Polyphenolic Profile and Biological Activity of Chinese Hawthorn (Crataegus pinnatifida BUNGE) Fruits
Abstract
:1. Introduction
2. Polyphenolic Profile of Chinese Hawthorn Fruits
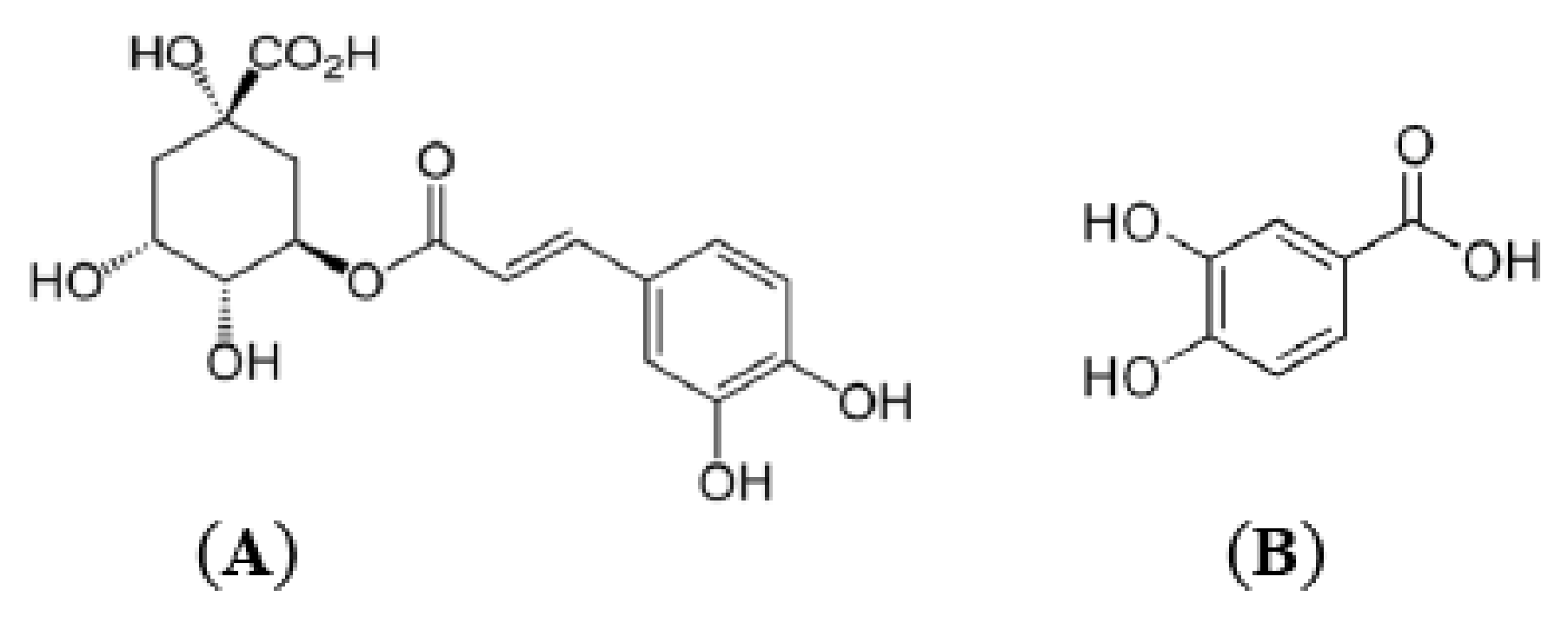
2.1. Phenolic Acids
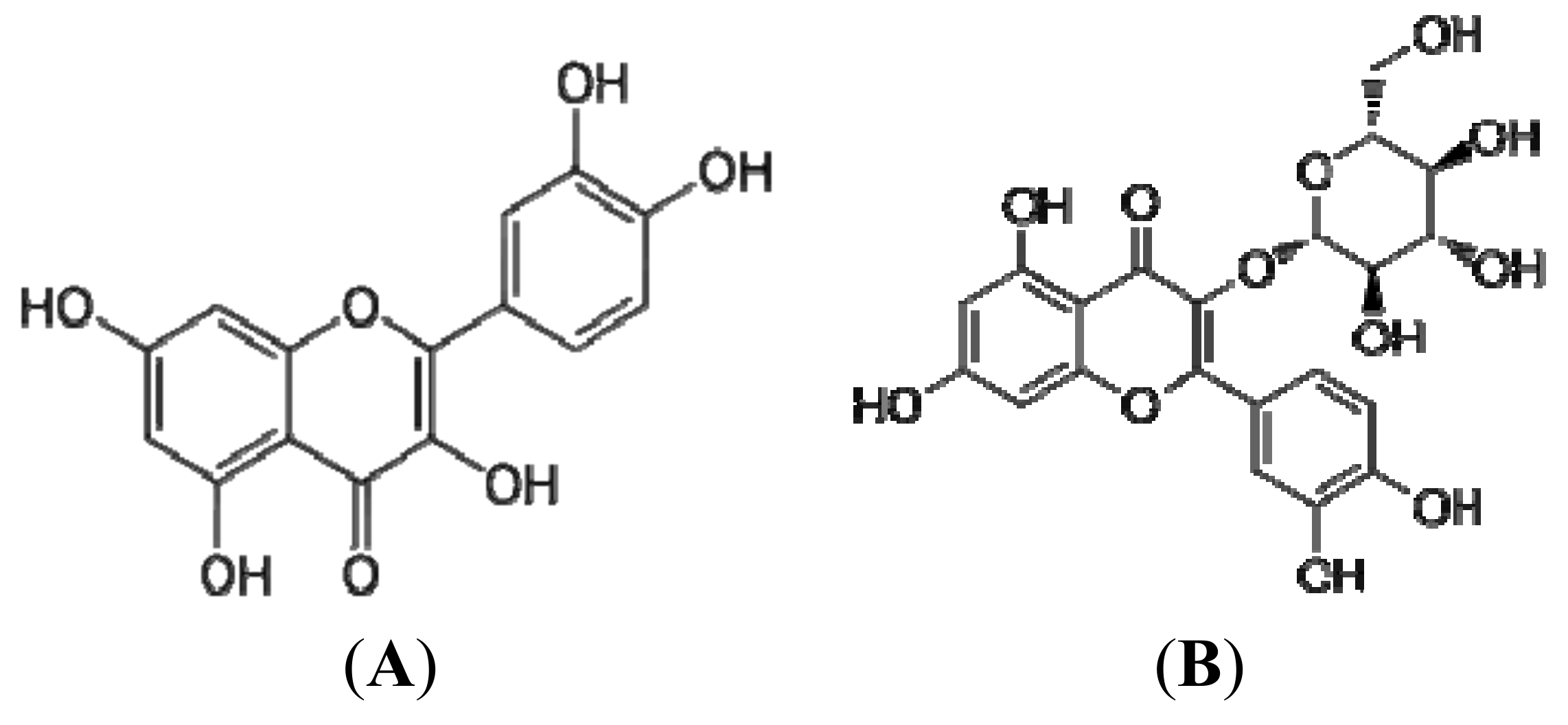
2.2. Flavonoids
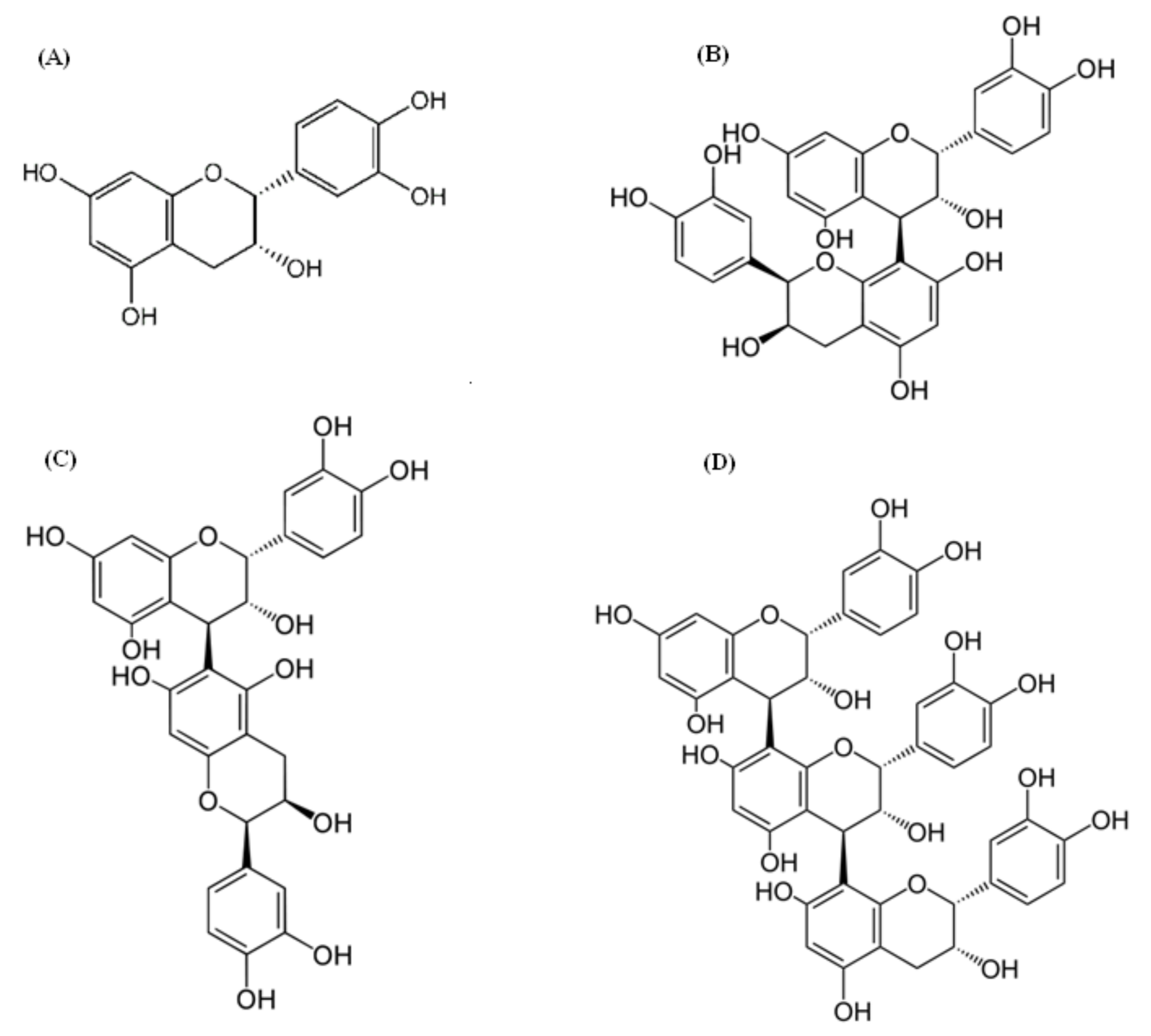
2.3. Procyanidins as the Main Component of Chinese Hawthorn Fruit
2.4. Methods for Quantification of Polyphenolic Compounds in Chinese Hawthorn Fruits
2.5. Stability of Polyphenolic Compounds in Chinese Hawthorn Fruits
2.6. Relationship between Individual Polyphenolic Compounds
3. Biological Activity of Polyphenols in Chinese Hawthorn Fruit
3.1. Antioxidant Activity of Chinese Hawthorn Fruit
3.2. Biological Activity of Fruits
3.2.1. Hypolipidaemic Effects
3.2.2. Effects on Cardiovascular Diseases and the Vascular System
3.2.3. Other Activities
4. Conclusions
Acknowledgements
References
- Weihmayer, T.; Ernst, E. Therapeutic effectiveness of Crateagus. Fortschr. Med. 1996, 114, 27–29. [Google Scholar]
- Tassel, M.C.; Kingston, R.; Gilroy, D.; Lehane, M.; Furey, A. Hawthorn (Crataegus spp.) in the treatment of cardiovascular disease. Pharmcogn. Rev. 2010, 4, 32–41. [Google Scholar]
- Fong, H.H.; Bauman, J.L. Hawthorn. J. Cardiovasc. Nurs. 2002, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wittig, J.; Leipolz, I.; Graefe, E.U.; Jaki, B.; Treutter, D.; Veit, M. Quantification of procyanidins in oral herbal medicinal products containing extracts of Crataegus species. Arzneimittelforschung 2002, 52, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.C.; Tian, B.F. China Fruit-Plant Monograph-Hawthorn Flora; China Forestry Publishing House: Beijing, China, 1996. [Google Scholar]
- Sanada, T.; Sato, Y. Genetic resources of Asian temperate fruits. In Asian Plants with Unique Horticultural Potential: Genetic Resources, Cultural Practices, and Utilization, Proceedings of The XXVI International Horticultural Congress, Toronto, Canada, 11–17 August 2002; Lee, J.M., Zhang, D., Eds.; ISHS: Leuven, Belgum, 2003; pp. 137–149. [Google Scholar]
- Liu, P.Z.; Kallio, H.; Lu, D.G.; Zhou, C.S.; Ou, S.Y.; Yang, B.R. Acids, Sugars, and Sugar Alcohols in Chinese Hawthorn (Crataegus spp.) Fruits. J. Agric. Food Chem. 2010, 58, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.R.; Liu, P.Z. Composition and health effects of phenolic compounds in hawthorn (Crataegus spp.) of different origins. J. Sci. Food Agric. 2012, 92, 1578–1590. [Google Scholar] [CrossRef] [PubMed]
- Kao, E.S.; Wang, C.J.; Lin, W.L.; Chu, C.Y.; Tseng, T.H. Effects of polyphenols derived from fruit of Crataegus pinnatifida on cell transformation, dermal edema and skin tumor formation by phorbol ester application. Food Chem. Toxicol. 2007, 45, 1795–1804. [Google Scholar] [CrossRef] [PubMed]
- Cui, T.; Li, J.Z.; Kayahara, H.; Ma, L.; Wu, L.X.; Nakamura, K. Quantification of the polyphenols and triterpene acids in Chinese hawthorn fruit by high-performance liquid chromatography. J. Agric. Food Chem. 2006, 54, 4574–4581. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.T.; Dao, J.; Shao, Z.H. Hawthorn: Potential roles in cardiovascular disease. Am. J. Chin. Med. 2005, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pittler, M.H.; Schmidt, K.; Ernst, E. Hawthorn extract for treating chronic heart failure: Meta-analysis of randomized trials. Am. J. Med. 2003, 114, 665–674. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Ho, W.K.K.; Huang, Y.; Chen, Z.Y. Hypocholesterolemic activity of hawthorn fruit is mediated by regulation of cholesterol-7 alpha-hydroxylase and acyl CoA: Cholesterol acyltransferase. Food Res. Int. 2002, 35, 885–891. [Google Scholar] [CrossRef]
- Gazdik, Z.; Krska, B.; Adam, V.; Saloun, J.; Pokorna, T.; Reznicek, V.; Horna, A.; Kizek, R. Electrochemical Determination of the Antioxidant Potential of Some Less Common Fruit Species. Sensors 2008, 8, 7564–7570. [Google Scholar] [CrossRef] [PubMed]
- Jurikova, T.; Rop, O.; Mlcek, J.; Sochor, J.; Balla, S.; Szekeres, L.; Hegedusova, A.; Hubalek, J.; Adam, V.; Kizek, R. Phenolic Profile of Edible Honeysuckle Berries (Genus Lonicera) and Their Biological Effects. Molecules 2012, 17, 61–79. [Google Scholar] [CrossRef] [PubMed]
- Rop, O.; Posolda, M.; Mlcek, J.; Reznicek, V.; Sochor, J.; Adam, V.; Kizek, R.; Sumczynski, D. Qualities of Native Apple Cultivar Juices Characteristic of Central Europe. Not. Bot. Horti. Agrobot. Cluj Na. 2012, 40, 222–228. [Google Scholar] [CrossRef]
- Cui, T.; Nakamura, K.; Tian, S.; Kayahara, H.; Tian, Y.L. Polyphenolic content and physiological activities of Chinese hawthorn extracts. Biosci. Biotechnol. Biochem. 2006, 70, 2948–2956. [Google Scholar] [CrossRef] [PubMed]
- Bernatoniene, J.; Masteikova, R.; Majiene, D.; Savickas, A.; Kevelaitis, E.; Bernatoniene, R.; Dvorackova, K.; Civinskiene, G.; Lekas, R.; Vitkevicius, K.; et al. Free radical-scavenging activities of Crataegus monogyna extracts. Med. Lith. 2008, 44, 706–712. [Google Scholar] [CrossRef]
- Tadic, V.M.; Dobric, S.; Markovic, G.M.; Dordevic, S.M.; Arsic, I.A.; Menkovic, N.R.; Stevic, T. Anti-inflammatory, gastroprotective, free-radical-scavenging, and antimicrobial activities of hawthorn berries ethanol extract. J. Agric. Food Chem. 2008, 56, 7700–7709. [Google Scholar] [CrossRef] [PubMed]
- Urbonaviciute, A.; Jakstas, V.; Kornygova, O.; Janulis, V.; Maruska, A. Capillary electrophoretic analysis of flavonoids in single-styled hawthorn (Crataegus monogyna Jacq.) ethanolic extracts. J. Chromatogr. A 2006, 1112, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Svedstrom, U.; Vuorela, H.; Kostiainen, R.; Huovinen, K.; Laakso, I.; Hiltunen, R. High-performance liquid chromatographic determination of oligomeric procyanidins from dimers up to the hexamer in hawthorn. J. Chromatogr. A 2002, 968, 53–60. [Google Scholar] [CrossRef]
- Svedstrom, U.; Vuorela, H.; Kostiainen, R.; Laakso, I.; Hiltunen, R. Fractionation of polyphenols in hawthorn into polymeric procyanidins, phenolic acids and flavonoids prior to high-performance liquid chromatographic analysis. J. Chromatogr. A 2006, 1112, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Svedstrom, U.; Vuorela, H.; Kostiainen, R.; Tuominen, J.; Kokkonen, J.; Rauha, J.P.; Laakso, I.; Hiltunen, R. Isolation and identification of oligomeric procyanidins from Crataegus leaves and flowers. Phytochemistry 2002, 60, 821–825. [Google Scholar] [CrossRef]
- Chang, Q.; Zuo, Z.; Harrison, F.; Chow, M.S.S. Hawhorn. J. Clin. Pharmacol. 2002, 42, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.Z.; Kallio, H.; Lu, D.G.; Zhou, C.S.; Yang, B.R. Quantitative analysis of phenolic compounds in Chinese hawthorn (Crataegus spp.) fruits by high performance liquid chromatography-electrospray ionisation mass spectrometry. Food Chem. 2011, 127, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.X.; Zeng, Y.K.; Zhou, Y.; Zang, S.L.; He, P.G.; Fang, Y.Z. Determination of active ingredients of hawthorn by capillary electrophoresis with electrochemical detection. Chin. J. Chem. 2006, 24, 1378–1383. [Google Scholar] [CrossRef]
- Zhang, P.C.; Xu, S.X. Flavonoid ketohexosefuranosides from the leaves of Crataegus pinnatifida Bge. var. major NEBr. Phytochemistry 2001, 57, 1249–1253. [Google Scholar] [CrossRef]
- Froehlicher, T.; Hennebelle, T.; Martin-Nizard, F.; Cleenewerck, P.; Hilbert, J.L.; Trotin, F.; Grec, S. Phenolic profiles and antioxidative effects of hawthorn cell suspensions, fresh fruits, and medicinal dried parts. Food Chem. 2009, 115, 897–903. [Google Scholar] [CrossRef]
- Nikolov, N.; Seligmann, O.; Wagner, H.; Horowitz, R.; Gentili, B. Neue flavonoid—Glykoside aus Crateagus monogyna und Crateagus pentagyna. Planta Med. 1982, 44, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Dauguet, J.C.; Bert, M.; Dolley, J.; Bekaert, A.; Lewin, G. 8-methoxy-kaempferol-3-neohesperidoside and other flavonoid from bee pollen of Crateagus monogyna. Phytochemistry 1993, 33, 1503–1505. [Google Scholar] [CrossRef]
- Schussler, M.; Hollzl, J.; Fricke, U. Myocardial effects of flavonoids from Crateagus species. Arzneimitteforschung 1995, 45, 842–845. [Google Scholar]
- He, F.; Pan, Q.H.; Shi, Y.; Duan, C.Q. Chemical Synthesis of Proanthocyanidins in Vitro and Their Reactions in Aging Wines. Molecules 2008, 13, 3007–3032. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Pan, Q.H.; Shi, Y.; Duan, C.Q. Biosynthesis and Genetic Regulation of Proanthocyanidins in Plants. Molecules 2008, 13, 2674–2703. [Google Scholar] [CrossRef] [PubMed]
- Weber, H.A.; Hodges, A.E.; Guthrie, J.R.; O’Brien, B.M.; Robaugh, D.; Clark, A.P.; Harris, R.K.; Algaier, J.W.; Smith, C.S. Comparison of proanthocyanidins in commercial antioxidants: Grape seed and pine bark extracts. J. Agric. Food Chem. 2007, 55, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.Y.; Feng, Y.X.; Quin, X.Q. Analysis of chemical constituents of hawthorn fruits and their quality evaluation. Yaoxue Xuebao 1995, 30, 138–143. [Google Scholar]
- Liu, W.M.; Chen, G.H.; Cui, T. Determination of flavones in Crataegus pinnatifida by capillary zone electrophoresis. J. Chromatogr. Sci. 2003, 41, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Sochor, J.; Skutkova, H.; Babula, P.; Zitka, O.; Cernei, N.; Rop, O.; Krska, B.; Adam, V.; Provaznik, I.; Kizek, R. Mathematical Evaluation of the Amino Acid and Polyphenol Content and Antioxidant Activities of Fruits from Different Apricot Cultivars. Molecules 2011, 16, 7428–7457. [Google Scholar] [CrossRef] [PubMed]
- Zitka, O.; Sochor, J.; Rop, O.; Skalickova, S.; Sobrova, P.; Zehnalek, J.; Beklova, M.; Krska, B.; Adam, V.; Kizek, R. Comparison of Various Easy-to-Use Procedures for Extraction of Phenols from Apricot Fruits. Molecules 2011, 16, 2914–2936. [Google Scholar] [CrossRef] [PubMed]
- Sochor, J.; Zitka, O.; Skutkova, H.; Pavlik, D.; Babula, P.; Krska, B.; Horna, A.; Adam, V.; Provaznik, I.; Kizek, R. Content of Phenolic Compounds and Antioxidant Capacity in Fruits of Apricot Genotypes. Molecules 2010, 15, 6285–6305. [Google Scholar] [CrossRef] [PubMed]
- Gazdik, Z.; Reznicek, V.; Adam, V.; Zitka, O.; Jurikova, T.; Krska, B.; Matuskovic, J.; Plsek, J.; Saloun, J.; Horna, A.; et al. Use of Liquid Chromatography with Electrochemical Detection for the Determination of Antioxidants in Less Common Fruits. Molecules 2008, 13, 2823–2836. [Google Scholar] [CrossRef] [PubMed]
- Rop, O.; Balik, J.; Reznicek, V.; Jurikova, T.; Skardova, P.; Salas, P.; Sochor, J.; Mlcek, J.; Kramarova, D. Chemical Characteristics of Fruits of Some Selected Quince (Cydonia oblonga Mill.) Cultivars. Czech. J. Food Sci. 2011, 29, 65–73. [Google Scholar] [CrossRef]
- Rop, O.; Jurikova, T.; Sochor, J.; Mlcek, J.; Kramarova, D. Antioxidant capacity, scavenging radical activity and selected chemical composition of native apple cultivars from central Europe. J. Food Qual. 2011, 34, 187–194. [Google Scholar] [CrossRef]
- Rop, O.; Reznicek, V.; Mlcek, J.; Jurikova, T.; Balik, J.; Sochor, J.; Kramarova, D. Antioxidant and radical oxygen species scavenging activities of 12 cultivars of blue honeysuckle fruit. Hortic. Sci. 2011, 38, 63–70. [Google Scholar] [CrossRef]
- Rop, O.; Sochor, J.; Jurikova, T.; Zitka, O.; Skutkova, H.; Mlcek, J.; Salas, P.; Krska, B.; Babula, P.; Adam, V.; et al. Effect of Five Different Stages of Ripening on Chemical Compounds in Medlar (Mespilus germanica L.). Molecules 2011, 16, 74–91. [Google Scholar] [CrossRef] [PubMed]
- Rop, O.; Mlcek, J.; Jurikova, T.; Valsikova, M.; Sochor, J.; Reznicek, V.; Kramarova, D. Phenolic content, antioxidant capacity, radical oxygen species scavenging and lipid peroxidation inhibiting activities of extracts of five black chokeberry (Aronia melanocarpa (Michx.) Elliot) cultivars. J. Med. Plants Res. 2010, 4, 2431–2437. [Google Scholar]
- Cheng, S.; Qiu, F.; Huang, J.; He, J.Q. Simultaneous determination of vitexin-2"-O-glucoside, vitexin-2"-O-rhamnoside, rutin, and hyperoside in the extract of hawthorn (Crataegus pinnatifida Bge.) leaves by RP-HPLC with ultraviolet photodiode array detection. J. Sep. Sci. 2007, 30, 717–721. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Huang, G.L.; Li, Y.L.; Wang, T.J.; Gao, C.K. Simultaneous Determination of Four Flavonoids in Extract of Hawthorn Leaves by Microemulsion Liquid Chromatography. Chin. J. Anal. Chem. 2009, 37, 1791–1794. [Google Scholar]
- Ying, X.X.; Li, H.B.; Xiong, Z.L.; Sun, Z.S.; Cai, S.; Zhu, W.L.; Bi, Y.J.; Li, F.M. LC determination of malondialdehyde concentrations in the human umbilical vein endothelial cell culture medium: Application to the antioxidant effect of Vitexin-2''-O-rhamnoside. Chromatographia 2008, 67, 679–686. [Google Scholar] [CrossRef]
- Wu, H.W.; Chen, M.L.; Fan, Y.C.; Elsebaei, F.; Zhu, Y. Determination of rutin and quercetin in Chinese herbal medicine by ionic liquid-based pressurized liquid extraction-liquid chromatography-chemiluminescence detection. Talanta 2012, 88, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Vierling, W.; Brand, N.; Gaedcke, F.; Sensch, K.H.; Schneider, E.; Scholz, M. Investigation of the pharmaceutical and pharmacological equivalence of different Hawthorn extracts. Phytomedicine 2003, 10, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Zuo, Z.; Chow, M.S.S.; Ho, W.K.K. Effect of storage temperature on phenolics stability in hawthorn (Crataegus pinnatifida var. major) fruits and a hawthorn drink. major) fruits and a hawthorn drink. Food Chem. 2006, 98, 426–430. [Google Scholar]
- Sochor, J.; Ryvolova, M.; Krystofova, O.; Salas, P.; Hubalek, J.; Adam, V.; Trnkova, L.; Havel, L.; Beklova, M.; Zehnalek, J.; et al. Fully Automated Spectrometric Protocols for Determination of Antioxidant Activity: Advantages and Disadvantages. Molecules 2010, 15, 8618–8640. [Google Scholar] [CrossRef] [PubMed]
- Sochor, J.; Babula, P.; Krska, B.; Horna, A.; Provaznik, I.; Hubalek, J.; Kizek, R. Evaluation of output signals from CoulArray detector for determination of antioxidant capacity of apricots samples. In Analysis of Biomedical Signals and Images; Jan, J., Jirik, R., Kolar, R., Kolarova, J., Kozumplik, J., Provaznik, I., Eds.; Brno Univ Technology Vut Press: Brno, Czech Republic, 2010; pp. 209–214. [Google Scholar]
- Pohanka, M.; Pavlis, O.; Ruttkay-Nedecky, B.; Sochor, J.; Sobotka, J.; Pikula, J.; Adam, V.; Kizek, R. Tularemia progression accompanied with oxidative stress and antioxidant alteration in spleen and liver of BALB/c mice. J. Microbiol. 2012, 50, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M.; Stetina, R.; Svobodova, H.; Ruttkay-Nedecky, B.; Jilkova, M.; Sochor, J.; Sobotka, J.; Adam, V.; Kizek, R. Sulfur mustard causes oxidative stress and depletion of antioxidants in muscles, livers, and kidneys of Wistar rats. Drug Chem. Toxicol. 2012, 23, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.Y.; Lee, M.J.; Liao, C.L.; Lin, W.L.; Yin, Y.F.; Tseng, T.H. Inhibitory effect of hot-water extract from dried fruit of Crataegus pinnatifida on low-density lipoprotein (LDL) oxidation in cell and cell-free systems. J. Agric. Food Chem. 2003, 51, 7583–7588. [Google Scholar] [CrossRef] [PubMed]
- Kao, E.S.; Wang, C.J.; Lin, W.L.; Yin, Y.F.; Wang, C.P.; Tseng, T.H. Anti-inflammatory potential of flavonoid contents from dried fruit of Crataegus pinnatifida in vitro and in vivo. J. Agric. Food Chem. 2005, 53, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Son, H.J.; Huang, C.; Lee, S.K.; Lohakare, J.; Wang, M.H. Comparison of Crateagus pinnatifida Bunge var. typica Schneider and C. pinnatifida Bunge fruits for antioxidant, anti-alpha-glucoside, and anti-inflammatory activities. Food Sci. Biotechnol. 2010, 19, 769–775. [Google Scholar] [CrossRef]
- Bahorun, T.; Gressier, B.; Trotin, F.; Brunet, C.; Dine, T.; Luyckx, M.; Vasseur, J.; Cazin, M.; Cazin, J.C.; Pinkas, M. Oxygen species scavenging activity of phenolic extracts from hawthorn fresh plant organs and pharmaceutical preparations. Arzneimittelforschung 1996, 46, 1086–1089. [Google Scholar] [PubMed]
- Rakotoarison-Greissier, D.A. Antioxidant activities of phenolic extracts from flowers, in vitro and cell suspension cultures of Crateagus monogyna. Pharmazie 1997, 52, 60–64. [Google Scholar]
- Jin, N.; Liu, T. Antioxidant capacity of procyanidins from hawthorn fruit. Food Ferment. Ind. 2007, 33, 45–47. [Google Scholar]
- Moon, H.I.; Kim, T.I.; Cho, H.S.; Kim, E.K. Identification of potential and selective collagenase, gelatinase inhibitors from Crataegus pinnatifida. Bioorg. Med. Chem. Lett. 2010, 20, 991–993. [Google Scholar] [CrossRef] [PubMed]
- Ammon, H.; Händel, M. Crateagus . Planta Med. 1981, 43, 209–239. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, M.; Haciseferogullari, H.; Marakoglu, T.; Arslan, D. Hawthorn (Crataegus spp.) fruit: Some physical and chemical properties. J. Food Eng. 2005, 69, 409–413. [Google Scholar] [CrossRef]
- Bahorun, T.; Aumjaud, E.; Ramphul, H.; Rycha, M.; Luximon-Ramma, A.; Trotin, F.; Aruoma, O.I. Phenolic constituents and antioxidant capacities of Crataegus monogyna (Hawthorn) callus extracts. Nahrung 2003, 47, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Y.; Zhang, Z.S.; Kwan, K.Y.; Zhu, M.; Ho, W.K.K.; Huang, Y. Endothelium-dependent relaxation induced by Hawthorn extract in rat mesenteric artery. Life Sci. 1998, 63, 1983–1991. [Google Scholar] [CrossRef]
- Quettier-Deleu, C.; Voiselle, G.; Fruchart, J.C.; Duriez, P.; Teissier, E.; Bailleul, F.; Vasseur, J.; Trotin, F. Hawthorn extracts inhibit LDL oxidation. Pharmazie 2003, 58, 577–581. [Google Scholar] [PubMed]
- Zhu, Y.P. Chinese Materia Medica: Chemistry, Pharmacology, and Applications; CRC Press: Boca Raton, FL, USA, 1998; p. 189. [Google Scholar]
- Rajendran, S.; Deepalakshmi, P.D.; Parasakthy, K.; Devarj, H.; Devaraji, N. Effect of tincture of Crateagus on the LDL-receptor activity of hepatic plasma membrane of rats fed an atherogenic diet. Atherosclerosis 1996, 123, 235–241. [Google Scholar] [CrossRef]
- Shanti, S.; Parasakthy, K.; Deepalakshmi, P.D.; Devaraj, S.N. Effect of tincture of Crateagus in rats. Indian J. Biochem. Biophys. 1994, 31, 143–146. [Google Scholar]
- Chen, J.B.; Xue, K.; Li, J.; Shi, D.; Krempin, M.; Zhu, M. The effect of an instant haw beverage on lipid levels, antioxidant enzyme and immune function in hyperlipidemia patients. Zhonghua Yu Fang Yi Xue Za Zhi 2002, 36, 172–175. [Google Scholar] [PubMed]
- Guan, Y.; Zhao, S. Yishou jiangzi (de-blood-lipid) tablets in the treatment of hyperlipidemia. J. Tradit. Chin. Med. 1995, 15, 178–179. [Google Scholar] [PubMed]
- Chen, Z.Y.; Jiao, R.; Ma, K.Y. Cholesterol-lowering nutraceuticals and functional foods. J. Agric. Food Chem. 2008, 56, 8761–8773. [Google Scholar] [CrossRef] [PubMed]
- Voneiff, M.; Brunner, H.; Haegeli, A.; Kreuter, U.; Martina, B.; Meier, B.; Schaffner, W. Hawthorn passion flower extract and improvement in physical exercise capacity of patients with dyspnea class-ii of the nyha functional classification. Acta Therap. 1994, 20, 47–66. [Google Scholar]
- Chen, J.D.; Wu, Y.Z.; Tao, Z.; Chen, Z.M.; Liu, X.P. Hawthorn (Shan Zha) drink and its lowering effect on blood lipid levels in humans and rats. World Rev. Nutr. Diet 1995, 77, 233–239. [Google Scholar]
- Luo, Y.C.; Chen, G.; Li, B.; Ji, B.P.; Xiao, Z.L.; Yi, G.; Tian, F. Dietary Intervention with AHP, a Functional Formula Diet, Improves Both Serum and Hepatic Lipids Profile in Dyslipidemia Mice. J. Food Sci. 2009, 74, H189–H195. [Google Scholar] [CrossRef] [PubMed]
- Min, S.W.; Jung, S.H.; Cho, K.H.; Kim, D.H. Antihyperlipidemic Effects of Red Ginseng, Crataegii Fructus and Their Main Constituents Ginsenoside Rg3 and Ursolic Acid in Mice. Biomol. Ther. 2008, 16, 364–369. [Google Scholar] [CrossRef]
- Ye, X.L.; Huang, W.W.; Chen, Z.; Li, X.G.; Lan, P.; Wang, L.; Gao, Y.; Zhao, Z.Q.; Chen, X. Synergic effect and structure-activity relationship of 3-hydroxy-3-methylglutaryl Coenzyme A reductase inhibitors from Crateagus pinnatifida Bge. J. Agric. Food Chem. 2010, 58, 3132–3138. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.K.K.; Chang, H.M. Method and compositions for lowering blood lipids, U.S. Patent 5,665,359, 9 September 1997. [Google Scholar]
- Kuo, D.H.; Yeh, C.H.; Shieh, P.C.; Cheng, K.C.; Chen, F.A.; Cheng, J.T. Effect of ShanZha, a Chinese herbal product, on obesity and dyslipidemia in hamsters receiving high-fat diet. J. Ethnopharmacol. 2009, 124, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.L.; Yan, J.; Qian, Y.; Wo, X.D.; Gao, L.P. Regulation of lipoprotein lipase expression by effect of hawthorn flavonoids on peroxisome proliferator response element pathway. J. Pharmacol. Sci. 2006, 100, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.S.; Chen, C.T.; Chen, L.J.; Cheng, K.C.; Yeh, C.H.; Cheng, J.T. Decrease of Blood Lipids Induced by Shan-Zha (Fruit of Crataegus pinnatifida) is Mainly Related to an Increase of PPAR alpha in Liver of Mice Fed High-Fat Diet. Horm. Metab. Res. 2011, 43, 625–630. [Google Scholar] [PubMed]
- Guo, R.; Pittler, M.H.; Ernst, E. Hawthorn extract for treating chronic heart failure. Cochrane Database of Syst. Rev. 2008, 1, 32. [Google Scholar] [CrossRef] [PubMed]
- Holubarsch, C.J.; Colucci, W.S.; Meinertz, T.; Gaus, W.; Tendera, M. Survival and Prognosis: Investigation of Crataegus Extract WS 1442 in congestive heart failure (SPICE)—Rationale, study design and study protocol. Eur. J. Heart Fail. 2000, 2, 431–437. [Google Scholar] [CrossRef]
- Hanack, T.; Bruckel, M.H. The treatment of mild stable forms of angina pectoris using Crategutt novo. Therapiewoche 1983, 33, 4331–4333. [Google Scholar]
- Hwang, H.S.; Bleske, B.E.; Ghannam, M.M.J.; Converso, K.; Russell, M.W.; Hunter, J.C.; Boluyt, M.O. Effects of hawthorn on cardiac remodeling and left ventricular dysfunction after 1 month of pressure overload-induced cardiac hypertrophy in rats. Cardiovasc. Drugs Ther. 2008, 22, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Vibes, J.; Lasserre, B.; Gleye, J.; Declume, C. Inhibition of thromboxane-a(2) biosynthesis in vitro by the main components of Crataegus oxyacantha (hawthorn) flower heads. Prostaglandins Leukot. Essent. Fatty Acids 1994, 50, 173–175. [Google Scholar] [CrossRef]
- Kim, S.H.; Kang, K.W.; Kim, K.W.; Kim, N.D. Procyanidins in Crataegus Extract evoke endothelium-dependent vasorelaxation in rat aorta. Life Sci. 2000, 67, 121–131. [Google Scholar] [CrossRef]
- Swinger, R.H.; Pietsch, M.; Frank, K.; Brixius, K. Crateagus special Extract WS 1442 increases force of contraction in human myocardium cAMP-independently. J. Cardiovasc. Pharmacol. 2000, 35, 700–707. [Google Scholar] [CrossRef]
- Dasgupta, A.; Kidd, L.; Poindexter, B.J.; Bick, R.J. Interference of Hawthorn on Serum Digoxin Measurements by Immunoassays and Pharmacodynamic Interaction With Digoxin. Arch. Pathol. Lab. Med. 2010, 134, 1188–1192. [Google Scholar] [PubMed]
- Iwaoka, E.; Noguchi, T.; Han, J.Y.; Lin, X.P.; Gao, M. Preventive effect of the Chinese herbal medicine “Myakuryu” on hypertension and stroke in stroke-prone spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol. 2007, 34, S51–S52. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, D.Q.; Li, Y.H.; Liu, H.; Liu, Z.B.; Zhao, C.F.; Hu, L.M.; Gao, X.M. Regulation effects on abnormal glucose and lipid metabolism of TZQ-F, a new kind of Traditional Chinese Medicine. J. Ethnopharmacol. 2010, 128, 575–582. [Google Scholar]
- Corder, R.; Warburton, R.C.; Khan, N.Q.; Brown, R.E.; Wood, E.G.; Lees, D.M. The procyanidin-induced pseudo laminar shear stress response: A new concept for the reversal of endothelial dysfunction. Clin. Sci. 2004, 107, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Xu, H.; Ngeh, L. The Evaluation of Chinese Therapeutic Food for the Treatment of Moderate Dyslipidemia. Evid. based Complement Altern. Med. 2012, 2012, 508683. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Chung, I.M.; Moon, H.I. Antiplasmodial procyanidins derivatives from Chinese Hawthorn. Immunopharmacol. Immunotoxicol. 2010, 32, 607–610. [Google Scholar] [CrossRef] [PubMed]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jurikova, T.; Sochor, J.; Rop, O.; Mlcek, J.; Balla, S.; Szekeres, L.; Adam, V.; Kizek, R. Polyphenolic Profile and Biological Activity of Chinese Hawthorn (Crataegus pinnatifida BUNGE) Fruits. Molecules 2012, 17, 14490-14509. https://doi.org/10.3390/molecules171214490
Jurikova T, Sochor J, Rop O, Mlcek J, Balla S, Szekeres L, Adam V, Kizek R. Polyphenolic Profile and Biological Activity of Chinese Hawthorn (Crataegus pinnatifida BUNGE) Fruits. Molecules. 2012; 17(12):14490-14509. https://doi.org/10.3390/molecules171214490
Chicago/Turabian StyleJurikova, Tunde, Jiri Sochor, Otakar Rop, Jiri Mlcek, Stefan Balla, Ladislav Szekeres, Vojtech Adam, and Rene Kizek. 2012. "Polyphenolic Profile and Biological Activity of Chinese Hawthorn (Crataegus pinnatifida BUNGE) Fruits" Molecules 17, no. 12: 14490-14509. https://doi.org/10.3390/molecules171214490
APA StyleJurikova, T., Sochor, J., Rop, O., Mlcek, J., Balla, S., Szekeres, L., Adam, V., & Kizek, R. (2012). Polyphenolic Profile and Biological Activity of Chinese Hawthorn (Crataegus pinnatifida BUNGE) Fruits. Molecules, 17(12), 14490-14509. https://doi.org/10.3390/molecules171214490





