Medicinal Plants: A Source of Anti-Parasitic Secondary Metabolites
Abstract
:1. Introduction
2. Human Parasites and Parasitic Diseases
| Parasite | Disease (estimated number of infections) | Vector (hosts); route of transmission | Distribution | Symptoms |
|---|---|---|---|---|
| Protozoa | ||||
| Apicomplexa | ||||
| Babesia spp. | Piroplasmosis (rare) | Ticks (Ixodes); bites | North America | Anaemia, damage of immune system |
| Plasmodium | Malaria (>250 million) | Mosquitos | Tropics and subtropics | Anaemia, enlarged liver and spleen, high fever, jaundice, haemorrhage, haemoglobinuria (“blackwater fever”); blockage of cerebral capillaries (P. falciparum) |
| (P. vivax , P. ovale , | ||||
| P. malariae , | ||||
| P. knowlesi , | ( Anopheles , Nyssorhynchus , | |||
| P. falciparum ) | Cellia , Kerteszia ); bites | |||
| Toxoplasma gondii | Toxoplasmosis | Main host are cats; infection of humans from faeces | Worldwide | Flu-like symptoms; cysts in muscle and neural tissues; encephalitis, serious danger for developing foetus (abortion, malformations) |
| Trypanosomatida | ||||
| Trypanosoma brucei; | African trypanosomiasis (>500,000) | Tsetse flies (Glossina); bites | Tropical Africa | Fever, rash, lymphoadenopathy, sleeping sickness (waste, comatose) |
| T. b. gambiense | ||||
| T. b. rhodesiense | ||||
| T. b. brucei | Nagana (only cattle) | Tsetse flies (Glossina); bites | Tropical Africa | Loss of cattle; symptoms as in humans |
| Trypanosoma cruzi | Chagas disease (10 million) | Bugs of the family Reduviidae (Rhodnius, Triatoma, Panstrongylus); bites | Central and South America | Local tissue lesions of eyes (Romana’s sign), myocarditis, cardiomegaly, megaoesophagus, megacolon |
| Leishmania donovani | Visceral leishmaniasis (kala-azar) (15 million) | Flies (Phlebotomus, | N-Africa, Eurasia | Enlargement of liver and spleen, fever, dermal lesions, dermal nodules |
| Lutzomyia ); bites | S America | |||
| Leishmania tropica | Cutaneous leishmaniasis (Old world) | Phlebotomus; bites | Eurasia, Africa | Ulcerative lesions, mucocutaneous lesions |
| L. major | ||||
| L. infantum | ||||
| Leishmania mexicana and several others | Cutaneous leishmaniasis (New world) | Lutzomyia; bites | Central and Southern America | Ulcerative lesions, mucocutaneous lesions |
| Amoebida | ||||
| Entamoeba histolytica and other species | Amoebiasis (70,000 deaths/year) | Infection from contaminated water or food | Worldwide | Dysentery, destruction of intestinal tissues, fever, liver and lung abscess |
| Diplomonadida | ||||
| Giardia lamblia | Giardiasis (2 million infections/year in USA) | Infection from contaminated water | Worldwide | Infection of duodenal and jejunal mucosa; diarrhoea, fever |
| Trichomonadida | ||||
| Trichomonas vaginalis | Trichomoniasis (180 million each year) | Sexual transmission | Worldwide | Mucosal tissue of genital tract |
| Trichomonas hominis | ||||
| METAZOA | ||||
| Nematoda | ||||
| Filarioidea | ||||
| Wucheria bancrofti Brugia spp. | Lymphatic filariases, elephantiasis (120 million) | Mosquitos (Aedes, Culex, Mansonia); bites | Tropical Africa, Asia, America | Infection of lymphatic system; enlargement of lymph nodes |
| Mansonella spp. | ||||
| Loa loa | Loaiasis (33 million) | Chrysops | Central Africa | Female worms migrate through tissues and the eye |
| Onchocerca volvulus | Skin filariases; onchocerciasis; river blindness (>17 million) | Flies (Simulium spp.); bites | Mostly tropical Africa and America | Formation of large nodules under skin or in eyes (causing blindness) |
| Trichuroidea | ||||
| Trichinella spiralis | Trichinosis (50 million) | Eating of infected muscles e.g., from pigs; bites | Worldwide | Invades muscular tissue, fever, myalgia, malaise and oedema |
| Trichurus trichiura | (500 million) | Infection from contaminated soil | Worldwide | Intestinal infection |
| Rhabditoidea | ||||
| Strongyloides stercoralis | Strongyloidiasis (70 million) | Infection from contaminated soil | Subtropics, tropics worldwide | Intestinal infection, anaemia; migrating larvae in skin |
| Ancylostomatoidea | ||||
| Ancylostoma duodenale; | Hookworm infection (700–900 million) | Infection from contaminated soil | Subtropics, tropics worldwide | Intestinal infection, anaemia, migrating larvae in skin |
| Necator americanus | ||||
| Oxyuroidea | ||||
| Enterobius vermicularis | Thread or pinworm (400 million) | Infection from contaminated humans | Worldwide | Intestinal infection |
| Ascaridoidea | ||||
| Ascaris lumbricoides | Ascariasis (800–1000 million) | Infection from contaminated soil | Worldwide | Intestinal infection, migrating larvae in various tissues |
| Dracunculoidea | ||||
| Dracunculus medinensis | Dracunculiasis; guinea worm infection (<3 million) | Copepods as intermediate host | Africa, Asia | Infects skin; female worms can reach a length of 100 cm |
| PlathelmintesTrematoda | ||||
| Schistosomatoidea | ||||
| Schistosoma mansoni | Schistosomiasis (200 million) | Water snails as intermediate host | Tropical and subtropical Africa, S America and E Asia | Dermatitis, infects liver, granuloma formation in liver, liver fibrosis, enlarged spleen |
| S. japonicum | ||||
| S. haematobium | Schistosomiasis (80 million) | Water snails as intermediate host | Africa | Infection of bladder, haematuria |
| Echinostomatoidea | ||||
| Fasciola hepatica | Fasciolopsiasis (2.4 million) | Water snail (Lymnaea) as host | Worldwide | Infection of liver |
| F. gigantica | ||||
| Opisthorchioidea | ||||
| Opisthorchis felineus | Opisthorchiasis, liver fluke (10 million) | Water snails as intermediate host; infection from infected fish | E Europe, Central and Eastern Asia | Infection of liver and gall bladder |
| O. viverrini | ||||
| O. sinensis (syn. Clonorchis) | Clonorchiasis (35 million) | Water snails as intermediate host; infection from infected fish | China, Japan | Infection of liver, fibrosis, carcinoma |
| Plagiorchioidea | ||||
| Paragoniumus westermani | Paragonimiasis (20 million) | Water snails and craps as intermediate hosts; infection from infected meat | Tropics of Africa, America and E Asia | Infection of internal organs, including lungs and brain |
| P. mexicanus and other species | ||||
| Platyhelminthes/Cestoda | ||||
| Diphyllobothrium latum and other cestodes | Diphyllobo-thriasis (fish tapeworm) (16 million) | Infection from infected fish | Worldwide | Intestinal infection; Vit B12 deficiency |
| Dipylidium caninum | Dog tapeworm (rare) | Dogs and cats are main hosts; fleas intermediate hosts which can infect humans; bites | Worldwide | Intestinal infection |
| Vampirolepis nana | Dwarf tapeworm (36 million) | Rodents are main hosts; insects intermediate hosts; bites | Worldwide | Intestinal infection |
| (syn . Hymenolepis) | ||||
| Taenia solium | Pork and beef tapeworm (80 million) | Infection from contaminated meat | Worldwide | Intestinal infection; cysts in various tissues (including brain) |
| T. asiatica | ||||
| T. saginata | ||||
| Echinococcus granulosus | Hydatidosis, echinococcosis (thousands) | Dogs, foxes; | Worldwide | Cyst (hydatid) formation in liver, lung or brain |
| E. multilocularis | Infection from faeces |
3. Antiparasitic Medicinal Plants and Their Secondary Metabolites
3.1. Mode of Action of Antiparasitic Cytotoxic Drugs
3.1.1. DNA, RNA
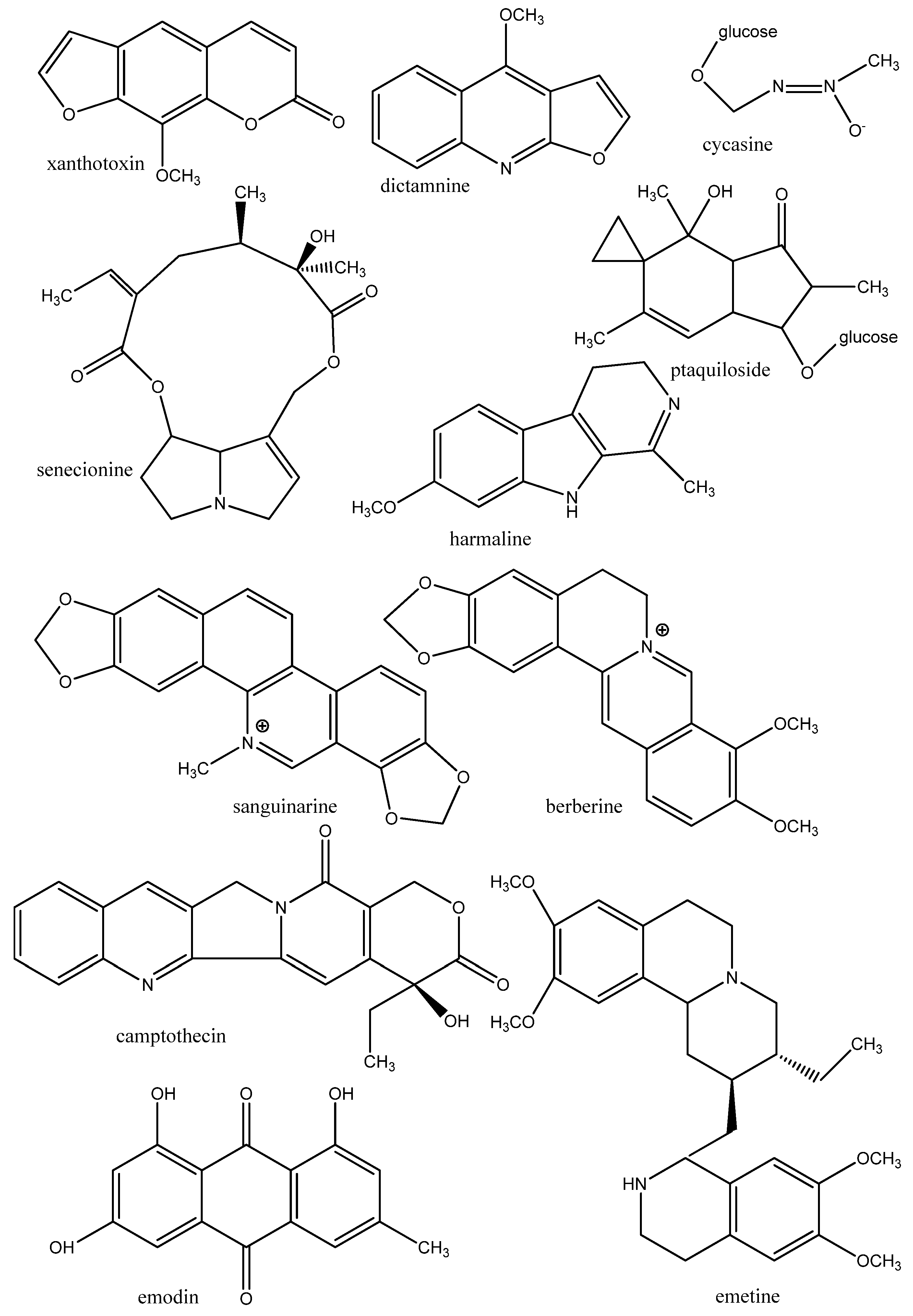
3.1.2. Proteins of the Cytoskeleton and Enzymes
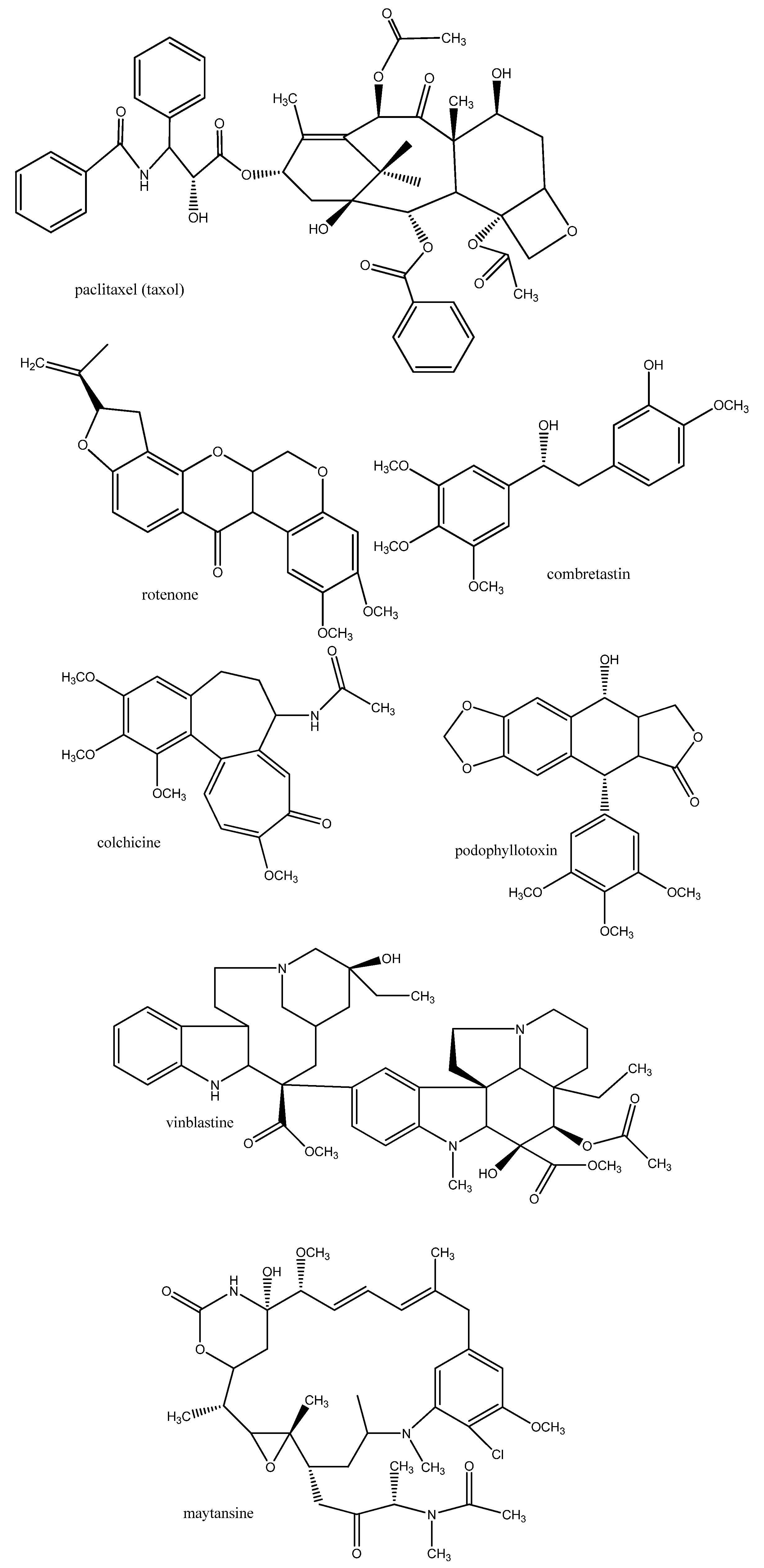
3.1.3. Biomembranes
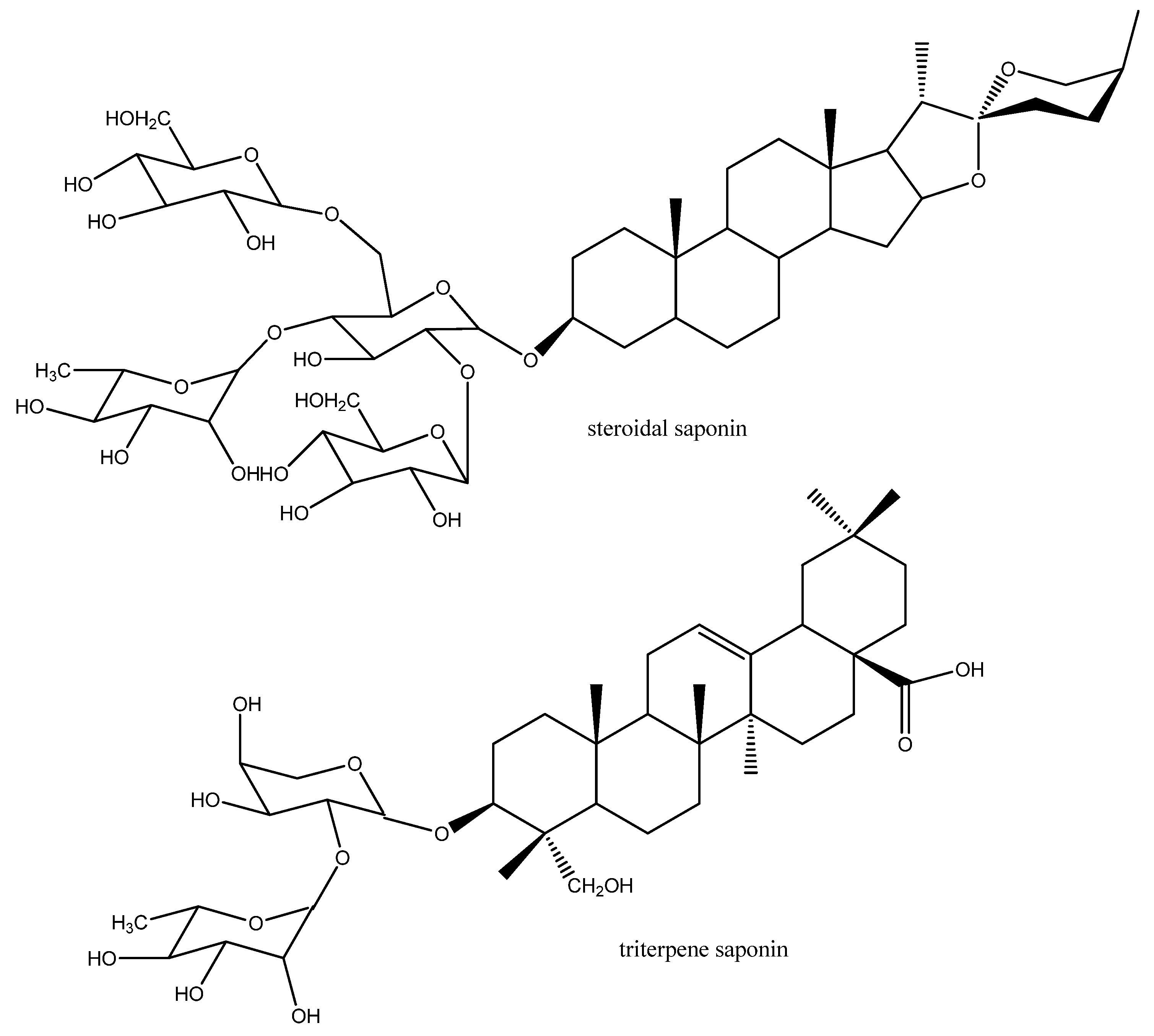
3.1.4. Nervous System
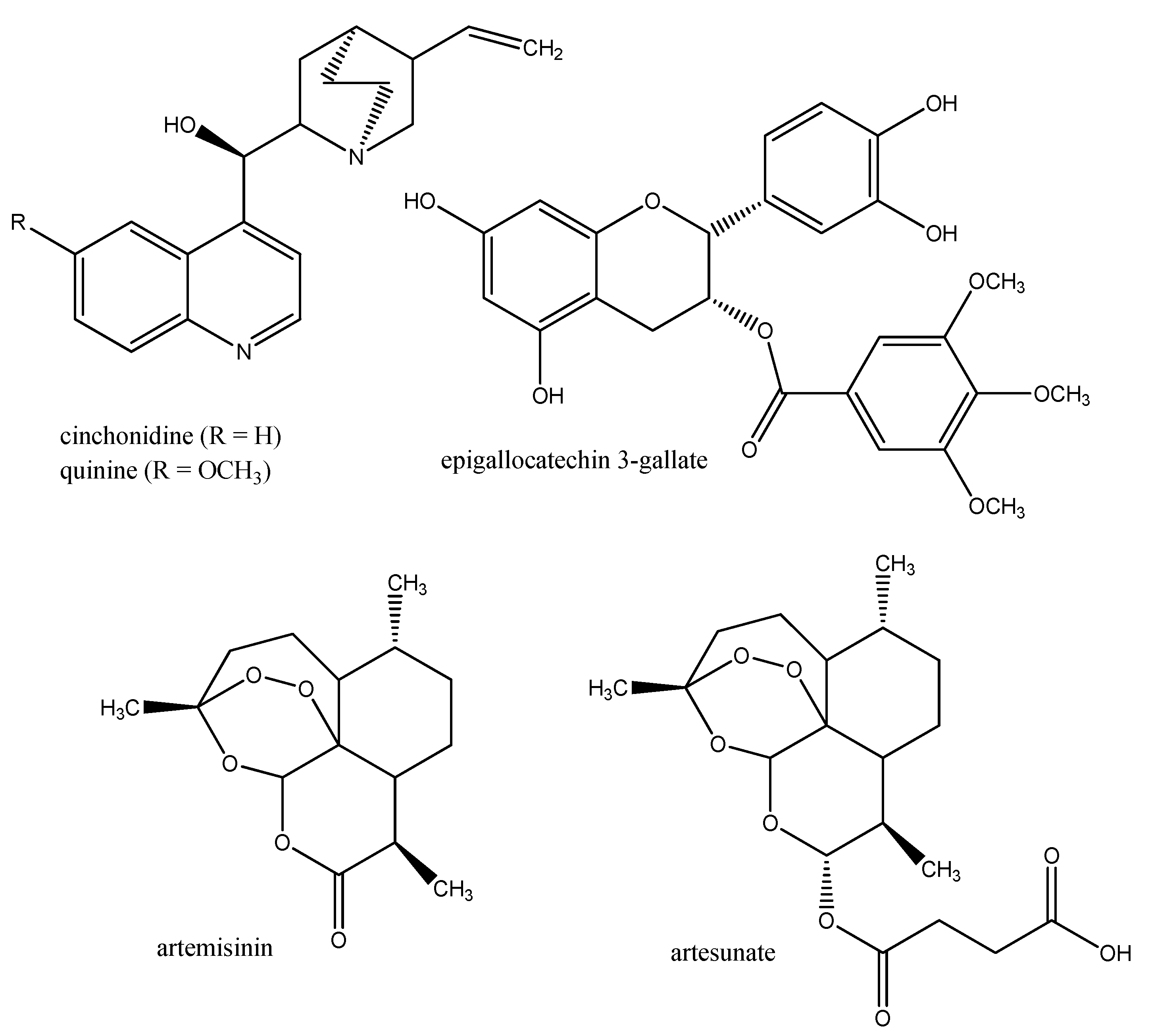
3.2. Anti-malaria Drugs
3.3. Drugs against Trypanosomes (Trypanosoma, Leishmania)
3.3.1. Trypanomiasis
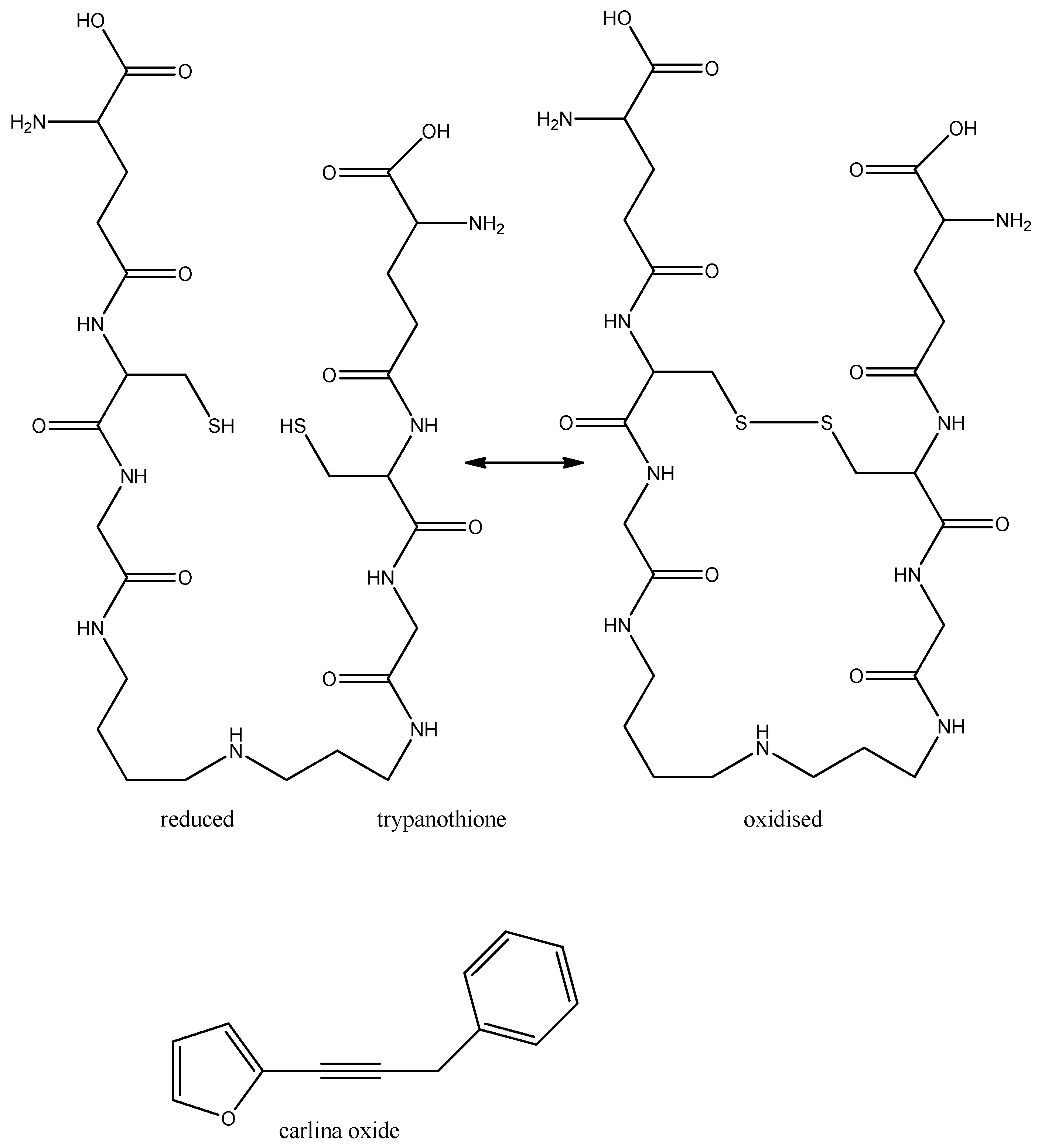
3.3.2. Chagas Disease
3.3.3. Leishmaniasis
3.4. Drugs against Trichomonas Vaginalis
3.5. Drugs against Worms (Nematoda, Cestoda, Trematoda)
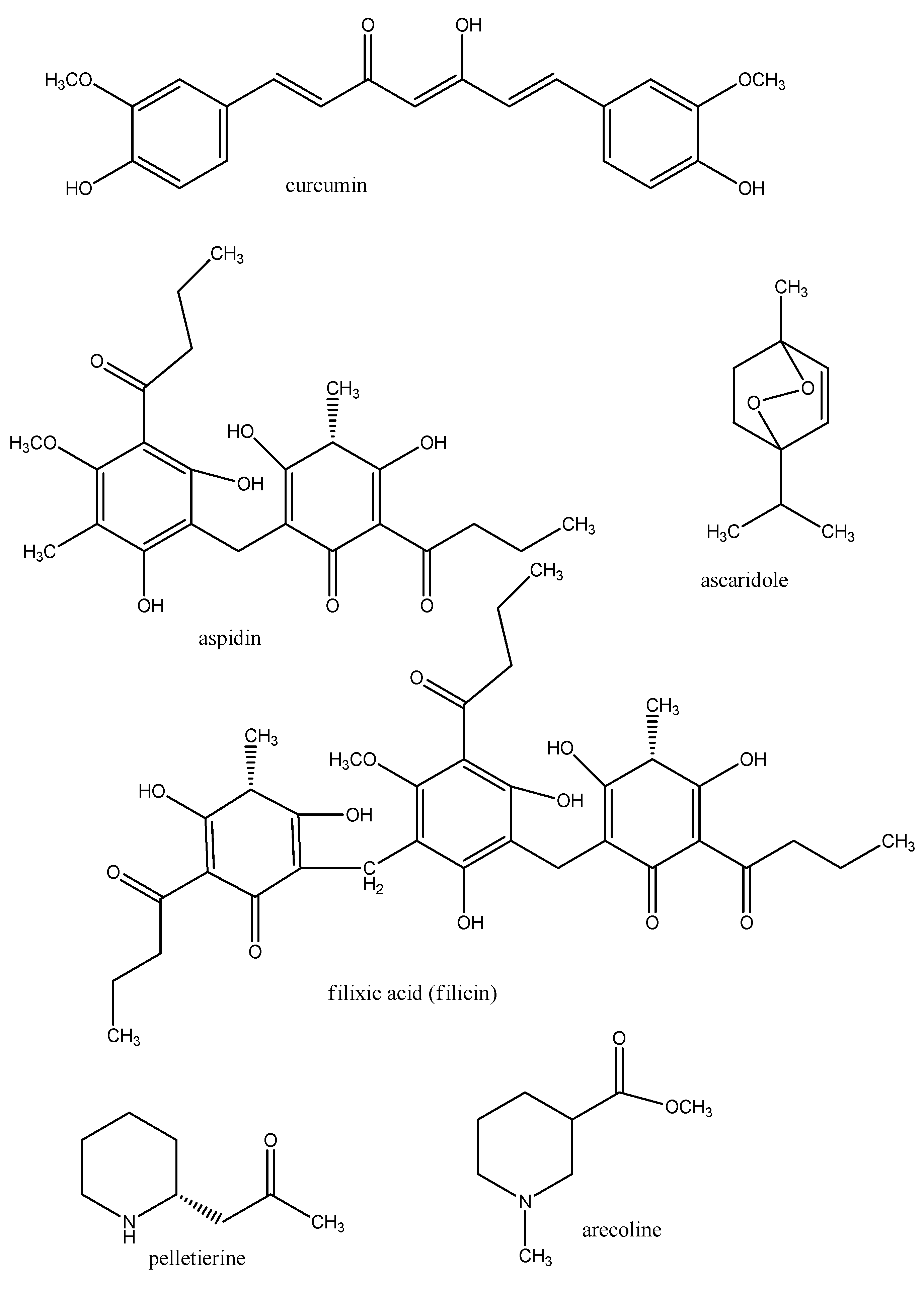
4. Conclusions
Acknowledgements
References
- Van Wyk, B.-E.; Wink, M. Medicinal Plants of the World: An Illustrated Scientific Guide to Important Medicinal Plants and Their Uses; Timber Press: Portland, OR, USA, 2004. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef]
- Wink, M. Functions and Biotechnology of Plant Secondary Metabolites; Wiley-Blackwell: Oxford, UK, 2010; Volume 39. [Google Scholar]
- Peters, W.; Pasvol, G. Atlas of Tropical Medicine and Parasitology, 6th ed; Mosby-Elsevier: Philadelphia, PA, USA, 2007. [Google Scholar]
- Nussbaum, K.; Honek, J.; Cadmus, C.; Efferth, T. Trypanosomatid parasites causing neglected diseases. Curr. Med. Chem. 2007, 17, 1594–1617. [Google Scholar]
- Wink, M. Production and application of phytochemicals from an agricultural perspective. In Phytochemistry and Agriculture, Proceedings Phytochemical Society Of Europe; van Beek, T.A., Breteler, H., Eds.; Oxford University Press: Oxford, UK, 1993; Volume 34, pp. 171–213. [Google Scholar]
- Pohlit, A.M.; Rezende, A.R.; Lopez Baldin, E.L.; Peporine Lopes, N.; de Andrade Neto, V.F. Plant extracts, isolated phytochemicals, and plant-derived agents which are lethal to arthropod vectors of human tropical diseases—A review. Planta Med. 2011, 77, 618–630. [Google Scholar] [CrossRef]
- Wink, M. Molecular modes of action of cytotoxic alkaloids- From DNA intercalation, spindle poisoning, topoisomerase inhibition to apoptosis and multiple drug resistance. Alkaloids 2007, 64, 1–48. [Google Scholar]
- Wink, M.; van Wyk, B.E. Mind-Altering and Poisonous Plants of the World; Timber Press: Portland, OR, USA, 2008. [Google Scholar]
- Wink, M.; Schimmer, O. Molecular modes of action of defensive secondary metabolites. In Functions and Biotechnology of Plant Secondary Metabolites; Wink, M., Ed.; Wiley-Blackwell: Oxford, UK, 2010; pp. 21–161. [Google Scholar]
- Schmeller, T.; Latz-Brüning, B.; Wink, M. Biochemical activities of berberine, palmatine and sanguinarine mediating chemical defence against microorganisms and herbivores. Phytochemistry 1997, 44, 257–266. [Google Scholar]
- Efferth, T.; Fu, Y.; Zu, Y.; Schwarz, G.; Newman, D.; Wink, M. Molecular target-guided tumor therapy with natural products derived from Traditional Chinese Medicine. Curr. Med. Chem. 2007, 14, 2024–2032. [Google Scholar] [CrossRef]
- Rosenkranz, V.; Wink, M. Alkaloids induce programmed cell death in bloodstream forms of trypanosomes (Trypanosoma b. brucei). Molecules 2008, 13, 2462–2473. [Google Scholar] [CrossRef]
- Stanton, R.A.; Gernert, K.M.; Nettles, J.H.; Aneja, R. Drugs that target dynamic microtubules: A new molecular perspective. Med. Res. Rev. 2011, 31, 443–481. [Google Scholar] [CrossRef]
- Möller, M.; Wink, M. Characteristics of apoptosis induction by the alkaloid emetine in human tumor cell lines. Planta Med. 2007, 73, 1–8. [Google Scholar]
- Wink, M. Evolutionary advantage and molecular modes of action of multi-component mixtures used in phytomedicine. Curr. Drug. Metab. 2008, 9, 996–1009. [Google Scholar] [CrossRef]
- Wink, M. Interference of alkaloids with neuroreceptors and ion channels. Stud. Nat. Prod. 2000, 21, 3–129. [Google Scholar] [CrossRef]
- Willcox, M. Improved traditional phytomedicines in current use for the clinical treatment of malaria. Planta Med. 2011, 77, 662–671. [Google Scholar] [CrossRef]
- Wink, M.; Ashour, M.; El-Readi, M.Z. Secondary metabolites inhibiting ABC transporters and reversing resistance of cancer cells and fungi to cytotoxic and antimicrobial agents. Front. Microbiol. 2012, 3, 1–15. [Google Scholar]
- Efferth, T.; Herrmann, F.; Tahrani, A.; Wink, M. Cytotoxic activity towards cancer cells of secondary constituents derived from Artemisia annua L. in comparison to its designated active constituent artemisinin. Phytomedicine 2011, 18, 959–969. [Google Scholar] [CrossRef]
- Kuhn, T.; Wang, Y. Artemisinin—An innovative cornerstone for anti-malarial therapy. Prog. Drug Res. 2008, 66, 385–422. [Google Scholar]
- Bero, J.; Quetin-Leclercq, J. Natural products published in 2009 from plants traditionally used to treat malaria. Planta Med. 2011, 77, 631–640. [Google Scholar] [CrossRef]
- Bero, J.; Frederich, M.; Quetin-Leclercq, J. Antimalarial compounds isolated from plants used in traditional medicine. J. Pharm. Pharmacol. 2009, 61, 1401–1433. [Google Scholar]
- Bilia, A.R. Non-nitrogenous plant-derived constituents with antiplasmodial activity. Nat. Prod. Comm. 2006, 1, 1181–1204. [Google Scholar]
- Caniato, T.; Puricelli, L. Review: Natural antimalarial agents (1995-2001). Crit. Rev. Plant Sci. 2003, 22, 79–105. [Google Scholar] [CrossRef]
- Fournet, A.; Munoz, V. Natural products as trypanocidal, antileishmanial and antimalarial drugs. Curr. Top. Med. Chem. 2002, 2, 1215–1238. [Google Scholar] [CrossRef]
- Haddad, M.; Sauvain, M.; Deharo, E. Curcuma as a parasiticidal agent: A review. Planta Med. 2011, 77, 672–678. [Google Scholar] [CrossRef]
- Hellmann, J.K.; Münter, S.; Wink, M.; Frischknecht, F. Synergistic and additive effects of epigallocatechin gallate and digitonin on Plasmodium sporozoite survival and motility. PLoS One 2010, 5, e8682. [Google Scholar]
- Kaur, K.; Jain, M.; Kaur, T.; Jain, R. Antimalarials from nature. Bioorg. Med. Chem. 2009, 17, 3229–3256. [Google Scholar] [CrossRef]
- Nogueira, C.R.; Lopes, L.M.X. Antiplasmodial natural products. Molecules 2011, 16, 2146–2190. [Google Scholar] [CrossRef]
- Phillipson, J.D. Natural products as drugs. Trans. R. Soc. Trop. Med. Hyg. 1994, 88, 17–19. [Google Scholar] [CrossRef]
- Saxena, S.; Pant, N.; Jain, D.C.; Bhakuni, R.S. Antimalarial agents from plant sources. Curr. Sci. 2003, 85, 1314–1329. [Google Scholar]
- Schwikkard, S.; van Heerden, F.R. Antimalarial activity of plant metabolites. Nat. Prod. Rep. 2003, 19, 675–692. [Google Scholar] [CrossRef]
- Wright, C.W. Plant derived antimalarial agents: new leads and challenges. Phytochem. Rev. 2005, 4, 55–61. [Google Scholar] [CrossRef]
- Eid, S.Y.; El-Readi, M.Z.; Wink, M. Digitonin synergistically enhances the cytotoxicity of plant secondary metabolites in cancer cells. Phytomedicine 2012. [Google Scholar] [CrossRef]
- Eid, S.Y.; El-Readi, M.Z.; Wink, M. Synergism of three-drug combinations of sanguinarine and other plant secondary metabolites with digitonin and doxorubicin in multi-drug resistant cancer cells. Phytomedicine 2012, in press. [Google Scholar]
- Gehrig, S.; Efferth, T. Development of drug resistance in Trypanosoma brucei rhodesiense and Trypanosoma brucei gambiense. Treatment of human African trypanosomiasis with natural products. Int. J. Mol. Med. 2008, 22, 411–419. [Google Scholar]
- Hannaert, V. Sleeping sickness pathogen (Trypanosoma brucei) and natural products: Therapeutic targets and screening systems. Planta Med. 2011, 77, 586–597. [Google Scholar] [CrossRef]
- Hoet, S.; Opperdoes, F.; Brun, R.; Quetin-Leclercq, J. Natural products active against African trypanosomes: a step towards new drugs. Nat. Prod. Rep. 2004, 21, 353–364. [Google Scholar] [CrossRef]
- Hoet, S.; Opperdoes, F.; Brun, R.; Adjakidje, V.; Quetin-Leclercq, J. In vitro antitrypanosomal activity of ethnopharmacologically selected Beninese plants. J. Ethnopharmacol. 2004, 91, 37–42. [Google Scholar] [CrossRef]
- Salem, M.M.; Werbovetz, K.A. Natural products from plants as drug candidates and lead compounds against leishmaniasis and trypanosomiasis. Curr. Med. Chem. 2006, 13, 2571–2598. [Google Scholar] [CrossRef]
- Merschjohann, K.; Sporer, F.; Steverding, D.; Wink, M. In vitro effect of alkaloids on bloodstream forms of Trypanosoma brucei and T. congolense. Planta Med. 2001, 67, 623–627. [Google Scholar] [CrossRef]
- Nibret, E.; Ashour, M.; Rubanza, C.D.; Wink, M. Antitrypanosomal and cytotoxic activities of some Tanzanian medicinal plants. Phytother. Res. 2010, 24, 45–47. [Google Scholar]
- Nibret, E.; Sporer, F.; Asres, K.; Wink, M. Antitrypanosomal and cytotoxic activities of pyrrolizidine alkaloid producing plants of Ethiopia. J. Pharm. Pharmacol. 2009, 61, 801–808. [Google Scholar]
- Nibret, E.; Wink, M. Chemical composition of essential oils from Hagenia abyssinica, Leonotis ocymifolia and Moringa stenopetala and structure activity relationships of isolated terpenoids in trypanosomes (Trypanosoma b. brucei) and human leucaemia cells (HL-60). Phytomedicine 2010, 17, 911–920. [Google Scholar] [CrossRef]
- Nibret, E.; Wink, M. In vitro antitrypanosomal and cytotoxic activities of four Artemisia species (Asteraceae) from Ethiopia. Phytomedicine 2010, 17, 369–374. [Google Scholar] [CrossRef]
- Herrmann, F.; Hamoud, R.; Sporer, F.; Tahrani, A.; Wink, M. Carlina oxide- a natural polyacetylene from Carlina acaulis (Asteraceae) with potent antitrypanosomal and antimicrobial properties. Planta Med. 2011, 77, 1905–1911. [Google Scholar] [CrossRef]
- Herrmann, F.; Tahrani, A.; Wink, M. Antitrypanosomal properties of Panax ginseng CA Meyer—New possibilities for a remarkable traditional drug! Phytother. Res. 2012. [Google Scholar] [CrossRef]
- Bodley, A.L.; Shapiro, T.A. Molecular and cytotoxic effects of camptothecin, a topoisomerase I inhibitor, on trypanosomes and Leishmani. Proc. Natl. Acad. Sci. USA 1995, 92, 3726–3730. [Google Scholar] [CrossRef]
- Ochola, D.O.; Prichard, R.K.; Lubega, C.W. Classical ligands bind tubulin of trypanosomes and inhibit their growth in vitro. J. Parasitol. 2002, 88, 600–604. [Google Scholar]
- Tempone, A.G.; de Oliveira, C.M.; Berlinck, R.G.S. Current approaches to discover marine antileishmanial natural products. Planta Med. 2011, 77, 572–585. [Google Scholar] [CrossRef]
- Polonio, T.; Efferth, T. Leishmaniasis: Drug resistance and natural products (review). Int. J. Mol. Med. 2008, 22, 277–286. [Google Scholar]
- Dube, A.; Gupta, R.; Singh, N. Reporter genes facilitating discovery of drugs targeting protozoan parasites. Trends Parasitol. 2009, 25, 432–439. [Google Scholar] [CrossRef]
- Gehrig, S.; Efferth, T. Development of drug resistance in Trichomonas vaginalis and its overcoming with natural products. Open Bioact. Comp. J. 2009, 2, 21–28. [Google Scholar] [CrossRef]
- Müllner, A.; Helfer, A.; Kotlyar, B.; Oswald, J.; Efferth, T. Chemistry and pharmacology of neglected helminthic diseases. Curr. Med. Chem. 2011, 18, 767–789. [Google Scholar] [CrossRef]
- Murthy, P.K.; Joseph, S.K.; Murthy, P.S.R. Plant products in the treatment and control of filariasis and other helminth infections and assay systems for antifilarial/anthelminthic activity. Planta Med. 2011, 77, 647–661. [Google Scholar] [CrossRef]
- Comley, J.C. New antifilaricicidal leads from plant? Trop. Med. Parasitol. 1990, 41, 1–9. [Google Scholar]
- Singh, N.P.; Singh, V.K. Streblus asper—An ancient Indian drug for the cure of filariasis. Acta Bot. Indica 1987, 15, 108–109. [Google Scholar]
- Chatterjee, R.K.; Fatma, N.; Murthy, P.K.; Sinha, P.; Kulreshtha, D.K.; Dhawan, B.N. Macrofilaricidal activity of the stem bark of Streblus asper and its major active constituents. Drug Dev. Res. 1992, 26, 67–78. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Nwude, N.; Ogunsui, R.A.; Aliu, Y.O. Screening of West African plants for anthelmintic activity. ILCA Bull. 1984, 17, 19–23. [Google Scholar]
- Liu, L.; Sporer, F.; Wink, M.; Jourdane, J.; Hennig, R.; Li, Y.L.; Ruppel, A. Anthraquinones in Rheum palmatum and Rumex dentatus (Polygonaceae) and phorbolesters from Jatropha curcas (Euphorbiaceae) with molluscicidal activity against schistosomias vector snails Oncomelania, Biomphalaria and Bulinus. Trop. Med. Intl. Health 1997, 2, 179–188. [Google Scholar]
- Kliks, M.M. Studies on the traditional herb anthelmintic Chenopodium ambrosioides L. Ethnopharmacological evaluation and clinical field trials. Soc. Sci. Med. 1985, 21, 16–21. [Google Scholar]
- Mueller, M.S.; Runyambo, S.; Wagner, I. Randomized controlled trial of a traditional preparation of Artemisia annua L. (Annual Wormwood) in the treatment of malaria. Trans. R. Soc. Trop. Med. Hyg. 2004, 98, 318–321. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wink, M. Medicinal Plants: A Source of Anti-Parasitic Secondary Metabolites. Molecules 2012, 17, 12771-12791. https://doi.org/10.3390/molecules171112771
Wink M. Medicinal Plants: A Source of Anti-Parasitic Secondary Metabolites. Molecules. 2012; 17(11):12771-12791. https://doi.org/10.3390/molecules171112771
Chicago/Turabian StyleWink, Michael. 2012. "Medicinal Plants: A Source of Anti-Parasitic Secondary Metabolites" Molecules 17, no. 11: 12771-12791. https://doi.org/10.3390/molecules171112771
APA StyleWink, M. (2012). Medicinal Plants: A Source of Anti-Parasitic Secondary Metabolites. Molecules, 17(11), 12771-12791. https://doi.org/10.3390/molecules171112771





