Cancer Chemoprevention by Carotenoids
Abstract
:Abbreviations
| ABCA1 | ATP-binding cassette transporter 1 |
| AFB1 | aflatoxin B1 |
| Akt | protein kinase B |
| AMD | age-related macular degeneration |
| AOM | azoxymethane |
| AP-1 | activator 1 |
| ARE | antioxidant response element |
| CAR | constitutive androstane receptor |
| Cdks | cyclin-dependent kinases |
| CHRP | β-cryptoxanthin- and hesperidin-rich powder |
| CMO-1 | β-carotene 15,15'-monooxygenase |
| COM2 | β-carotene 9',10'-monooxygenase |
| COX | cyclooxygenase |
| CUSM | citrus unshiu segment membrane |
| CVD | cardiovascular disease |
| CYP | cytochrome P450 |
| DMH | 1,2-dimethylhydrazine |
| EGF | early growth response gene |
| ERK | extracellular signal-regulated kinase |
| GJIC | gap junctional intercellular communication |
| GSK3β | glycogen synthase kinase 3β |
| GSTs | glutathione S-transferases |
| HDL | high-density lipoproteins |
| HO-1 | heme oxygenase-1 |
| IGF | insulin growth factor |
| IGFBPs | IGF binding proteins |
| IL | interleukin |
| LDL | low-density lipoproteins |
| MJ | satsuma mandarin (Citrus unshiu Marc) juice |
| MMP | matrix metalloproteinases |
| NF-kB | nuclear factor kappaB |
| 4-NQO | 4-nitroquinoline 1-oxide |
| NQO1 | NAD(P)H:quinone oxidoreductase |
| Nrf2 | NF-E2-related factor 2 |
| OH-BBN | N-butyl-N(4-hydroxybutyl)nitrosamine |
| PPARs | peroxisome proliferator-activated receptors |
| PSA | prostate-specific antigen |
| RAR | retinoic acid receptor |
| ROS | reactive oxygen species |
| RXR | retinoid X receptor |
| SXR/PXR | steroid and xenobiotic receptor/pregnane X receptor |
| TCF/LEF | transcription factors T cell factor/lymphoid enhancer factor |
| TNF | tumor necrosis factor |
| TRE | TPA response element |
| UV | ultraviolet |
| VDR | vitamin D3 receptor |
1. Introduction
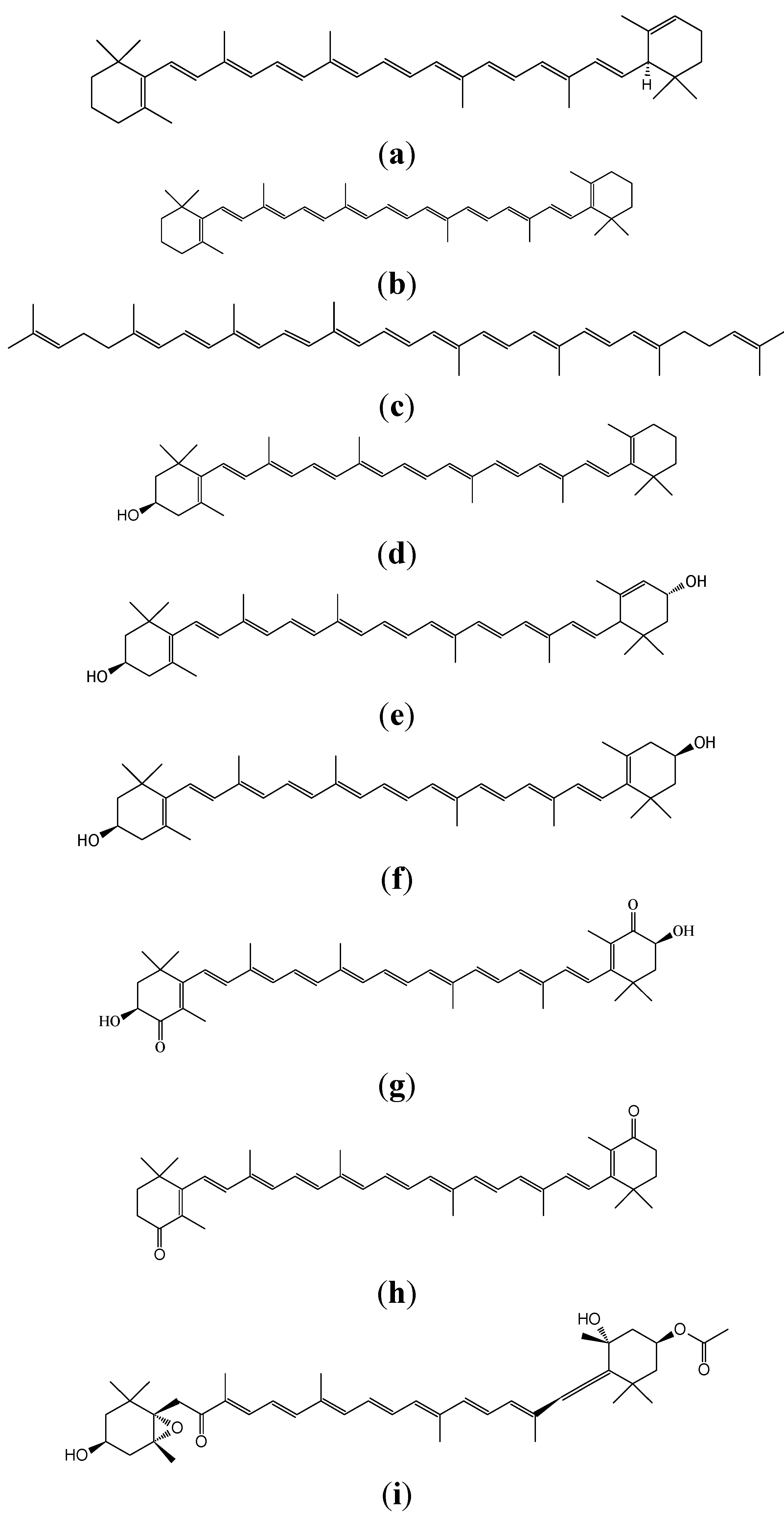
| Carotenoids | Dietary Sources | Function | Effects |
|---|---|---|---|
| α-Carotene | Yellow-orange vegetables (carrots, sweet totatoes, pumpkin) and Dark-green vegetables (broccoli, green beans, spinach) | Provitamin A activity; Anti-oxidant | Immune- enhancement; Stimulate cell to cell communication; Decreases risk of some cancers |
| β-Carotene | Green leafy vegetables and orange and yellow fruits and vegetables (carrots, apricots, spinach, sweet potetoes, pumpkin, pepper, kale, cantaloupe) | Provitamin A activity; Antioxidant | Immune-enhancement; Decreases risk of some cancers and some cardiovascular events; high-dose supplementation may increase the risk of lung cancer among smokers |
| Lycopene | Tomatoes, water melon, apricot, peaches | Anti-oxidant | Decreases risk of some cancers and some cardiovascular events, diabetes, and osteoporosis |
| β-Cyptoxanthin | Orange fruits (mandarin orange and papaya, etc.), corn, peas, and egg yolks | Provitamin A activity; Anti-oxidant | Anti-inflammatory effects; Inhibits risks of some cancer and cardiovascular events; Immune enhancement |
| Lutein/Zeaxanthin | Dark green leafy vegetables (spinach, kale), red peppers, maize, tomatoes, corn, and egg yolks | Anti-photosensitizing agent and photosynthetic pigment; Acts as antioxidants and blue light filters | Decrease age-related macular degeneration, cataract, and risk of cardiovascular disease and certain cancers |
| Astaxanthin | Green algae, salmon, trout, crustacea | Antioxidant; Coloration | Prevent certain cancers, cataract, diabetes, and inflammatory neurodegenerative and cardiovascular diseases |
| Canthaxanthin | Salmon, crustacea | Antioxidant; Coloration | Immune enhancement; Decreases risk of some cancers |
| Focoxanthin | Brown algae, heterokonts | Antioxidant | Anti-cancer, anti-allergic, anti-obese, anti-inflammatory, and anti-osteoporotic activities |
2. Definition of Carotenoids
3. Absorption, Metabolism, and Bioavailability of Carotenes and Xanthophylls
4. Distribution and Nature of Certain Carotenoids
4.1. Hydrocarbone Carotenoids
4.2. Lycopene
4.3. Lutein and Zeaxanthin
4.4. β-Cryptoxanthin
4.5. Astaxanthin
4.6. Canthaxanthin
4.7. Fucoxanthin
5. Clinical Trials with Long-Term β-Carotene Supplementation
| Studies | Study Designs | Ref. No. | |||
|---|---|---|---|---|---|
| Population | Intervention | Duration | Cancer outcome | ||
| ATBC | 29,133 Finish male smokers (50–69 years of age) | β-carotene, 20 mg/day; vitamin E, 50 mg/day | 5–8 years | 18% increase in lung cancer; 8% increase in mortality | 13 |
| CARET | 18,314 men and women and asbestoss workers (45–74 years of age) | β-carotene, 30 mg/day; vitamin A, 25,000 IU | <4 years | 28% increase in lung cancer; 17% increase in deaths | 15 |
| PHS | 22,071 male physicians (40–84 years of age) | β-carotene, 50 mg on alternate days | 12 years | No effect of supplementation in incidence of cancer | 14 |
| Linxian | 29,584 men and women, vitamin and mineral deficient (40–69 years of age) | β-carotene, 15 mg/day; selenium, 50 mg/day; α-tocopherol, 30 mg/day | 5 years | 13% decrease in total cancers; 9% decrease in overall deaths | 84 |
| Women’s Health Study | 39,876 female health professionals (over 45 years of age) | β-carotene, 50 mg on alternate days | 4.1 years (2.1 years’ treatment and 2.0 years’ follow-up) | No effect of supplementation in incidence of cancer | 87 |
6. Cancer Chemoprevention by Carotenoids in Preclinical Studies
6.1. α- and β-Carotene
6.2. β-Cryptoxanthin
6.3. Lycopene
6.4. Lutein and Zeaxanthin
6.5. Astaxanthin
6.6. Canthaxanthin
6.7. Fucoxanthin
7. Mechanisms of Cancer Chemoprevention by Carotenoids
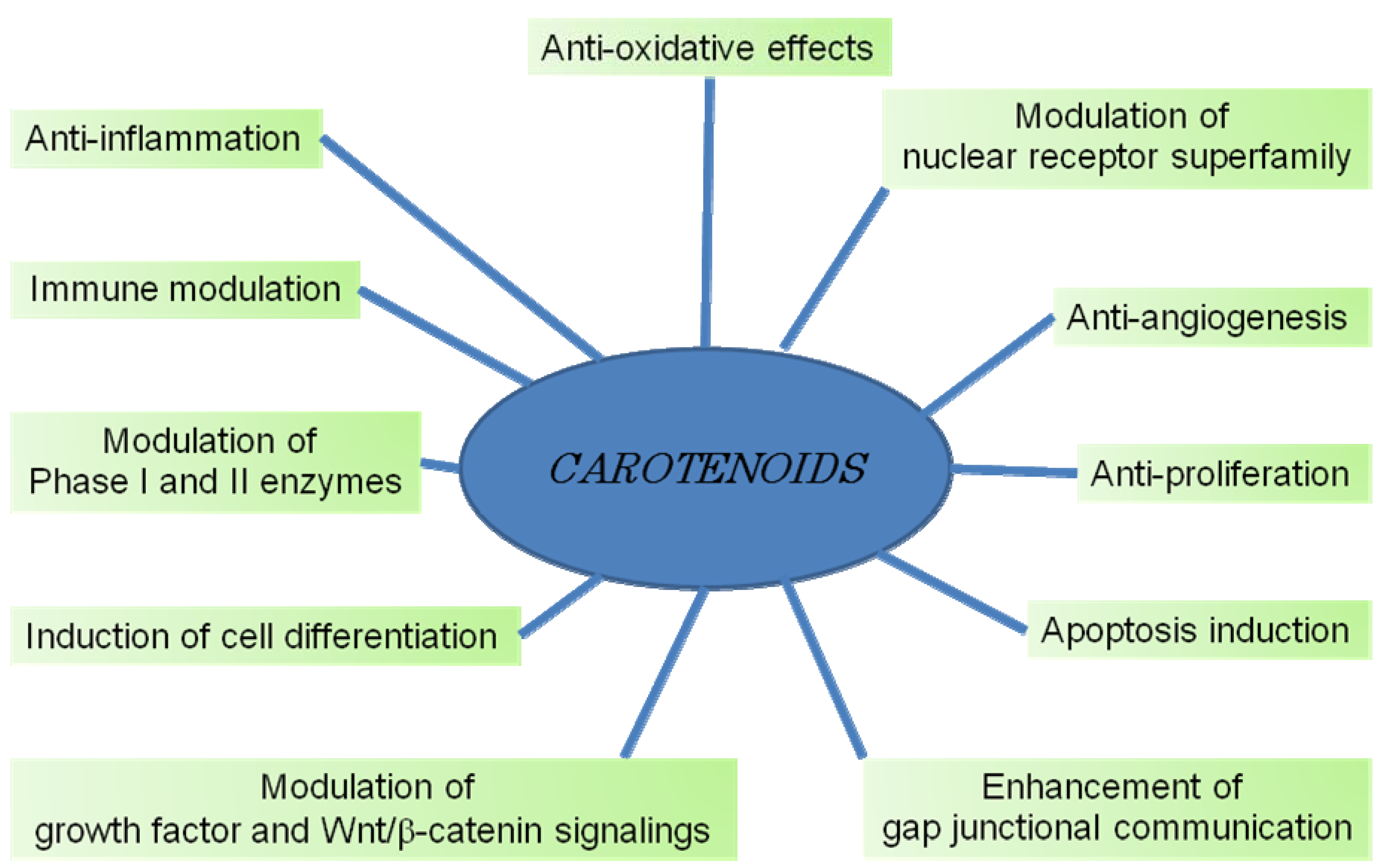
7.1. Gap Junctional Intercellular Communication
7.2. Growth Factor Signaling
7.3. Cell Cycle Progression
7.4. Differentiation-Related Proteins
7.5. RAR
7.6. PPAR
7.7. Xenobiotic and other Orphan Nuclear Receptors
7.8. Antioxidant Response Element
7.9. AP-1 Transcriptional Complex
7.10. Wnt/β-Catenin Pathway
7.11. Inflammatory Cytokines
8. Conclusions
Acknowledgments
Conflict of Interest
References
- Sporn, M.B.; Suh, N. Chemoprevention of cancer. Carcinogenesis 2000, 21, 525–530. [Google Scholar] [CrossRef]
- Sporn, M.B.; Suh, N. Chemoprevention: An essential approach to controlling cancer. Nat. Rev. Cancer 2002, 2, 537–543. [Google Scholar] [CrossRef]
- Chesson, A.; Collins, A. Assessment of the role of diet in cancer prevention. Cancer Lett. 1997, 114, 237–245. [Google Scholar] [CrossRef]
- Tanaka, T. Chemoprevention of human cancer: Biology and therapy. Crit. Rev. Oncol. Hematol. 1997, 25, 139–174. [Google Scholar] [CrossRef]
- Tanaka, T. Effect of diet on human carcinogenesis. Crit. Rev. Oncol. Hematol. 1997, 25, 73–95. [Google Scholar] [CrossRef]
- Lee, B.M.; Park, K.K. Beneficial and adverse effects of chemopreventive agents. Mutat. Res. 2003, 523–524, 265–278. [Google Scholar]
- Potter, J.D. Cancer prevention: Epidemiology and experiment. Cancer Lett. 1997, 114, 7–9. [Google Scholar] [CrossRef]
- Wargovich, M.J. Experimental evidence for cancer preventive elements in foods. Cancer Lett. 1997, 114, 11–17. [Google Scholar] [CrossRef]
- Eastwood, M.A. Interaction of dietary antioxidants in vivo: How fruit and vegetables prevent disease? QJM 1999, 92, 527–530. [Google Scholar]
- Holick, C.N.; Michaud, D.S.; Stolzenberg-Solomon, R.; Mayne, S.T.; Pietinen, P.; Taylor, P.R.; Virtamo, J.; Albanes, D. Dietary carotenoids, serum β-carotene, and retinol and risk of lung cancer in the a-tocopherol,β-carotene cohort study. Am. J. Epidemiol. 2002, 156, 536–547. [Google Scholar]
- Rock, C.L. Carotenoid update. J. Am. Diet Assoc. 2003, 103, 423–425. [Google Scholar] [CrossRef]
- Zhang, S.; Hunter, D.J.; Forman, M.R.; Rosner, B.A.; Speizer, F.E.; Colditz, G.A.; Manson, J.E.; Hankinson, S.E.; Willett, W.C. Dietary carotenoids and vitamins A, C, and E and risk of breast cancer. J. Natl. Cancer Inst. 1999, 91, 547–556. [Google Scholar] [CrossRef]
- Heinonen, O.P.; Albanes, D. The effect of vitamin E and β carotene on the incidence of lung cancer and other cancers in male smokers. The a-Tocopherol, β Carotene Cancer Prevention Study Group. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar]
- Hennekens, C.H.; Buring, J.E.; Manson, J.E.; Stampfer, M.; Rosner, B.; Cook, N.R.; Belanger, C.; LaMotte, F.; Gaziano, J.M.; Ridker, P.M.; et al. Lack of effect of long-term supplementation with β carotene on the incidence of malignant neoplasms and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1145–1149. [Google Scholar] [CrossRef]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L.; Valanis, B.; Williams, J.H.; et al. Effects of a combination of β carotene and vitamin A on lung cancer and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1150–1155. [Google Scholar] [CrossRef]
- Chew, B.P.; Park, J.S.; Wong, M.W.; Wong, T.S. A comparison of the anticancer activities of dietary β-carotene, canthaxanthin and astaxanthin in mice in vivo. Anticancer Res. 1999, 19, 1849–1853. [Google Scholar]
- Levy, J.; Bosin, E.; Feldman, B.; Giat, Y.; Miinster, A.; Danilenko, M.; Sharoni, Y. Lycopene is a more potent inhibitor of human cancer cell proliferation than eitherα-carotene or β-carotene. Nutr. Cancer 1995, 24, 257–266. [Google Scholar] [CrossRef]
- Murakoshi, M.; Nishino, H.; Satomi, Y.; Takayasu, J.; Hasegawa, T.; Tokuda, H.; Iwashima, A.; Okuzumi, J.; Okabe, H.; Kitano, H.; et al. Potent preventive action of a-carotene against carcinogenesis: Spontaneous liver carcinogenesis and promoting stage of lung and skin carcinogenesis in mice are suppressed more effectively by a-carotene than by β-carotene. Cancer Res. 1992, 52, 6583–6587. [Google Scholar]
- Park, J.S.; Chew, B.P.; Wong, T.S. Dietary lutein from marigold extract inhibits mammary tumor development in BALB/c mice. J. Nutr. 1998, 128, 1650–1656. [Google Scholar]
- Jalal, F.; Nesheim, M.C.; Agus, Z.; Sanjur, D.; Habicht, J.P. Serum retinol concentrations in children are affected by food sources of β-carotene, fat intake, and anthelmintic drug treatment. Am. J. Clin. Nutr. 1998, 68, 623–629. [Google Scholar]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar]
- Clevidence, B.A.; Bieri, J.G. Asociation of carotenoids with human plasma lipoproteins. In Methods in Enzymology; Abelson, J.N., Simon, M.I., Eds.; Academic Press: San Diego, CA, USA, 1993; pp. 33–46. [Google Scholar]
- Wang, W.; Connor, S.L.; Johnson, E.J.; Klein, M.L.; Hughes, S.; Connor, W.E. Effect of dietary lutein and zeaxanthin on plasma carotenoids and their transport in lipoproteins in age-related macular degeneration. Am. J. Clin. Nutr. 2007, 85, 762–769. [Google Scholar]
- Kotake-Nara, E.; Nagao, A. Absorption and metabolism of xanthophylls. Mar. Drugs 2011, 9, 1024–1037. [Google Scholar] [CrossRef]
- Nagao, A. Absorption and metabolism of dietary carotenoids. Biofactors 2011, 37, 83–87. [Google Scholar] [CrossRef]
- Khachik, F.; Spangler, C.J.; Smith, J.C., Jr.; Canfield, L.M.; Steck, A.; Pfander, H. Identification, quantification, and relative concentrations of carotenoids and their metabolites in human milk and serum. Anal. Chem. 1997, 69, 1873–1881. [Google Scholar] [CrossRef]
- Goodman, D.S.; Huang, H.S. Biosynthesis of vitamin a with rat intestinal enzymes. Science 1965, 149, 879–880. [Google Scholar]
- Olson, J.A.; Hayaishi, O. The enzymatic cleavage of β-carotene into vitamin A by soluble enzymes of rat liver and intestine. Proc. Natl. Acad. Sci. USA 1965, 54, 1364–1370. [Google Scholar] [CrossRef]
- Wang, X.D.; Krinsky, N.I. The bioconversion of β-carotene into retinoids. Subcell. Biochem. 1998, 30, 159–180. [Google Scholar]
- Wang, X.D.; Tang, G.W.; Fox, J.G.; Krinsky, N.I.; Russell, R.M. Enzymatic conversion of β-carotene into β-apo-carotenals and retinoids by human, monkey, ferret, and rat tissues. Arch. Biochem. Biophys. 1991, 285, 8–16. [Google Scholar] [CrossRef]
- Kiefer, C.; Hessel, S.; Lampert, J.M.; Vogt, K.; Lederer, M.O.; Breithaupt, D.E.; von Lintig, J. Identification and characterization of a mammalian enzyme catalyzing the asymmetric oxidative cleavage of provitamin A. J. Biol. Chem. 2001, 276, 14110–14116. [Google Scholar]
- Lakshmanan, M.R.; Pope, J.L.; Olson, J.A. The specificity of a partially purified carotenoid cleavage enzyme of rabbit intestine. Biochem. Biophys. Res. Commun. 1968, 33, 347–352. [Google Scholar] [CrossRef]
- Wang, X.D.; Russell, R.M.; Liu, C.; Stickel, F.; Smith, D.E.; Krinsky, N.I. β-oxidation in rabbit liver in vitro and in the perfused ferret liver contributes to retinoic acid biosynthesis from β-apocarotenoic acids. J. Biol. Chem. 1996, 271, 26490–26498. [Google Scholar]
- Ferrucci, L.; Perry, J.R.; Matteini, A.; Perola, M.; Tanaka, T.; Silander, K.; Rice, N.; Melzer, D.; Murray, A.; Cluett, C.; et al. Common variation in the β-carotene 15,15'-monooxygenase 1 gene affects circulating levels of carotenoids: A genome-wide association study. Am. J. Hum. Genet. 2009, 84, 123–133. [Google Scholar]
- Leung, W.C.; Hessel, S.; Meplan, C.; Flint, J.; Oberhauser, V.; Tourniaire, F.; Hesketh, J.E.; von Lintig, J.; Lietz, G. Two common single nucleotide polymorphisms in the gene encoding β-carotene 15,15'-monoxygenase alter β-carotene metabolism in female volunteers. FASEB J. 2009, 23, 1041–1053. [Google Scholar] [CrossRef]
- Armstrong, G.A.; Hearst, J.E. Carotenoids 2: Genetics and molecular biology of carotenoid pigment biosynthesis. FASEB J. 1996, 10, 228–237. [Google Scholar]
- Demmig-Adams, B.; Gilmore, A.M.; Adams, W.W., 3rd. Carotenoids 3: In vivo function of carotenoids in higher plants. FASEB J. 1996, 10, 403–412. [Google Scholar]
- van Breemen, R.B.; Pajkovic, N. Multitargeted therapy of cancer by lycopene. Cancer Lett. 2008, 269, 339–351. [Google Scholar] [CrossRef]
- Huang, C.S.; Fan, Y.E.; Lin, C.Y.; Hu, M.L. Lycopene inhibits matrix metalloproteinase-9 expression and down-regulates the binding activity of nuclear factor-kappa B and stimulatory protein-1. J. Nutr. Biochem. 2007, 18, 449–456. [Google Scholar]
- Huang, C.S.; Liao, J.W.; Hu, M.L. Lycopene inhibits experimental metastasis of human hepatoma SK-Hep-1 cells in athymic nude mice. J. Nutr. 2008, 138, 538–543. [Google Scholar]
- Huang, C.S.; Shih, M.K.; Chuang, C.H.; Hu, M.L. Lycopene inhibits cell migration and invasion and upregulates Nm23-H1 in a highly invasive hepatocarcinoma, SK-Hep-1 cells. J. Nutr. 2005, 135, 2119–2123. [Google Scholar]
- Yang, C.M.; Yen, Y.T.; Huang, C.S.; Hu, M.L. Growth inhibitory efficacy of lycopene and β-carotene against androgen-independent prostate tumor cells xenografted in nude mice. Mol. Nutr. Food Res. 2011, 55, 606–612. [Google Scholar] [CrossRef]
- Landrum, J.T.; Bone, R.A. Lutein, zeaxanthin, and the macular pigment. Arch. Biochem. Biophys. 2001, 385, 28–40. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu. Rev. Nutr. 2003, 23, 171–201. [Google Scholar] [CrossRef]
- Miller, N.J.; Sampson, J.; Candeias, L.P.; Bramley, P.M.; Rice-Evans, C.A. Antioxidant activities of carotenes and xanthophylls. FEBS Lett. 1996, 384, 240–242. [Google Scholar] [CrossRef]
- di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar]
- Junghans, A.; Sies, H.; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Biophys. 2001, 391, 160–164. [Google Scholar]
- Humphries, J.M.; Khachik, F. Distribution of lutein, zeaxanthin, and related geometrical isomers in fruit, vegetables, wheat, and pasta products. J. Agric. Food. Chem. 2003, 51, 1322–1327. [Google Scholar] [CrossRef]
- Moeller, S.M.; Jacques, P.F.; Blumberg, J.B. The potential role of dietary xanthophylls in cataract and age-related macular degeneration. J. Am. Coll. Nutr. 2000, 19, 522–527. [Google Scholar]
- Hadden, W.L.; Watkins, R.H.; Levy, L.W.; Regalado, E.; Rivadeneira, D.M.; van Breemen, R.B.; Schwartz, S.J. Carotenoid composition of marigold (Tagetes erecta) flower extract used as nutritional supplement. J. Agric. Food Chem. 1999, 47, 4189–4194. [Google Scholar] [CrossRef]
- Nelis, H.J.; de Leenheer, A.P. Microbial sources of carotenoid pigments used in foods and feeds. J. Appl. Bacteriol. 1991, 70, 181–191. [Google Scholar]
- Mangels, A.R.; Holden, J.M.; Beecher, G.R.; Forman, M.R.; Lanza, E. Carotenoid content of fruits and vegetables: An evaluation of analytic data. J. Am. Diet Assoc. 1993, 93, 284–296. [Google Scholar] [CrossRef]
- Sugiura, M.; Matsumoto, H.; Kato, M.; Ikoma, Y.; Yano, M.; Nagao, A. Multiple linear regression analysis of the seasonal changes in the serum concentration of β-cryptoxanthin. J. Nutr. Sci. Vitaminol. (Tokyo) 2004, 50, 196–202. [Google Scholar]
- Kaplan, L.A.; Lau, J.M.; Stein, E.A. Carotenoid composition, concentrations, and relationships in various human organs. Clin. Physiol. Biochem. 1990, 8, 1–10. [Google Scholar]
- Nair, P.P.; Lohani, A.; Norkus, E.P.; Feagins, H.; Bhagavan, H.N. Uptake and distribution of carotenoids, retinol, and tocopherols in human colonic epithelial cells in vivo. Cancer Epidemiol. Biomark. Prev. 1996, 5, 913–916. [Google Scholar]
- Sugiura, M.; Nakamura, M.; Ikoma, Y.; Yano, M.; Ogawa, K.; Matsumoto, H.; Kato, M.; Ohshima, M.; Nagao, A. High serum carotenoids are inversely associated with serum gamma-glutamyltransferase in alcohol drinkers within normal liver function. J. Epidemiol. 2005, 15, 180–186. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ikoma, Y.; Yano, M.; Ogawa, K.; Matsumoto, H.; Kato, M.; Ohshima, M.; Nagao, A. Serum carotenoid concentrations are inversely associated with serum aminotransferases in hyperglycemic subjects. Diabetes Res. Clin. Pract. 2006, 71, 82–91. [Google Scholar] [CrossRef]
- Nishino, H.; Tokuda, H.; Murakoshi, M.; Satomi, Y.; Masuda, M.; Onozuka, M.; Yamaguchi, S.; Takayasu, J.; Tsuruta, J.; Okuda, M.; et al. Cancer prevention by natural carotenoids. Biofactors 2000, 13, 89–94. [Google Scholar]
- Tanaka, T.; Kohno, H.; Murakami, M.; Shimada, R.; Kagami, S.; Sumida, T.; Azuma, Y.; Ogawa, H. Suppression of azoxymethane-induced colon carcinogenesis in male F344 rats by mandarin juices rich in β-cryptoxanthin and hesperidin. Int. J. Cancer 2000, 88, 146–150. [Google Scholar] [CrossRef]
- Rauscher, R.; Edenharder, R.; Platt, K.L. In vitro antimutagenic and in vivo anticlastogenic effects of carotenoids and solvent extracts from fruits and vegetables rich in carotenoids. Mutat. Res. 1998, 413, 129–142. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Ando, F.; Shimokata, H.; Yano, M. Dietary patterns of antioxidant vitamin and carotenoid intake associated with bone mineral density: Findings from post-menopausal Japanese female subjects. Osteoporos. Int. 2011, 22, 143–152. [Google Scholar] [CrossRef]
- Sugiura, M.; Ogawa, K.; Yano, M. Effect of chronic administration of fruit extract (Citrus unshiu Marc.) on glucose tolerance in GK rats, a model of type 2 diabetes. Biosci. Biotechnol. Biochem. 2006, 70, 293–295. [Google Scholar]
- Uchiyama, S.; Yamaguchi, M. Oral administration of β-cryptoxanthin prevents bone loss in ovariectomized rats. Int. J. Mol. Med. 2006, 17, 15–20. [Google Scholar]
- Takayanagi, K. Prevention of adiposity by oral administration of β-cryptoxanthin. Front. Neurol. 2011, 2, 67. [Google Scholar]
- Takayanagi, K.; Morimoto, S.I.; Shirakura, Y.; Mukai, K.; Sugiyama, T.; Tokuji, Y.; Ohnishi, M. Mechanism of visceral fat reduction in Tsumura Suzuki Obese, Diabetes (TSOD) mice orally administered β-cryptoxanthin from Satsuma mandarin oranges (Citrus unshiu Marc). J. Agric. Food Chem. 2011, 59, 12342–12351. [Google Scholar] [CrossRef]
- Katsuura, S.; Imamura, T.; Bando, N.; Yamanishi, R. β-Carotene and β-cryptoxanthin but not lutein evoke redox and immune changes in RAW264 murine macrophages. Mol. Nutr. Food Res. 2009, 53, 1396–1405. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Weitzmann, M.N. The bone anabolic carotenoid β-cryptoxanthin enhances transforming growth factor-β1-induced SMAD activation in MC3T3 preosteoblasts. Int. J. Mol. Med. 2009, 24, 671–675. [Google Scholar]
- Paterson, E.; Gordon, M.H.; Niwat, C.; George, T.W.; Parr, L.; Waroonphan, S.; Lovegrove, J.A. Supplementation with fruit and vegetable soups and beverages increases plasma carotenoid concentrations but does not alter markers of oxidative stress or cardiovascular risk factors. J. Nutr. 2006, 136, 2849–2855. [Google Scholar]
- Yuan, J.P.; Peng, J.; Yin, K.; Wang, J.H. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol. Nutr. Food Res. 2011, 55, 150–165. [Google Scholar] [CrossRef]
- Pashkow, F.J.; Watumull, D.G.; Campbell, C.L. Astaxanthin: A novel potential treatment for oxidative stress and inflammation in cardiovascular disease. Am. J. Cardiol. 2008, 101, 58–68. [Google Scholar]
- Hulisz, D.T.; Boles, G.L. Clinical review of canthaxanthin (‘tanning pills’). Am. Pharm. 1993, NS33, 44–46. [Google Scholar]
- Palozza, P.; Krinsky, N.I. Astaxanthin and canthaxanthin are potent antioxidants in a membrane model. Arch. Biochem. Biophys. 1992, 297, 291–295. [Google Scholar] [CrossRef]
- Terao, J. Antioxidant activity of β-carotene-related carotenoids in solution. Lipids 1989, 24, 659–661. [Google Scholar] [CrossRef]
- Zhang, L.X.; Cooney, R.V.; Bertram, J.S. Carotenoids up-regulate connexin43 gene expression independent of their provitamin A or antioxidant properties. Cancer Res. 1992, 52, 5707–5712. [Google Scholar]
- Dembitsky, V.M.; Maoka, T. Allenic and cumulenic lipids. Prog. Lipid Res. 2007, 46, 328–375. [Google Scholar] [CrossRef]
- Nomura, T.; Kikuchi, M.; Kubodera, A.; Kawakami, Y. Proton-donative antioxidant activity of fucoxanthin with 1,1-diphenyl-2-picrylhydrazyl (DPPH). Biochem. Mol. Biol. Int. 1997, 42, 361–370. [Google Scholar]
- Yan, X.; Chuda, Y.; Suzuki, M.; Nagata, T. Fucoxanthin as the major antioxidant in Hijikia fusiformis, a common edible seaweed. Biosci. Biotechnol. Biochem. 1999, 63, 605–607. [Google Scholar] [CrossRef]
- Woo, M.N.; Jeon, S.M.; Shin, Y.C.; Lee, M.K.; Kang, M.A.; Choi, M.S. Anti-obese property of fucoxanthin is partly mediated by altering lipid-regulating enzymes and uncoupling proteins of visceral adipose tissue in mice. Mol. Nutr. Food Res. 2009, 53, 1603–1611. [Google Scholar]
- Beppu, F.; Niwano, Y.; Tsukui, T.; Hosokawa, M.; Miyashita, K. Single and repeated oral dose toxicity study of fucoxanthin (FX), a marine carotenoid, in mice. J. Toxicol. Sci. 2009, 34, 501–510. [Google Scholar] [CrossRef]
- Iio, K.; Okada, Y.; Ishikura, M. Bacterial reverse mutation test and micronucleus test of fucoxanthin oil from microalgae. Shokuhin Eiseigaku Zasshi 2011, 52, 190–193. [Google Scholar] [CrossRef]
- Beppu, F.; Niwano, Y.; Sato, E.; Kohno, M.; Tsukui, T.; Hosokawa, M.; Miyashita, K. In vitro and in vivo evaluation of mutagenicity of fucoxanthin (FX) and its metabolite fucoxanthinol (FXOH). J. Toxicol. Sci. 2009, 34, 693–698. [Google Scholar] [CrossRef]
- Block, G.; Patterson, B.; Subar, A. Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutr. Cancer 1992, 18, 1–29. [Google Scholar] [CrossRef]
- Blot, W.J.; Li, J.Y.; Taylor, P.R.; Guo, W.; Dawsey, S.; Wang, G.Q.; Yang, C.S.; Zheng, S.F.; Gail, M.; Li, G.Y.; et al. Nutrition intervention trials in Linxian, China: Supplementation with specific vitamin/mineral combinations, cancer incidence, and disease-specific mortality in the general population. J. Natl. Cancer Inst. 1993, 85, 1483–1492. [Google Scholar] [CrossRef]
- Ziegler, R.G.; Mayne, S.T.; Swanson, C.A. Nutrition and lung cancer. Cancer Causes Control 1996, 7, 157–177. [Google Scholar]
- Wang, X.D.; Russell, R.M. Procarcinogenic and anticarcinogenic effects of β-carotene. Nutr. Rev. 1999, 57, 263–272. [Google Scholar]
- Lee, I.M.; Cook, N.R.; Manson, J.E.; Buring, J.E.; Hennekens, C.H. β-Carotene supplementation and incidence of cancer and cardiovascular disease: The women’s health study. J. Natl. Cancer Inst. 1999, 91, 2102–2106. [Google Scholar] [CrossRef]
- Kelloff, G.J.; Boone, C.W.; Crowell, J.A.; Steele, V.E.; Lubet, R.A.; Doody, L.A.; Malone, W.F.; Hawk, E.T.; Sigman, C.C. New agents for cancer chemoprevention. J. Cell. Biochem. Suppl. 1996, 26, 1–28. [Google Scholar]
- Tanaka, T. Chemoprevention of oral carcinogenesis. Eur. J. Cancer B Oral Oncol. 1995, 31B, 3–15. [Google Scholar] [CrossRef]
- Tanaka, T.; Mori, H. Inhibition of colon carcinogenesis by non-nutritive constituents in foods. J. Toxicol. Pathol. 1996, 9, 139–149. [Google Scholar] [CrossRef]
- Tanaka, T.; Sugie, S. Inhibition of colon carcinogenesis by dietary non-nutritive compounds. J. Toxicol. Pathol. 2008, 20, 215–235. [Google Scholar] [CrossRef]
- Davies, K.J. Oxidative stress: The paradox of aerobic life. Biochem. Soc. Symp. 1995, 61, 1–31. [Google Scholar]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef]
- Ames, B.N.; Shigenaga, M.K. Oxidants are a major contributor to aging. Ann. N.Y. Acad. Sci. 1992, 663, 85–96. [Google Scholar]
- Donaldson, M.S. Nutrition and cancer: A review of the evidence for an anti-cancer diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef]
- Tanaka, T.; Makita, H.; Ohnishi, M.; Hirose, Y.; Wang, A.; Mori, H.; Satoh, K.; Hara, A.; Ogawa, H. Chemoprevention of 4-nitroquinoline 1-oxide-induced oral carcinogenesis by dietary curcumin and hesperidin: comparison with the protective effect of β-carotene. Cancer Res. 1994, 54, 4653–4659. [Google Scholar]
- Narisawa, T.; Fukaura, Y.; Hasebe, M.; Ito, M.; Aizawa, R.; Murakoshi, M.; Uemura, S.; Khachik, F.; Nishino, H. Inhibitory effects of natural carotenoids, a-carotene, β-carotene, lycopene and lutein, on colonic aberrant crypt foci formation in rats. Cancer Lett. 1996, 107, 137–142. [Google Scholar] [CrossRef]
- Faure, H.; Fayol, V.; Galabert, C.; Grolier, P.; Le Moel, G.; Steghens, J.P.; van Kappel, A.; Nabet, F. Carotenoids: 1. Metabolism and physiology. Ann. Biol. Clin. (Paris) 1999, 57, 169–183. [Google Scholar]
- Tanaka, T.; Sugiura, H.; Inaba, R.; Nishikawa, A.; Murakami, A.; Koshimizu, K.; Ohigashi, H. Immunomodulatory action of citrus auraptene on macrophage functions and cytokine production of lymphocytes in female BALB/c mice. Carcinogenesis 1999, 20, 1471–1476. [Google Scholar]
- Narisawa, T.; Fukaura, Y.; Oshima, S.; Inakuma, T.; Yano, M.; Nishino, H. Chemoprevention by the oxygenated carotenoid β-cryptoxanthin of N-methylnitrosourea-induced colon carcinogenesis in F344 rats. Jpn. J. Cancer Res. 1999, 90, 1061–1065. [Google Scholar] [CrossRef]
- Kohno, H.; Taima, M.; Sumida, T.; Azuma, Y.; Ogawa, H.; Tanaka, T. Inhibitory effect of mandarin juice rich in β-cryptoxanthin and hesperidin on 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced pulmonary tumorigenesis in mice. Cancer Lett. 2001, 174, 141–150. [Google Scholar] [CrossRef]
- Tanaka, T.; Makita, H.; Kawabata, K.; Mori, H.; Kakumoto, M.; Satoh, K.; Hara, A.; Sumida, T.; Fukutani, K.; Ogawa, H. Modulation of N-methyl-N-amylnitrosamine-induced rat oesophageal tumourigenesis by dietary feeding of diosmin and hesperidin, both alone and in combination. Carcinogenesis 1997, 18, 761–769. [Google Scholar]
- Tanaka, T.; Makita, H.; Kawabata, K.; Mori, H.; Kakumoto, M.; Satoh, K.; Hara, A.; Sumida, T.; Ogawa, H. Chemoprevention of azoxymethane-induced rat colon carcinogenesis by the naturally occurring flavonoids, diosmin and hesperidin. Carcinogenesis 1997, 18, 957–965. [Google Scholar]
- Tanaka, T.; Makita, H.; Ohnishi, M.; Hirose, Y.; Wang, A.; Mori, H.; Satoh, K.; Hara, A.; Ogawa, H. Chemoprevention of 4-nitroquinoline 1-oxide-induced oral carcinogenesis by dietary curcumin and hesperidin: Comparison with the protective effect of β-carotene. Cancer Res. 1994, 54, 4653–4659. [Google Scholar]
- Tanaka, T.; Makita, H.; Ohnishi, M.; Mori, H.; Satoh, K.; Hara, A.; Sumida, T.; Fukutani, K.; Ogawa, H. Chemoprevention of 4-nitroquinoline 1-oxide-induced oral carcinogenesis in rats by flavonoids diosmin and hesperidin, each alone and in combination. Cancer Res. 1997, 57, 246–252. [Google Scholar]
- Yang, M.; Tanaka, T.; Hirose, Y.; Deguchi, T.; Mori, H.; Kawada, Y. Chemopreventive effects of diosmin and hesperidin on N-butyl-N-(4-hydroxybutyl)nitrosamine-induced urinary-bladder carcinogenesis in male ICR mice. Int. J. Cancer 1997, 73, 719–724. [Google Scholar]
- Kohno, H.; Maeda, M.; Honjo, S.; Murakami, M.; Shimada, R.; Masuda, S.; Sumida, T.; Azuma, Y.; Ogawa, H.; Tanaka, T. Prevention of colonic preneoplastic lesions by the β-cryptoxanthin and hesperidin rich powder prepared from Citrus unshiu Marc. Juice in male F344 rats. J. Toxicol. Pathol. 1999, 12, 209–215. [Google Scholar] [CrossRef]
- Tanaka, T.; Tanaka, T.; Tanaka, M.; Kuno, T. Cancer chemoprevention by citrus pulp and juices containing high amounts of β-cryptoxanthin and hesperidin. J. Biomed. Biotechnol. 2012, . [Google Scholar] [CrossRef]
- Suzuki, R.; Kohno, H.; Yasui, Y.; Hata, K.; Sugie, S.; Miyamoto, S.; Sugawara, K.; Sumida, T.; Hirose, Y.; Tanaka, T. Diet supplemented with citrus unshiu segment membrane suppresses chemically induced colonic preneoplastic lesions and fatty liver in male db/db mice. Int. J. Cancer 2007, 120, 252–258. [Google Scholar] [CrossRef]
- Tanaka, T.; Yasui, Y.; Ishigamori-Suzuki, R.; Oyama, T. Citrus compounds inhibit inflammation- and obesity-related colon carcinogenesis in mice. Nutr. Cancer 2008, 60, S70–S80. [Google Scholar] [CrossRef]
- Narisawa, T.; Fukaura, Y.; Hasebe, M.; Nomura, S.; Oshima, S.; Sakamoto, H.; Inakuma, T.; Ishiguro, Y.; Takayasu, J.; Nishino, H. Prevention of N-methylnitrosourea-induced colon carcinogenesis in F344 rats by lycopene and tomato juice rich in lycopene. Jpn. J. Cancer Res. 1998, 89, 1003–1008. [Google Scholar] [CrossRef]
- Sharoni, Y.; Giron, E.; Rise, M.; Levy, J. Effects of lycopene-enriched tomato oleoresin on 7,12-dimethyl-benz[a]anthracene-induced rat mammary tumors. Cancer Detect. Prev. 1997, 21, 118–123. [Google Scholar]
- Nagasawa, H.; Mitamura, T.; Sakamoto, S.; Yamamoto, K. Effects of lycopene on spontaneous mammary tumour development in SHN virgin mice. Anticancer Res. 1995, 15, 1173–1178. [Google Scholar]
- Kim, D.J.; Takasuka, N.; Kim, J.M.; Sekine, K.; Ota, T.; Asamoto, M.; Murakoshi, M.; Nishino, H.; Nir, Z.; Tsuda, H. Chemoprevention by lycopene of mouse lung neoplasia after combined initiation treatment with DEN, MNU and DMH. Cancer Lett. 1997, 120, 15–22. [Google Scholar] [CrossRef]
- Astorg, P.; Gradelet, S.; Berges, R.; Suschetet, M. Dietary lycopene decreases the initiation of liver preneoplastic foci by diethylnitrosamine in the rat. Nutr. Cancer 1997, 29, 60–68. [Google Scholar] [CrossRef]
- Okajima, E.; Tsutsumi, M.; Ozono, S.; Akai, H.; Denda, A.; Nishino, H.; Oshima, S.; Sakamoto, H.; Konishi, Y. Inhibitory effect of tomato juice on rat urinary bladder carcinogenesis after N-butyl-N-(4-hydroxybutyl)nitrosamine initiation. Jpn. J. Cancer Res. 1998, 89, 22–26. [Google Scholar] [CrossRef]
- Cohen, L.A.; Zhao, Z.; Pittman, B.; Khachik, F. Effect of dietary lycopene on N-methylnitrosourea-induced mammary tumorigenesis. Nutr. Cancer 1999, 34, 153–159. [Google Scholar] [CrossRef]
- Hu, K.Q.; Liu, C.; Ernst, H.; Krinsky, N.I.; Russell, R.M.; Wang, X.D. The biochemical characterization of ferret carotene-9',10'-monooxygenase catalyzing cleavage of carotenoids in vitro and in vivo. J. Biol. Chem. 2006, 281, 19327–19338. [Google Scholar] [CrossRef]
- Lian, F.; Smith, D.E.; Ernst, H.; Russell, R.M.; Wang, X.D. Apo-10'-lycopenoic acid inhibits lung cancer cell growth in vitro, and suppresses lung tumorigenesis in the A/J mouse model in vivo. Carcinogenesis 2007, 28, 1567–1574. [Google Scholar] [CrossRef]
- Lian, F.; Wang, X.D. Enzymatic metabolites of lycopene induce Nrf2-mediated expression of phase II detoxifying/antioxidant enzymes in human bronchial epithelial cells. Int. J. Cancer 2008, 123, 1262–1268. [Google Scholar] [CrossRef]
- Yang, C.M.; Huang, S.M.; Liu, C.L.; Hu, M.L. Apo-8'-lycopenal induces expression of HO-1 and NQO-1 via the ERK/p38-Nrf2-ARE pathway in human HepG2 cells. J. Agric. Food Chem. 2012, 60, 1576–7585. [Google Scholar] [CrossRef]
- Hsing, A.W.; Comstock, G.W.; Abbey, H.; Polk, B.F. Serologic precursors of cancer. Retinol, carotenoids, and tocopherol and risk of prostate cancer. J. Natl. Cancer Inst. 1990, 82, 941–946. [Google Scholar]
- Schuurman, A.G.; Goldbohm, R.A.; Brants, H.A.; van den Brandt, P.A. A prospective cohort study on intake of retinol, vitamins C and E, and carotenoids and prostate cancer risk (Netherlands). Cancer Causes Control 2002, 13, 573–582. [Google Scholar] [CrossRef]
- Giovannucci, E.; Ascherio, A.; Rimm, E.B.; Stampfer, M.J.; Colditz, G.A.; Willett, W.C. Intake of carotenoids and retinol in relation to risk of prostate cancer. J. Natl. Cancer Inst. 1995, 87, 1767–1776. [Google Scholar] [CrossRef]
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Stampfer, M.J.; Willett, W.C. A prospective study of tomato products, lycopene, and prostate cancer risk. J. Natl. Cancer Inst. 2002, 94, 391–398. [Google Scholar] [CrossRef]
- Wu, K.; Erdman, J.W., Jr.; Schwartz, S.J.; Platz, E.A.; Leitzmann, M.; Clinton, S.K.; DeGroff, V.; Willett, W.C.; Giovannucci, E. Plasma and dietary carotenoids, and the risk of prostate cancer: A nested case-control study. Cancer Epidemiol. Biomark. Prev. 2004, 13, 260–269. [Google Scholar] [CrossRef]
- Kucuk, O.; Sarkar, F.H.; Sakr, W.; Djuric, Z.; Pollak, M.N.; Khachik, F.; Li, Y.W.; Banerjee, M.; Grignon, D.; Bertram, J.S.; et al. Phase II randomized clinical trial of lycopene supplementation before radical prostatectomy. Cancer Epidemiol. Biomark. Prev. 2001, 10, 861–868. [Google Scholar]
- Bowen, P.; Chen, L.; Stacewicz-Sapuntzakis, M.; Duncan, C.; Sharifi, R.; Ghosh, L.; Kim, H.S.; Christov-Tzelkov, K.; van Breemen, R. Tomato sauce supplementation and prostate cancer: Lycopene accumulation and modulation of biomarkers of carcinogenesis. Exp. Biol. Med. (Maywood) 2002, 227, 886–893. [Google Scholar]
- Cohen, L.A. A review of animal model studies of tomato carotenoids, lycopene, and cancer chemoprevention. Exp. Biol. Med. (Maywood) 2002, 227, 864–868. [Google Scholar]
- Michaud, D.S.; Feskanich, D.; Rimm, E.B.; Colditz, G.A.; Speizer, F.E.; Willett, W.C.; Giovannucci, E. Intake of specific carotenoids and risk of lung cancer in 2 prospective US cohorts. Am. J. Clin. Nutr. 2000, 72, 990–997. [Google Scholar]
- Le Marchand, L.; Hankin, J.H.; Bach, F.; Kolonel, L.N.; Wilkens, L.R.; Stacewicz-Sapuntzakis, M.; Bowen, P.E.; Beecher, G.R.; Laudon, F.; Baque, P.; et al. An ecological study of diet and lung cancer in the South Pacific. Int. J. Cancer 1995, 63, 18–23. [Google Scholar]
- Chew, B.P.; Park, J.S. Carotenoid action on the immune response. J. Nutr. 2004, 134, 257–261. [Google Scholar]
- Kawashima, T. A marine carotenoid, fucoxanthin, induces regulatory T cells and inhibits Th17 cell differentiation in vitro. Biosci. Biotechnol. Biochem. 2011, 75, 2066–2069. [Google Scholar] [CrossRef]
- Iannone, A.; Rota, C.; Bergamini, S.; Tomasi, A.; Canfield, L.M. Antioxidant activity of carotenoids: An electron-spin resonance study on β-carotene and lutein interaction with free radicals generated in a chemical system. J. Biochem. Mol. Toxicol. 1998, 12, 299–304. [Google Scholar] [CrossRef]
- Sujak, A.; Gabrielska, J.; Grudzinski, W.; Borc, R.; Mazurek, P.; Gruszecki, W.I. Lutein and zeaxanthin as protectors of lipid membranes against oxidative damage: The structural aspects. Arch. Biochem. Biophys. 1999, 371, 301–307. [Google Scholar] [CrossRef]
- Haegele, A.D.; Gillette, C.; O’Neill, C.; Wolfe, P.; Heimendinger, J.; Sedlacek, S.; Thompson, H.J. Plasma xanthophyll carotenoids correlate inversely with indices of oxidative DNA damage and lipid peroxidation. Cancer Epidemiol. Biomark. Prev. 2000, 9, 421–425. [Google Scholar]
- Stahl, W.; Junghans, A.; de Boer, B.; Driomina, E.S.; Briviba, K.; Sies, H. Carotenoid mixtures protect multilamellar liposomes against oxidative damage: Synergistic effects of lycopene and lutein. FEBS Lett. 1998, 427, 305–308. [Google Scholar] [CrossRef]
- Gonzalez de Mejia, E.; Loarca-Pina, G.; Ramos-Gomez, M. Antimutagenicity of xanthophylls present in Aztec Marigold (Tagetes erecta) against 1-nitropyrene. Mutat. Res. 1997, 389, 219–226. [Google Scholar] [CrossRef]
- Gonzalez de Mejia, E.; Ramos-Gomez, M.; Loarca-Pina, G. Antimutagenic activity of natural xanthophylls against aflatoxin B1 in Salmonella typhimurium. Environ. Mol. Mutagen. 1997, 30, 346–353. [Google Scholar] [CrossRef]
- Park, J.S.; Chew, B.P.; Wong, T.S.; Zhang, J.X.; Magnuson, N.S. Dietary lutein but not astaxanthin or β-carotene increases pim-1 gene expression in murine lymphocytes. Nutr. Cancer 1999, 33, 206–212. [Google Scholar]
- Tsubono, Y.; Tsugane, S.; Gey, K.F. Plasma antioxidant vitamins and carotenoids in five Japanese populations with varied mortality from gastric cancer. Nutr. Cancer 1999, 34, 56–61. [Google Scholar] [CrossRef]
- Slattery, M.L.; Benson, J.; Curtin, K.; Ma, K.N.; Schaeffer, D.; Potter, J.D. Carotenoids and colon cancer. Am. J. Clin. Nutr. 2000, 71, 575–582. [Google Scholar]
- Wingerath, T.; Sies, H.; Stahl, W. Xanthophyll esters in human skin. Arch. Biochem. Biophys. 1998, 355, 271–274. [Google Scholar] [CrossRef]
- Stahl, W.; Heinrich, U.; Jungmann, H.; Sies, H.; Tronnier, H. Carotenoids and carotenoids plus vitamin E protect against ultraviolet light-induced erythema in humans. Am. J. Clin. Nutr. 2000, 71, 795–798. [Google Scholar]
- Hunter, D.J.; Manson, J.E.; Colditz, G.A.; Stampfer, M.J.; Rosner, B.; Hennekens, C.H.; Speizer, F.E.; Willett, W.C. A prospective study of the intake of vitamins C, E, and A and the risk of breast cancer. N. Engl. J. Med. 1993, 329, 234–240. [Google Scholar] [CrossRef]
- Potischman, N.; McCulloch, C.E.; Byers, T.; Nemoto, T.; Stubbe, N.; Milch, R.; Parker, R.; Rasmussen, K.M.; Root, M.; Graham, S.; et al. Breast cancer and dietary and plasma concentrations of carotenoids and vitamin A. Am. J. Clin. Nutr. 1990, 52, 909–915. [Google Scholar]
- Rohan, T.E.; McMichael, A.J.; Baghurst, P.A. A population-based case-control study of diet and breast cancer in Australia. Am. J. Epidemiol. 1988, 128, 478–489. [Google Scholar]
- Sumantran, V.N.; Zhang, R.; Lee, D.S.; Wicha, M.S. Differential regulation of apoptosis in normal versus transformed mammary epithelium by lutein and retinoic acid. Cancer Epidemiol. Biomark. Prev. 2000, 9, 257–263. [Google Scholar]
- Freudenheim, J.L.; Marshall, J.R.; Vena, J.E.; Laughlin, R.; Brasure, J.R.; Swanson, M.K.; Nemoto, T.; Graham, S. Premenopausal breast cancer risk and intake of vegetables, fruits, and related nutrients. J. Natl. Cancer Inst. 1996, 88, 340–348. [Google Scholar] [CrossRef]
- Dorgan, J.F.; Sowell, A.; Swanson, C.A.; Potischman, N.; Miller, R.; Schussler, N.; Stephenson, H.E., Jr. Relationships of serum carotenoids, retinol, a-tocopherol, and selenium with breast cancer risk: Results from a prospective study in Columbia, Missouri (United States). Cancer Causes Control 1998, 9, 89–97. [Google Scholar] [CrossRef]
- Zhang, S.; Tang, G.; Russell, R.M.; Mayzel, K.A.; Stampfer, M.J.; Willett, W.C.; Hunter, D.J. Measurement of retinoids and carotenoids in breast adipose tissue and a comparison of concentrations in breast cancer cases and control subjects. Am. J. Clin. Nutr. 1997, 66, 626–632. [Google Scholar]
- Toniolo, P.; van Kappel, A.L.; Akhmedkhanov, A.; Ferrari, P.; Kato, I.; Shore, R.E.; Riboli, E. Serum carotenoids and breast cancer. Am. J. Epidemiol. 2001, 153, 1142–1147. [Google Scholar] [CrossRef]
- Yeum, K.J.; Ahn, S.H.; Rupp de Paiva, S.A.; Lee-Kim, Y.C.; Krinsky, N.I.; Russell, R.M. Correlation between carotenoid concentrations in serum and normal breast adipose tissue of women with benign breast tumor or breast cancer. J. Nutr. 1998, 128, 1920–1926. [Google Scholar]
- Anonymous. Eskimo diets and diseases. Lancet 1983, 1, 1139–1141.
- Bates, C.; van Dam, C.; Horrobin, D.F.; Morse, N.; Huang, Y.S.; Manku, M.S. Plasma essential fatty acids in pure and mixed race American Indians on and off a diet exceptionally rich in salmon. Prostaglandins Leukot. Med. 1985, 17, 77–84. [Google Scholar] [CrossRef]
- Tanaka, T.; Morishita, Y.; Suzui, M.; Kojima, T.; Okumura, A.; Mori, H. Chemoprevention of mouse urinary bladder carcinogenesis by the naturally occurring carotenoid astaxanthin. Carcinogenesis 1994, 15, 15–19. [Google Scholar] [CrossRef]
- Tanaka, T.; Makita, H.; Ohnishi, M.; Mori, H.; Satoh, K.; Hara, A. Chemoprevention of rat oral carcinogenesis by naturally occurring xanthophylls, astaxanthin and canthaxanthin. Cancer Res. 1995, 55, 4059–4064. [Google Scholar]
- Tanaka, T.; Kawamori, T.; Ohnishi, M.; Makita, H.; Mori, H.; Satoh, K.; Hara, A. Suppression of azoxymethane-induced rat colon carcinogenesis by dietary administration of naturally occurring xanthophylls astaxanthin and canthaxanthin during the postinitiation phase. Carcinogenesis 1995, 16, 2957–2963. [Google Scholar] [CrossRef]
- Yasui, Y.; Hosokawa, M.; Mikami, N.; Miyashita, K.; Tanaka, T. Dietary astaxanthin inhibits colitis and colitis-associated colon carcinogenesis in mice via modulation of the inflammatory cytokines. Chem. Biol. Interact. 2011, 193, 79–87. [Google Scholar] [CrossRef]
- McCarty, M.F. Minimizing the cancer-promotional activity of cox-2 as a central strategy in cancer prevention. Med. Hypotheses 2011, 78, 45–57. [Google Scholar] [CrossRef]
- Nagendraprabhu, P.; Sudhandiran, G. Astaxanthin inhibits tumor invasion by decreasing extracellular matrix production and induces apoptosis in experimental rat colon carcinogenesis by modulating the expressions of ERK-2, NFkB and COX-2. Invest. New Drugs 2011, 29, 207–224. [Google Scholar] [CrossRef]
- Gradelet, S.; Le Bon, A.M.; Berges, R.; Suschetet, M.; Astorg, P. Dietary carotenoids inhibit aflatoxin B1-induced liver preneoplastic foci and DNA damage in the rat: Role of the modulation of aflatoxin B1 metabolism. Carcinogenesis 1998, 19, 403–411. [Google Scholar] [CrossRef]
- Hix, L.M.; Frey, D.A.; McLaws, M.D.; Østerlie, M.; Lockwood, S.F.; Bertram, J.S. Inhibition of chemically-induced neoplastic transformation by a novel tetrasodium diphosphate astaxanthin derivative. Carcinogenesis 2005, 26, 1634–1641. [Google Scholar]
- Black, H.S.; Mathews-Roth, M.M. Protective role of butylated hydroxytoluene and certain carotenoids in photocarcinogenesis. Photochem. Photobiol. 1991, 53, 707–716. [Google Scholar] [CrossRef]
- Santamaria, L.; Bianchi, A.; Arnaboldi, A.; Ravetto, C.; Bianchi, L.; Pizzala, R.; Andreoni, L.; Santagati, G.; Bermond, P. Chemoprevention of indirect and direct chemical carcinogenesis by carotenoids as oxygen radical quenchers. Ann. N.Y. Acad. Sci. 1988, 534, 584–596. [Google Scholar]
- Grubbs, C.J.; Eto, I.; Juliana, M.M.; Whitaker, L.M. Effect of canthaxanthin on chemically induced mammary carcinogenesis. Oncology 1991, 48, 239–245. [Google Scholar] [CrossRef]
- Onogi, N.; Okuno, M.; Matsushima-Nishiwaki, R.; Fukutomi, Y.; Moriwaki, H.; Muto, Y.; Kojima, S. Antiproliferative effect of carotenoids on human colon cancer cells without conversion to retinoic acid. Nutr. Cancer 1998, 32, 20–24. [Google Scholar] [CrossRef]
- Bertram, J.S.; Pung, A.; Churley, M.; Kappock, T.J., IV; Wilkins, L.R.; Cooney, R.V. Diverse carotenoids protect against chemically induced neoplastic transformation. Carcinogenesis 1991, 12, 671–678. [Google Scholar] [CrossRef]
- Mathews-Roth, M.M.; Krinsky, N.I. Carotenoid dose level and protection against UV-B induced skin tumors. Photochem. Photobiol. 1985, 42, 35–38. [Google Scholar] [CrossRef]
- Hanusch, M.; Stahl, W.; Schulz, W.A.; Sies, H. Induction of gap junctional communication by 4-oxoretinoic acid generated from its precursor canthaxanthin. Arch. Biochem. Biophys. 1995, 317, 423–428. [Google Scholar] [CrossRef]
- Gradelet, S.; Astorg, P.; Leclerc, J.; Chevalier, J.; Vernevaut, M.F.; Siess, M.H. Effects of canthaxanthin, astaxanthin, lycopene and lutein on liver xenobiotic-metabolizing enzymes in the rat. Xenobiotica 1996, 26, 49–63. [Google Scholar] [CrossRef]
- Jewell, C.; O’Brien, N.M. Effect of dietary supplementation with carotenoids on xenobiotic metabolizing enzymes in the liver, lung, kidney and small intestine of the rat. Br. J. Nutr. 1999, 81, 235–242. [Google Scholar]
- Palozza, P.; Maggiano, N.; Calviello, G.; Lanza, P.; Piccioni, E.; Ranelletti, F.O.; Bartoli, G.M. Canthaxanthin induces apoptosis in human cancer cell lines. Carcinogenesis 1998, 19, 373–376. [Google Scholar]
- Goralczyk, R.; Buser, S.; Bausch, J.; Bee, W.; Zuhlke, U.; Barker, F.M. Occurrence of birefringent retinal inclusions in cynomolgus monkeys after high doses of canthaxanthin. Invest. Ophthalmol. Vis. Sci. 1997, 38, 741–752. [Google Scholar]
- Leyon, H.; Ros, A.M.; Nyberg, S.; Algvere, P. Reversibility of canthaxanthin deposits within the retina. Acta Ophthalmol. (Copenh.) 1990, 68, 607–611. [Google Scholar]
- Das, S.K.; Hashimoto, T.; Kanazawa, K. Growth inhibition of human hepatic carcinoma HepG2 cells by fucoxanthin is associated with down-regulation of cyclin D. Biochim. Biophys. Acta 2008, 1780, 743–749. [Google Scholar] [CrossRef]
- Hosokawa, M.; Kudo, M.; Maeda, H.; Kohno, H.; Tanaka, T.; Miyashita, K. Fucoxanthin induces apoptosis and enhances the antiproliferative effect of the PPARgamma ligand, troglitazone, on colon cancer cells. Biochim. Biophys. Acta 2004, 1675, 113–119. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, P.; Hamada, M.; Takahashi, S.; Xing, G.; Liu, J.; Sugiura, N. Potential chemoprevention effect of dietary fucoxanthin on urinary bladder cancer EJ-1 cell line. Oncol. Rep. 2008, 20, 1099–1103. [Google Scholar]
- Yonekura, L.; Kobayashi, M.; Terasaki, M.; Nagao, A. Keto-carotenoids are the major metabolites of dietary lutein and fucoxanthin in mouse tissues. J. Nutr. 2010, 140, 1824–1831. [Google Scholar] [CrossRef]
- Kim, J.M.; Araki, S.; Kim, D.J.; Park, C.B.; Takasuka, N.; Baba-Toriyama, H.; Ota, T.; Nir, Z.; Khachik, F.; Shimidzu, N.; et al. Chemopreventive effects of carotenoids and curcumins on mouse colon carcinogenesis after 1,2-dimethylhydrazine initiation. Carcinogenesis 1998, 19, 81–85. [Google Scholar] [CrossRef]
- Nishino, H.; Murakoshi, M.; Tokuda, H.; Satomi, Y. Cancer prevention by carotenoids. Arch. Biochem. Biophys. 2009, 483, 165–168. [Google Scholar]
- Okuzumi, J.; Takahashi, T.; Yamane, T.; Kitao, Y.; Inagake, M.; Ohya, K.; Nishino, H.; Tanaka, Y. Inhibitory effects of fucoxanthin, a natural carotenoid, on N-ethyl-N'-nitro-N-nitrosoguanidine- induced mouse duodenal carcinogenesis. Cancer Lett. 1993, 68, 159–168. [Google Scholar] [CrossRef]
- Yoshiko, S.; Hoyoku, N. Fucoxanthin, a natural carotenoid, induces G1 arrest and GADD45 gene expression in human cancer cells. In Vivo 2007, 21, 305–309. [Google Scholar]
- Miyashita, K.; Nishikawa, S.; Beppu, F.; Tsukui, T.; Abe, M.; Hosokawa, M. The allenic carotenoid fucoxanthin, a novel marine nutraceutical from brown seaweeds. J. Sci. Food Agric. 2011, 91, 1166–1174. [Google Scholar] [CrossRef]
- Liu, C.L.; Huang, Y.S.; Hosokawa, M.; Miyashita, K.; Hu, M.L. Inhibition of proliferation of a hepatoma cell line by fucoxanthin in relation to cell cycle arrest and enhanced gap junctional intercellular communication. Chem. Biol. Interact. 2009, 182, 165–172. [Google Scholar] [CrossRef]
- Liu, C.L.; Chiu, Y.T.; Hu, M.L. Fucoxanthin enhances HO-1 and NQO1 expression in murine hepatic BNL CL.2 cells through activation of the Nrf2/ARE system partially by its pro-oxidant activity. J. Agric. Food Chem. 2011, 59, 11344–11351. [Google Scholar]
- Murakami, C.; Takemura, M.; Sugiyama, Y.; Kamisuki, S.; Asahara, H.; Kawasaki, M.; Ishidoh, T.; Linn, S.; Yoshida, S.; Sugawara, F.; et al. Vitamin A-related compounds, all-trans retinal and retinoic acids, selectively inhibit activities of mammalian replicative DNA polymerases. Biochim. Biophys. Acta 2002, 1574, 85–92. [Google Scholar] [CrossRef]
- Zhang, L.X.; Cooney, R.V.; Bertram, J.S. Carotenoids enhance gap junctional communication and inhibit lipid peroxidation in C3H/10T1/2 cells: relationship to their cancer chemopreventive action. Carcinogenesis 1991, 12, 2109–2114. [Google Scholar] [CrossRef]
- LeRoith, D.; Roberts, C.T., Jr. The insulin-like growth factor system and cancer. Cancer Lett. 2003, 195, 127–137. [Google Scholar] [CrossRef]
- Voskuil, D.W.; Vrieling, A.; van’t Veer, L.J.; Kampman, E.; Rookus, M.A. The insulin-like growth factor system in cancer prevention: Potential of dietary intervention strategies. Cancer Epidemiol. Biomark. Prev. 2005, 14, 195–203. [Google Scholar]
- Mantzoros, C.S.; Tzonou, A.; Signorello, L.B.; Stampfer, M.; Trichopoulos, D.; Adami, H.O. Insulin-like growth factor 1 in relation to prostate cancer and benign prostatic hyperplasia. Br. J. Cancer 1997, 76, 1115–1118. [Google Scholar] [CrossRef]
- Hankinson, S.E.; Willett, W.C.; Colditz, G.A.; Hunter, D.J.; Michaud, D.S.; Deroo, B.; Rosner, B.; Speizer, F.E.; Pollak, M. Circulating concentrations of insulin-like growth factor-I and risk of breast cancer. Lancet 1998, 351, 1393–1396. [Google Scholar]
- Ma, J.; Pollak, M.N.; Giovannucci, E.; Chan, J.M.; Tao, Y.; Hennekens, C.H.; Stampfer, M.J. Prospective study of colorectal cancer risk in men and plasma levels of insulin-like growth factor (IGF)-I and IGF-binding protein-3. J. Natl. Cancer Inst. 1999, 91, 620–625. [Google Scholar] [CrossRef]
- Yu, H.; Spitz, M.R.; Mistry, J.; Gu, J.; Hong, W.K.; Wu, X. Plasma levels of insulin-like growth factor-I and lung cancer risk: A case-control analysis. J. Natl. Cancer Inst. 1999, 91, 151–156. [Google Scholar]
- Hata, K.; Kubota, M.; Shimizu, M.; Moriwaki, H.; Toshiya, K.; Tanaka, T.; Hara, A.; Hirose, Y. C57BL/KsJ-db/db-ApcMin/+ mice exhibit an increased incidence of intestinal neoplasms. Int. J. Mol. Sci. 2011, 12, 8133–8145. [Google Scholar] [CrossRef]
- Karas, M.; Amir, H.; Fishman, D.; Danilenko, M.; Segal, S.; Nahum, A.; Koifmann, A.; Giat, Y.; Levy, J.; Sharoni, Y. Lycopene interferes with cell cycle progression and insulin-like growth factor I signaling in mammary cancer cells. Nutr. Cancer 2000, 36, 101–111. [Google Scholar] [CrossRef]
- Karas, M.; Danilenko, M.; Fishman, D.; LeRoith, D.; Levy, J.; Sharoni, Y. Membrane-associated insulin-like growth factor-binding protein-3 inhibits insulin-like growth factor-I-induced insulin-like growth factor-I receptor signaling in ishikawa endometrial cancer cells. J. Biol. Chem. 1997, 272, 16514–16520. [Google Scholar]
- Amir, H.; Karas, M.; Giat, J.; Danilenko, M.; Levy, R.; Yermiahu, T.; Levy, J.; Sharoni, Y. Lycopene and 1,25-dihydroxyvitamin D3 cooperate in the inhibition of cell cycle progression and induction of differentiation in HL-60 leukemic cells. Nutr. Cancer 1999, 33, 105–112. [Google Scholar] [CrossRef]
- Murakoshi, M.; Takayasu, J.; Kimura, O.; Kohmura, E.; Nishino, H.; Iwashima, A.; Okuzumi, J.; Sakai, T.; Sugimoto, T.; Imanishi, J.; et al. Inhibitory effects of a-carotene on proliferation of the human neuroblastoma cell line GOTO. J. Natl. Cancer Inst. 1989, 81, 1649–1652. [Google Scholar]
- Stivala, L.A.; Savio, M.; Quarta, S.; Scotti, C.; Cazzalini, O.; Rossi, L.; Scovassi, I.A.; Pizzala, R.; Melli, R.; Bianchi, L.; et al. The antiproliferative effect of β-carotene requires p21waf1/cip1 in normal human fibroblasts. Eur. J. Biochem. 2000, 267, 2290–2296. [Google Scholar] [CrossRef]
- Satomi, Y.; Nishino, H. Implication of mitogen-activated protein kinase in the induction of G1 cell cycle arrest and gadd45 expression by the carotenoid fucoxanthin in human cancer cells. Biochim. Biophys. Acta 2009, 1790, 260–266. [Google Scholar] [CrossRef]
- Ford, N.A.; Elsen, A.C.; Zuniga, K.; Lindshield, B.L.; Erdman, J.W., Jr. Lycopene and apo-12'-lycopenal reduce cell proliferation and alter cell cycle progression in human prostate cancer cells. Nutr. Cancer 2011, 63, 256–263. [Google Scholar] [CrossRef]
- Nahum, A.; Hirsch, K.; Danilenko, M.; Watts, C.K.; Prall, O.W.; Levy, J.; Sharoni, Y. Lycopene inhibition of cell cycle progression in breast and endometrial cancer cells is associated with reduction in cyclin D levels and retention of p27(Kip1) in the cyclin E-cdk2 complexes. Oncogene 2001, 20, 3428–3436. [Google Scholar] [CrossRef]
- Gross, M.D.; Bishop, T.D.; Belcher, J.D.; Jacobs, D.R., Jr. Induction of HL-60 cell differentiation by carotenoids. Nutr. Cancer 1997, 27, 169–173. [Google Scholar] [CrossRef]
- Tarantilis, P.A.; Morjani, H.; Polissiou, M.; Manfait, M. Inhibition of growth and induction of differentiation of promyelocytic leukemia (HL-60) by carotenoids from Crocus sativus L. Anticancer Res. 1994, 14, 1913–1918. [Google Scholar]
- Danilenko, M.; Wang, X.; Studzinski, G.P. Carnosic acid and promotion of monocytic differentiation of HL60-G cells initiated by other agents. J. Natl. Cancer Inst. 2001, 93, 1224–1233. [Google Scholar] [CrossRef]
- Steiner, M.; Priel, I.; Giat, J.; Levy, J.; Sharoni, Y.; Danilenko, M. Carnosic acid inhibits proliferation and augments differentiation of human leukemic cells induced by 1,25-dihydroxyvitamin D3 and retinoic acid. Nutr. Cancer 2001, 41, 135–144. [Google Scholar]
- Stahl, W.; von Laar, J.; Martin, H.D.; Emmerich, T.; Sies, H. Stimulation of gap junctional communication: Comparison of acyclo-retinoic acid and lycopene. Arch. Biochem. Biophys. 2000, 373, 271–274. [Google Scholar] [CrossRef]
- Ben-Dor, A.; Nahum, A.; Danilenko, M.; Giat, Y.; Stahl, W.; Martin, H.D.; Emmerich, T.; Noy, N.; Levy, J.; Sharoni, Y. Effects of acyclo-retinoic acid and lycopene on activation of the retinoic acid receptor and proliferation of mammary cancer cells. Arch. Biochem. Biophys. 2001, 391, 295–302. [Google Scholar] [CrossRef]
- Muto, Y.; Moriwaki, H.; Omori, M. In vitro binding affinity of novel synthetic polyprenoids (polyprenoic acids) to cellular retinoid-binding proteins. Gann 1981, 72, 974–977. [Google Scholar]
- Araki, H.; Shidoji, Y.; Yamada, Y.; Moriwaki, H.; Muto, Y. Retinoid agonist activities of synthetic geranyl geranoic acid derivatives. Biochem. Biophys. Res. Commun. 1995, 209, 66–72. [Google Scholar] [CrossRef]
- Butler, R.; Mitchell, S.H.; Tindall, D.J.; Young, C.Y. Nonapoptotic cell death associated with S-phase arrest of prostate cancer cells via the peroxisome proliferator-activated receptor gamma ligand, 15-deoxy-delta12,14-prostaglandin J2. Cell Growth Differ. 2000, 11, 49–61. [Google Scholar]
- Kubota, T.; Koshizuka, K.; Williamson, E.A.; Asou, H.; Said, J.W.; Holden, S.; Miyoshi, I.; Koeffler, H.P. Ligand for peroxisome proliferator-activated receptor gamma (troglitazone) has potent antitumor effect against human prostate cancer both in vitro and in vivo. Cancer Res. 1998, 58, 3344–3352. [Google Scholar]
- Mueller, E.; Smith, M.; Sarraf, P.; Kroll, T.; Aiyer, A.; Kaufman, D.S.; Oh, W.; Demetri, G.; Figg, W.D.; Zhou, X.P.; et al. Effects of ligand activation of peroxisome proliferator-activated receptor gamma in human prostate cancer. Proc. Natl. Acad. Sci. USA 2000, 97, 10990–10995. [Google Scholar]
- Yasui, Y.; Kim, M.; Tanaka, T. PPAR Ligands for Cancer Chemoprevention. PPAR Res. 2008, 2008, 548919. [Google Scholar]
- Jaiswal, A.K. Regulation of genes encoding NAD(P)H:quinone oxidoreductases. Free Radic. Biol. Med. 2000, 29, 254–262. [Google Scholar] [CrossRef]
- Venugopal, R.; Jaiswal, A.K. Nrf1 and Nrf2 positively and c-Fos and Fra1 negatively regulate the human antioxidant response element-mediated expression of NAD(P)H:quinone oxidoreductase1 gene. Proc. Natl. Acad. Sci. USA 1996, 93, 14960–14965. [Google Scholar] [CrossRef]
- Wang, X.D.; Liu, C.; Bronson, R.T.; Smith, D.E.; Krinsky, N.I.; Russell, M. Retinoid signaling and activator protein-1 expression in ferrets given β-carotene supplements and exposed to tobacco smoke. J. Natl. Cancer Inst. 1999, 91, 60–66. [Google Scholar]
- Xie, W.; Evans, R.M. Orphan nuclear receptors: The exotics of xenobiotics. J. Biol. Chem. 2001, 276, 37739–37742. [Google Scholar]
- Altucci, L.; Gronemeyer, H. The promise of retinoids to fight against cancer. Nat. Rev. Cancer 2001, 1, 181–193. [Google Scholar] [CrossRef]
- Pavan, B.; Biondi, C.; Dalpiaz, A. Nuclear retinoic acid receptor β as a tool in chemoprevention trials. Curr. Med. Chem. 2006, 13, 3553–3563. [Google Scholar] [CrossRef]
- Shimizu, M.; Sakai, H.; Shirakami, Y.; Iwasa, J.; Yasuda, Y.; Kubota, M.; Takai, K.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Acyclic retinoid inhibits diethylnitrosamine-induced liver tumorigenesis in obese and diabetic C57BLKS/J- +Leprdb/+Leprdb mice. Cancer Prev. Res. (Phila.) 2011, 4, 128–136. [Google Scholar]
- Moriwaki, H.; Shimizu, M.; Okuno, M.; Nishiwaki-Matsushima, R. Chemoprevention of liver carcinogenesis with retinoids: Basic and clinical aspects. Hepatol. Res. 2007, 37, S299–S302. [Google Scholar] [CrossRef]
- Fabricius, E.M.; Kruse-Boitschenko, U.; Schneeweiss, U.; Wildner, G.P.; Hoffmeister, B.; Raguse, J.D. Model examination of chemoprevention with retinoids in squamous cell carcinomas of the head and neck region and suitable biomarkers for chemoprevention. Int. J. Oncol. 2011, 39, 1083–1097. [Google Scholar]
- Prakash, P.; Liu, C.; Hu, K.Q.; Krinsky, N.I.; Russell, R.M.; Wang, X.D. β-Carotene and β-apo-14'-carotenoic acid prevent the reduction of retinoic acid receptor β in benzo[a]pyrene-treated normal human bronchial epithelial cells. J. Nutr. 2004, 134, 667–673. [Google Scholar]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Schmidt, M.V.; Brune, B.; von Knethen, A. The nuclear hormone receptor PPARgamma as a therapeutic target in major diseases. ScientificWorldJournal 2010, 10, 2181–2197. [Google Scholar] [CrossRef]
- Youssef, J.; Badr, M. Peroxisome proliferator-activated receptors and cancer: Challenges and opportunities. Br. J. Pharmacol. 2011, 164, 68–82. [Google Scholar] [CrossRef]
- Sarraf, P.; Mueller, E.; Smith, W.M.; Wright, H.M.; Kum, J.B.; Aaltonen, L.A.; de la Chapelle, A.; Spiegelman, B.M.; Eng, C. Loss-of-function mutations in PPAR gamma associated with human colon cancer. Mol. Cell 1999, 3, 799–804. [Google Scholar] [CrossRef]
- Simone, R.E.; Russo, M.; Catalano, A.; Monego, G.; Froehlich, K.; Boehm, V.; Palozza, P. Lycopene inhibits NF-kB-mediated IL-8 expression and changes redox and PPARgamma signalling in cigarette smoke-stimulated macrophages. PLoS One 2011, 6, e19652. [Google Scholar]
- Yang, C.M.; Lu, I.H.; Chen, H.Y.; Hu, M.L. Lycopene inhibits the proliferation of androgen-dependent human prostate tumor cells through activation of PPARγ-LXRα-ABCA1 pathway. J. Nutr. Biochem. 2012, 23, 8–17. [Google Scholar] [CrossRef]
- Xie, W.; Barwick, J.L.; Simon, C.M.; Pierce, A.M.; Safe, S.; Blumberg, B.; Guzelian, P.S.; Evans, R.M. Reciprocal activation of xenobiotic response genes by nuclear receptors SXR/PXR and CAR. Genes Dev. 2000, 14, 3014–3023. [Google Scholar] [CrossRef]
- Moore, L.B.; Goodwin, B.; Jones, S.A.; Wisely, G.B.; Serabjit-Singh, C.J.; Willson, T.M.; Collins, J.L.; Kliewer, S.A. St. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 7500–7502. [Google Scholar]
- Ramos-Gomez, M.; Kwak, M.K.; Dolan, P.M.; Itoh, K.; Yamamoto, M.; Talalay, P.; Kensler, T.W. Sensitivity to carcinogenesis is increased and chemoprotective efficacy of enzyme inducers is lost in nrf2 transcription factor-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 3410–3415. [Google Scholar]
- Kim, Y.C.; Masutani, H.; Yamaguchi, Y.; Itoh, K.; Yamamoto, M.; Yodoi, J. Hemin-induced activation of the thioredoxin gene by Nrf2. A differential regulation of the antioxidant responsive element by a switch of its binding factors. J. Biol. Chem. 2001, 276, 18399–18406. [Google Scholar]
- Dhakshinamoorthy, S.; Jaiswal, A.K. Functional characterization and role of INrf2 in antioxidant response element-mediated expression and antioxidant induction of NAD(P)H:quinone oxidoreductase1 gene. Oncogene 2001, 20, 3906–3917. [Google Scholar] [CrossRef]
- Hayes, J.D.; McMahon, M. Molecular basis for the contribution of the antioxidant responsive element to cancer chemoprevention. Cancer Lett. 2001, 174, 103–113. [Google Scholar] [CrossRef]
- Kwak, M.K.; Egner, P.A.; Dolan, P.M.; Ramos-Gomez, M.; Groopman, J.D.; Itoh, K.; Yamamoto, M.; Kensler, T.W. Role of phase 2 enzyme induction in chemoprotection by dithiolethiones. Mutat. Res. 2001, 480–481, 305–315. [Google Scholar]
- Kong, A.N.; Owuor, E.; Yu, R.; Hebbar, V.; Chen, C.; Hu, R.; Mandlekar, S. Induction of xenobiotic enzymes by the MAP kinase pathway and the antioxidant or electrophile response element (ARE/EpRE). Drug Metab. Rev. 2001, 33, 255–271. [Google Scholar] [CrossRef]
- Xu, C.; Li, C.Y.; Kong, A.N. Induction of phase I, II and III drug metabolism/transport by xenobiotics. Arch. Pharm. Res. 2005, 28, 249–268. [Google Scholar] [CrossRef]
- Morimitsu, Y.; Nakagawa, Y.; Hayashi, K.; Fujii, H.; Kumagai, T.; Nakamura, Y.; Osawa, T.; Horio, F.; Itoh, K.; Iida, K.; et al. A sulforaphane analogue that potently activates the Nrf2-dependent detoxification pathway. J. Biol. Chem. 2002, 277, 3456–3463. [Google Scholar]
- Angel, P.; Karin, M. The role of Jun, Fos and the AP-1 complex in cell-proliferation and transformation. Biochim. Biophys. Acta 1991, 1072, 129–157. [Google Scholar]
- Albanese, C.; Johnson, J.; Watanabe, G.; Eklund, N.; Vu, D.; Arnold, A.; Pestell, R.G. Transforming p21ras mutants and c-Ets-2 activate the cyclin D1 promoter through distinguishable regions. J. Biol. Chem. 1995, 270, 23589–23597. [Google Scholar]
- Palozza, P.; Parrone, N.; Catalano, A.; Simone, R. Tomato lycopene and inflammatory cascade: Basic interactions and clinical implications. Curr. Med. Chem. 2010, 17, 2547–2563. [Google Scholar] [CrossRef]
- Polakis, P. Wnt signaling and cancer. Genes Dev. 2000, 14, 1837–1851. [Google Scholar]
- Schulenburg, A.; Cech, P.; Herbacek, I.; Marian, B.; Wrba, F.; Valent, P.; Ulrich-Pur, H. CD44-positive colorectal adenoma cells express the potential stem cell markers musashi antigen (msi1) and ephrin B2 receptor (EphB2). J. Pathol. 2007, 213, 152–160. [Google Scholar] [CrossRef]
- Cohen, P.; Frame, S. The renaissance of GSK3. Nat. Rev. Mol. Cell Biol. 2001, 2, 769–776. [Google Scholar] [CrossRef]
- Korkaya, H.; Paulson, A.; Charafe-Jauffret, E.; Ginestier, C.; Brown, M.; Dutcher, J.; Clouthier, S.G.; Wicha, M.S. Regulation of mammary stem/progenitor cells by PTEN/Akt/β-catenin signaling. PLoS Biol. 2009, 7, e1000121. [Google Scholar]
- Tang, F.Y.; Shih, C.J.; Cheng, L.H.; Ho, H.J.; Chen, H.J. Lycopene inhibits growth of human colon cancer cells via suppression of the Akt signaling pathway. Mol. Nutr. Food Res. 2008, 52, 646–654. [Google Scholar] [CrossRef]
- Lin, M.C.; Wang, F.Y.; Kuo, Y.H.; Tang, F.Y. Cancer chemopreventive effects of lycopene: Suppression of MMP-7 expression and cell invasion in human colon cancer cells. J. Agric. Food Chem. 2011, 59, 11304–11318. [Google Scholar] [CrossRef]
- Tanaka, T.; Suzuki, R. Inflammation and cancer. In Cancer: Disease Progression and Chemoprevention; Tanaka, T., Ed.; Research Signpost: Kerala, India, 2007; pp. 27–44. [Google Scholar]
- Tanaka, T.; Kohno, H.; Suzuki, R.; Yamada, Y.; Sugie, S.; Mori, H. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003, 94, 965–973. [Google Scholar] [CrossRef]
- Kim, H. Inhibitory mechanism of lycopene on cytokine expression in experimental pancreatitis. Ann. N.Y. Acad. Sci. 2011, 1229, 99–102. [Google Scholar]
- Erkan, M.; Reiser-Erkan, C.; Michalski, C.W.; Kleeff, J. Tumor microenvironment and progression of pancreatic cancer. Exp. Oncol. 2010, 32, 128–131. [Google Scholar]
- Nitsche, C.; Simon, P.; Weiss, F.U.; Fluhr, G.; Weber, E.; Gartner, S.; Behn, C.O.; Kraft, M.; Ringel, J.; Aghdassi, A.; et al. Environmental risk factors for chronic pancreatitis and pancreatic cancer. Dig. Dis. 2011, 29, 235–242. [Google Scholar] [CrossRef]
- Gallicchio, L.; Boyd, K.; Matanoski, G.; Tao, X.G.; Chen, L.; Lam, T.K.; Shiels, M.; Hammond, E.; Robinson, K.A.; Caulfield, L.E.; et al. Carotenoids and the risk of developing lung cancer: A systematic review. Am. J. Clin Nutr. 2008, 88, 372–383. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer Chemoprevention by Carotenoids. Molecules 2012, 17, 3202-3242. https://doi.org/10.3390/molecules17033202
Tanaka T, Shnimizu M, Moriwaki H. Cancer Chemoprevention by Carotenoids. Molecules. 2012; 17(3):3202-3242. https://doi.org/10.3390/molecules17033202
Chicago/Turabian StyleTanaka, Takuji, Masahito Shnimizu, and Hisataka Moriwaki. 2012. "Cancer Chemoprevention by Carotenoids" Molecules 17, no. 3: 3202-3242. https://doi.org/10.3390/molecules17033202
APA StyleTanaka, T., Shnimizu, M., & Moriwaki, H. (2012). Cancer Chemoprevention by Carotenoids. Molecules, 17(3), 3202-3242. https://doi.org/10.3390/molecules17033202




