3.1. General
All chemicals and solvents were from commercial sources and used without further treatment unless specified otherwise. The purity of all compounds was examined by high performance liquid chromatography (ODS column, 250 mm × 4.6 mm, 5 μm particle; mobile phase: acetonitrile in water; flow rate: 1.0 mL/min at 25 °C). 1H-NMR and 13C-NMR spectra were obtained using Me4Si as an internal standard on Bruker 400 or Varian 300 NMR instruments. Mass spectra were recorded on a Qstar LC/MS instrument. Chromatography was performed with commercial silica gel (300–400 mesh).
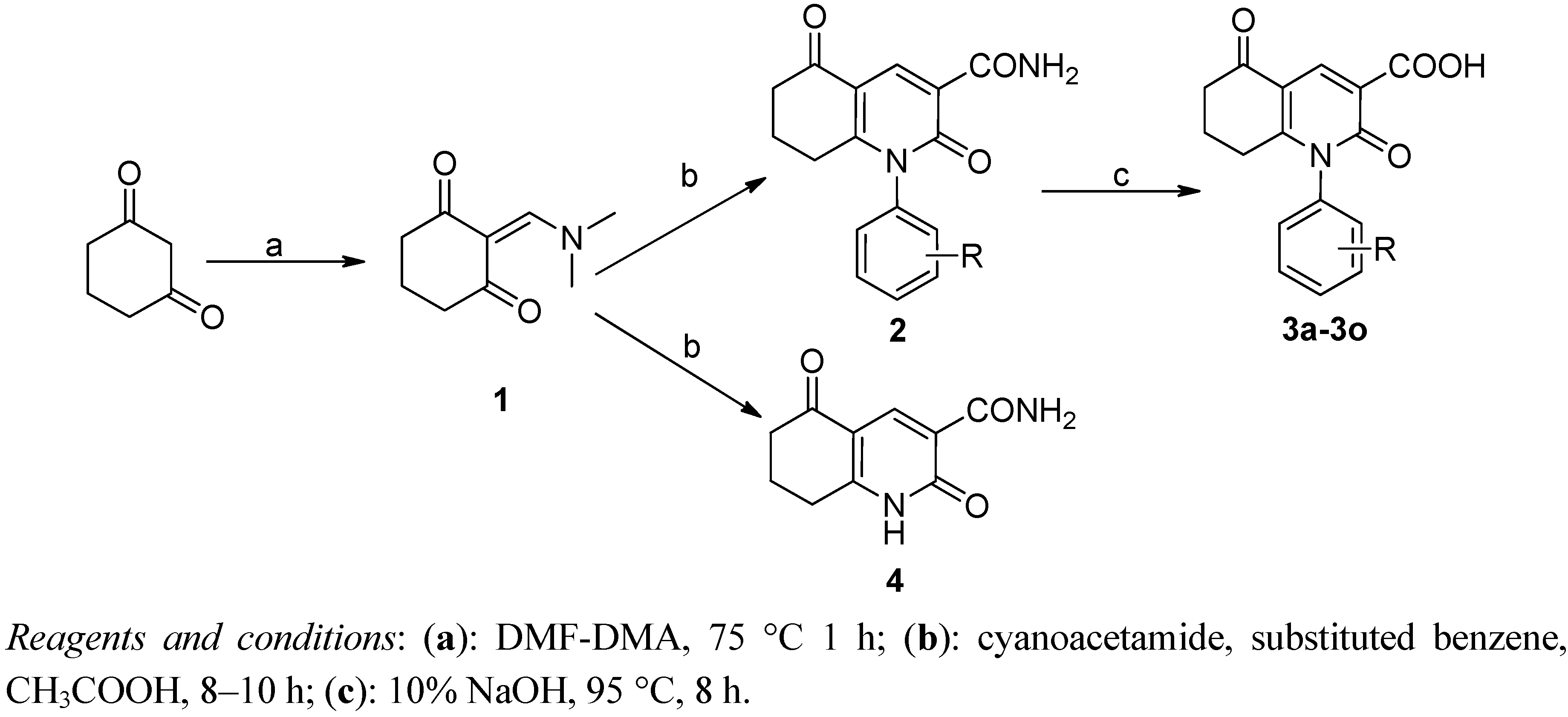
3.1.1. Preparation of 2-((Dimethylamino) methylene)cyclohexane-1,3-dione (1)
To 1,3-cyclohexandione (50 g) was added DMF-DMA (140 mL) with stirring. The resulting solution was heated to 75 °C for one hour. Solvent was removed and the residue was recrystallized from EtOAc to yield slight yellow crystals. Yield: 90%, mp: 117–118 °C.
3.1.2. General Procedure for the Preparation of 2
1-Phenyl-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (2a). Compound 1 (10 g) was dissolved in the mixture of 2-propanol (70 mL) and piperidine (0.1 g, 0.11 mmol) was added to the reaction. The mixture was stirred at room temperature for 5 h and to the mixture was added aniline (8.0 g) in acetic acid (40 mL). After 3 h, crystallization of a yellow solid occurred, which was filtered and recrystallized from methanol to yield a white solid. Yield: 61%, mp: >300.0 °C. MS m/z: 283 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 2.068–2.171 (m, 2H), 2.569–2.591 (m, 2H), 7.253–7.278 (m, 2H, Ar-H), 7.591–7.686 (m, 3H, Ar-H), 9.182 (s, 1H), 13.227 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 21.088, 29.514, 36.556, 116.537, 116.730, 127.243 (2C), 130.698 (2C), 130.819, 136.006, 143.862, 161.063, 164.239, 165.456, 192.657.
3.1.3. General Procedure for the Preparation of 3
1-Phenyl-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3a). To 2a (8.0 g) was added NaOH (10%, 50 mL) with stirring. The resulting solution was heated to 95 °C for 8 h and allowed to cool to room temperature. The pH of the solution was adjusted to 2 with diluted HCl, and crystallization of a yellow solid occurred, which was filtered and recrystallized in methanol to yield a white solid. Yield: 85%, HPLC purity: 99.4%. mp: 259.8–260.6 °C. MS m/z: 283 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.985–2.170 (m, 2H), 2.421–2.441 (m, 2H), 2.462–2.540 (m, 2H), 6.569–6.602 (d, 1H, J = 9.9 Hz), 7.197–7.262 (m, 2H, Ar-H), 7.480–7.587 (m, 3H, Ar-H), 8.049–8.081 (d, 1H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.472, 28.924, 36.470, 114.684, 119.004, 127.771 (2C), 129.449, 130.133 (2C), 136.950, 137.491, 157.133, 163.212, 194.063.
1-(2'-Chlorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3b). Yield: 87%, HPLC purity: 99.1%, mp: 247.3–248.1 °C, MS m/z: 317 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 2.101–2.581 (m, 2H), 2.606–2.692 (m, 2H), 2.498–2.529 (m, 2H), 7.4263 (m, 1H, Ar-H), 7.531–7.675 (m, 2H, Ar-H), 7.682–7.707 (m, 1H, Ar-H), 9.208 (s, 1H), 13.016 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.759, 28.372, 36.161, 114.577, 117.385, 129.316, 130.262, 130.728, 130.895, 132.017, 134.374, 141.705, 161.373, 162.540, 164.531, 193.551.
1-(3'-Chlorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3c). Yield: 91%, HPLC purity: 99.2%, mp: 247.3–248.1 °C, MS m/z: 317 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.965–1.996(m, 2H), 2.498–2.529 (m, 4H), 7.444 (s, 1H, Ar-H), 7.451–7.667 (m, 3H, Ar-H), 8.686 (s, 1H), 13.222 (s, 1H).13C-NMR (100 MHz, DMSO-d6): δ 20.736, 29.272, 36.207, 114.829, 116.645, 127.089, 128.286, 130.133, 131.780, 134.229, 138.150, 141.477, 163.036, 163.119, 164.622, 193.772.
1-(4'-Chlorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3d). Yield: 88%, HPLC purity: 99.0%, mp: 244.2–244.9 °C, MS m/z: 317 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.085–2.171 (m, 2H), 2.286–2.374 (m, 2 H), 2.572–2.641 (m, 2H), 7.215–7.263 (m, 2H, Ar-H), 7.610–7.638 (m, 2H, Ar-H), 9.154 (s, 1H), 13.034 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.883, 29.374, 36.310, 116.456, 116.569, 128.632 (2C), 130.929 (2C), 134.171, 136.804, 143.762, 160.726, 16 3.861, 165.127, 192.295.
1-(2'-Methylphenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3e). Yield: 91%, HPLC purity: 99.0%, mp: 237.0–238.2 °C, MS m/z: 297 (M+). 1H-NMR (300-MHz, DMSO-d6): δ 2.055–2.199 (m, 5H), 2.316–2.415 (m, 1H), 2.531–2.699 (m, 3H, CH3), 7.132–7.157 (m, 1H, Ar-H), 7.263–7.538 (m, 3H, Ar-H), 9.202 (s, 1H), 13.232 (s, 1H). 13C-NMR (100 M Hz, DMSO-d6): δ 17.005, 20.705, 28.586, 36.169, 115.272, 116.553, 127.829, 127.913, 130.209, 131.643, 134.870, 135.969, 141.530, 162.769, 162.837, 164.615, 193.787.
1-(3'-Methylphenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3f). Yield: 87%, HPLC purity: 99.2%, mp: 219.2–219.9 °C, MS m/z: 297 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 2.407 (m, 3H, CH3), 2.254 (m, 6H), 7.220–7.504 (m, 4H, Ar-H), 8.705 (s, 1H), 13.381 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.667, 21.087, 29.333, 36.207, 115.104, 116.149, 127.638 (2C), 130.636 (2C), 134.252, 139.638, 141.355, 163.539, 163.837, 164.683, 193.848.
1-(4'-Methylphenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3g). Yield: 86%, HPLC purity: 99.1%, mp: 244.2–244.9 °C, MS m/z: 297 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 2.387–2.503 (m, 3H, CH3), 2.503–2.520 (m, 6H), 7.294–7.426 (m, 4H, Ar-H), 8.704 (s, 1H), 13.403 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.667, 21.087, 29.333, 36.2 07, 115.104, 116.149, 127.638 (2C), 130.636 (2C), 134.252, 139.638, 141.355, 163.837, 16 4.683, 193.848.
1-(2'-Fluorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3h). Yield: 87%, HPLC purity: 99.1%, mp: 210.4–212.2 °C, MS m/z: 301 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.986–2.01 6m, 2H), 2.457–2.544 (m, 4H), 7.462–7.601 (m, 4H, Ar-H), 8.673 (s, 1H), 13.099 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.636, 28.555, 36.077, 114.715, 116.988, 117.179, 117.248, 126.051, 130.239, 132.528, 132.604, 141.644, 161.479, 162.799, 164.508, 193.536.
1-(4'-Fluorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3i). Yield: 89%, HPLC purity: 99.1%, mp: 260.4–261.3 °C, MS m/z: 301 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 2.059–2.170 (m, 2H), 2.571–2.641 (m, 4H), 7.147–7.263 (m, 4H, Ar-H), 8.698 (s, 1H), 13.298 (s, 1H).13C-NMR (100 MHz, DMSO-d6): δ 20.422, 29.035, 35.918, 114.652, 116.243, 122.999, 130.074 (2C), 132.931 (2C), 135.934, 141.172, 162.83, 164.356, 193.500.
1-(4'-Bromophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3j). Yield: 86%, HPLC purity: 99.1%, mp: 256.0–257.3 °C, MS m/z: 364 (M+).1H-NMR (300 MHz, DMSO-d6): δ 2.059–2.170 (m, 2H), 2.571–2.641 (m, 4H), 7.147–7.263(m, 2H, Ar-H), 7.768–7.797 (m, 2H, Ar-H), 9.161 (s, 1H), 13.232 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.422, 29.035, 35.918, 114.652, 116.243, 122.999, 130.074 (2C), 132.931 (2C), 135.934, 141.172, 162.83, 164.356, 193.500.
1-(4'-Methoxyphenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinolinecarboxylic acid (3k). Yield: 90%, HPLC purity: 99.0%, mp: 254.5–255.6 °C, MS m/z: 313 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.066–2.170 (m, 2H), 2.585–2.637 (m, 4H), 3.897 (s, 3H, OH), 7.097–7.262 (m, 4H, Ar-H), 9.173 (s, 1H), 13.271 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.659, 2.349, 36.192, 55.798, 115.127, 115.256 (2C), 116.019, 129.057 (2C), 129.209, 141.316, 160.060, 163.875, 164.104, 164.668, 193.841.
1-(2',4’-Dichlorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinoline carboxylicacid (3l). Yield: 91%, HPLC purity: 99.0%, mp: 263.0–266.3 °C, MS m/z: 353 (M+). 1H-NMR (400 MHz, MSO-d6): δ 1.956–2.040 (m, 2H), 2.472–2.631 (m, 4H), 7.147–7.263 (m, 1H, Ar-H), 7.727–7.754 (m, 1H, Ar-H), 8.020–8.025(m, 1H, Ar-H), 8.669 (s, 1H), 13.040 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.443, 28.011, 35.838, 114.171, 129.215, 130.092, 131.343, 132.014, 133.243, 135.432, 141.405, 160.615, 162.125, 164.185, 193.174.
1-(2',4’-Diethylphenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinoline carboxylicacid (3m). Yield: 87%, HPLC purity: 99.0%, mp: 240.5–241.1 °C, MS m/z: 311 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.049 (s, 3H, CH3), 2.076–2.191 (m, 2H), 2.431 (s, 3H, CH3), 2.335–2.397(m, 1H), 2.527–2.708 (m, 3H), 6.995–7.021 (m, 1H, Ar-H), 7.233–7.274 (m, 1H, Ar-H), 9.202(s, 1H), 13.316 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 17.432, 21.188, 21.447, 28.945, 36.656, 116.655, 116.768, 126.742, 129.116, 132.971, 134.163, 141.232, 143.916, 161.133, 164.331, 165.070, 192.673.
1-(3',4'-Dichlorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinoline carboxylicacid (3n). Yield: 86%, HPLC purity: 99.1%, mp: 224.0–225.2 °C, MS m/z: 353 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.956–2.006 (m, 2H), 2.468–3.352 (m, 4H), 7.505–7.527 (m, H, Ar-H), 7.890–7.931 (m, 2H, Ar-H), 8.671 (s, H), 13153 (s, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 20.913, 29.423, 36.379, 114.917, 117.029, 129.020, 130.706, 132.245, 132.645, 133.148, 136.856, 141.684, 162.907, 163.218, 164.747, 193.874.
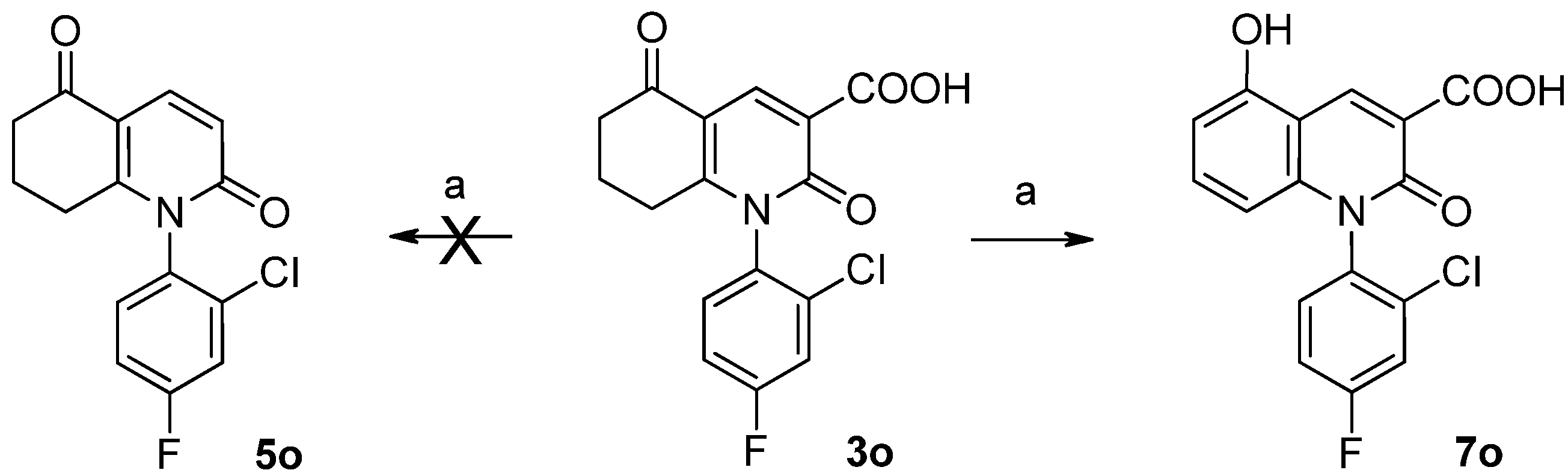
1-(2'-Chloro-4'-fluorophenyl)-1,2,5,6,7,8-hexahydro-2,5-dioxo-3-quinoline carboxylicacid (3o). Yield: 86%, HPLC purity: 99.0%, mp: 337.8–338.7 °C, MS m/z: 336 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.956–1.988 (m, 2H), 2.469–2.506(m, 4H), 7.661–7.853 (m, 3H), 8.435–8.444 (1H), 8.806 (COOH).13C-NMR (100 MHz, DMSO-d6): δ 20.512, 28.812, 35.922, 113.927, 117.764, 118.984, 120.533, 129.123, 130.512, 134.028, 140.002, 156.198, 158.677, 161.492, 162.232, 163.521, 193.579.
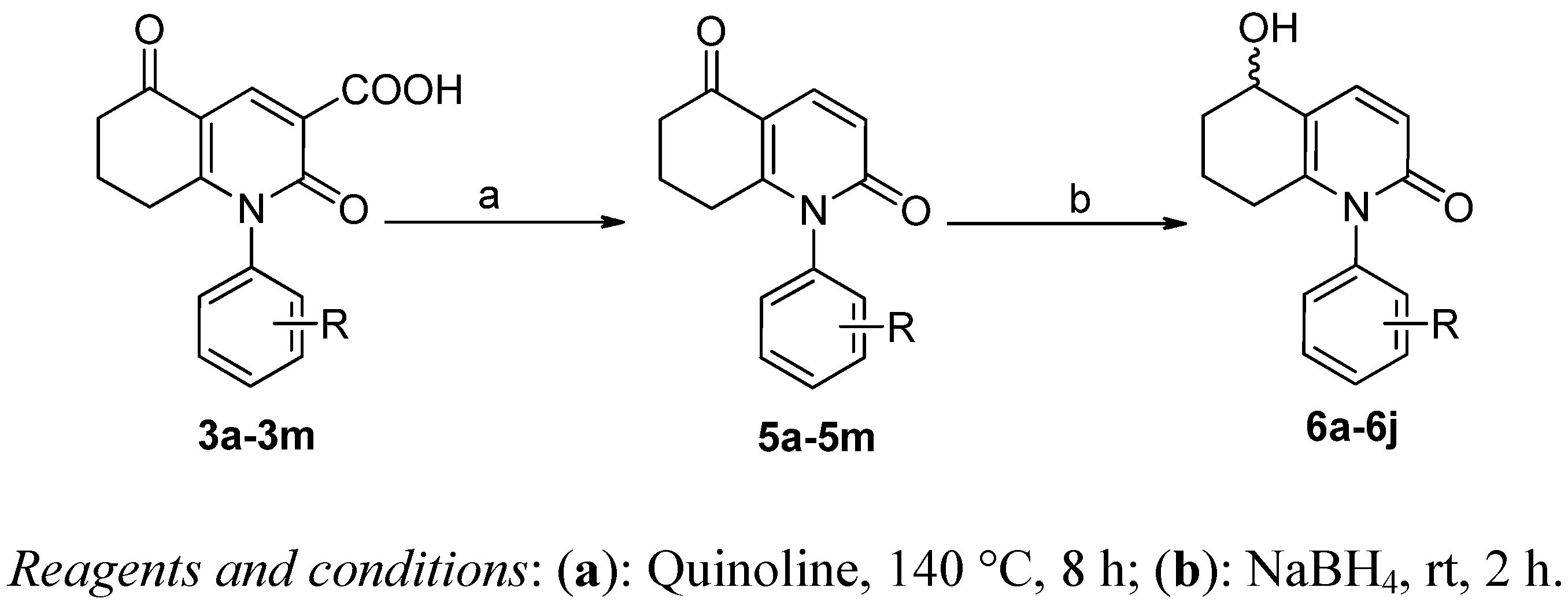
3.1.4. General Procedure for the Preparation of 5
1-Phenyl-7,8-dihydro-2,5(1H,6H)-quinolinedione (5a). To dried quinolone (20 mL) and 3a (5.0 g) copper (0.1 g) was added. The mixture was stirred and heated to 140–150 °C and refluxed for 8 h. After the suspension was cooled in the air to room temperature, 5a was obtained upon column chromatography (EtOAc/petroleum ether) to yield a white solid. Yield: 53%, HPLC purity: 99.1%, mp: 138.4–139.3 °C, MS m/z: 239 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.985–2.170 (m, 2H), 2.421–2.441 (m, 2H), 2.462–2.540 (m, 2H), 6.569–6.602 (d, 1H, J = 9.9 Hz), 7.197–7.262 (m, 2H, Ar-H), 7.480–7.587 (m, 3H, Ar-H), 8.049–8.081 (d, 1H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.472, 28.924, 36.470, 114.684, 119.004, 127.771 (2C), 129.449, 130.133 (2C), 136.950, 137.491, 157.133, 163.212, 194.063.
1-(2'-Chlorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5b). Yield: 36%, HPLC purity: 99.2%, mp: 161.2–161.9 °C, MS m/z: 273 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.019–2.104 (m, 2H), 2.342–2.508 (m, 2H), 2.517–2.618 (m, 2H), 6.575–6.607 (d, 1H, J = 8.8 Hz), 7.265–7.306 (m, 1H, Ar-H), 7.444–7.505 (m, 2H, Ar-H), 7.444–7.505 (m, 2H, Ar-H), 7.587–7.643 (m, 1H, Ar-H), 8.066–8.098 (d, 1 H, J= 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.193, 27.981, 36.222, 114.059, 118.255, 129.110, 130.583, 130.644, 131.330, 131.468, 135.267, 137.258, 158.275, 161.578, 194.008.
1-(3'-Chlorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5c). Yield: 36%, HPLC purity: 99.3%, mp: 184.3–185.4 °C, MS m/z: 273 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.932 (m, 2H), 2.404–2.437 (m, 4H), 6.463–6.487 (d, 1H, J = 9.6 Hz), 7.339–7.366 (s, 1H, Ar-H), 7.564 (m, 1H, Ar-H), 7.564–7.608 (m, 2H, Ar-H), 7.925–7.948 (d, 1H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.125, 28.692, 36.192, 113.876, 118.010, 127.470, 128.675, 129.438, 131.460, 134.000, 136.922, 139.112, 158.557, 162.257, 194.062.
1-(4'-Chlorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5d). Yield: 41%, HPLC purity: 99.6%, mp: 144.0–144.5 °C, MS m/z: 273 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.007–2.069 (m, 2H), 2.424–2.533 (m, 4H), 6.558–6.582 (d, 1H, J = 9.6 Hz), 7.148–7.260 (m, 2H, Ar-H), 7.519–7.539 (m, 2H, Ar-H), 8.041–8.065 (d, 1 H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.581, 29.100, 36.560, 114.997, 119.174, 129.389 (2C), 130.567 (2C), 135.743, 137.304, 156.917, 163.224, 194.068.
1-(2'-Methylphenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5e). Yield: 40%, HPLC purity: 99.1%, mp: 144.1–144.9 °C, MS m/z: 253 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 1.974–2.094 (m, 5H), 2.167–2.264 (m, 1H), 2.444–2.549 (m, 3H, CH3), 6.582–6.614 (d, 1H, J= 9.6 Hz), 7.088–7.111 (m, 1H, Ar-H), 7.265–7.417 (m, 3H, Ar-H), 8.065–8.098 (d, 1H, J = 9.9 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 17.371, 21.257, 28.257, 36.476, 114.747, 119.009, 127.501, 127.741, 129.707, 131.622, 134.972, 136.593, 136.933, 157.006, 162.603, 194.061.
1-(3'-Methylphenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5f). Yield: 39%, HPLC purity: 99.1%, mp: 122.6–123.0 °C. MS m/z: 253 (M+).1H-NMR (300 MHz, DMSO-d6): δ 1.983–2.068 (m, 2H), 2.425–2.475 (m, 3H, CH3), 2.491–2.536 (m, 4 H), 6.563–6.595 (d, 1H, J = 9.6 Hz), 6.988–7.014 (m, 2H, Ar-H), 7.263–7.318 (m, 1H, Ar-H), 7.412–7.463 (m, 1H, Ar-H), 8.038–8.070 (d, 1H, J = 9.6 Hz).13C-NMR (100 MHz, DMSO-d6): δ 21.653, 21.703, 29.115, 36.720, 114 .868, 119.186, 124.869, 128.404, 130.511, 137.123, 137.618, 140.584, 157.482, 163.527, 194.360
1-(4'-Methyphenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5g). Yield: 40%, HPLC purity: 99.0%, mp: 196.5–197.2 °C, MS m/z: 253 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 1.980–2.065 (m, 2H), 2.437–2.478 (m, 3H, CH3), 2.491–2.535 (m, 4H), 6.563–6.595 (d, 1H, J= 9.6 Hz), 7.068–7.095 (m, 2H, Ar-H), 7.263–7.366 (m, 2H, Ar-H), 8.036–8.068 (d, 1H, J = 9.6 Hz).13C-NMR (100 MHz, DMSO-d6): δ 21.467, 21.076, 36.627, 114.815, 119.092, 127.551 (2C), 130.951 (2C), 134.934, 137.021, 139.724, 157.520, 163.546, 194.309.
1-(2'-Fluorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5h). Yield: 41%, HPLC purity: 99.0%, mp: 207.8–208.7 °C, MS m/z: 257 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.913–1.992 (m, 2H), 2.269–2.618 (m, 4H), 6.499–6.523 (d, 1H, J = 9.6 Hz), 7.349–7.632 (m, 4H, Ar-H), 7.955–7.979 (d, 1H, J = 9.9 Hz).13C-NMR (100 MHz, DMSO-d6): δ 21.102, 28.097, 36.169, 114.219, 116.866 , 117.988, 24.907, 125.860, 130.651, 131.948, 137.321, 155.979, 158.443, 161.716, 194.047.
1-(4'-Fluorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5i). Yield: 42%, HPLC purity: 99.0%, mp: 177.2–178.2 °C, MS m/z: 257 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.911–1.943 (m, 2H), 2.387–2.406 (m, 4H), 6.454–6.478 (d, 1H, J = 9.6 Hz), 7.398–7.415 (m, 4H, Ar-H), 7.921–7.945 (d, 1H, J = 9.9 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 20.352, 28.972, 35.846, 114.682, 116.070, 116.658, 116.886, 129.993, 130.084, 132.678, 141.085, 160.996, 163.125, 163.201, 163.201, 163.453, 164.277, 193.426.
1-(4'-Bromophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5j). Yield: 40%, HPLC purity: 99.2%, mp: 211.8–212.9 °C, MS m/z: 319 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 2.004–2.088 (m, 2H), 2.426–2.547 (m, 4H), 6.560–6.592 (d, 1H, J = 9.6 Hz), 7.088–7.266 (m, 2H, Ar-H), 7.681–7.709 (m, 2H, Ar-H), 8.045–8.078 (d, 1H, J = 9.9 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 21.525, 29.052, 36.508, 114.926, 119.090, 123.748, 129.666 (2C), 133.516 (2C), 136.499, 137.245, 156.823, 163.089, 194.000.
1-(4'-Methoxyphenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5k). Yield: 45%, HPLC purity: 99.2%, mp: 165.3–165.8 °C, MS m/z: 269 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.884–1.947 (m, 2H), 2.396–2.428 (m, 4H), 6.432–6.456 (d, 1H, J= 9.6 Hz), 7.076–7.098 (m, 2H, Ar-H), 7.219–7.242 (m, 2H, Ar-H), 7.902–7.927 (d, 1H, J= 10.0 Hz).13C-NMR (100 MHz, DMSO-d6): δ 21.132, 28.784, 36.199, 55.714, 113.799, 115.012 (2C), 117.911, 29.438 (2C), 130.308, 136.625, 159.290, 159.549, 162.631, 194.146.
1-(2',4’-Dichlorophenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5l). Yield: 35%, HPLC purity: 99.2%, mp: 200.5–201.7 °C, MS m/z: 307 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.904–2.004 (m, 2H), 2.191–2.264 (m, 1H), 2.429–2.548 (m, 4H), 6.503–6.527 (d, 1H, J = 9.6 Hz), 6.956–6.975 (d, 1H, Ar-H), 7.669–7.696 (m, 1H, Ar-H), 7.965–7.971 (d, 1H, Ar-H), 7.956–7.980 (d, 1H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 20.840, 27.508, 35.876, 113.820, 117.871, 128.963, 129.909, 131.671, 132.373, 134.044, 134.860, 137.042, 157.838, 161.133, 193.586.
1-(2',4’-Dimethyphenyl)-7,8-dihydro-2,5(1H,6H)-quinolinedione (5m). Yield: 45%, HPLC purity: 99.0%, mp: 178.5–179.0 °C, MS m/z: 267 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 1.968–2.083 (m, 2H), 2.034 (s, 3H, CH3), 2.189–2.262 (m, 1H), 2.380 (s, 3H, CH3), 2.441–2.572 (m, 3H), 6.570–6.594 (d, 1H, J = 9.6 Hz), 6.956–6.975 (m, 1H, Ar-H), 7.151–7.260 (m, 2H, Ar-H), 8.044–8.068 (d, 1H, J = 9.6 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 17.462, 21.381, 21.653, 28.432, 36.639, 114.867, 119.100, 127.321, 128.595, 132.503, 134.072, 134.633, 137.053, 139.880, 157.432, 162.943, 194.269.
3.1.5. General Procedure for the Preparation of 6
5-Hydroxy-1-phenyl-5,6,7,8-tetrahydroquinolin-2(1H)-one (6a). To a solution of ethanol (20 mL) and 5a (1.0 g) was added NaBH4 (0.02 mol). The mixture was stirred at room temperature for 5 h. Ethanolwas removed completely and a white solid was obtained. The white solid was dissolved in water (30 mL), and pH was adjusted to 4 with diluted HCl. Crystallization of a white solid occurred, which was filtered and recrystallized in methanol to afford a white solid. Yield: 87%, HPLC purity: 99.1%, mp: 218.5–220.0 °C, MS m/z: 241 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.635–1.681 (m, 1H), 1.707–1.900 (m, 4H), 2.010–2.338 (m, 2H), 4.653 (s, 1H, OH), 6.5677–6.598 (m, 1H, J= 9.3 Hz), 7.129–7.262 (m, 2H, Ar-H), 7.446–7.521 (m, 3H, Ar-H), 7.545–7.607 (m, 1H).13C-NMR (100 MHz, DMSO-d6): δ21.4 72, 28.924, 36.470, 114.684, 119.004, 127.771 (2C), 129.449, 130.133 (2C), 136.950, 137.491, 157.133, 163.212, 194.063.
5-Hydroxy-1-(4'-chlorophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6d). Yield: 82%, HPLC purity: 99.1%, mp: 160.9–161.7 °C, MS m/z: 275 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.659–1.701 (m, 1H), 1.806–1.884 (m, 1H), 2.071–2.139 (m, 4H), 2.010–2.338 (m, 2H), 4.648 (s, 1H, OH), 6.565–6.589 (m, 1H, J= 9.6 Hz), 7.086–7.266 (m, 2H, Ar-H), 7.474–7.490 (m, 2H, Ar-H), 7.506–7.513 (m, 1H). 13C-NMR (100 MHz, DMSO-d6): δ 17.411, 28.694, 30.975, 66.243, 116.769, 118.043, 119.096 (2C), 122.231, 124.802, 134.834, 136.711, 139.595, 141.563 (2C), 144.996.
5-Hydroxy-1-(2'-fluorophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6h). Yield: 87%, HPLC purity: 98.1%, mp: 186.9–188.8 °C, MS m/z: 259 (M−H)+. 1H-NMR (400 MHz, DMSO-d6): δ 1.732 (m, 6H), 4.439–4.450 (m, 1H), 5.187–5.203 (m, 1H), 6.395–6.418 (m, 1H, J = 9.2 Hz), 7.341–7.369 (m, 2H, Ar-H), 7.432–7.457m, 1H, J = 9.4 Hz), 7.537–7.560 (m, 2H, Ar-H). 13C-NMR (100 MHz, DMSO-d6): δ 17.493, 28.059, 30.935, 64.616, 116.622, 116.820, 117.576, 117.812, 125.868, 131.201, 142.796, 144.726, 156.154, 158.619, 161.701.
5-Hydroxy-1-(4'-fluorophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6i). Yield: 78%, HPLC purity: 99.1%, mp: 186.9–188.8 °C, MS m/z: 259 (M+).1H-NMR (400 MHz, DMSO-d6): δ 1.732 (m, 6H), 4.439–4.450 (m, 1H), 5.187–5.203 (m, 1H), 6.395–6.418 (m, 1H, J = 9.2 Hz), 7.341–7.369 (m, 2H, Ar-H), 7.432–7.457 (m, 1H, J = 9.4 Hz), 7.537–7.560 (m, 2H, Ar-H). 13C-NMR (100 MHz, DMSO-d6): δ 17.493, 28.059, 30.935, 64.616, 116.622, 116.820, 117.576, 117.812, 125.868, 131.201, 142.796, 144.726, 156.154, 158.619, 161.700.
5-Hydroxy-1-(4'-bromophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6j). Yield: 91%, HPLC purity: 99.2%, mp: 179.5–180.6 °C, MS m/z: 319 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.721–2.010 (m, 6H), 4.439–4.462 (m, 1H), 5.174–5.189 (m, 1H), 6.367–6.390 (m, 1H, J= 9.2 Hz), 7.177–7.198 (m, 2H, Ar-H), 7.511–7.535 (m, 1H, J = 9.4 Hz), 7.699–7.717 (m, 2H, Ar-H). 13C-NMR (100 MHz, DMSO-d6): δ 17.730, 28.624, 30.966, 64.761, 117.637, 121.825, 131.063 (2C), 132.742 (2C), 138.044, 142.354, 144.711, 162.151.
5-Hydroxy-1-(4'-methoxyphenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6k). Yield: 75%, HPLC purity: 99.0%, mp: 181.5–181.9 °C, MS m/z: 271 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.514–1.749 (m, 4H), 2.015 (m, 2H), 3.803 (S, 3H, OCH3), 4.431–4.442 (m, 1H), 5.142–5.157 (m, 2H), 6.367–6.343 (m, 1H, J= 9.2 Hz), 6.926–7.185 (m, 4H, Ar-H), 7.481–7.504 (m, 1H, J = 9.2 Hz).13C-NMR (100 MHz, DMSO-d6): δ 17.730, 28.677, 31.019, 55.653, 64.830, 114.829, 114.875, 117.377, 117.576, 129.621 (2C), 131.254, 142.041, 145.436, 159.114, 162.524.
5-Hydroxy-1-(2',4’-dichlorophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6l). Yield: 91%, HPLC purity: 99.2%, mp: 210.9–217.4 °C, MS m/z: 309 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.533–1.640 (m, 2H), 1.696–1.833 (m, 3H), 2.104–2.151 (m, 2H), 3.849–3.864 (m, 1H), 4.454–4.491 (m, 1 H), 6.396–6.420 (m, 1H, J = 9.3 Hz), 7.457–7.479 (m, 1H, Ar-H), 7.553–7.577 (m, 1H, J = 9.4 Hz), 7.594–7.621 (m, 1H, Ar-H), 7.890–7.896 (m, 2H, Ar-H). 13C-NMR (100 MHz, DMSO-d6): δ 17.233, 27.471, 30.614, 64.212, 117.285, 117.499, 128.591, 129.606, 131.856, 132.611, 134.046, 135.045, 142.376, 143.841, 161.006.
5-Hydroxy-1-(2',4’-dimethyphenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (6m). Yield: 92%, HPLC purity: 99.1%, mp: 148.9–149.5 °C, MS m/z: 269 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 1.724–1.739 (m, 5H), 1.865–1.904 (s, 3H, CH3), 1.986–2.089 (m, 1H), 2.322 (S, 3H, CH3), 4.435–4.446 (m, 1H), 5.119–5.160 (m, 1H), 6.354–6.377 (m, 1H, J = 9.3 Hz), 6.926–7.185 (m, 4H, Ar-H), 7.536–7.513 (m, 1H, J = 9.4 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 17.524, 28.227, 30.295, 64.616, 116.622, 116.820, 117.576, 117.812, 125.868, 131.201, 142.796, 144.726, 156.154, 158.619, 161.624.
3.1.6. 1-(2-Chloro-4-fluorophenyl)-5-hydroxy-2-oxo-1,2-dihydroquinoline-3-carboxylic acid (7o)
To dried quinoline (20 mL) was added 3o (5.0 g) and copper (0.1 g). The mixture was stirred and heated to 140–150 °C and refluxed for 8 h. When the suspension was cooled in the air to room temperature, compound 7o was obtained, which upon column chromatography (EtOAc/petroleum ether) to yield a white solid. Yield: 34%, HPLC purity: 99.0%, mp: 181.5–181.9 °C, MS m/z: 334 (M+). 1H-NMR (400 MHz, DMSO-d6): δ 6.003–6.024 (d, 1H), 6.742–6.762 (d, 1H), 7.345–7.386 (d, 1 H), 7.462–7.480 (d, 1H), 7.664–686 (d, 1H), 7.709–7.751 (d, 1H), 7.812–7.824 (d, 1H), 9.154 (S, 1H), 11.073 (S, 1H).13C-NMR (100 MHz, DMSO-d6): δ 106.199, 107.946, 109.136, 117.993, 118.214, 119.731, 123.947, 130.023, 131.023, 131.374, 134.143, 134.662, 134.700, 138.957, 142.794, 156.007, 156.243, 158.479, 161.408, 163.918.
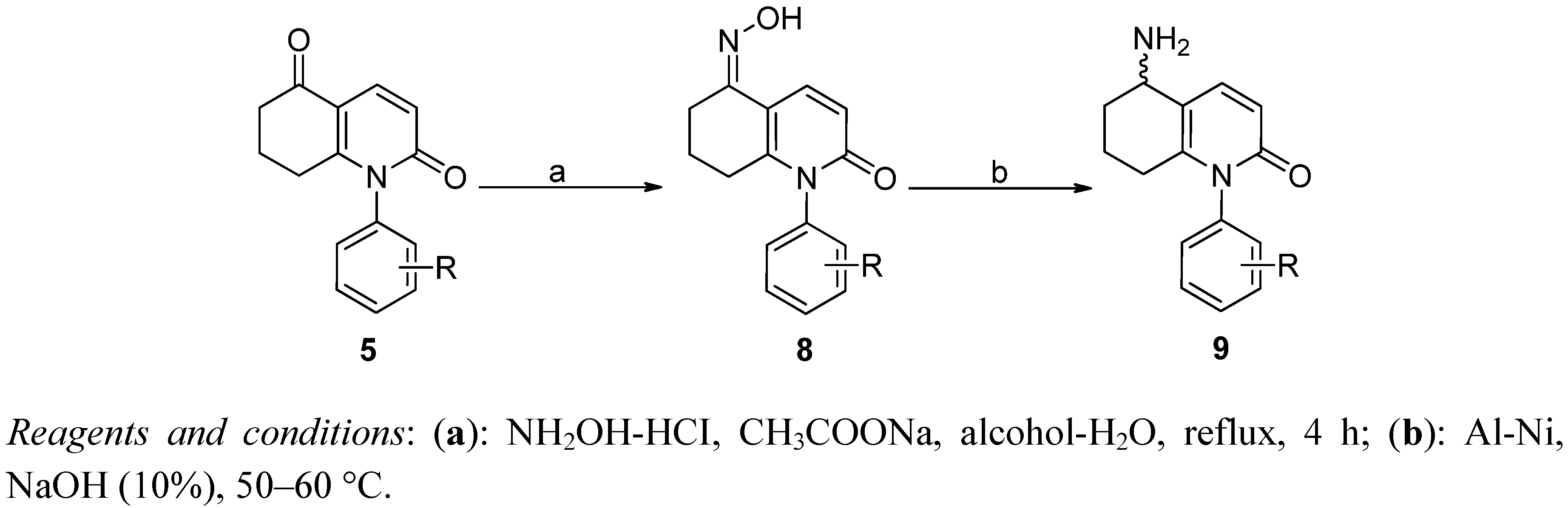
3.1.7. General Procedure for the Preparation of 8i and 8k
5-Hydroxyimimo-1-(4-methoxyphenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (8i). To 5i (1 g), hydroxylammonium chloride (280 mg), anhydrous sodium acetate (330 mg), and water-ethanol (1:1, 50 mL) were added, the mixture was stirred and heated to reflux for 4 h. The white solid which formed was filtered off. Yield: 82%, HPLC: 99.4%; mp: 112.9–113.2 °C, MS m/z: 284 (M+). 1H-NMR (500 MHz, DMSO-d6): δ 1.638–1.688 (m, 2H), 2.177–2.202(m, 2H), 2.495–2.538(m, 2H), 3.813 (m, 3H), 6.393–6.413 (m, 1H, J = 10 Hz), 7.047–7.065 (m, 2H, Ar-H), 7.161–7.178 (m, 2H, Ar-H), 7.942–7.961 (m, 1H, J = 10Hz), 10.887 (m, 1H, OH). 13C-NMR (125 MHz, DMSO-d6): δ 20.268, 21.729, 28.288, 55.864, 110.832, 115.058 (2C), 118.561, 129.792, 131.020, 136.087 (2C), 148.473, 151.206, 159.490, 162.524.
5-Hydroxyimimo-1-(4-fluorophenyl)-5,6,7,8-tetrahydroquinolin-2(1H)-one (8k). Yield: 82%, HPLC: 97.0%; mp: 121.9–122.2 °C, MS m/z: 272 (M+). 1H-NMR (500 MHz DMSO-d6): δ 1.663–1.688 (m, 2H), 2.166–2.190(m, 2H), 2.499–2.544 (m, 2H), 6.415–6.435 (m, 1H, J = 10 Hz), 7.336–7.386 (m, 4H, Ar-H), 7.962–7.981 (m, 1H, J = 10 Hz), 10.914 (m, 1H, OH). 13C-NMR (125 MHz DMSO-d6): δ 20.237, 21.692, 28.260, 111.004, 116.689, 116.871, 118.604, 120.983, 131.053, 136.660, 148.050, 151.113, 161.195, 162.362, 163.146.
3.1.8. 5-Amino-1-phenyl-5,6,7,8-tetrahydroquinolin-2(1H)-one (9a)
To the suspension of 5a (478 mg) and Al-Ni (1:1, 340 mg) in ethanol (30 mL) was quickly added NaOH (10%, 6 mL) and kept stirring at 50 °C. After the disappearance of starting materials, the solution was cooled, filtered, and extracted with dichloromethane. After removal of the solvent, recrystallization from EtOAc, slight white crystals were obtained. Yield: 35%, HPLC purity: 99.0%; mp: 186.5–187.3 °C. MS m/z: 241 (M+). 1H-NMR (300 MHz, DMSO-d6): δ 1.597–1.676 (m, 2H), 1.753–1.907 (m, 2H), 2.044–2.170 (m, 2H), 3.849–3.864 (m, 1H), 6.563–6.594 (m, 1H, J = 9.3Hz), 7.143–7.265 (m, 2H, Ar-H), 7.413–7.484 (m, 3H, Ar-H), 7.501–7.532 (m, 1H, J = 9.3 Hz). 13C-NMR (100 MHz, DMSO-d6): δ 18.239, 29.010, 32.367, 47.815, 118.330, 119.152, 128.211, 128.918, 129.982, 130.011, 138.688, 141.438, 144.507, 163.522.