Volatile Organic Compounds from Centaurium erythraea Rafn (Croatia) and the Antimicrobial Potential of Its Essential Oil
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Headspace C. erythraea VOC Composition
| No. | Compound | RIHP-5MS | RIHP-FFAP | Area percentage (%)A B | |
|---|---|---|---|---|---|
| 1. | 2-Methylpentane * | <900 | / | 0.5 | 0.6 |
| 2. | 3-Methylpentane * | <900 | / | 1.1 | 1.7 |
| 3. | Hexane | <900 | < 900 | 0.6 | 2.0 |
| 4. | Ethyl acetate | <900 | 901 | - | 0.8 |
| 5. | Methylcyclopentane | <900 | / | 1.7 | 1.9 |
| 6. | Cyclohexane | <900 | / | 0.5 | 0.8 |
| 7. | Pentanal | <900 | / | - | 0.8 |
| 8. | Toluene | <900 | 1067 | 4.5 | 18.0 |
| 9. | Hexanal | <900 | 1105 | 1.5 | 4.8 |
| 10. | 1,4-Dimethylbenzene * | <900 | / | - | 0.4 |
| 11. | Heptanal | 905 | / | - | 0.4 |
| 12. | α-Thujene | 933 | / | - | 0.6 |
| 13. | α-Pinene | 941 | 1040 | - | 1.0 |
| 14. | Propylbenzene (Isocumene) | 960 | / | - | 0.6 |
| 15. | m-Ethyltoluene * | 968 | / | 0.9 | 1.7 |
| 16. | 1,2,4-Trimethylbenzene (Pseudocumene) | 976 | / | 0.9 | 1.0 |
| 17. | Sabinene | 982 | 1140 | 0.6 | 1.3 |
| 18. | β-Pinene | 984 | 1130 | - | 1.1 |
| 19. | o-Ethyltoluene * | 987 | / | 0.8 | 0.8 |
| 20. | 2-Pentylfuran | 996 | 1249 | 1.0 | 2.4 |
| 21. | 1,2,3-trimethylbenzene (Hemimellitene) | 1002 | / | 1.8 | 2.5 |
| 22. | p-Cymene | 1032 | 1293 | 2.8 | 3.5 |
| 23. | Limonene | 1036 | 1220 | 1.5 | 1.8 |
| 24. | 1,8-Cineole | 1039 | 1231 | 1.1 | 0.9 |
| 25. | γ-Terpinene | 1066 | / | 2.2 | 2.2 |
| 26. | α-Terpinolene | 1094 | / | - | 0.5 |
| 27. | Undecane | 1100 | 1100 | - | 0.7 |
| 28. | Linalool | 1107 | 1560 | 2.8 | 0.9 |
| 29. | Nonanal | 1109 | / | 2.9 | 1.8 |
| 30. | β-Thujone | 1112 | 1450 | 2.8 | 1.2 |
| 31. | Camphor | 1152 | 1548 | 5.1 | 2.0 |
| 32. | Menthone | 1162 | 1490 | 6.1 | 4.3 |
| 33. | Isomenthone | 1172 | 1519 | 0.9 | 1.6 |
| 34. | Menthol | 1183 | 1658 | 2.9 | 6.7 |
| 35. | Terpinen-4-ol | 1186 | 1623 | 10.3 | 6.9 |
| 36. | Naphthalene | 1190 | / | 8.2 | 1.8 |
| 37. | α-Terpineol | 1198 | 1717 | 1.5 | 0.6 |
| 38. | Decanal | 1211 | 1518 | 2.3 | 1.1 |
| 39. | Verbenone | 1217 | / | 0.7 | 0.4 |
| 40. | Bornyl acetate | 1295 | 1601 | 0.9 | 0.4 |
| 41. | Safrole | 1295 | / | 2.7 | - |
| 42. | Menthyl acetate | 1299 | / | 1.6 | 0.9 |
| 43. | Tridecane | 1300 | 1300 | 0.9 | 1.0 |
| 44. | α-Copaene | 1382 | 1510 | 2.0 | 0.8 |
| 45. | (E)-β-Damascenone | 1390 | 1848 | 1.1 | - |
| 46. | Tetradecane | 1400 | 1400 | 3.2 | 1.4 |
| 47. | Longifolene | 1409 | 1592 | 1.0 | 1.6 |
| 48. | β-Caryophyllene | 1425 | 1618 | 3.7 | 1.6 |
| 49. | trans-β-Farnesene | 1464 | 1677 | 2.5 | 0.7 |
| 50. | Hexadecane | 1600 | 1600 | 1.8 | 1.4 |
| 51. | Heptadecane | 1700 | 1700 | 1.0 | - |
| 52. | Nonadecane | 1900 | 1900 | 0.8 | - |
| Total identified (%) | 93.7 | 93.9 | |||
| No. | Compound | RIHP-5MS | RIHP-FFAP | Peak area (%)C D E | ||
|---|---|---|---|---|---|---|
| 1. | Ethyl acetate | <900 | 901 | - | - | 1.3 |
| 2. | Hexanal | <900 | 1105 | - | - | 1.1 |
| 3. | 4-Methyloctane * | <900 | / | - | 0.3 | - |
| 4. | 1,4-Dimethylbenzene * | <900 | / | - | 0.2 | - |
| 5. | Nonane | 900 | 900 | - | 8.6 | 0.2 |
| 6. | Undecane | 1100 | 1100 | - | 0.6 | - |
| 7. | Linalool | 1107 | 1560 | 3.0 | - | 3.9 |
| 8. | Nonanal | 1109 | / | 0.2 | - | 0.5 |
| 9. | β-Thujone | 1112 | 1450 | 0.8 | - | 1.1 |
| 10. | Camphor | 1152 | 1548 | 1.5 | - | 1.8 |
| 11. | Menthone | 1162 | 1490 | 2.5 | - | 3.3 |
| 12. | Isomenthone | 1172 | 1519 | 0.3 | - | 0.9 |
| 13. | Borneol | 1175 | 1723 | 1.4 | - | 1.6 |
| 14. | Menthol | 1183 | 1658 | 7.0 | - | 8.8 |
| 15. | Terpinen-4-ol | 1186 | 1623 | 1.2 | - | 1.5 |
| 16. | α-Terpineol | 1198 | 1717 | 1.1 | - | 1.4 |
| 17. | Methyl chavicol (Estragole) | 1205 | 1697 | 2.6 | 1.1 | 0.9 |
| 18. | Decanal | 1211 | 1518 | 0.1 | - | 0.3 |
| 19. | Pulegone | 1248 | 1677 | 0.2 | - | 0.8 |
| 20. | Carvone | 1252 | 1767 | 1.5 | - | 2.0 |
| 21. | Piperitone | 1263 | 1761 | 0.7 | - | 0.3 |
| 22. | Geraniol | 1264 | 1865 | 0.2 | - | - |
| 23. | Bornyl acetate | 1295 | 1601 | 0.7 | - | 0.9 |
| 24. | trans-Anethole | 1293 | 1860 | 3.6 | 1.5 | 1.0 |
| 25. | Menthyl acetate | 1299 | / | 0.3 | - | 0.7 |
| 26. | Thymol | 1307 | 2152 | 2.6 | - | 5.8 |
| 27. | Carvacrol | 1318 | 2189 | 6.1 | - | 13.8 |
| 28. | trans, trans-Deca-2,4-dienal | 1325 | 1839 | 0.2 | - | - |
| 29. | α-Copaene | 1382 | 1510 | 0.3 | 0.6 | - |
| 30. | (E)-β-Damascenone | 1390 | 1848 | 2.3 | 2.8 | |
| 31. | Tetradec-1-ene | 1397 | / | - | 0.5 | - |
| 32. | Tetradecane | 1400 | 1400 | - | 0.3 | - |
| 33. | Longifolene | 1409 | 1592 | - | 0.5 | - |
| 34. | Methyleugenol | 1414 | / | 0.3 | - | - |
| 35. | β-Caryophyllene | 1425 | 1618 | 0.9 | 1.1 | - |
| 36. | α-Humulene | 1460 | 1691 | - | 0.5 | - |
| 37. | Geranyl acetone | 1461 | 1875 | 1.3 | - | 0.7 |
| 38. | trans-β-Farnesene | 1464 | 1677 | 1.3 | 1.0 | - |
| 39. | α-Amorphene | 1483 | 1707 | 0.2 | 0.5 | - |
| 40. | Ar-curcumene | 1490 | 1791 | 0.2 | 0.7 | - |
| 41. | (E)-β-Ionone | 1493 | 1958 | 1.4 | - | 2.0 |
| 42. | α-Selinene | 1500 | 1747 | - | 0.8 | - |
| 43. | α-Muurolene | 1506 | 1745 | 0.3 | - | - |
| 44. | Pentadecane | 1500 | / | - | 1.1 | - |
| 45. | β-Bisabolene | 1515 | 1743 | 0.2 | 0.7 | - |
| 46. | γ-Cadinene | 1521 | 1814 | 0.3 | - | - |
| 47. | δ-Cadinene | 1531 | 1777 | 0.8 | 1.9 | - |
| 48. | Myristicin | 1533 | 2271 | - | - | 0.3 |
| 49. | α-Calacorene | 1551 | 1938 | 0.3 | 0.8 | - |
| 50. | Spathulenol | 1586 | 2088 | 1.3 | - | 0.9 |
| 51. | Caryophyllene oxide | 1589 | 1998 | 0.9 | - | 1.7 |
| 52. | Dodecanoic acid | 1594 | / | 1.0 | - | - |
| 53. | Hexadec-1-ene | 1598 | / | - | 1.7 | - |
| 54. | Viridiflorol | 1599 | 2059 | 0.8 | - | 1.1 |
| 55. | Hexadecane | 1600 | 1600 | - | 1.0 | - |
| 56. | α-Cadinol | 1665 | / | 0.7 | - | 1.6 |
| 57. | Ethyldibenzothiophene (isomer I) * | 1682 | / | 0.7 | - | - |
| 58. | Acorenone B | 1696 | / | 0.9 | - | 1.2 |
| 59. | Heptadecane | 1700 | 1700 | 0.3 | 1.1 | - |
| 60. | Farnesol * | 1710 | / | 0.5 | - | 0.6 |
| 61. | Pentadecanal | 1723 | / | 0.5 | - | 0.8 |
| 62. | Chromolaenin | 1727 | / | - | 2.1 | - |
| 63. | Ethyldibenzothiophene (isomer II) * | 1729 | / | - | 0.6 | - |
| 64. | Ethyldibenzothiophene (isomer III) * | 1734 | / | 0.2 | 1.5 | - |
| 65. | Aristolone | 1767 | / | 0.4 | - | 0.8 |
| 66. | Tetradecanoic acid | 1791 | / | 1.5 | 2.3 | 0.7 |
| 67. | Octadec-1-ene | 1799 | / | 2.6 | - | |
| 68. | Octadecane | 1800 | 1800 | 0.2 | 1.1 | - |
| 69. | Neophytadiene | 1849 | 1925 | 1.4 | 4.5 | - |
| 70. | Hexahydroxyfarnesyl acetone (Phytone) | 1857 | 2082 | 4.0 | - | 6.6 |
| 71. | Pentadecanoic acid | 1896 | / | 0.2 | - | - |
| 72. | Nonadecane | 1900 | 1900 | 0.7 | 4.6 | - |
| 73. | (E,E)-Farnesyl acetone | 1928 | 0.9 | - | 1.5 | |
| 74. | Methyl hexadecanoate | 1937 | / | 0.3 | - | 0.7 |
| 75. | Isophytol | 1960 | / | 0.3 | - | 0.2 |
| 76. | Hexadecanoic acid | 1963 | / | 5.7 | 2.2 | 6.8 |
| 77. | Eicos-3-ene * | 1998 | / | - | 2.3 | - |
| 78. | Eicosane | 2000 | 2000 | - | 1.5 | - |
| 79. | Heneicosane | 2100 | 2100 | 2.2 | 8.7 | - |
| 80. | Phytol * | 2135 | / | 1.9 | - | - |
| 81. | Linoleic acid | 2165 | / | 3.9 | - | 5.3 |
| 82. | Docosane | 2200 | 2200 | 0.5 | 2.6 | - |
| 83. | Tricosane | 2300 | 2300 | 6.8 | 25.7 | - |
| 84. | Tetracosane | 2400 | 2400 | 1.3 | 4.7 | - |
| 85. | 3-Methylbutyl 3-methylbutanoate | / | 1306 | 0.2 | - | - |
| 86. | α-Thujone | / | 1469 | 0.3 | - | 0.6 |
| 87. | Dihydroedulan II | / | 1514 | 0.3 | - | - |
| 88. | Dihydroedulan I | / | 1544 | 0.5 | - | - |
| 89. | Cuminal | / | 1815 | 0.2 | - | 0.3 |
| Total identified (%) | 89.4 | 94.1 | 91.1 | |||
2.2. C. erythraea Essential Oil Composition
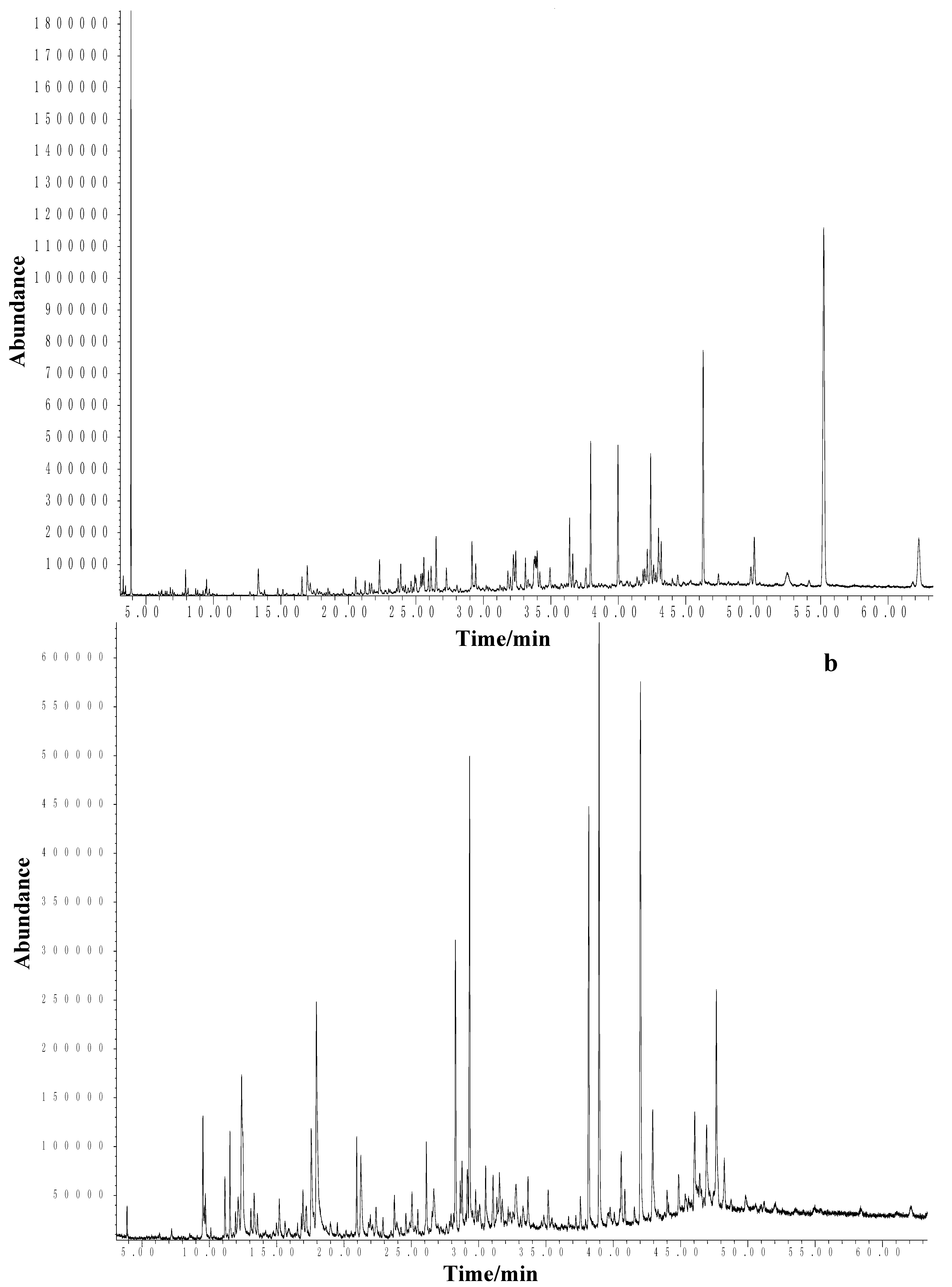
2.3. Unlocking Antimicrobial Potential of the Essential Oil
| Culture | Essential oil | Antibiotics | ||||
|---|---|---|---|---|---|---|
| Ampicilin | Norfloxacin | Ofloxacin | Tetracycline | |||
| Gram negative | Escherichia coli | 13 ± 0.577 * | 15 ± 2.828 | 26 ± 0 | 25 ± 0 | 20 ± 1.414 |
| Pseudomonas fluorescens | 0 ± 0 | ** | 40 ± 5.657 | 31 ± 0 | 22 ± 1.414 | |
| Salmonella enteritidis | 13 ± 1 | 24 ± 0 | 27 ± 0 | 26.5 ± 0.707 | 24.5 ± 0.707 | |
| Gram positive | Bacillus cereus | 7 ± 0.693 | 13 ± 0.707 | 22 ± 0 | 24 ± 0.707 | 22 ± 0.707 |
| Listeria monocytogenes | 0 ± 0 | 37 ± 0 | 30.5 ± 0.707 | 28.5 ± 0.707 | 37.5 ± 2.121 | |
| Staphylococcus aureus | 8 ± 0.289 | 45 ± 1.414 | 29.5 ± 3.536 | 29.5 ± 2.121 | 30.5 ± 3.536 | |
3. Experimental
3.1. Plant Material, Solvents and Isolation of the Essential Oil
3.2. Microcolumn Oil Fractionation
3.3. Headspace Solid-Phase Microextraction (HS-SPME)
3.4. Gas Chromatography and Mass Spectrometry
3.5. Strains and Antibacterial Testing
4. Conclusions
Acknowledgements
References and Notes
- Tahraoui, A.; Israili, Z.H.; Lyoussi, B. Acute and sub-chronic toxicity of a lyophilised aqueous extract of Centaurium erythraea in rodents. J. Ethnopharmacol. 2010, 132, 48–55. [Google Scholar] [CrossRef]
- Nikolova-Damyanova, B.; Handjieva, N. Quantitative determination of swertiamarin and gentiopicroside in Centarium erythrea and C. turcicum by densitometry. Phytochem. Anal. 1996, 7, 140–142. [Google Scholar] [CrossRef]
- Kumarasamy, Y.; Nahar, L.; Cox, P.J.; Jaspars, M.; Sarker, S.D. Bioactivity of secoiridoid glycosides from Centaurium erythraea. Phytomedicine 2003, 10, 344–347. [Google Scholar] [CrossRef]
- Kumarasamy, Y.; Nahar, L.; Sarker, S.D. Bioactivity of gentiopicroside from the aerial parts of Centaurium erythraea. Fitoterapia 2003, 74, 151–154. [Google Scholar] [CrossRef]
- Valentão, P.; Andrade, P.B.; Silva, A.M.S.; Moreira, M.M.; Seabra, R.M. Isolation and structural elucidation of 5-formyl-2,3-dihydroisocumarins from Centaurium erythraea aerial parts. Nat. Prod. Res. 2003, 15, 361–364. [Google Scholar]
- Sefi, M.; Fetoui, H.; Lachkar, N.; Tahraoui, A.; Lyoussi, B.; Boudawara, T.; Zeghal, N. Centarium erythrea (Gentianaceae) leaf extract alleviates streptozotocin-induced oxidative stress and β-cell damage in rat pancreas. J. Ethnopharmacol. 2011, 135, 243–250. [Google Scholar] [CrossRef]
- Valentão, P.; Fernandes, E.; Carvalho, F.; Andrade, P.B.; Seabre, R.M.; Bastos, M.L. Antioxidant Activity of Centaurium erythraea Infusion Evidenced by Its Superoxide Radical Scavenging and Xanthine Oxidase Inhibitory Activity. J. Agric. Food Chem. 2001, 49, 3476–3479. [Google Scholar] [CrossRef]
- Berkan, T.; Ustünes, L.; Lermioglu, F.; Ozer, A. Antiinflammatory, analgesic and antipyretic effects of an aqueous extract of Erythraea centaurium. Planta Med. 1991, 57, 34–37. [Google Scholar] [CrossRef]
- Kirbağ, S.; Zengin, F.; Kursat, M. Antimicrobial activities of extracts of some plants. Pak. J. Bot. 2009, 41, 2067–2070. [Google Scholar]
- Jovanović, O.; Radulović, N.; Stojanović, G.; Palić, R.; Zlatković, B.; Gudžić, B. Chemical composition of the essential oil of Centaurium erythraea Rafn (Gentianaceae) from Serbia. J. Essent. Oil Res. 2009, 21, 317–322. [Google Scholar] [CrossRef]
- Heiden, A.C.; Kobel, K.; Komenda, M.; Koppmann, R.; Shao, M.; Wildt, J. Toluene emissions from plants. Geophys. Res. Lett. 1999, 26, 1283–1286. [Google Scholar] [CrossRef]
- Azuma, H.; Toyota, M.; Asakawa, Y.; Kawano, S. Naphthalene—A constituent of Magnolia flowers. Phytochemistry 1996, 42, 999–1004. [Google Scholar] [CrossRef]
- Chen, J.; Henderson, G. Naphthalene in Formosan Subterranean Termite Carton Nests. J. Agric. Food Chem. 1998, 46, 2337–2339. [Google Scholar] [CrossRef]
- Daisy, B.H.; Strobel, G.A.; Castillo, U.; Ezra, D.; Sears, J.; Weaver, D.K.; Runyon, J.B. Naphthalene, an insect repellent, is produced by Muscodor vitigenus, a novel endophytic fungs. Microbiology 2002, 148, 3737–3741. [Google Scholar]
- Larick, D.K.; Hedrick, H.B.; Bailey, M.E.; Williams, J.E.; Hancock, D.L.; Garner, G.B.; Morrow, R.E. Flavour constituents of beef as influenced by forage- and grain-feeding. J. Food Sci. 1987, 52, 245–251. [Google Scholar] [CrossRef]
- Ramachandran, R.; Khan, Z.R.; Caballero, P.; Juliano, B.O. Olfactory sensitivity of two sympatric species of rice leaf folders (Lepidoptera: Pyralidae) to plant volatiles. J. Chem. Ecol. 1990, 16, 2647–2666. [Google Scholar] [CrossRef]
- Gorgen, G.; Boland, W. Biosynthesis of 1-alkenes in higher plants: stereochemical implications. A model study with Carthamus tinctorius (Asteraceae). Eur. J. Biochem. 1989, 185, 237–242. [Google Scholar] [CrossRef]
- Martinsm, C.M.C.; Mesquita, S.M.M.; Vaz, W.L.C. Cuticular waxes of the holm [Quercus ilex L. subsp. ballota (Desf.) Samp.] and cork (Q. suber L.) oaks. Phytochem. Anal. 1999, 10, 1–5. [Google Scholar] [CrossRef]
- Loayza, I.; Abujder, D.; Aranda, R.; Jakupovic, J.; Collin, G.; Deslauriers, H.; Jean, F.-I. Essential oils of Baccharis salicifolia, B. latifolia and B. dracunculifolia. Phytochemistry 1995, 38, 381–389. [Google Scholar]
- Dorman, H.J.; Deans, S.G. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef]
- Cosentino, S.; Tuberoso, C.I.G.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas, F. In vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Appl. Microbiol. 1999, 29, 130–135. [Google Scholar] [CrossRef]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of Antibacterial Action of Three Monoterpenes. Antimicrob. Agents Ch. 2005, 49, 2474–2478. [Google Scholar] [CrossRef]
- Rattanachaikunsopon, P.; Phumkhachorn, P. Antimicrobial activity of basil (Ocimum basilicum) oil against Salmonella enteritidis in vitro and in food. Biosci. Biotech. Bioch. 2010, 74, 1200–1204. [Google Scholar]
- De Martino, L.; De Feo, V.; Formisano, C.; Mignola, E.; Senatore, F. Chemical Composition and Antimicrobial Activity of the Essential Oils from Three Chemotypes of Origanum vulgare L. ssp. hirtum (Link) Ietswaart Growing Wild in Campania (Southern Italy). Molecules 2009, 14, 2735–2746. [Google Scholar] [CrossRef]
- Pintore, G.; Usai, M.; Bradesi, P.; Juliano, C.; Boatto, G.; Tomi, F.; Chessa, M.; Cerri, R.; Casanova, J. Chemical composition and antimicrobial activity of Rosmarinus officinalis L. oils from Sardinia and Corsica. Flavour Frag. J. 2002, 17, 15–19. [Google Scholar] [CrossRef]
- Wilkinson, J.M.; Hipwell, M.; Ryan, T.; Cavanagh, H.M.A. Bioactivity of Backhousiacitriodora: Antibacterial and antifungal activity. J. Agric. Food Chem. 2003, 51, 76–81. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods - a review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Magiatisa, P.; Skaltsounisa, A.-L.; Chinoua, I.; Haroutounianb, S.A. Chemical Composition and in vitro Antimicrobial Activity of the Essential Oils of Three Greek Achillea Species. Z. Naturforsch. 2002, 57c, 287–290. [Google Scholar]
- Jerković, I.; Mastelić, J.; Miloš, M.; Juteau, F.; Masotti, V.; Viano, J. Chemical variability of Artemisia vulgaris L. essential oils originated from Mediterranean area of France and Croatia. Flavour Frag. J. 2003, 18, 436–440. [Google Scholar] [CrossRef]
- Jerković, I.; Marijanović, Z.; Gugić, M.; Roje, M. Chemical Profile of the Organic Residue from Ancient Amphora Found in the Adriatic Sea Determined by Direct GC and GC-MS Analysis. Molecules 2011, 16, 7936–7948. [Google Scholar] [CrossRef]
- El-Sayed, A.M. The Pherobase: Database of Insect Pheromones and Semiochemicals. Available online: http://www.pherobase.com/ (accessed on 9 December 2011).
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Wanger, A. Disk Diffusion Test and Gradient Methodologies; CRC Press: Boca Raton, FL, USA, 2007; pp. 53–58. [Google Scholar]
- Sample Availability: Contact the corresponding author.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jerković, I.; Gašo-Sokač, D.; Pavlović, H.; Marijanović, Z.; Gugić, M.; Petrović, I.; Kovač, S. Volatile Organic Compounds from Centaurium erythraea Rafn (Croatia) and the Antimicrobial Potential of Its Essential Oil. Molecules 2012, 17, 2058-2072. https://doi.org/10.3390/molecules17022058
Jerković I, Gašo-Sokač D, Pavlović H, Marijanović Z, Gugić M, Petrović I, Kovač S. Volatile Organic Compounds from Centaurium erythraea Rafn (Croatia) and the Antimicrobial Potential of Its Essential Oil. Molecules. 2012; 17(2):2058-2072. https://doi.org/10.3390/molecules17022058
Chicago/Turabian StyleJerković, Igor, Dajana Gašo-Sokač, Hrvoje Pavlović, Zvonimir Marijanović, Mirko Gugić, Ivana Petrović, and Spomenka Kovač. 2012. "Volatile Organic Compounds from Centaurium erythraea Rafn (Croatia) and the Antimicrobial Potential of Its Essential Oil" Molecules 17, no. 2: 2058-2072. https://doi.org/10.3390/molecules17022058
APA StyleJerković, I., Gašo-Sokač, D., Pavlović, H., Marijanović, Z., Gugić, M., Petrović, I., & Kovač, S. (2012). Volatile Organic Compounds from Centaurium erythraea Rafn (Croatia) and the Antimicrobial Potential of Its Essential Oil. Molecules, 17(2), 2058-2072. https://doi.org/10.3390/molecules17022058







