Phenolic Alkaloids from Menispermum dauricum Rhizome Protect against Brain Ischemia Injury via Regulation of GLT-1, EAAC1 and ROS Generation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of PAM on OGD-Reoxygenation Induced Cell Injury
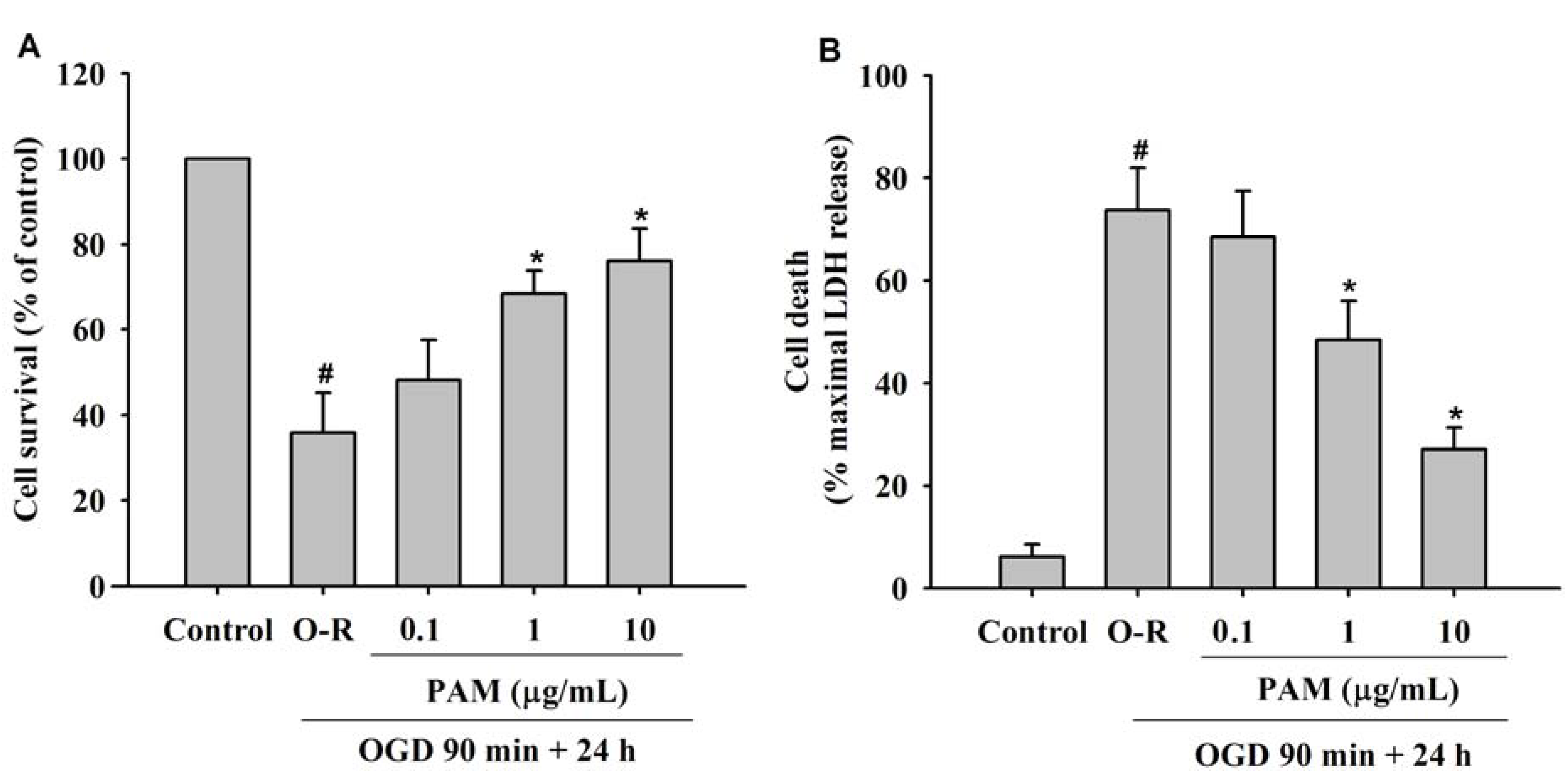
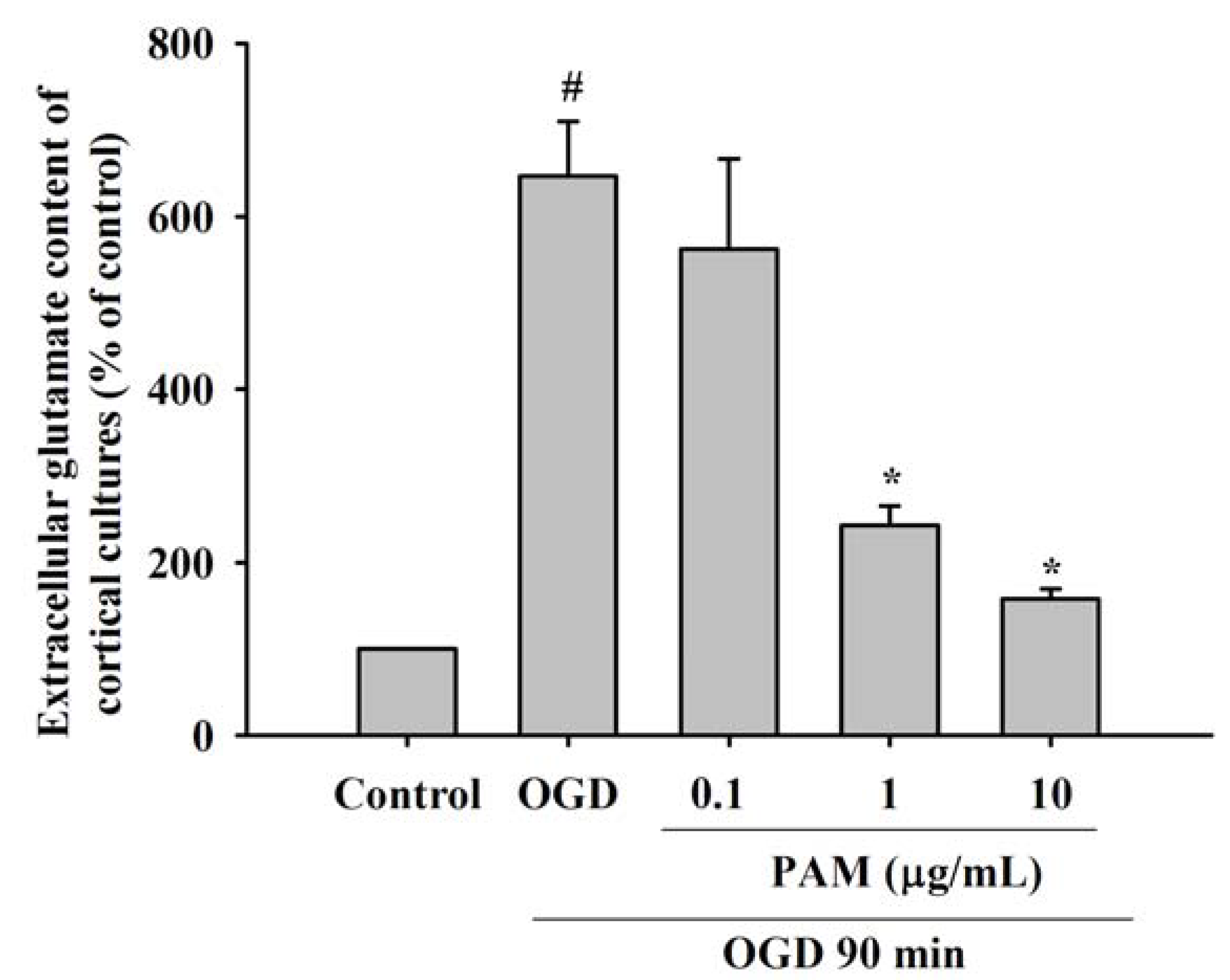
2.2. Effects of PAM on OGD-Evoked Increase of Extracellular Glutamate Content
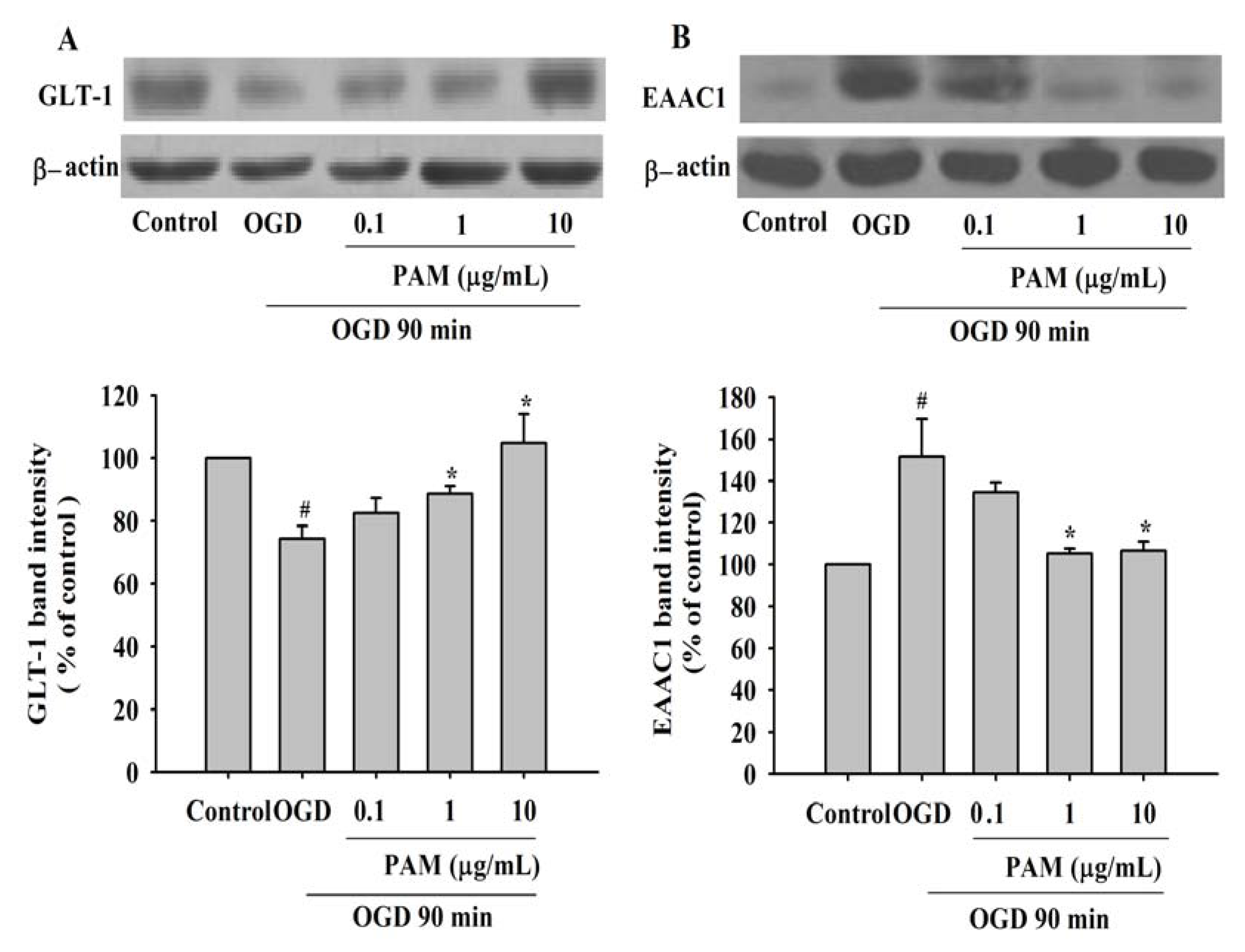
2.3. Effects of PAM on the Level of GLT-1 and EAAC1 after OGD 90 min
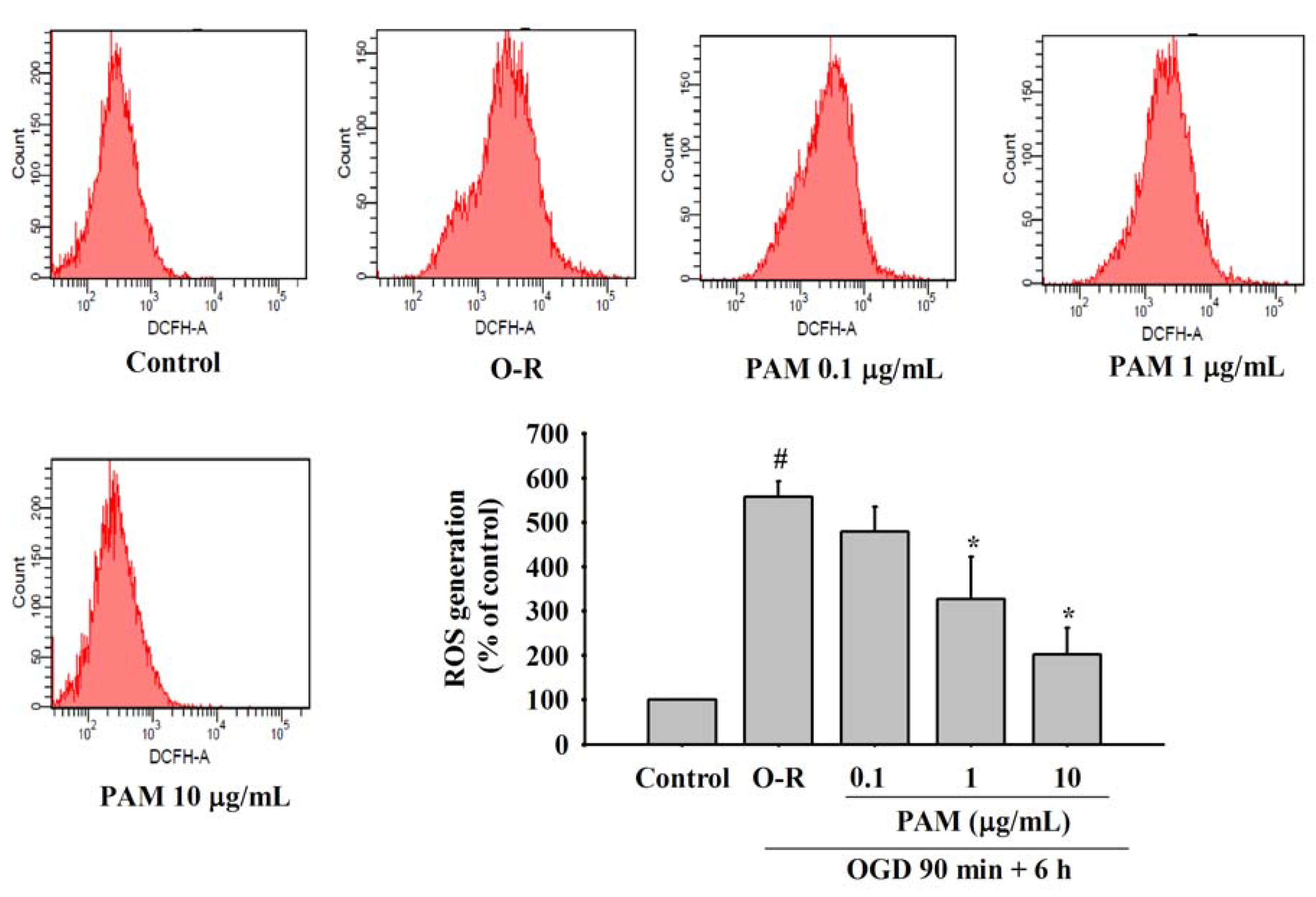
2.4. Effects of PAM on OGD-Reoxygenation Evoked Increase of Intracellular ROS Generation
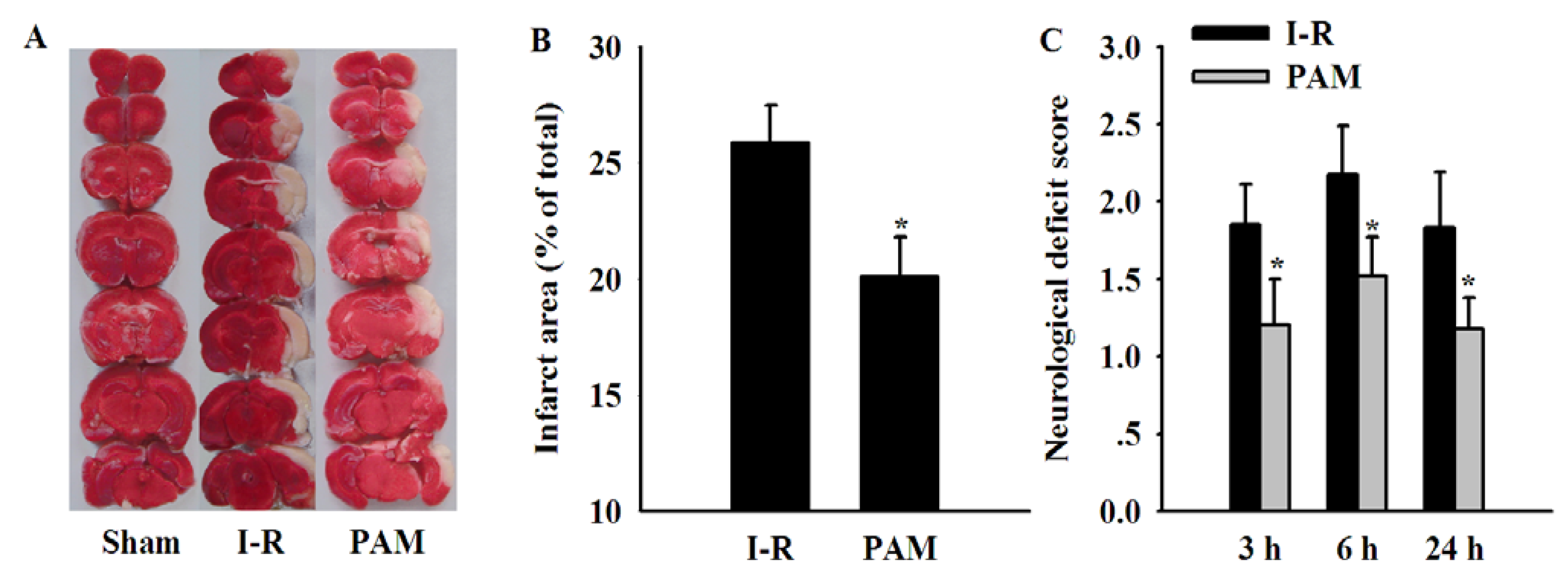
2.5. Effects of PAM on Cerebral Infarct Area and Neurological Deficit Score
3. Experimental
3.1. Chemicals
3.2. Preparation of PAM
3.3. Primary Rat Cortical Cultures and Oxygen-Glucose Deprivation Followed by Reoxygenation
3.4. MTT Reduction Test and LDH Assay
3.5. Glutamate Assay in the Culture Medium
3.6. Western Blot Analysis of GLT-1 and EAAC1
3.7. Intracellular ROS Measurement
3.8. Animals
3.9. Cerebral Ischemia-Reperfusion Model in Rat and Drug Administration
3.10. Neurological Deficit Evaluation and Infarct Size Measurement
3.11. Statistical Analysis
4. Conclusions
Acknowledgements
- Samples Availability: Samples of the compound PAM is available from the authors.
References and Notes
- Doyle, K.P.; Simon, R.P.; Stenzel-Poore, M.P. Mechanisms of ischemic brain damage. Neuropharmacology 2008, 55, 310–318. [Google Scholar] [CrossRef]
- Lo, E.H.; Dalkara, T.; Moskowitz, M.A. Mechanisms, challenges and opportunities in stroke. Nat. Rev. Neurosci. 2003, 4, 399–415. [Google Scholar]
- Gibson, C.J.; Meyer, R.C.; Hamm, R.J. Traumatic brain injury and the effects of diazepam, diltiazem, and MK-801 on GABA-A receptor subunit expression in rat hippocampus. J. Biomed. Sci. 2010, 17. [Google Scholar] [CrossRef]
- Rothstein, J.D.; Martin, L.; Levey, A.L.; Dykes-Hoberg, M.; Jin, L.; Wu, D.; Nash, N.; Kuncl, R.W. Localization of neuronal and glial glutamate transporters. Neuron 1994, 13, 713–725. [Google Scholar] [CrossRef]
- Lehre, K.P.; Levy, L.M.; Ottersen, O.P.; Storm-Mathisen, J.; Danbort, N.C. Differential expression of two glutamate transporters in rat brain: Quantitative and immunocytochemical observations. J. Neurosci. 1995, 5, 1836–1853. [Google Scholar]
- Nigam, S.; Schewe, T. Phospholipase A(2)s and lipid peroxidation. Biochim. Biophys. Acta 1488, 167–181. [Google Scholar]
- Chan, P.H. Mitochondria and neuronal death/survival signaling pathways in cerebral ischemia. Neurochem. Res. 2004, 29, 1943–1949. [Google Scholar] [CrossRef]
- Green, A.R. Pharmacological approaches to acute ischaemic stroke: Reperfusion certainly, neuroprotection possibly. Brit. J. Pharmacol. 2008, 153 (Suppl. 1), 325–338. [Google Scholar]
- Liu, Q.N.; Zhang, L.; Gong, P.L.; Yang, X.Y.; Zeng, F.D. Inhibitory effects of dauricine on early afterdepolarizations and L-type calcium current. Can. J. Physiol. Pharm. 2009, 87, 954–962. [Google Scholar] [CrossRef]
- Liu, Q.N.; Zhang, L.; Gong, P.L.; Yang, X.Y.; Zeng, F.D. Daurisoline suppressed early afterdepolarizations and inhibited L-type calcium current. Am. J. Chin. Med. 2010, 38, 37–49. [Google Scholar] [CrossRef]
- Kong, X.Y.; Gong, P.L. Effect of phenolic alkaloids of Menispermum dauricum on thrombosis and platelet aggregation. Yao Xue Xue Bao 2005, 40, 916–919. [Google Scholar]
- Qian, J.Q. Cardiovascular pharmacological effects of bisbenzylisoquinoline alkaloid derivatives. Acta Pharmacol. Sin. 2002, 23, 1086–1092. [Google Scholar]
- Liu, J.G.; Li, R.; Liu, G.Q. l-S.R-daurisoline protects cultured hippocampal neurons against glutamate neurotoxicity by reducing nitric oxide production. Zhongguo Yao Li Xue Bao 1999, 20, 21–26. [Google Scholar]
- Yang, X.Y.; Jiang, S.Q.; Zhang, L.; Liu, Q.N.; Gong, P.L. Inhibitory effect of dauricine on inflammatory process following focal cerebral ischemia/reperfusion in rats. Am. J. Chin. Med. 2007, 35, 477–486. [Google Scholar] [CrossRef]
- Luo, H.; Peng, M.; Ye, H.; Chen, L.; Peng, A.; Tang, M.; Zhang, F.; Shi, J. Predictable and linear scale-up of four phenolic alkaloids separation from the roots of Menispermum dauricum using high-performance counter-current chromatography. J. Chromatogr. B 2010, 878, 1929–1933. [Google Scholar] [CrossRef]
- Wang, F.; Qu, L.; Lv, Q.; Guo, L.J. Effect of phenolic alkaloids from Menispermum dauricum on myocardial-cerebral ischemia-reperfusion injury in rabbits. Acta Pharmacol. Sin. 2001, 22, 1130–1134. [Google Scholar]
- Panickar, K.S.; Norenberg, M.D. Astrocytes in cerebral ischemic injury: Morphological and general considerations. Glia 2005, 50, 287–298. [Google Scholar] [CrossRef]
- Kawakami, Z.; Kanno, H.; Ueki, T.; Terawaki, K.; Tabuchi, M.; Ikarashi, Y.; Kase, Y. Neuroprotective effects of yokukansan, a traditional Japanese medicine, on glutamate-mediated excitotoxicity in cultured cells. Neuroscience 2009, 159, 1397–1407. [Google Scholar] [CrossRef]
- Ikeda, M.; Nakazawa, T.; Abe, K.; Kaneko, T.; Yamatsu, K. Extracellular accumulation of glutamate in the hippocampus induced by ischemia is not calcium dependent—In vitro and in vivo evidence. Neurosci. Lett. 1989, 96, 202–206. [Google Scholar] [CrossRef]
- Budd, S.L.; Nicholls, D.G. Mitochondria, calcium regulation, and acute glutamate excitotoxicity in cultured cerebellar granule cells. J. Neurochem. 1996, 67, 2282–2291. [Google Scholar]
- Khodorov, B. Glutamate-induced deregulation of calcium homeostasis and mitochondrial dysfunction in mammalian central neurones. Prog. Biophys. Mol. Biol. 2004, 86, 279–351. [Google Scholar] [CrossRef]
- David, J.R.; Takeo, O.; David, A. Glutamate release in severe brain ischaemia is mainly by reversed uptake. Nature 2000, 403, 316–321. [Google Scholar] [CrossRef]
- Rao, V.L.; Bowen, K.K.; Dempsey, R.J. Transient focal cerebral ischemia down-regulates glutamate transporters GLT-1 and EAAC1 expression in rat brain. Neurochem. Res. 2001, 26, 497–502. [Google Scholar] [CrossRef]
- Esposito, F.; Ammendola, R.; Faraonio, R.; Russo, T.; Cimino, F. Redox control of signal transduction, gene expression and cellular senescence. Neurochem. Res. 2004, 29, 617–628. [Google Scholar] [CrossRef]
- Hattori, I.; Takagi, Y.; Nakamura, H.; Nozaki, K.; Bai, J.; Kondo, N.; Sugino, T.; Nishimura, M.; Hashimoto, N.; Yodoi, J. Intravenous administration of thioredoxin decreases brain damage following transient focal cerebral ischemia in mice. Antioxid. Redox. Signal. 2004, 6, 81–87. [Google Scholar] [CrossRef]
- Taylor, J.M.; Crack, P.J. Impact of oxidative stress on neuronal survival. Clin. Exp. Pharmacol. Physiol. 2004, 31, 397–406. [Google Scholar] [CrossRef]
- Hall, E.D. Lipid Peroxidation. In Primer on Cerebrovascular Disease, 1st; Welch, K.M.A., Caplan, L.R., Reis, D.J., Siesjo, B.K., Wier, B., Eds.; Academic Press: San Diego, CA, USA, 1997; pp. 200–204. [Google Scholar]
- Siesjo, B.K. Pathophysiology and treatment of focal cerebral ischemia. Part I: Pathophysiology. J. Neurosurgery 1992, 77, 169–184. [Google Scholar]
- Altay, T.; McLaughlin, B.; Wu, J.Y.; Park, T.S.; Gidday, J.M. Slit modulates cerebrovascular inflammation and mediates neuroprotection against global cerebral ischemia. Exp. Neurol. 2007, 207, 186–194. [Google Scholar] [CrossRef]
- Huang, X.; Li, Q.; Li, H.; Guo, L. Neuroprotective and antioxidative effect of cactus polysaccharides in vivo and in vitro. Cell. Mol. Neurobiol. 2009, 29, 1211–1221. [Google Scholar] [CrossRef]
- Cheng, Z.Y.; He, W.; Zhou, X.X.; Lv, Q.; Xu, X.L.; Yang, S.S.; Zhao, C.M.; Guo, L.J. Cordycepin protects against cerebral ischemia/reperfusion injury in vivo and in vitro. Eur. J. Pharmacol. 2011, 664, 20–28. [Google Scholar] [CrossRef]
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar] [CrossRef]
- He, Z.; Sun, T.; Hu, H.Z.; Xu, X.L.; Guo, L.J. Effects of phenolic alkaloids of Menispermum dauricum on evoked potentials in hippocampus CA3 region in ischemic rats. Acta Med. Univ. Sci. Technol. Huazhong 2005, 34, 266–269. [Google Scholar]
- Swanson, R.A.; Shiraishi, K.; Morton, M.T.; Sharp, F.R. Methionine sulfoximine reduces cortical infarct size in rats after middle cerebral artery occlusion. Stroke 1990, 21, 322–327. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, B.; Chen, Y.; Sun, X.; Zhou, M.; Ding, J.; Zhan, J.-J.; Guo, L.-J. Phenolic Alkaloids from Menispermum dauricum Rhizome Protect against Brain Ischemia Injury via Regulation of GLT-1, EAAC1 and ROS Generation. Molecules 2012, 17, 2725-2737. https://doi.org/10.3390/molecules17032725
Zhao B, Chen Y, Sun X, Zhou M, Ding J, Zhan J-J, Guo L-J. Phenolic Alkaloids from Menispermum dauricum Rhizome Protect against Brain Ischemia Injury via Regulation of GLT-1, EAAC1 and ROS Generation. Molecules. 2012; 17(3):2725-2737. https://doi.org/10.3390/molecules17032725
Chicago/Turabian StyleZhao, Bo, Yang Chen, Xi Sun, Mei Zhou, Jie Ding, Jin-Jin Zhan, and Lian-Jun Guo. 2012. "Phenolic Alkaloids from Menispermum dauricum Rhizome Protect against Brain Ischemia Injury via Regulation of GLT-1, EAAC1 and ROS Generation" Molecules 17, no. 3: 2725-2737. https://doi.org/10.3390/molecules17032725
APA StyleZhao, B., Chen, Y., Sun, X., Zhou, M., Ding, J., Zhan, J.-J., & Guo, L.-J. (2012). Phenolic Alkaloids from Menispermum dauricum Rhizome Protect against Brain Ischemia Injury via Regulation of GLT-1, EAAC1 and ROS Generation. Molecules, 17(3), 2725-2737. https://doi.org/10.3390/molecules17032725



