Rapid and Sensitive LC-MS/MS Method for the Determination of Metoprolol in Beagle Dog Plasma with a Simple Protein Precipitation Treatment and Its Pharmacokinetic Applications
Abstract
:1. Introduction
2. Results and Discussion
2.1. LC-MS/MS Analysis
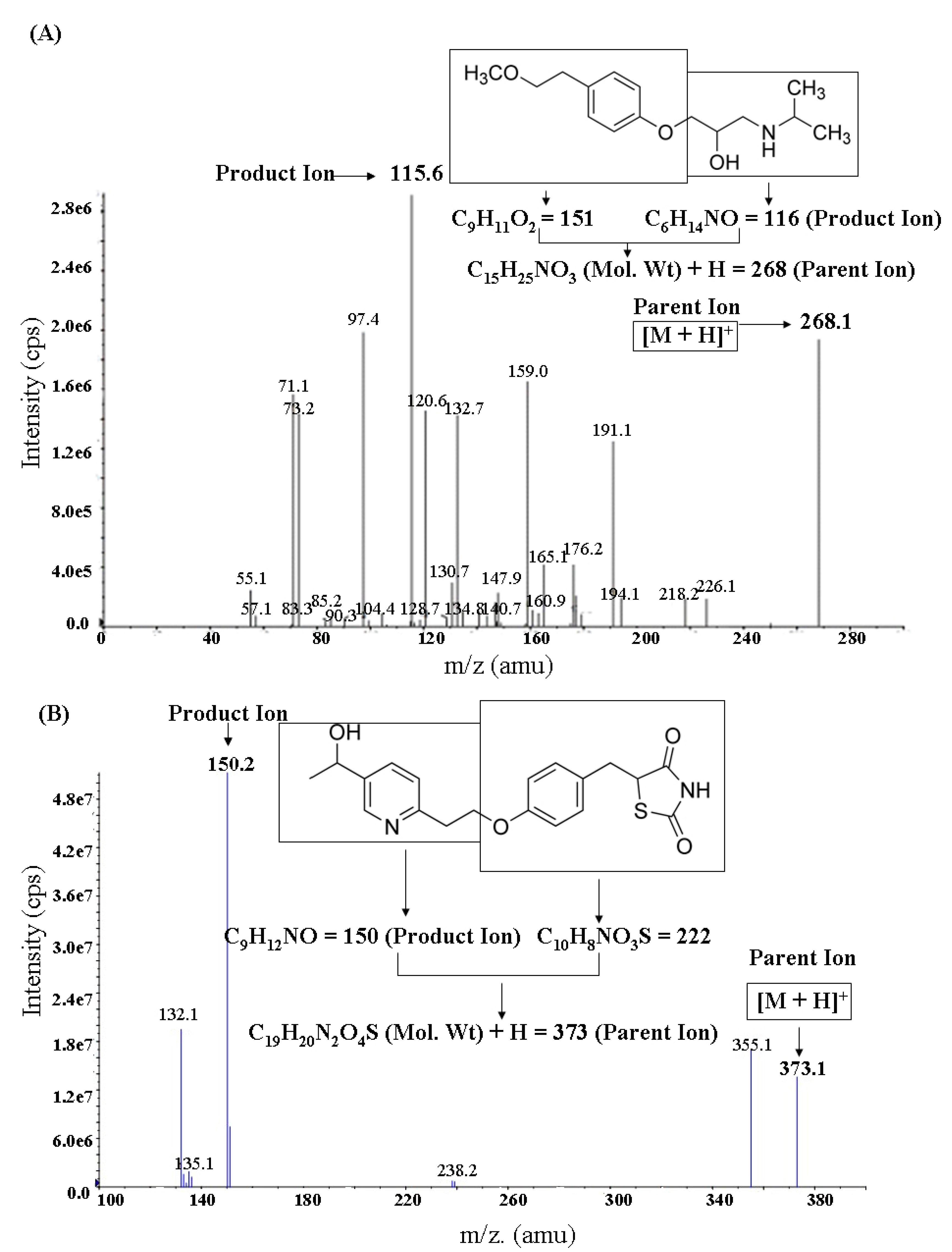
2.2. Preparation of Plasma Samples
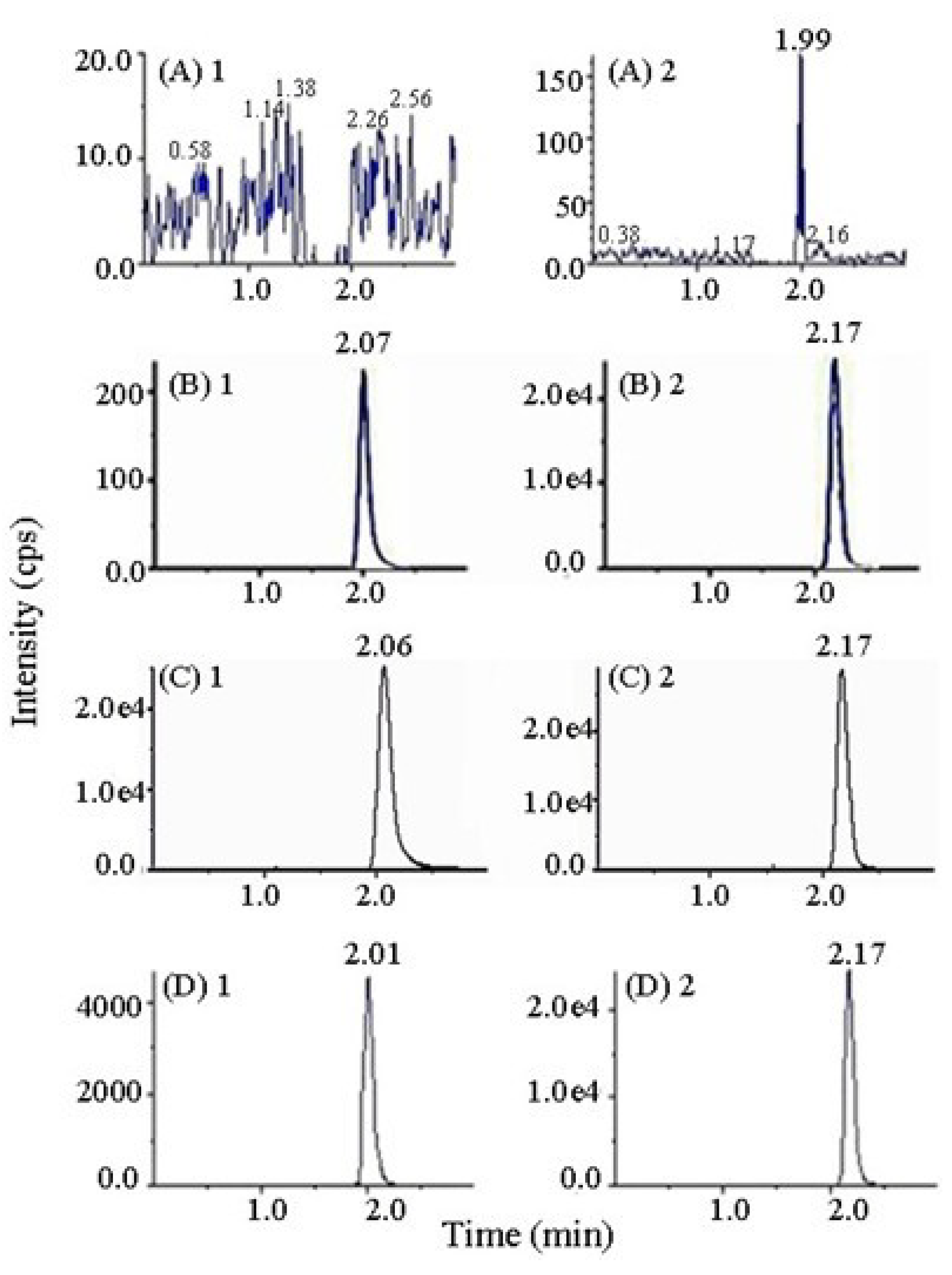
2.3. Method Validation
2.3.1. Selectivity
2.3.2. Linearity and Lower Limit of Quantification
 (n = 5). The lower limit of quantification (LLOQ) was 3.03 ng/mL. The precision and accuracy for the LLOQ was 8.72% and 99.96% ± 8.71% respectively (Table 1), which was acceptable for the analysis of biological samples.
(n = 5). The lower limit of quantification (LLOQ) was 3.03 ng/mL. The precision and accuracy for the LLOQ was 8.72% and 99.96% ± 8.71% respectively (Table 1), which was acceptable for the analysis of biological samples.2.3.3. Precision and Accuracy
| Concentration Added (ng/mL) | Concentration Observed (ng/mL) | Intra-day RSD (%) | Inter-Day RSD (%) | Accuracy (%) | ||
|---|---|---|---|---|---|---|
| First Day | Second Day | Third Day | ||||
| 3.03 | 3.03 ± 0.26 | 8.72 | - | - | - | 99.96 ± 8.71 |
| 6.06 | 5.84 ± 0.29 | 2.59 | 5.57 | 5.15 | 5.01 | 96.40 ± 4.82 |
| 181.68 | 172.96 ± 10.20 | 3.01 | 4.50 | 2.54 | 5.89 | 95.20 ± 5.61 |
| 363.36 | 347.91 ± 29.62 | 10.65 | 10.44 | 2.78 | 8.51 | 95.75 ± 8.15 |
2.3.4. Recovery and Matrix Effect
| Drug | Concentration Added (ng/mL) | Recovery (R) | Matrix Effect (ME) | ||
|---|---|---|---|---|---|
| R (%) | RSD (%) | ME (%) | RSD (%) | ||
| MP | 6.06 | 76.06 ± 3.38 | 4.45 | 93.67 ± 8.88 | 9.48 |
| 181.68 | 93.41 ± 2.47 | 2.64 | 100.85 ± 6.27 | 6.22 | |
| 363.36 | 95.25 ± 2.93 | 3.07 | 104.19 ± 5.71 | 5.48 | |
| IS | 86.40 | 93.71 ± 2.29 | 2.44 | 97.54 ± 2.15 | 2.20 |
2.3.5. Stability
| Conditions | Concentration Added (ng/mL) | Concentration Observed (ng/mL) | Accuracy (%) | RSD (%) |
|---|---|---|---|---|
| Long-term ( two months) | 6.06 | 5.97 ± 0.48 | 98.49 ± 7.91 | 8.03 |
| 181.68 | 177.06 ± 4.73 | 97.46 ± 2.60 | 2.67 | |
| 363.36 | 354.33 ± 6.27 | 97.52 ± 1.72 | 1.77 | |
| Short-term (24 h) | 6.06 | 6.18 ± 0.23 | 102.13 ± 3.80 | 3.69 |
| 181.68 | 177.24 ± 1.60 | 97.56 ± 0.89 | 0.91 | |
| 363.36 | 360.98 ± 26.25 | 99.30 ± 7.25 | 7.27 | |
| Frozen-thaw | 6.06 | 5.75 ± 0.36 | 95.01 ± 5.94 | 6.19 |
| 181.68 | 171.11 ± 8.31 | 94.18 ± 4.57 | 4.86 | |
| 363.36 | 342.22 ± 9.13 | 94.18 ± 2.51 | 2.66 | |
| Post-preparative (24 h) | 6.06 | 5.74 ± 0.35 | 94.77 ± 5.87 | 6.16 |
| 181.68 | 176.92 ± 1.80 | 97.51 ± 1.22 | 1.02 | |
| 363.36 | 354.17 ± 7.67 | 97.47 ± 2.11 | 2.17 |
2.4. Pharmacokinetics Study of MP Tartrate Tablets in Beagle Dogs
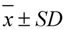 , n = 6).
, n = 6).
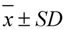 , n = 6).
, n = 6).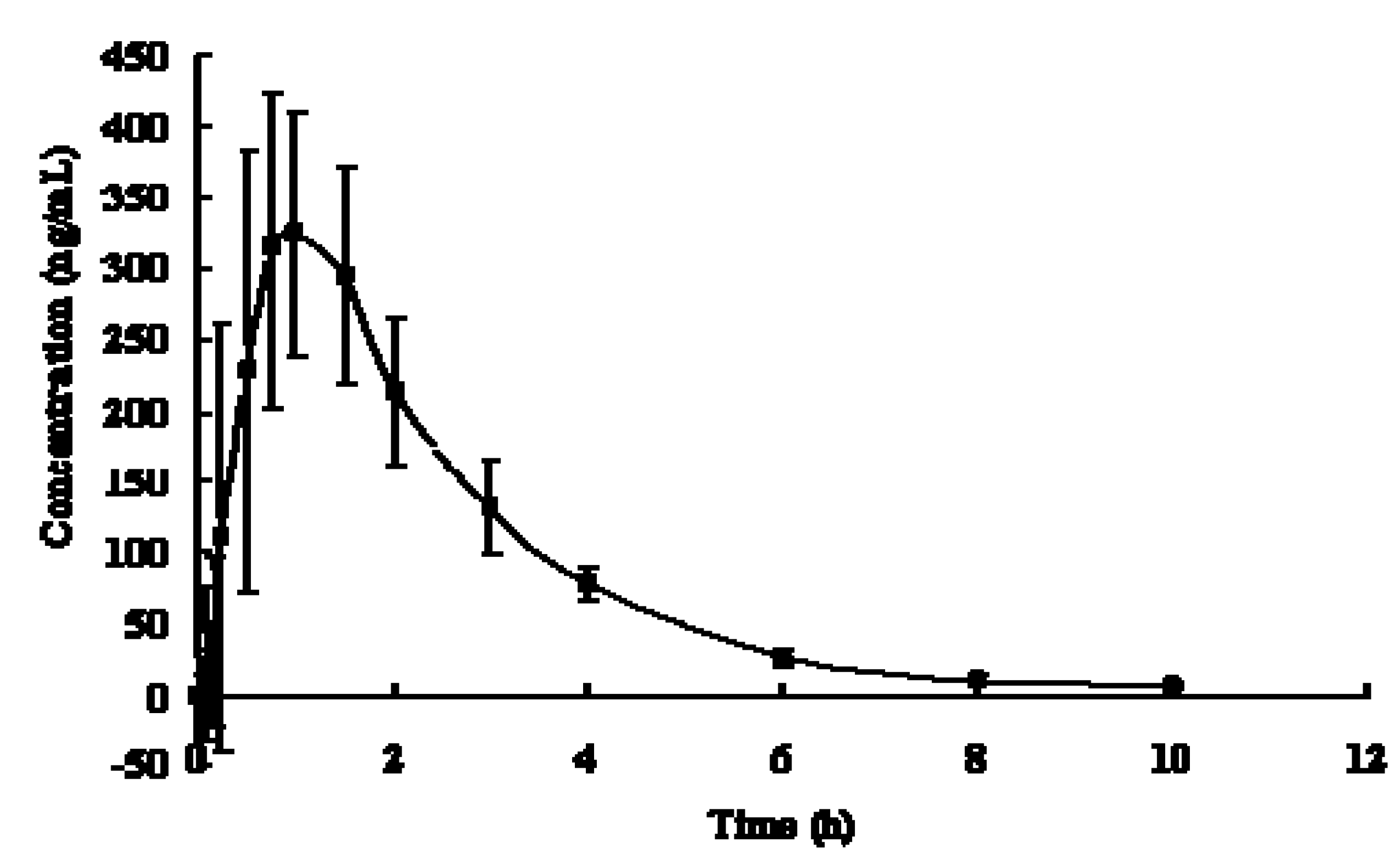
| Pharmacokinetic Parameters | Mean ± SD |
|---|---|
| AUC0-t (µg/L·h) | 919.88 ± 195.67 |
| AUC0-∞ (µg/L·h) | 934.42 ± 200.92 |
| MRT0-t (h) | 2.40 ± 0.19 |
| MRT0-∞ (h) | 2.56 ± 0.17 |
| t1/2 (h) | 1.73 ± 0.49 |
| Tmax (h) | 0.96 ± 0.33 |
| Cmax (ng/mL) | 349.12 ± 78.04 |
3. Experimental
3.1. Drugs and Reagents
3.2. Animals
3.3. LC-MS/MS Conditions and Instrumentation
3.4. Preparation of Standard and Quality Control Working Solutions
3.5. Preparation of Plasma Samples
3.6. Method Validation
3.6.1. Selectivity
3.6.2. Linearity and Lower Limit of Quantification
3.6.3. Precision and Accuracy
 is the average observed concentration of QC samples. The accuracy was calculated by comparing the observed concentration (
is the average observed concentration of QC samples. The accuracy was calculated by comparing the observed concentration ( 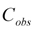 ) with the nominal concentrations (
) with the nominal concentrations ( 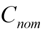 ) of analytes in QC samples according to formula (2):
) of analytes in QC samples according to formula (2):
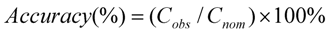
3.6.4. Recovery and Matrix Effect
3.6.5. Stability
3.7. Pharmacokinetics Study of MP Tartrate Tablets in Beagle Dogs
 ) and the time to reach
) and the time to reach 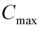 (
(  ) were estimated directly from the experimental observation values. The area under the plasma concentration-time curve from time zero to time of the last measurable concentration (
) were estimated directly from the experimental observation values. The area under the plasma concentration-time curve from time zero to time of the last measurable concentration ( 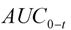 ) was calculated by the trapezoidal rule. The AUC extrapolated to infinity (
) was calculated by the trapezoidal rule. The AUC extrapolated to infinity ( 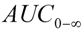 ) was calculated by formula (3), in which
) was calculated by formula (3), in which  is the last measurable plasma concentration,
is the last measurable plasma concentration, 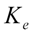 is the terminal elimination rate constant. The
is the terminal elimination rate constant. The 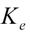 > was calculated from the slope of the terminal logarithm plot of plasma concentration versus time curve using linear least-squares regression method. The terminal elimination half-life (
> was calculated from the slope of the terminal logarithm plot of plasma concentration versus time curve using linear least-squares regression method. The terminal elimination half-life ( 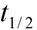 ) was calculated by formula (4):
) was calculated by formula (4):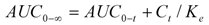
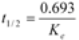
4. Conclusions
Acknowledgments
- Samples Availability: Please contact the authors.
References and Notes
- Zhang, F.; Cui, M.X.; Zhang, Z.Q. Quantitative determination of metoprolol by liquid chromatography mass spectrometry in rat plasma. Bull. Acad. Mil. Med. Sci. 2009, 33, 543–545. [Google Scholar]
- Gu, J.F.; Zhang, N.; Xi, Y.W.; Li, L.J.; Huang, G.H. Studies on explosion pulsatile-release tablets of metoprolol fumarate in vitro. Chin. Pharm. J. 2006, 41, 1560–1564. [Google Scholar]
- Jagdale, S.C.; Chede, S.M.; Gulwady, R.; Kuchekar, B.S.; Lokhande, P.D.; Shah, T.P.; Chabukswar, A.R. Pulsatile multiparticulate drug delivery system for metoprolol succinate. Arch. Pharm. Res. 2011, 34, 369–376. [Google Scholar] [CrossRef]
- Fang, J.; Semple, H.A.; Song, J. Determination of metoprolol, and its four metabolites in dog plasma. J. Chromatogr. B 2004, 809, 9–14. [Google Scholar] [CrossRef]
- Huang, G.H.; Zhang, N.; Gao, Y.H.; Zou, M.; He, D.W. The study of pharmacokinetics of metoprolol succinate sustained release tablets in dogs. J. Shandong Univ. (Health Sci.) 2004, 42, 56–59. [Google Scholar]
- Mistry, B.; Leslie, J.; Eddington, N.E. A sensitive assay of metoprolol and its major metabolite α-hydroxy metoprolol in human plasma and determination of dextromethorphan and its metabolite dextrorphan in urine with high performance liquid chromatography and fluorometric detection. J. Pharm. Biomed.Anal. 1998, 16, 1041–1049. [Google Scholar] [CrossRef]
- Musch, G.; Buelens, Y.; Massart, D.L. A strategy for the determination of beta blockers in plasma using solid-phase extraction in combination with high-performance liquid chromatography. J. Pharm. Biomed. Anal. 1989, 7, 483–497. [Google Scholar] [CrossRef]
- Wang, J.; Fu, L.Q.; Guan, Y.B.; Fang, Y.; Liu, Z.Y. Bioequivalence of metoprolol tartrate tablets in healthy volunteers. Chin. J. Clin. Pharmacol. Ther. 2006, 11, 1043–1047. [Google Scholar]
- Mistry, B.; Leslie, J.L.; Eddington, N.D. Enantiomeric separation of metoprolol and α-hydroxymetoprolol by liquid chromatography and fluorescence detection using a chiral stationary phase. J. Chromatogr. B 2001, 758, 153–161. [Google Scholar] [CrossRef]
- Zhang, B.K.; Yuan, H.Y.; Guo, J.P.; Li, H.D.; Peng, W.X.; Wang, F.; Lu, A.W. Study on relative bioavailability, pharmacokinetics and pharmacodynamics of metoprolol tartrate tablet. Chin. Hosp. Pharm. J. 2001, 21, 589–592. [Google Scholar]
- Guo, X.H.; Wang, X.; Wang, W.G. Pharmacokinetics and bioavailability of felodipine-metoprolol transdermal patch vs. market sustained-release tablets in rabbits. Chin. J. Drug Appl. Monit. 2008, 5, 6–15. [Google Scholar]
- Yang, P.H.; Tu, X.D. Studies on the membrane controlled release metoprolol tablet. J. Chin. Pharm. Univ. 1991, 22, 341–345. [Google Scholar]
- Guo, Y.L.; Wang, W.G.; Zhu, C.L.; Gan, Y. Preparation and in vitro-in vivo evaluation of metoprolol tartrate pulsatile controlled-release tablets. Chin. J. Pharm. 2010, 41, 424–429. [Google Scholar]
- Löbenberg, R.; Kim, J.S.; Amidon, G.L. Pharmacokinetics of an immediate release, a controlled release and a two pulse dosage form in dogs. Eur. J. Pharm. Biopharm. 2005, 60, 17–23. [Google Scholar] [CrossRef]
- Sarkar, A.K.; Ghosh, D.; Das, A.; Selvan, P.S.; Gowda, K.V.; Mandal, U.; Bose, A.; Agarwal, S.; Bhaumik, U.; Pal, T.K. Simultaneous determination of metoprolol succinate and amlodipine besylate in human plasma by liquid chromatography-tandem mass spectrometry method and its application in bioequivalence study. J. Chromatogr. B 2008, 873, 77–85. [Google Scholar] [CrossRef]
- Senthamil Selvan, P.; Pal, T.K. Chromatography-tandem mass spectrometry method for the simultaneous quantitation of metoprolol succinate and simvastatin in human plasma. J. Pharm. Biomed. Anal. 2009, 49, 780–785. [Google Scholar] [CrossRef]
- Venkateswarlu, P.; Kumar, B.N.; Seshaiah, K.; Prasad, W. Selective and sensitive method for the determination of metoprolol in human plasma using liquid chromatography coupled with tandem mass spectrometry. Acta Pharm. 2010, 60, 177–184. [Google Scholar] [CrossRef]
- Dong, S.Y.; Tong, X.H.; Li, J.C.; Wu, H.P.; Jiang, Z.W. Pharmacokinetics and bioequivalence of metoprolol sustained release tablets in Chinese healthy volunteers by LC-MS/MS. Chin. J. New Drugs Clin. Rem. 2008, 27, 429–433. [Google Scholar]
- Jensen, B.P.; Sharp, C.F.; Gardiner, S.J.; Begg, E.J. Development and validation of a stereoselective liquid chromatography-tandem mass spectrometry assay for quantification of S-and R-metoprolol in human plasma. J. Chromatogr. B 2008, 865, 48–54. [Google Scholar] [CrossRef]
- Yu, C.; Zou, M.J.; Shi, Y.J.; Cheng, G. Preparation of metoprolol tartrate pulsatile controlled-release pellets and its pharmacokinetics in beagle dogs. Chin. J. Pharm. 2011, 9, 30–37. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, S.; Wang, X.; Peng, K.; Ma, Z.; Zhang, X.; Fu, S.; Li, X.; Li, L.; Hong, A.; Jiang, J. Rapid and Sensitive LC-MS/MS Method for the Determination of Metoprolol in Beagle Dog Plasma with a Simple Protein Precipitation Treatment and Its Pharmacokinetic Applications. Molecules 2012, 17, 2663-2674. https://doi.org/10.3390/molecules17032663
Li S, Wang X, Peng K, Ma Z, Zhang X, Fu S, Li X, Li L, Hong A, Jiang J. Rapid and Sensitive LC-MS/MS Method for the Determination of Metoprolol in Beagle Dog Plasma with a Simple Protein Precipitation Treatment and Its Pharmacokinetic Applications. Molecules. 2012; 17(3):2663-2674. https://doi.org/10.3390/molecules17032663
Chicago/Turabian StyleLi, Sha, Xingli Wang, Kelong Peng, Zhiguo Ma, Xiaoqi Zhang, Shaolian Fu, Xiaofei Li, Linlin Li, Aihua Hong, and Jie Jiang. 2012. "Rapid and Sensitive LC-MS/MS Method for the Determination of Metoprolol in Beagle Dog Plasma with a Simple Protein Precipitation Treatment and Its Pharmacokinetic Applications" Molecules 17, no. 3: 2663-2674. https://doi.org/10.3390/molecules17032663
APA StyleLi, S., Wang, X., Peng, K., Ma, Z., Zhang, X., Fu, S., Li, X., Li, L., Hong, A., & Jiang, J. (2012). Rapid and Sensitive LC-MS/MS Method for the Determination of Metoprolol in Beagle Dog Plasma with a Simple Protein Precipitation Treatment and Its Pharmacokinetic Applications. Molecules, 17(3), 2663-2674. https://doi.org/10.3390/molecules17032663




