In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study
Abstract
:1. Introduction
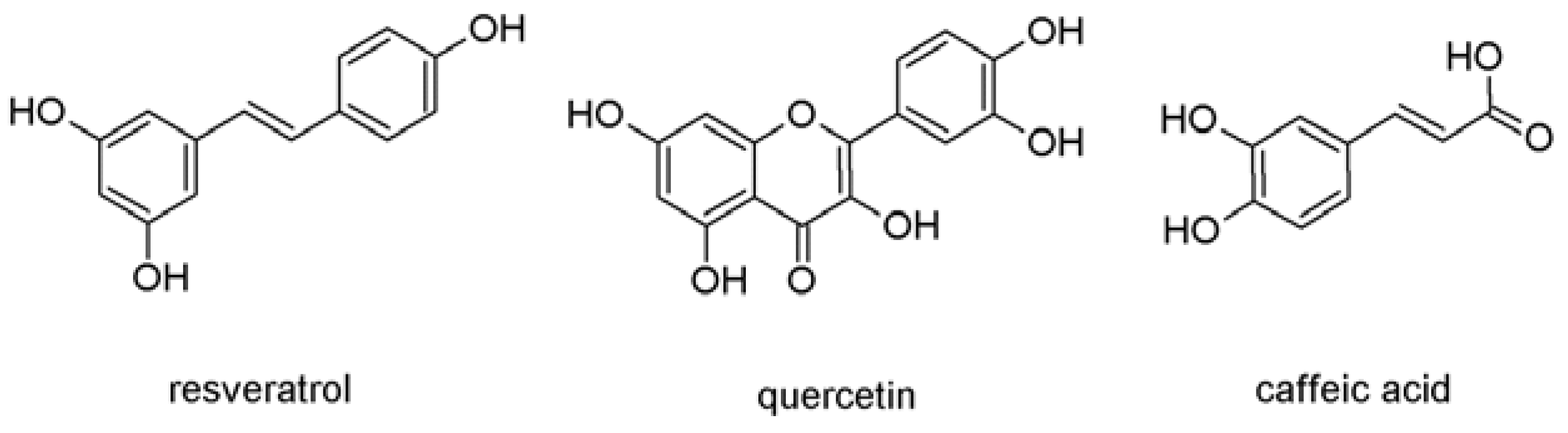
2. Results and Discussion
2.1. Antioxidant Effects of Single Wine Polyphenols and Their Mixtures
| SRD | NRD | FRAP | RP | DPPH | ABTS | NO | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | IC50 (µM) | r | IC50 (µM) | r | IC50 (µM) | r | IC50 (µM) | r | IC50 (µM) | r | IC50 (µM) | r | IC50 (µM) | r |
| Quercetin | 3.82 | 0.95 | 3.61 | 0.97 | 55.08 | 0.99 | 253.51 | 0.98 | 4.36 | 0.99 | 2.87 | 0.97 | 54.94 | 0.94 |
| Resveratrol | 192.53 | 0.98 | 107.08 | 0.98 | 162.09 | 0.99 | 780.42 | 0.99 | 98.02 | 0.97 | 3.98 | 0.96 | 95.22 | 0.96 |
| Caffeic acid | 9.93 | 0.99 | 7.79 | 0.97 | 77.73 | 0.94 | 132.29 | 0.99 | 4.48 | 0.99 | 6.82 | 0.97 | 48.39 | 0.97 |
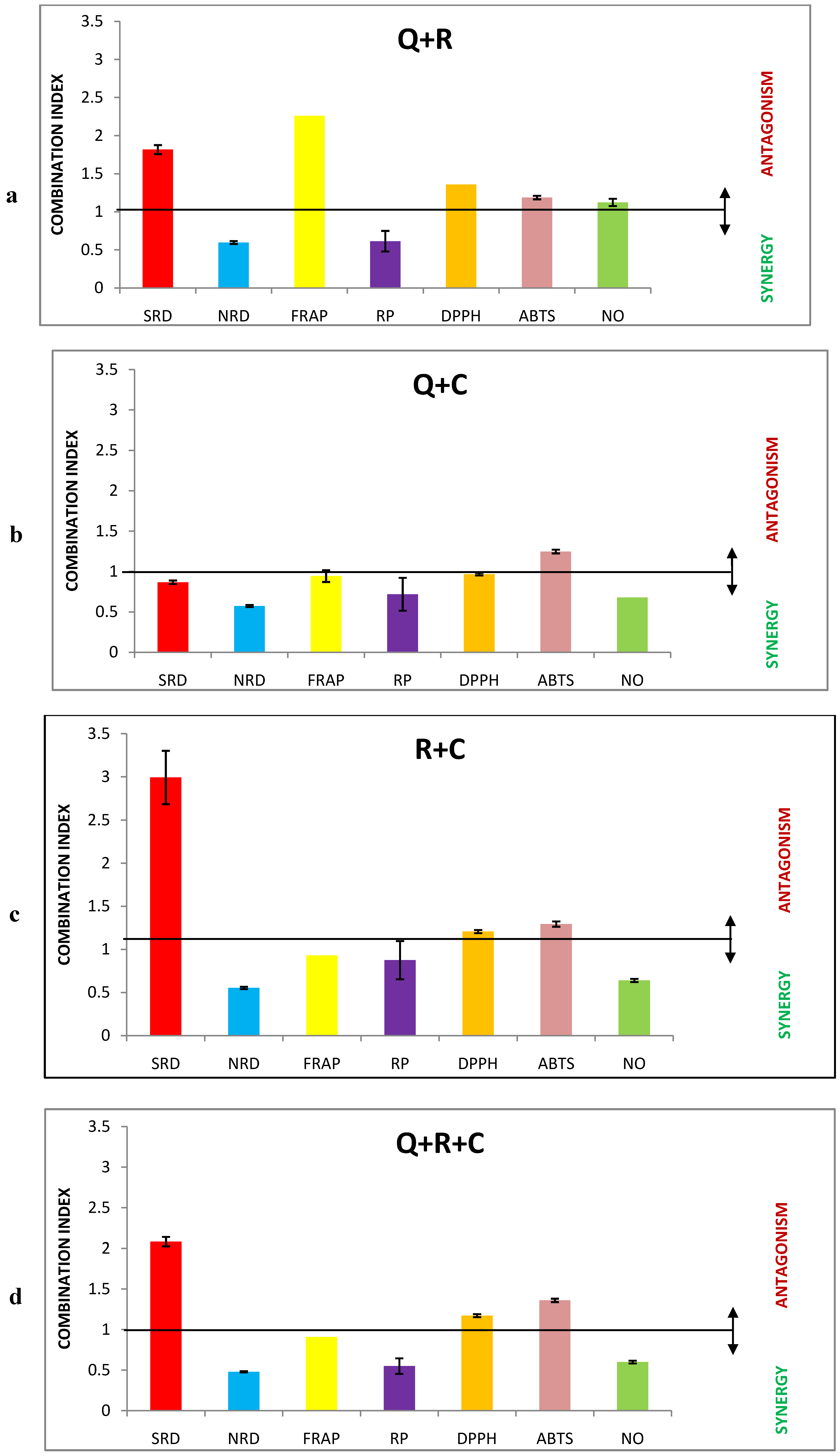
2.2. Interaction Analysis of Antioxidant Activities of Polyphenolic Mixtures
| Mixture | IC50 (µM) | Absolute contributions (µM) | r | DRI | |
|---|---|---|---|---|---|
| SRD | Q+R | 13.6 | 6.8 + 6.8 | 0.99 | 0.6:28.3 |
| Q+C | 4.8 | 2.4 + 2.4 | 0.98 | 1.6:4.2 | |
| R+C | 56.3 | 28.15 + 28.15 | 0.92 | 6.8:0.4 | |
| Q+R+C | 17.0 | 5.66 + 5.66 + 5.66 | 0.95 | 0.7:34.0:1.8 | |
| NRD | Q+R | 4.2 | 2.1 + 2.1 | 0.89 | 1.7:51.4 |
| Q+C | 2.8 | 1.4 + 1.4 | 0.97 | 2.6:5.5 | |
| R+C | 8.0 | 4.0 + 4.0 | 0.96 | 26.7:1.9 | |
| Q+R+C | 3.5 | 1.16 + 1.16 + 1.16 | 0.99 | 3.1:92.4:6.7 | |
| FRAP | Q+R | 185.7 | 92.85 + 92.85 | 0.99 | 0.6:1.7 |
| Q+C | 60.8 | 30.4 + 30.4 | 0.93 | 1.8:2.6 | |
| R+C | 97.8 | 48.9 + 48.9 | 0.99 | 3.3:1.6 | |
| Q+R+C | 73.3 | 24.43 + 24.43 + 24.43 | 0.98 | 2.3:6.6:3.2 | |
| RP | Q+R | 231.0 | 15.5 + 15.5 | 0.99 | 2.2:6.8 |
| Q+C | 122.7 | 61.35 + 61.35 | 0.99 | 4.1:2.2 | |
| R+C | 193.8 | 96.9 + 96.9 | 0.99 | 8.1:1.4 | |
| Q+R+C | 128.1 | 42.7 + 42.7 + 42.7 | 0.99 | 5.9:18.3:3.1 | |
| DPPH | Q+R | 11.4 | 5.7 + 5.7 | 0.99 | 0.8:17.3 |
| Q+C | 4.3 | 2.15 + 2.15 | 0.97 | 2.0:2.1 | |
| R+C | 10.4 | 5.2 + 5.2 | 0.99 | 19.0:0.9 | |
| Q+R+C | 7.6 | 2.53 + 2.53 + 2.53 | 0.98 | 1.7:38.7:1.8 | |
| ABTS | Q+R | 13.6 | 6.8 + 6.8 | 0.99 | 1.4:2.0 |
| Q+C | 4.8 | 2.4 + 2.4 | 0.98 | 1.1:2.7 | |
| R+C | 56.3 | 28.15 + 28.15 | 0.92 | 1.2:2.1 | |
| Q+R+C | 17.0 | 8.5 + 8.5 | 0.95 | 1.6:2.2:3.7 | |
| NO | Q+R | 78.2 | 39.1 + 39.1 | 0.93 | 1.4:2.4 |
| Q+C | 34.9 | 17.45 + 17.45 | 0.95 | 3.1:2.8 | |
| R+C | 41.0 | 20.5 + 20.5 | 0.97 | 4.6:2.4 | |
| Q+R+C | 36.4 | 12.13 + 12.13 + 12.13 | 0.92 | 4.5:7.8:4.0 |
3. Experimental
3.1. Instrument and Software
3.2. Chemicals
3.3. Site-Specific Hydroxyl Radical-Mediated 2-Deoxy-D-ribose Degradation (SRD)
3.4. Nonsite-Specific Hydroxyl Radical-Mediated 2-Deoxy-D-Ribose Degradation (NRD)
3.5. Ferric Ion Reducing Antioxidant Power Assay (FRAP)
3.6. Ferric Ion Reducing Power Assay (RP)
3.7. DPPH Scavenging Assay
3.8. ABTS Scavenging Assay
3.9. NO Scavenging Assay
3.10. Statistical and Interaction Analysis
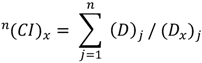
4. Conclusions
Acknowledgments
Conflict of Interest
- Sample Availability: Samples of the compounds are available from the authors.
References
- Gülçin, I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid). Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef]
- Obrenovich, M.E.; Nair, N.G.; Beyaz, A.; Aliev, G.; Reddy, V.P. The role of polyphenolic antioxidants in health, disease, and aging. Rejuvenation Res. 2010, 13, 631–643. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Hanhineva, K.; Törrönen, R.; Bondia-Pons, I.; Pekkinen, J.; Kolehmainen, M.; Mykkänen, H.; Poutanen, K. Impact of dietary polyphenols on carbohydrate metabolism. Int. J. Mol. Sci. 2010, 11, 1365–1402. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. Engl. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Soleas, G.J.; Diamandis, E.P.; Goldberg, D.M. Wine as a biological fluid: History, production, and role in disease prevention. J. Clin. Lab. Anal. 1997, 11, 287–313. [Google Scholar] [CrossRef]
- Biavatti, M.W. Synergy: An old wisdom, a new paradigm for pharmacotherapy. Braz. J. Pharm. Sci. 2009, 45, 371–378. [Google Scholar]
- Herrmann, F.; Wink, M. Synergistic interactions of saponins and monoterpenes in HeLa cells, Cos7 cells and in erythrocytes. Phytomedicine 2011, 18, 1191–1196. [Google Scholar] [CrossRef]
- Alén-Ruiz, F.; García-Falcón, M.S.; Pérez-Lamela, M.C.; Martínez-Carballo, E.; Simal-Gándara, J. Influence of major polyphenols on antioxidant activity in Mencía and Brancellao red wines. Food Chem. 2009, 113, 53–60. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C.; Aruoma, O. The deoxyribose method: A simple “test tube” assay for determination of rate constants for reactionsof hydroxyl radicals. Anal. Biochem. 1987, 165, 215–219. [Google Scholar]
- Chobot, V. Simultaneous detection of pro- and antioxidative effects in the variants of the deoxyribose degradation assay. J. Agric. Food Chem. 2010, 58, 2088–2094. [Google Scholar] [CrossRef]
- Belguendouz, L.; Fremont, L.; Linard, A. Resveratrol inhibits metal ion-dependent and independent peroxidation of porcine low-density lipoproteins. Biochem. Pharmacol. 1997, 53, 1347–1355. [Google Scholar]
- Perron, N.R.; Brumaghim, J.L. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 2009, 53, 75–100. [Google Scholar] [CrossRef]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef]
- Hider, R.C.; Liu, Z.D.; Khodr, H.H. Metal chelation of polyphenols. Methods Enzymol. 2001, 335, 190–203. [Google Scholar]
- Jameson, G.N.L.; Linert, W. The oxidation of 6-hydroxydopamine in aqueous solution. Part 3. Kinetics and mechanism of the oxidation with iron(III). J. Chem. Soc.Perkin Trans. 2 2001, 2, 569–575. [Google Scholar] [CrossRef]
- Litwinienko, G.; Ingold, K.U. Abnormal solvent effects on hydrogen atom abstractions. 1. The reactions of phenols with 2,2-diphenyl-1-picrylhydrazyl (dpph*) in alcohols. J. Org. Chem. 2003, 68, 3433–3438. [Google Scholar] [CrossRef]
- Villano, D.; Fernández-Pachón, M.S.; Moyá, M.L.; Troncoso, A.M.; García-Parrilla, M.C. Radical scavenging ability of polyphenolic compounds towards DPPH free radical. Talanta 2007, 71, 230–235. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationship of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Sueishi, Y.; Hori, M.; Kita, M.; Kotake, Y. Nitric oxide (NO) scavenging capacity of natural antioxidants. Food Chem. 2011, 129, 866–870. [Google Scholar] [CrossRef]
- Číž, M.; Pavelková, M.; Gallová, L.; Králová, J.; Kubala, L.; Lojek, A. The influence of wine polyphenols on reactive oxygen and nitrogen species production by murine macrophages RAW 264.7. Physiol. Res. 2008, 57, 393–402. [Google Scholar]
- De la Puerta, R.; Martínez Domínguez, M.E.; Ruíz-Gutíerrez, V.; Flavill, J.A.; Hoult, J.R. Effects of virgin olive oil phenolics on scavenging of reactive nitrogen species and upon nitrergic neurotransmission. Life Sci. 2001, 69, 1213–1222. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Peyrat-Maillard, M.N.; Cuvelier, M.E.; Berset, C. Antioxidant activity of phenolic compounds in 2,2'-azobis (2-amidinopropane) dihydrochloride (AAPH)-induced oxidation: Synergistic and antagonistic effects. J. Am. Oil Chem. Soc. 2003, 80, 1007–1012. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.F. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar]
- Tian, B.; Hua, Y. Concentration dependence of prooxidant and antioxidant effects of aloin and aloe-emodin on DNA. Food Chem. 2005, 91, 413–418. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determination by the use of a stable free radical. Nature 1958, 181, 1119–1200. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Marcocci, L.; Maguire, J.J.; Droy-Lefaix, M.T.; Packer, L. The nitric oxide-scavenging properties of Ginkgo biloba extract EGB 761. Biochem. Biophys. Res. Commun. 1994, 201, 748–755. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kurin, E.; Mučaji, P.; Nagy, M. In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study. Molecules 2012, 17, 14336-14348. https://doi.org/10.3390/molecules171214336
Kurin E, Mučaji P, Nagy M. In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study. Molecules. 2012; 17(12):14336-14348. https://doi.org/10.3390/molecules171214336
Chicago/Turabian StyleKurin, Elena, Pavel Mučaji, and Milan Nagy. 2012. "In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study" Molecules 17, no. 12: 14336-14348. https://doi.org/10.3390/molecules171214336
APA StyleKurin, E., Mučaji, P., & Nagy, M. (2012). In Vitro Antioxidant Activities of Three Red Wine Polyphenols and Their Mixtures: An Interaction Study. Molecules, 17(12), 14336-14348. https://doi.org/10.3390/molecules171214336




