bezerra, D.P.; Ribeiro, R.A.; Cavada, B.S. Vatairea macrocarpa lectin induces paw edema with leukocyte infiltration. Protein Pept. Lett. 2004, 11, 195–200. [Google Scholar] [CrossRef]
- Alencar, N.M.; Assreuy, A.M.; Havt, A.; Benevides, R.G.; Moura, T.R.; Sousa, R.B.; Ribeiro, R.A.; Cunha, F.Q.; Cavada, B.S. Vatairea macrocarpa (Leguminosae) lectin activates cultured macrophages to release chemotactic mediators. Naunyn Schmiedebergs Arch. Pharmacol. 2007, 374, 275–282. [Google Scholar] [CrossRef]
- Assreuy, A.M.; Shibuya, M.D.; Martins, G.J.; Souza, M.L.; Cavada, B.S.; Moreira, R.A.; Oliveira, J.T.A.; Ribeiro, R.A.; Flores, C.A. Anti-inflammatory effect of glucose-mannose binding lectins isolated from Brazilian beans. Mediators Inflamm. 1997, 6, 201–210. [Google Scholar] [CrossRef]
- Assreuy, A.M.; Martins, G.J.; Moreira, M.E.; Brito, G.A.; Cavada, B.S.; Ribeiro, R.A.; Flores, C.A. Prevention of cyclophosphamide-induced hemorrhagic cystitis by glucose-mannose binding plant lectins. J. Urol. 1999, 161, 1988–1993. [Google Scholar] [CrossRef]
- Alencar, N.M.; Teixeira, E.H.; Assreuy, A.M.; Cavada, B.S.; Flores, C.A.; Ribeiro, R.A. Leguminous lectins as tools for studying the role of sugar residues in leukocyte recruitment. Mediators Inflamm. 1999, 8, 107–113. [Google Scholar] [CrossRef]
- Alencar, N.M.; Cavalcante, C.F.; Vasconcelos, M.P.; Leite, K.B.; Aragão, K.S.; Assreuy, A.M.; Nogueira, N.A.; Cavada, B.S.; Vale, M.R. Anti-inflammatory and antimicrobial effect of lectin from Lonchocarpus sericeus seeds in an experimental rat model of infectious peritonitis. J. Pharm. Pharmacol. 2005, 57, 919–922. [Google Scholar]
- Napimoga, M.H.; Cavada, B.S.; Alencar, N.M.; Mota, M.L.; Bittencourt, F.S.; Alves-Filho, J.C.; Grespan, R.; Gonçalves, R.B.; Clemente-Napimoga, J.T.; Freitas, A.; et al. Lonchocarpus sericeus lectin decreases leukocyte migration and mechanical hypernociception by inhibiting cytokine and chemokines production. Int. Immunopharmacol. 2007, 7, 824–835. [Google Scholar]
- Figueiredo, J.G.; Bitencourt, F.S.; Beserra, I.G.; Teixeira, C.S.; Luz, P.B.; Bezerra, E.H.; Mota, M.R.; Assreuy, A.M.; Cunha, F.Q.; Cavada, B.S.; et al. Antinociceptive activity and toxicology of the lectin from Canavalia boliviana seeds in mice. Naunyn. Schmiedebergs Arch. Pharmacol. 2009, 380, 407–414. [Google Scholar] [CrossRef]
- Pires, A.F.; Assreuy, A.M.; Lopes, E.A.; Celedônio, N.R.; Soares, C.E.; Rodrigues, N.V.; Sousa, P.L.; Benevides, R.G.; Nagano, C.S.; Cavada, B.S.; et al. Opioid-like antinociceptive effects of oral administration of a lectin purified from the seeds of Canavalia brasiliensis. Fundam. Clin. Pharmacol. 2011. x. [Google Scholar] [CrossRef]
- Naeem, A.; Haque, S.R.; Khan, H. Purification and characterization of a novel β-D-galactosides- specific lectin from Clitoria ternatea. Protein J. 2007, 26, 403–413. [Google Scholar] [CrossRef]
- Singer, S.J. Some early history of membrane. Annu. Rev. Physiol. 2004, 66, 1–27. [Google Scholar] [CrossRef]
- Gabius, H.J.; André, S.; Jiménez-Barbero, J.; Romero, A.; Solís, D. From lectin structure to functional glycomics: Principles of the sugar code. Trends Biochem. Sci. 2011, 36, 298–313. [Google Scholar] [CrossRef]
- Oliveira, M.D.L.; Andrade, C.A.S.; Santos-Magalhães, N.S.; Coelho, L.C.B.B.; Teixeira, J.A.; Cunha, M.G.C.; Correia, M.T.S. Purification of a lectin from Eugenia uniflora L. seeds and its potential antibacterial activity. Lett. Appl. Microbiol. 2008, 46, 371–376. [Google Scholar] [CrossRef]
- Guzman-Partida, A.M.; Robles-Burgueno, M.R.; Ortega-Nieblas, M.; Vazquez-Moreno, I. Purification and characterization of complex carbohydrate specific isolectins from wild legume seeds: Acacia constricta is (vinorama) highly homologous to Phaseolus vulgaris lectins. Biochimie 2004, 86, 335–342. [Google Scholar] [CrossRef]
- Boleti, A.P.A.; Ventura, C.A.; Justo, G.Z.; Silva, R.A.; Sousa, A.C.T.; Ferreira, C.V.; Yano, T.; Macedo, M.L. Pouterin, a novel potential cytotoxic lectin-like protein with apoptosis-inducing activity in tumorigenic mammalian cells. Toxicon 2008, 51, 1321–1330. [Google Scholar] [CrossRef]
- Barbosa, P.S.F.; Martins, A.M.C.; Toyama, M.H.; Joazeiro, P.P.; Beriam, L.O.S.; Fonteles, M.C.; Monteiro, H.A.S. Purification and biological effects of a C-type lectin isolated from Bothrops moojeni. J. Venom. Anim. Toxins 2010, 16, 493–504. [Google Scholar] [CrossRef]
- Santi-Gadelha, T.; Rocha, B.A.M.; Oliveira, C.C.; Aragão, K.S.; Marinho, E.S.; Gadelha, C.A.A.; Toyama, M.H.; Pinto, V.P.T.; Nagano, C.S.; Delatorre, P.; et al. Purification of a PHA-like chitin binding lectin from Acacia farnesiana seeds: A time-dependent oligomerization protein. Appl. Biochem. Biotechnol. 2008, 150, 97–111. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebersand, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar]
- Souza, J.D.; Silva, M.B.R.; Argolo, A.C.C.; Napoleão, T.H.; Sá, R.A.; Correia, M.T.S.; Paiva, P.M.G.; Silva, M.D.C.; Coelho, L.C.B.B. A new Bauhinia monandra galactose-specific lectin purified in milligram quantities from secondary roots with antifungal and termiticidal activities. Int. Biodeter. Biodegr. 2011, 65, 696–702. [Google Scholar] [CrossRef]
- Oliveira, T.J.; Melo, V.M.; Câmara, M.F.; Vasconcelos, I.M.; Beltramini, L.M.; Machado, O.L. Purification and physicochemical characterization of a cotyledonary lectin from Luetzelburgia auriculata. Phytochemistry 2002, 61, 301–310. [Google Scholar]
- Ferreira, A.P.; Corrêa, T.; Cunha, R.; Marques, M.J.; Montesano, M.A.; Souza, M.A.; Teixeira, H.C. Human serum antibody reactivity towards Paracoccidioides brasiliensis antigens treated with sodium metaperiodate. Rev. Soc. Bras. Med. Trop. 2008, 41, 325–329. [Google Scholar] [CrossRef]
- Dresch, R.R.; Haeser, A.S.; Lerner, C.; Mothes, B.; Vozári-Hampe, M.M.; Henriques, A.T. Detection of lectinic activity and hemolytic activity in extracts of native sponges (Porifera) of atlantic coast of Brazil. Rev. Bras. Farmacogn. 2005, 15, 16–22. [Google Scholar] [CrossRef]
- Prokof’eva, N.G.; Utkina, N.K.; Chaikina, E.L.; Makarchenko, A.E. Biological activities of marine sesquiterpenoid quinones: Structure-activity relationships in cytotoxic and hemolytic assays. Comp. Biochem. Phys. B 2004, 139, 169–173. [Google Scholar]
- Hatakeyama, T.; Kohzaki, H.; Nagatomo, H.; Yamasaki, N. Purification and characterization of four Ca2+-dependent lectins from the marine invertebrate Cucumaria echinata. J Biochem. 1994, 116, 209–214. [Google Scholar]
- Kuramoto, T.; Uzuyama, H.; Hatakeyama, T.; Tamura, T.; Nakashima, T.; Yamaguchi, K.; Oda, T. Cytotoxicity of a GalNAc-specific C-type lectin CEL-I toward various cell lines. J. Biochem. 2005, 137, 41–50. [Google Scholar] [CrossRef]
- Stai, H.Y.; Chen, Y.F.; Wu, T.S. Anti-inflammatory and analgesic activities of extract from roots of Angelica pubescens. Planta Med. 1995, 61, 1–8. [Google Scholar] [CrossRef]
- Schowb, M.; Dubost, M.C. Entendre La Douleur; Pharmapost: Rc Montargis, France, 1984; p. 99. [Google Scholar]
- Garcia, M.D.; Fernandez, M.A.; Alvarez, A.; Saenz, M.T. Antinociceptive and anti-inflammatory effect of the aqueous extract from leaves of Pimenta racemosa var. ozua (Mirtaceae). J. Ethnopharmacol. 2004, 91, 29–73. [Google Scholar]
- Tjolsen, A.; Berge, O.G.; Hunskaar, S.; Rosland, J.H.; Hole, K. The formalin test: An evaluation of the method. Pain 1992, 51, 5–17. [Google Scholar] [CrossRef]
- Tjolsen, A.; Hole, K. Animal models of analgesia. In The Pharmacology of Pain; Besson, M.J., Deckson, A., Eds.; Springer-Verlag: Berlin, Germany, 1997; pp. 21–41. [Google Scholar]
- Malmberg, A.B.; Yaksh, T.L. Cyclooxygenase inhibition and the spinal release of prostaglandin E2 and amino acids evoked by paw formalin injection: A microdialysis study in unanesthetized rats. J. Neurosci. 1995, 15, 2768–2776. [Google Scholar]
- Omote, K.; Kawamata, T.; Kawamata, M.; Namiki, A. Formalin-induced nociception activates a monoaminergic descending inhibitory system. Brain Res. 1998, 814, 194–198. [Google Scholar] [CrossRef]
- Santos, A.R.; De Campos, R.O.; Miguel, O.G.; Cechinel-Filho, V.; Yunes, R.A.; Calixto, J.B. The involvement of K+ channels and Gi/o protein in the antinociceptive action of the gallic acid ethyl ester. Eur. J. Pharmacol. 1999, 379, 7–17. [Google Scholar] [CrossRef]
- Vanegas, H.; Schaible, H.G. Prostaglandins and cyclooxygenases [correction of cycloxygenases] in the spinal cord. Prog. Neurobiol. 2001, 64, 327–363. [Google Scholar] [CrossRef]
- Gaertner, M.; Müller, L.; Roos, J.F.; Cani, G.; Santos, A.R.; Niero, R.; Calixto, J.B.; Yunes, R.A.; Delle Monache, F.; Cechinel-Filho, V. Analgesic triterpenes from Sebastianiaschottiana roots. Phytomedicine 1999, 6, 41–44. [Google Scholar] [CrossRef]
- Hunskaar, S.; Hole, K. The formalin test in mice: Dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar] [CrossRef]
- Shibata, M.; Ohkubo, T.; Takahashi, H.; Inoki, R. Modified formalin test: Characteristic biphasic pain response. Pain 1989, 38, 347–352. [Google Scholar] [CrossRef]
- Kulinsky, V.I. Biochemical aspects of inflammation. Biochemistry 2007, 72, 733–746. [Google Scholar]
- Vinegar, R.; Schreiber, W.; Hugo, R. Biphasic development of carrageenan edema in rats. J. Pharmacol. 1969, 166, 95–103. [Google Scholar]
- Souza, G.E.; Cunha, F.Q.; Mello, R.; Ferreira, S.H. Neutrophil migration induced by inflammatory stimuli is reduced by macrophage depletion. Agents Actions 1988, 24, 377–380. [Google Scholar] [CrossRef]
- Levine, J.D.; Lau, W.; Kwiat, G.; Goetzl, E.J. Leukotriene B4 produces hyperalgesia that is dependent on polymorphonuclear leukocytes. Science 1984, 225, 743–745. [Google Scholar]
- Verri, W.A., Jr.; Schivo, I.R.; Cunha, T.M.; Liew, F.Y.; Ferreira, S.H.; Cunha, F.Q. Interleukin-18 induces mechanical hypernociception in rats via endothelin acting on ETB receptors in a morphine-sensitive manner. J. Pharmacol. Exp. Ther. 2004, 310, 710–717. [Google Scholar] [CrossRef]
- Santodomingo-Garzón, T.; Cunha, T.M.; Verri, W.A., Jr.; Valério, D.A.; Parada, C.A.; Poole, S.; Ferreira, S.H.; Cunha, F.Q. Atorvastatin inhibits inflammatory hypernociception. Br. J. Pharmacol. 2006, 149, 14–22. [Google Scholar] [CrossRef]
- Bitencourt, F.S.; Figueiredo, J.G.; Mota, M.R.L.; Bezerra, C.C.R.; Silvestre, P.P.; Vale, M.R.; Nascimento, K.S.; Sampaio, A.H.; Nagano, C.S.; Saker-Sampaio, S.; et al. Antinociceptive and anti-inflammatory effects of a mucin-binding agglutinin isolated from the red marine alga Hypnea cervicornis. Naunyn. Schmiedebergs Arch. Pharmacol. 2008, 377, 139–148. [Google Scholar] [CrossRef]
- Bradford, M.M.A. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principles of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the bacteriophage t4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Kapitany, R.A.; Zebrowski, E.J. A high resolution PAS stain for polyacrylamide gel electrophoresis. Anal. Biochem. 1973, 56, 361–369. [Google Scholar] [CrossRef]
- Koster, R.; Anderson, M.; Debeer, E.J. Acetic-acid for analgesil screening. Fed. Proc. 1959, 18, 412. [Google Scholar]
- Landucci, E.C.; Antunes, E.; Donato, J.L.; Faro, R.; Hyslop, S.; Marangoni, S.; Oliveira, B.; Cirino, G.; de Nucci, G. Inhibition of carrageenin-induced rat paw edema by crotapotin, a polypeptide complexed with phospholipase A2. Br. J. Pharmacol. 1995, 114, 578–583. [Google Scholar] [CrossRef]
- Wilhelm, D.L. The mediation of increased vascular permeability in inflammation. Pharmacol. Rev. 1962, 14, 251–280. [Google Scholar]
- Souza, G.E.; Ferreira, S.H. Blockade by antimacrophage serum of the migration of PMN neutrophils into the inflamed peritoneal cavity. Agents Actions 1985, 17, 97–103. [Google Scholar] [CrossRef]
- Rangel, M.; Malpezzi, E.L.A.; Susini, S.M.M.; Freitas, J.C. Hemolytic activity in extracts of the diatom Nitzsnhia. Toxicon 1997, 35, 305–309. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Leite, J.F.M.; Assreuy, A.M.S.; Mota, M.R.L.; Bringel, P.H.d.S.F.; E Lacerda, R.R.; Gomes, V.d.M.; Cajazeiras, J.B.; Do Nascimento, K.S.; Pessôa, H.d.L.F.; Gadelha, C.A.d.A.; et al. Antinociceptive and Anti-inflammatory Effects of a Lectin-Like Substance from Clitoria fairchildiana R. Howard Seeds. Molecules 2012, 17, 3277-3290. https://doi.org/10.3390/molecules17033277
Leite JFM, Assreuy AMS, Mota MRL, Bringel PHdSF, E Lacerda RR, Gomes VdM, Cajazeiras JB, Do Nascimento KS, Pessôa HdLF, Gadelha CAdA, et al. Antinociceptive and Anti-inflammatory Effects of a Lectin-Like Substance from Clitoria fairchildiana R. Howard Seeds. Molecules. 2012; 17(3):3277-3290. https://doi.org/10.3390/molecules17033277
Chicago/Turabian StyleLeite, Joana Filomena Magalhães, Ana Maria Sampaio Assreuy, Mário Rogério Lima Mota, Pedro Henrique de Souza Ferreira Bringel, Rodrigo Rodrigues E Lacerda, Vinícius de Morais Gomes, João Batista Cajazeiras, Kyria Santiago Do Nascimento, Hilzeth de Luna Freire Pessôa, Carlos Alberto de Almeida Gadelha, and et al. 2012. "Antinociceptive and Anti-inflammatory Effects of a Lectin-Like Substance from Clitoria fairchildiana R. Howard Seeds" Molecules 17, no. 3: 3277-3290. https://doi.org/10.3390/molecules17033277
APA StyleLeite, J. F. M., Assreuy, A. M. S., Mota, M. R. L., Bringel, P. H. d. S. F., E Lacerda, R. R., Gomes, V. d. M., Cajazeiras, J. B., Do Nascimento, K. S., Pessôa, H. d. L. F., Gadelha, C. A. d. A., Delatorre, P., Cavada, B. S., & Santi-Gadelha, T. (2012). Antinociceptive and Anti-inflammatory Effects of a Lectin-Like Substance from Clitoria fairchildiana R. Howard Seeds. Molecules, 17(3), 3277-3290. https://doi.org/10.3390/molecules17033277




