Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications
Abstract
:1. Introduction
2. Cyclodextrin-Porphyrin Assemblies
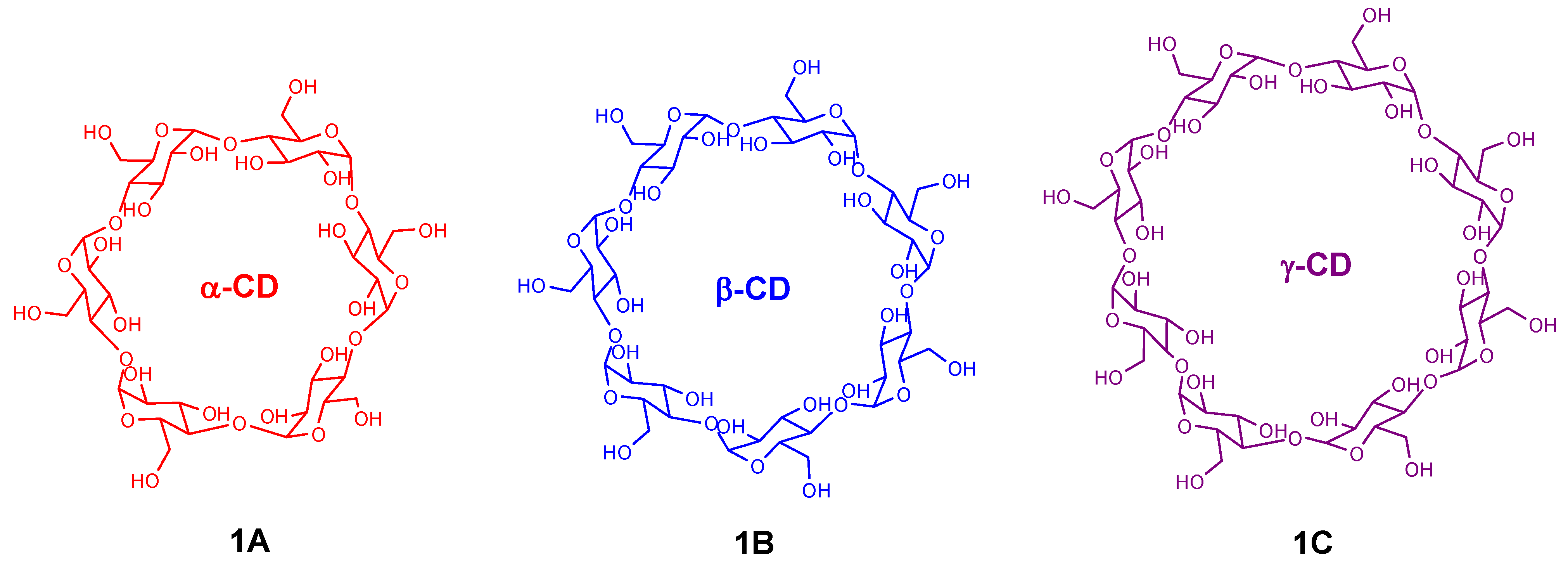
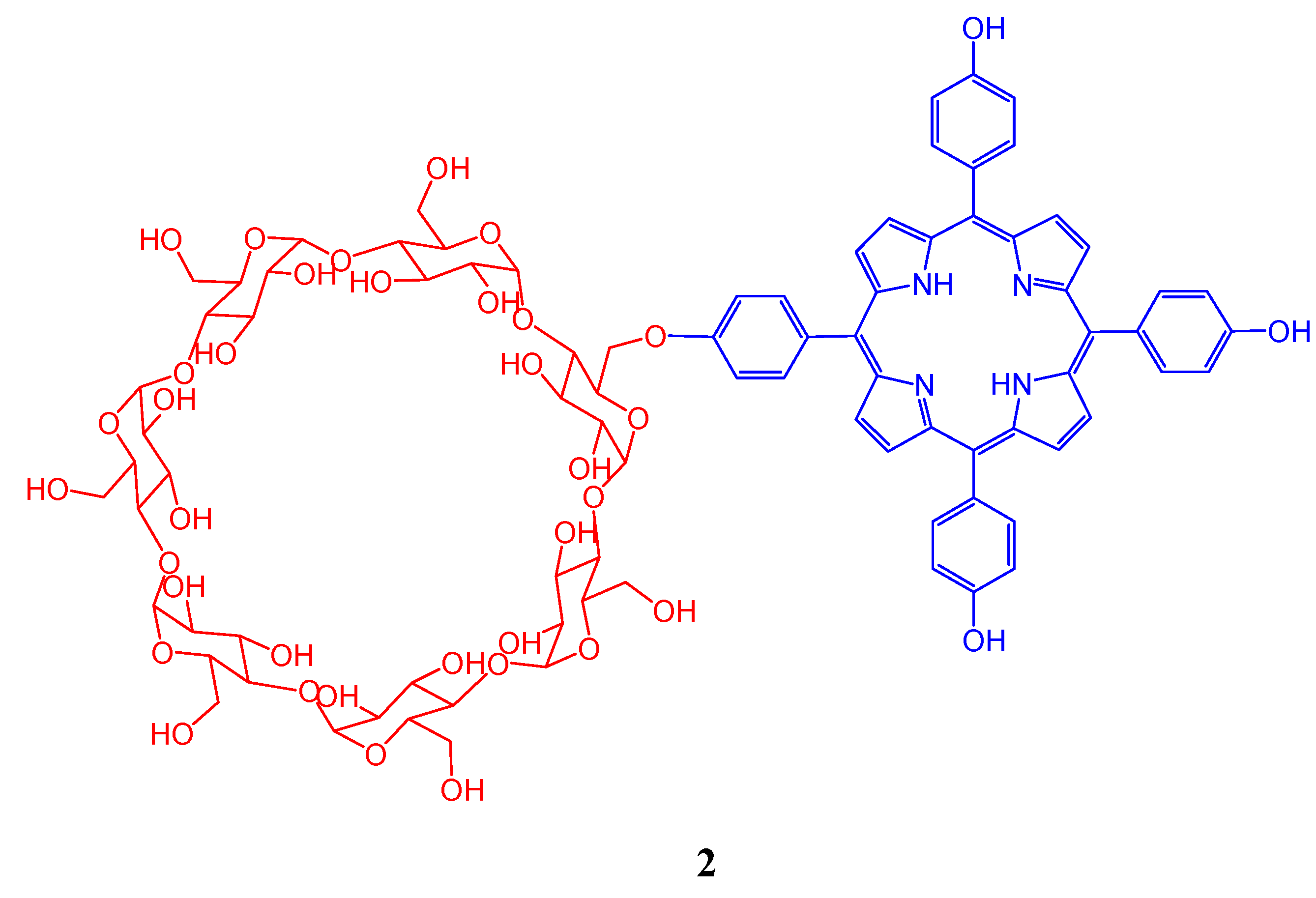
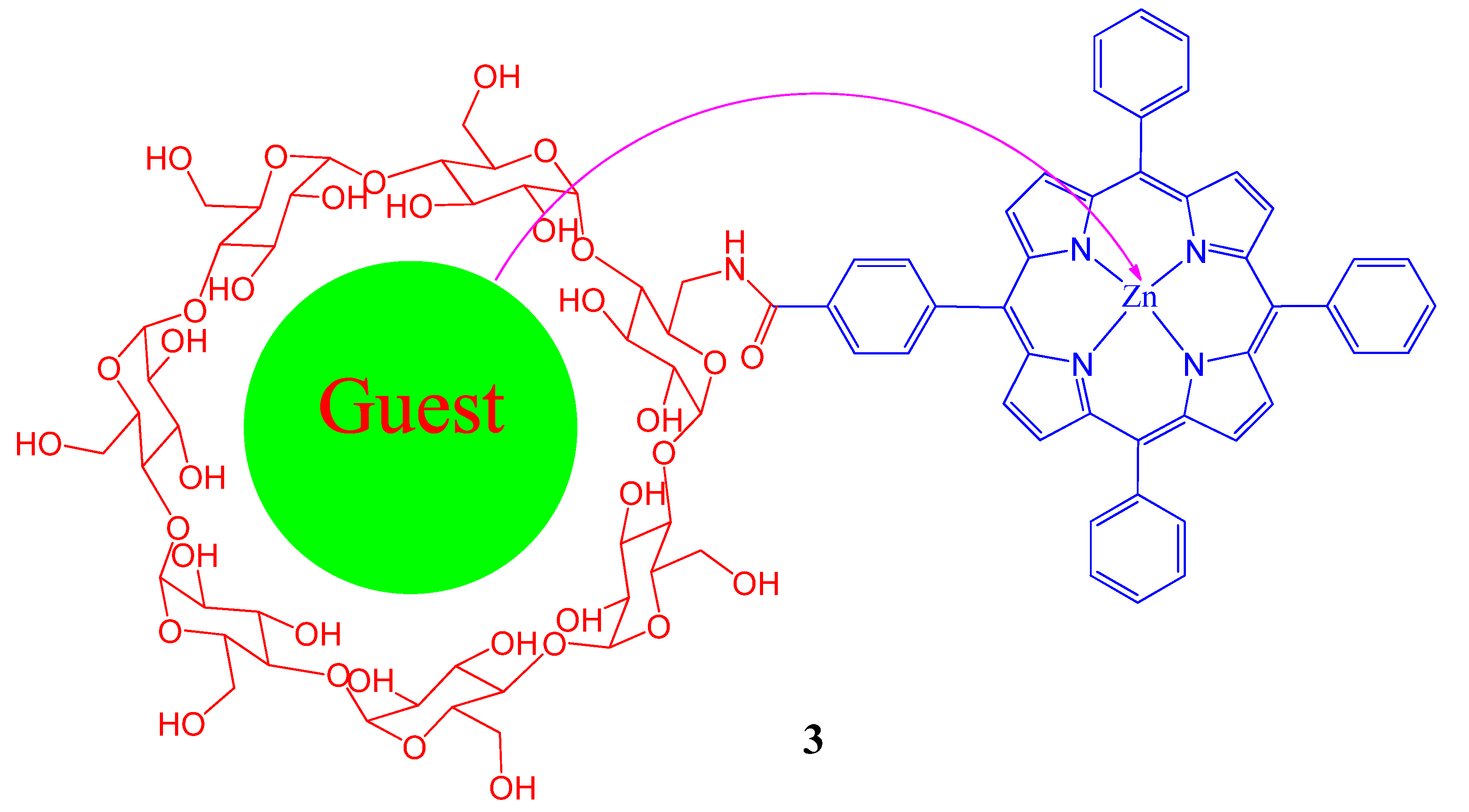
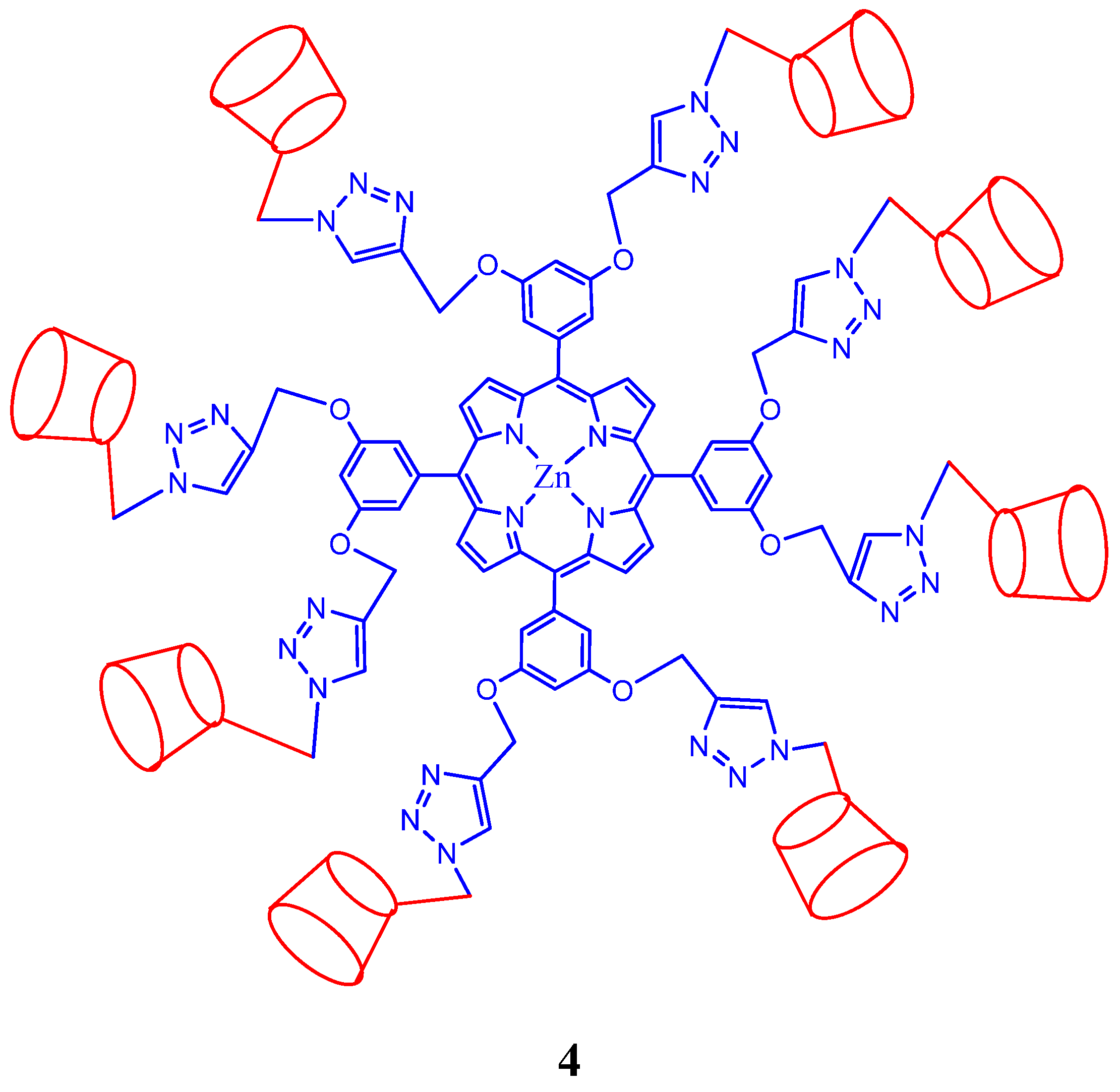
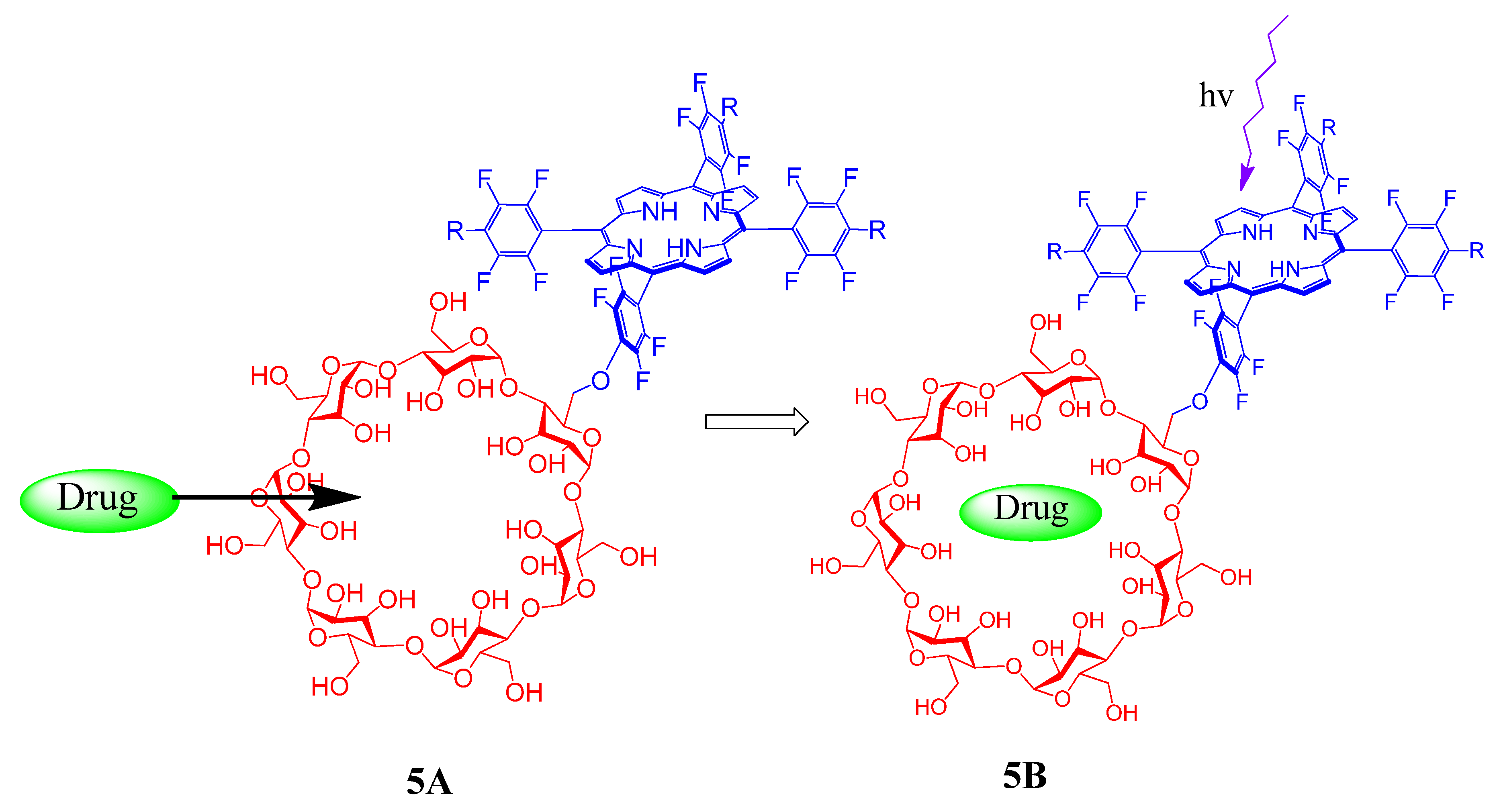
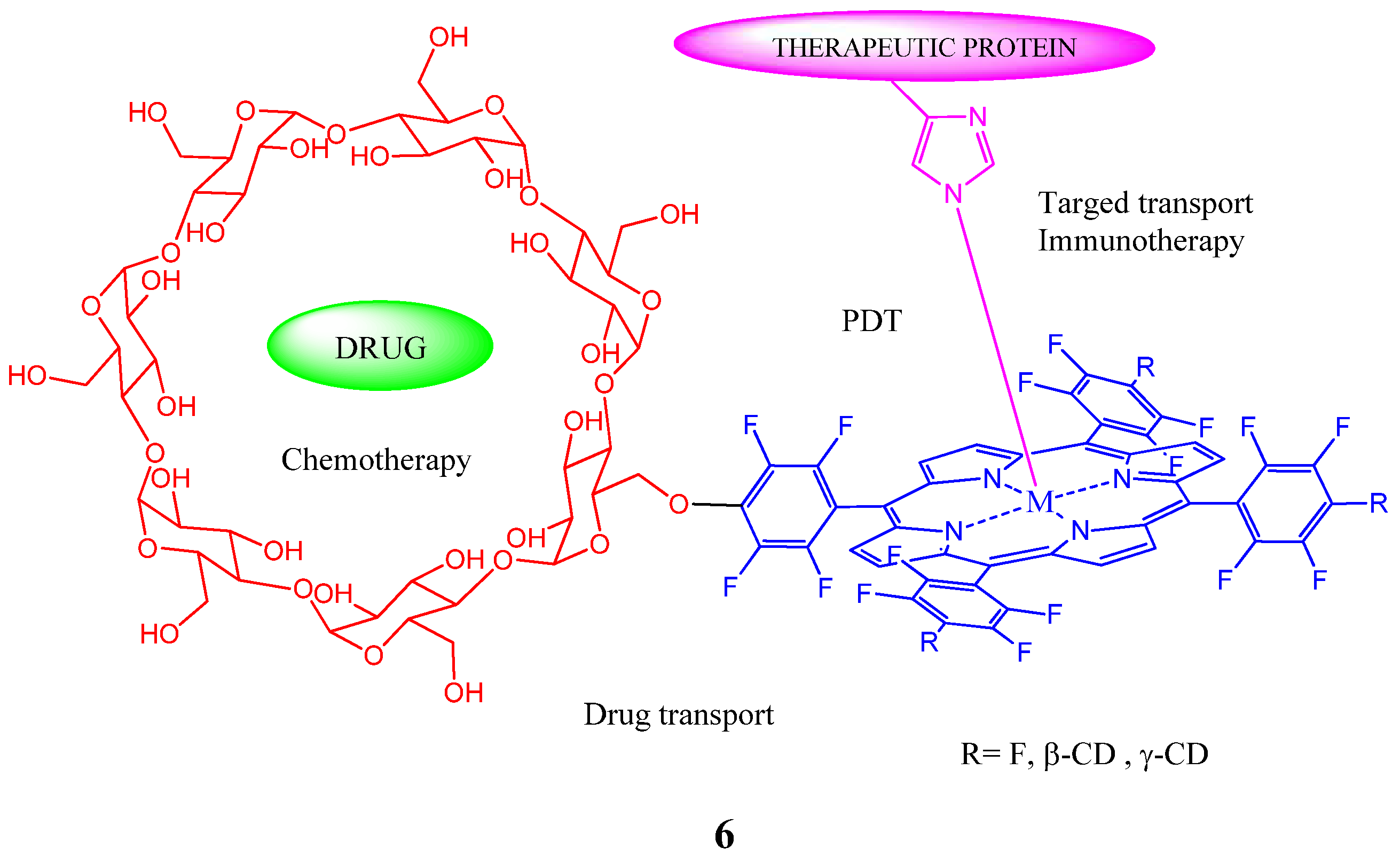
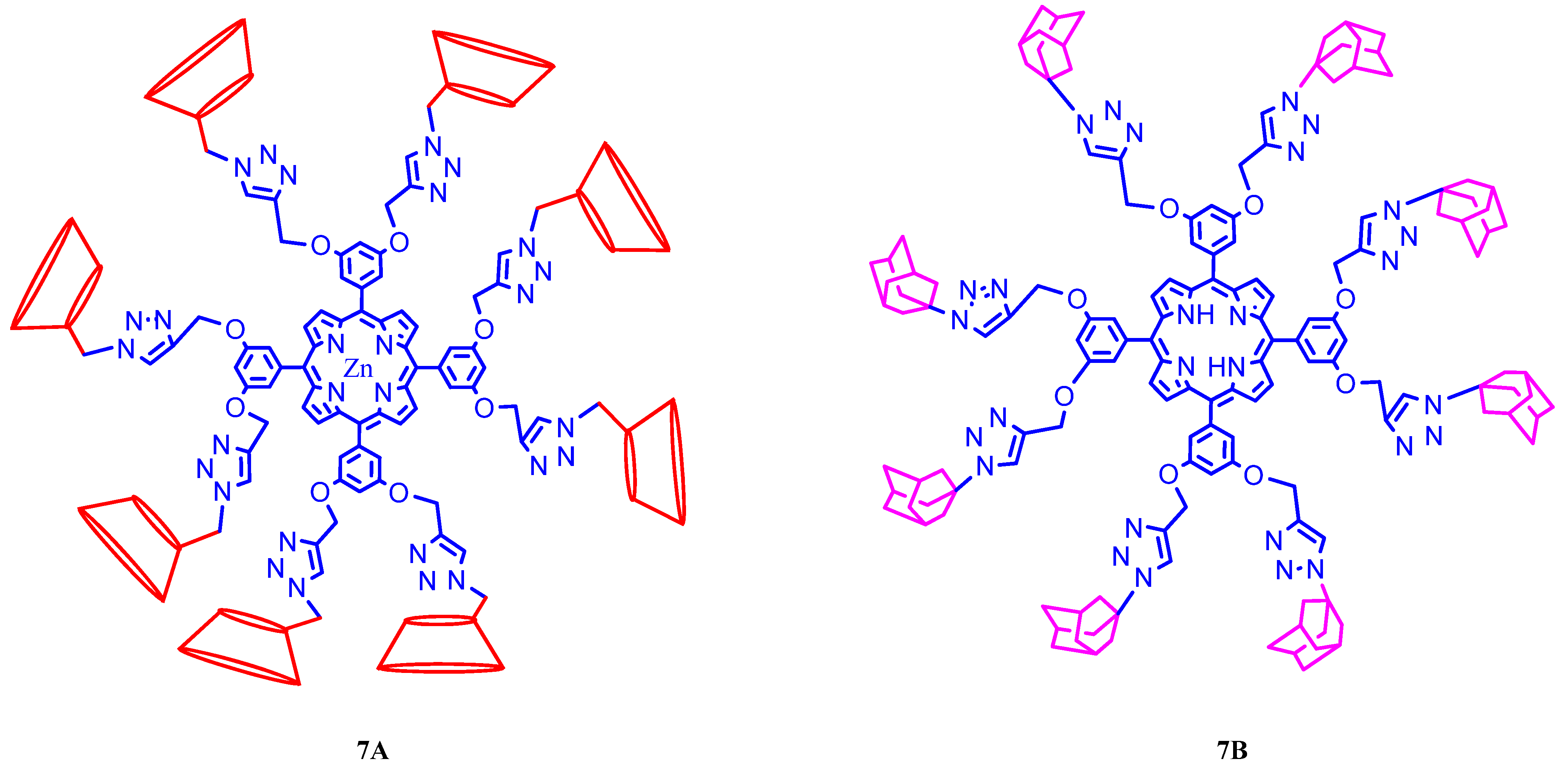
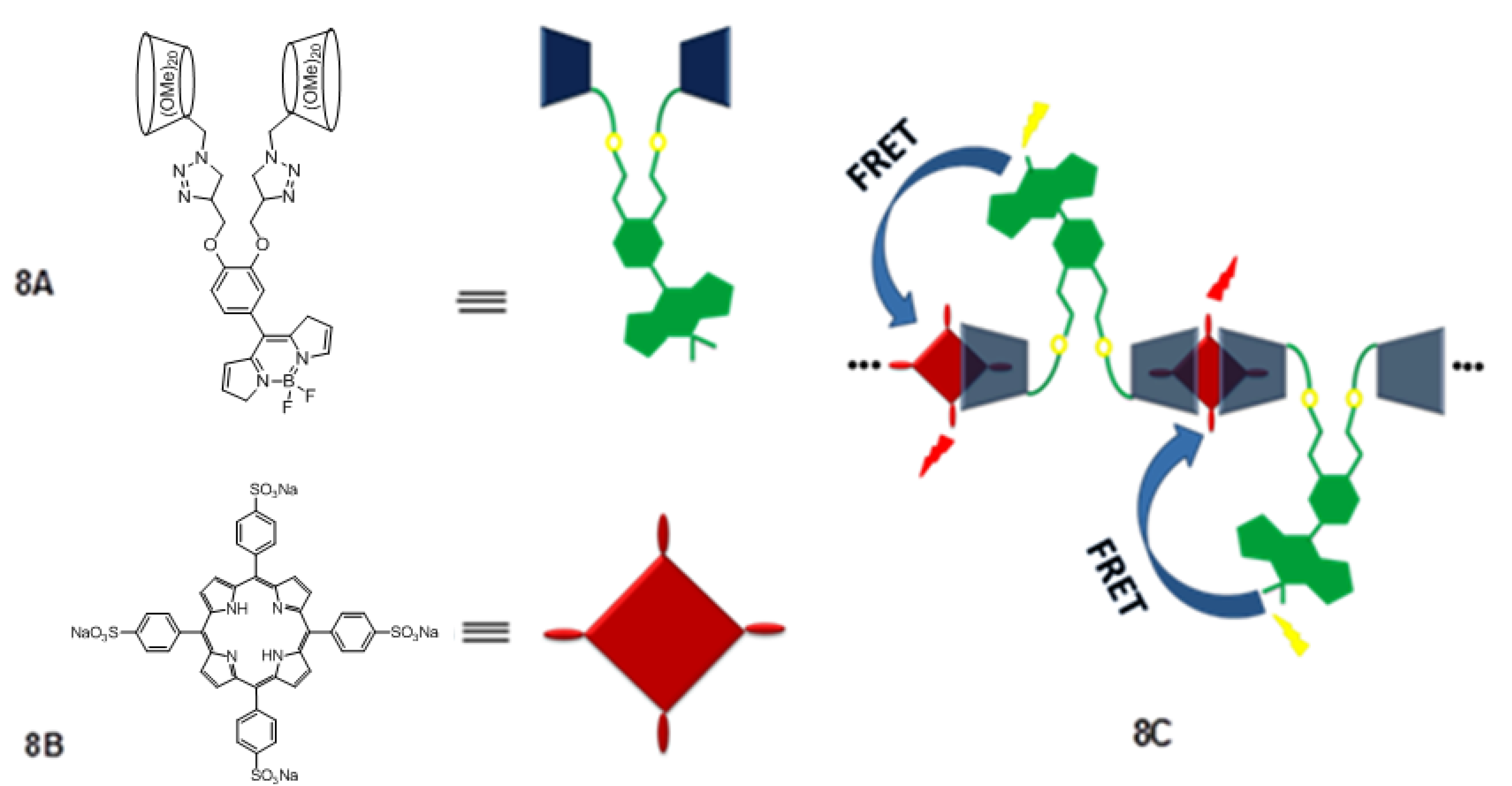
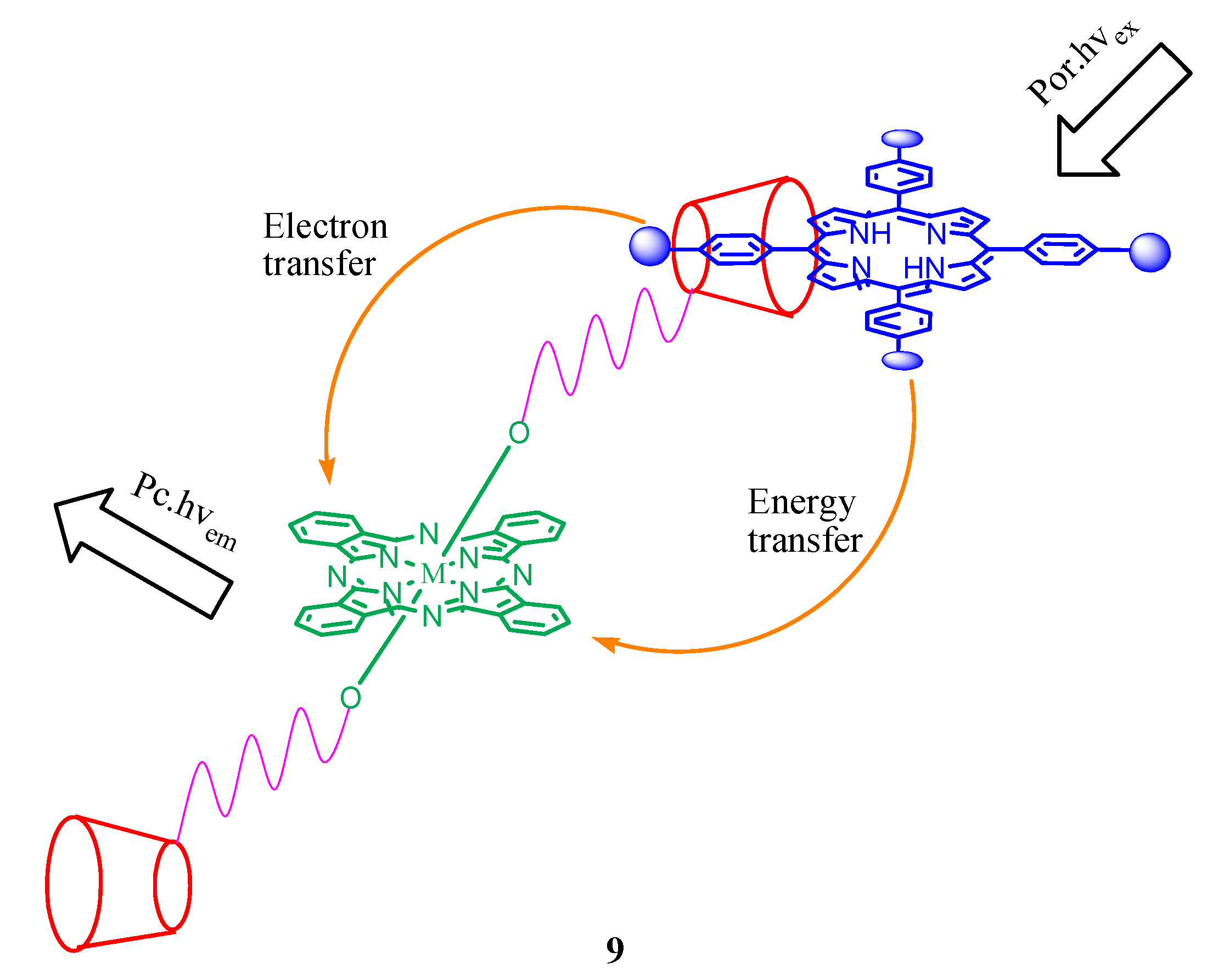
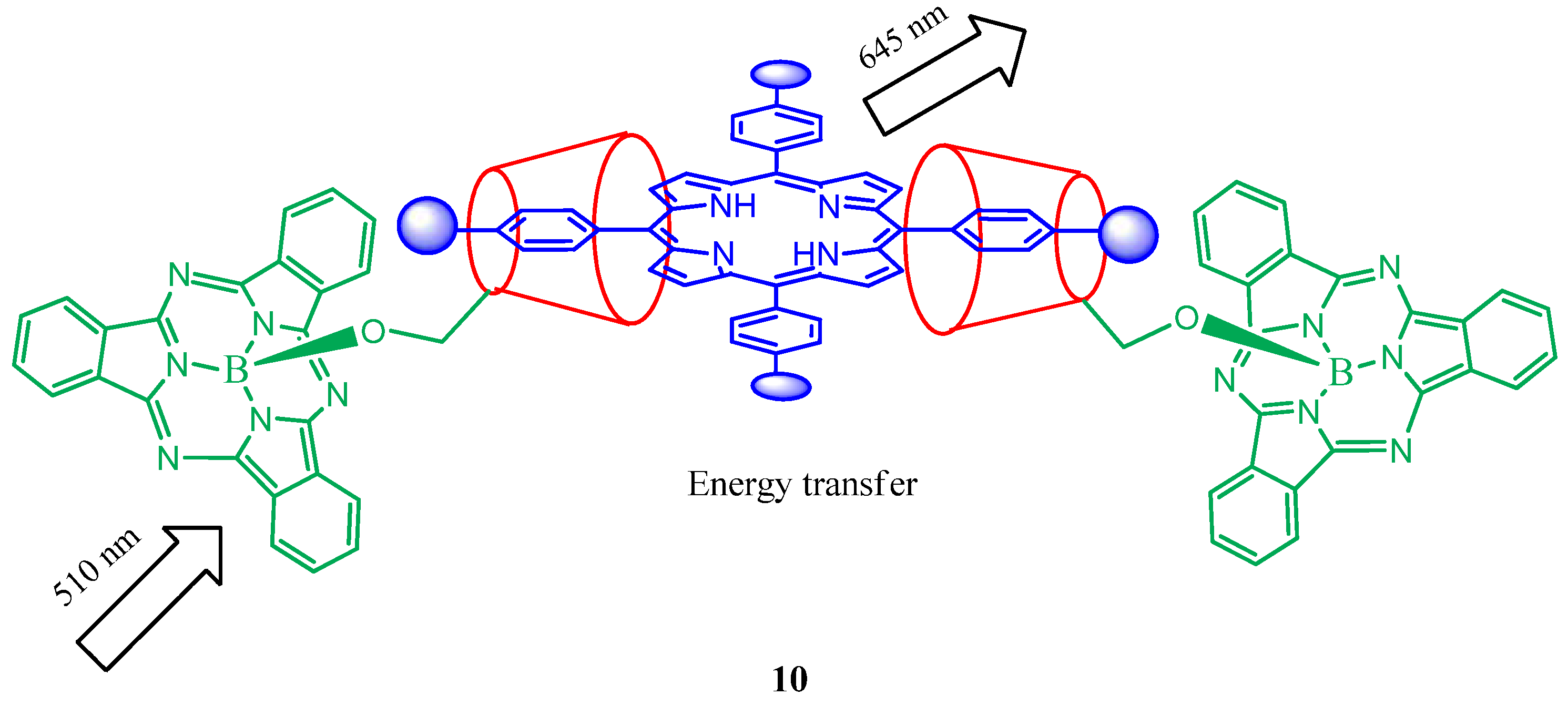
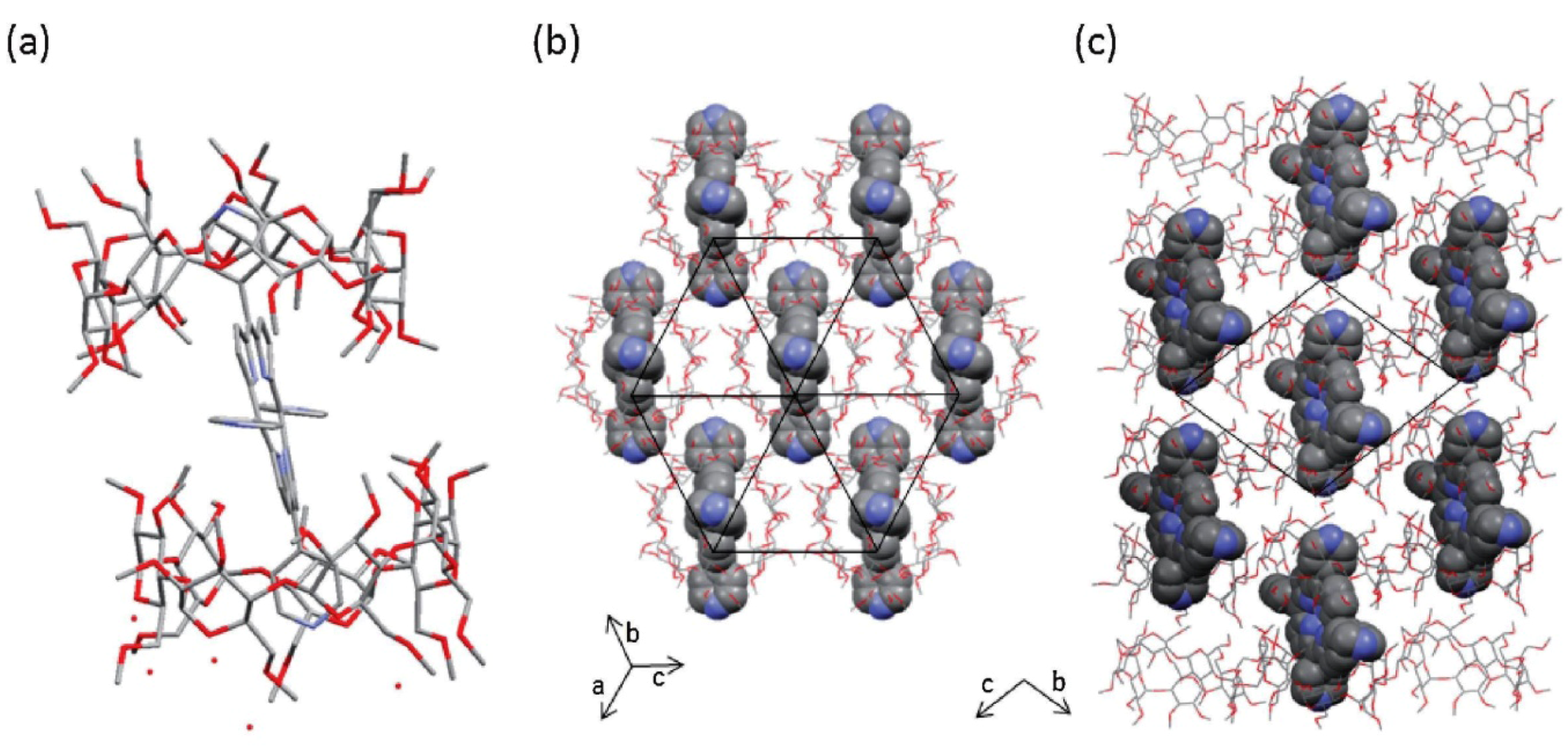
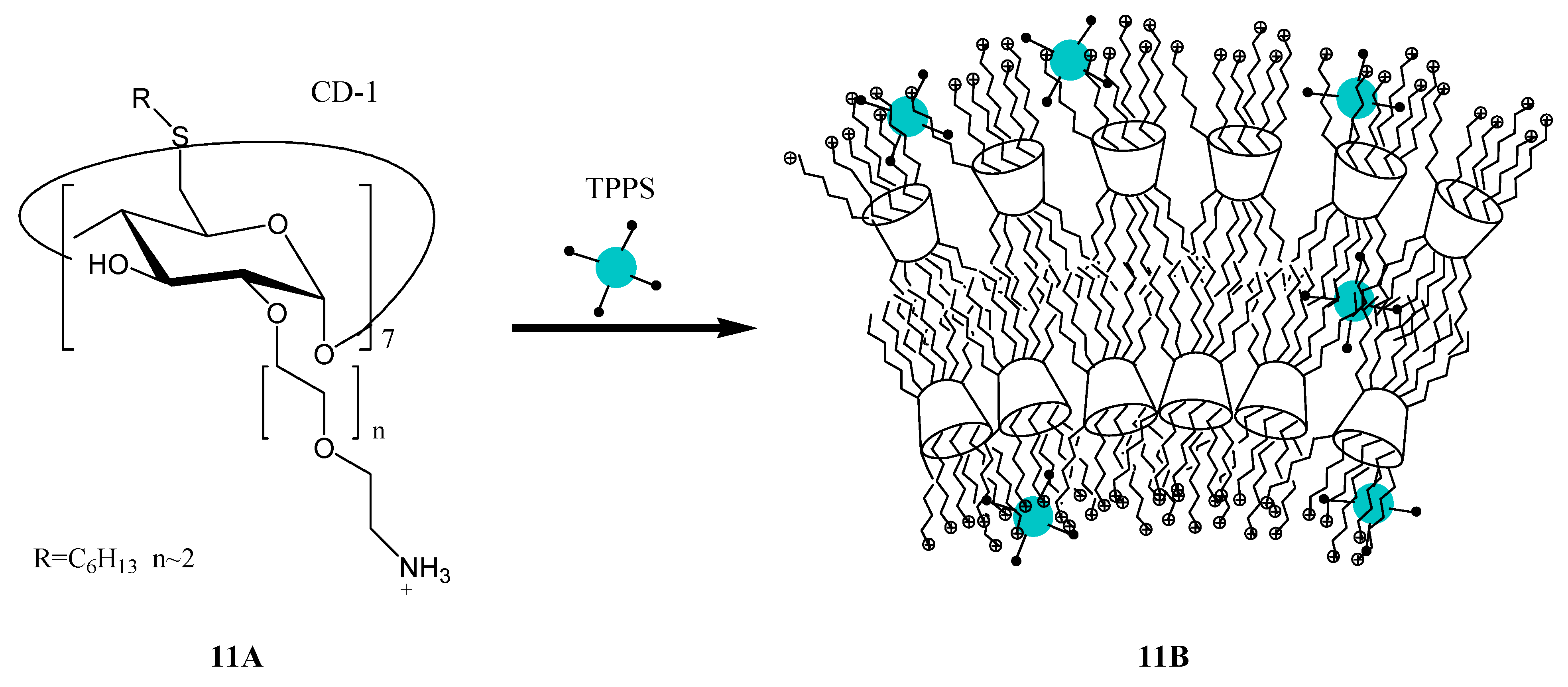

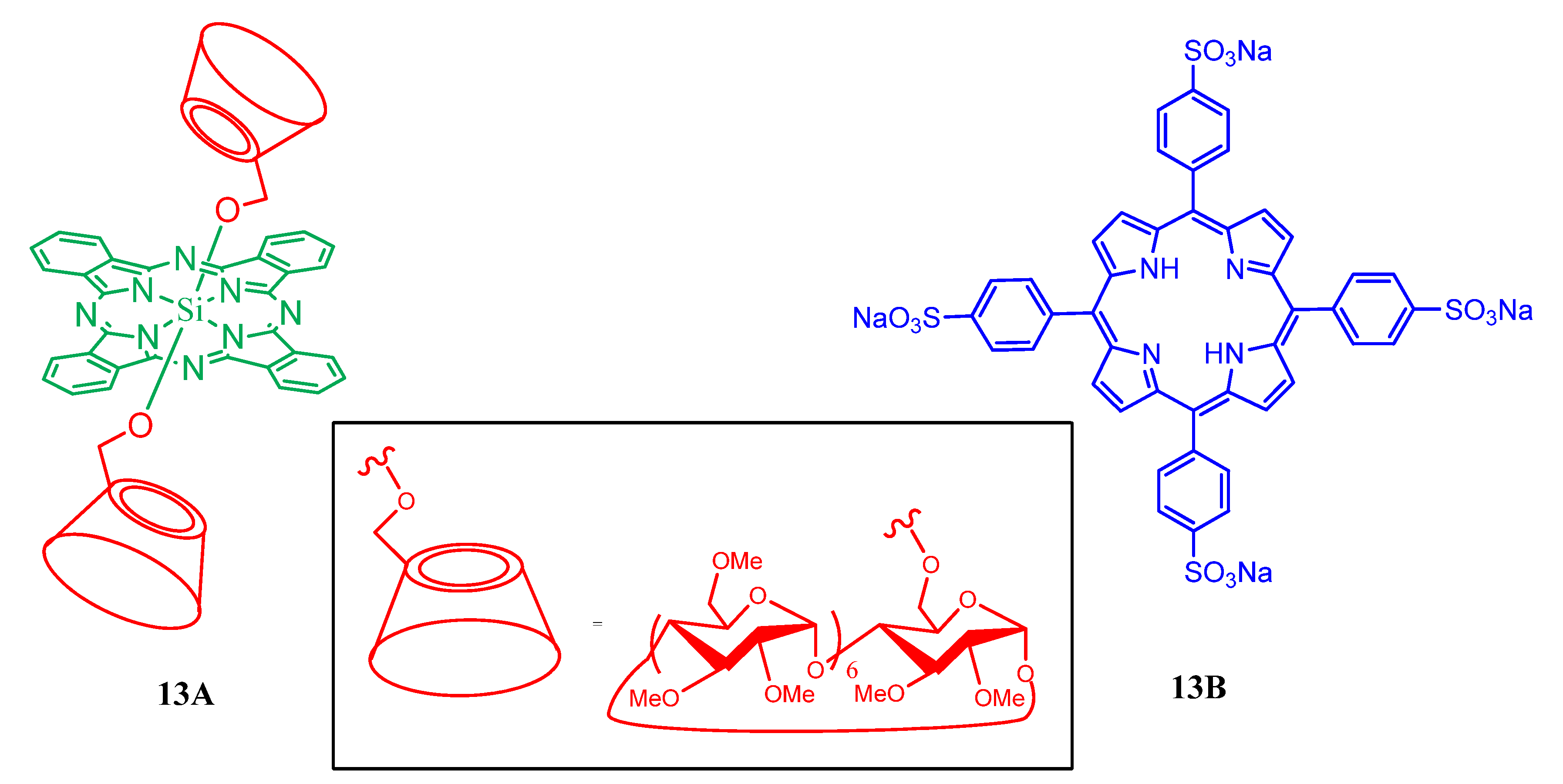
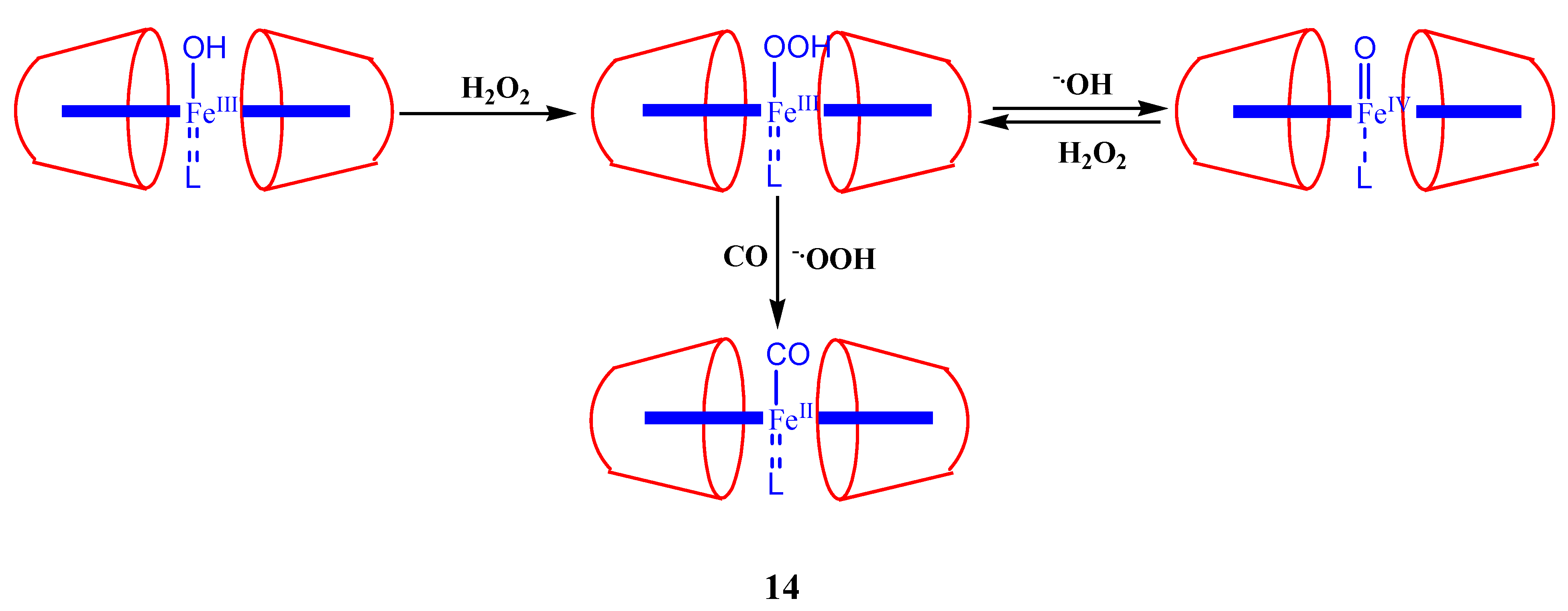
3. Calixarene-Porphyrin Assemblies
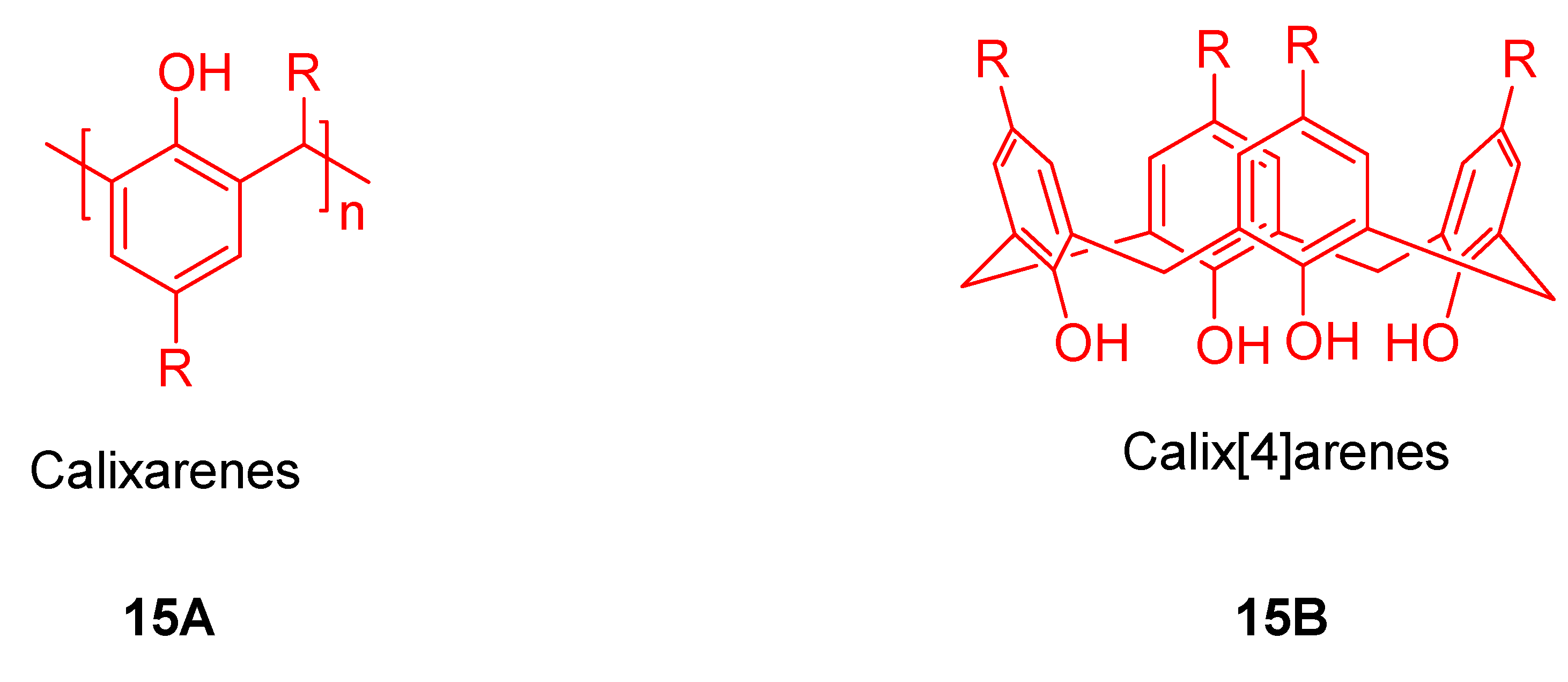
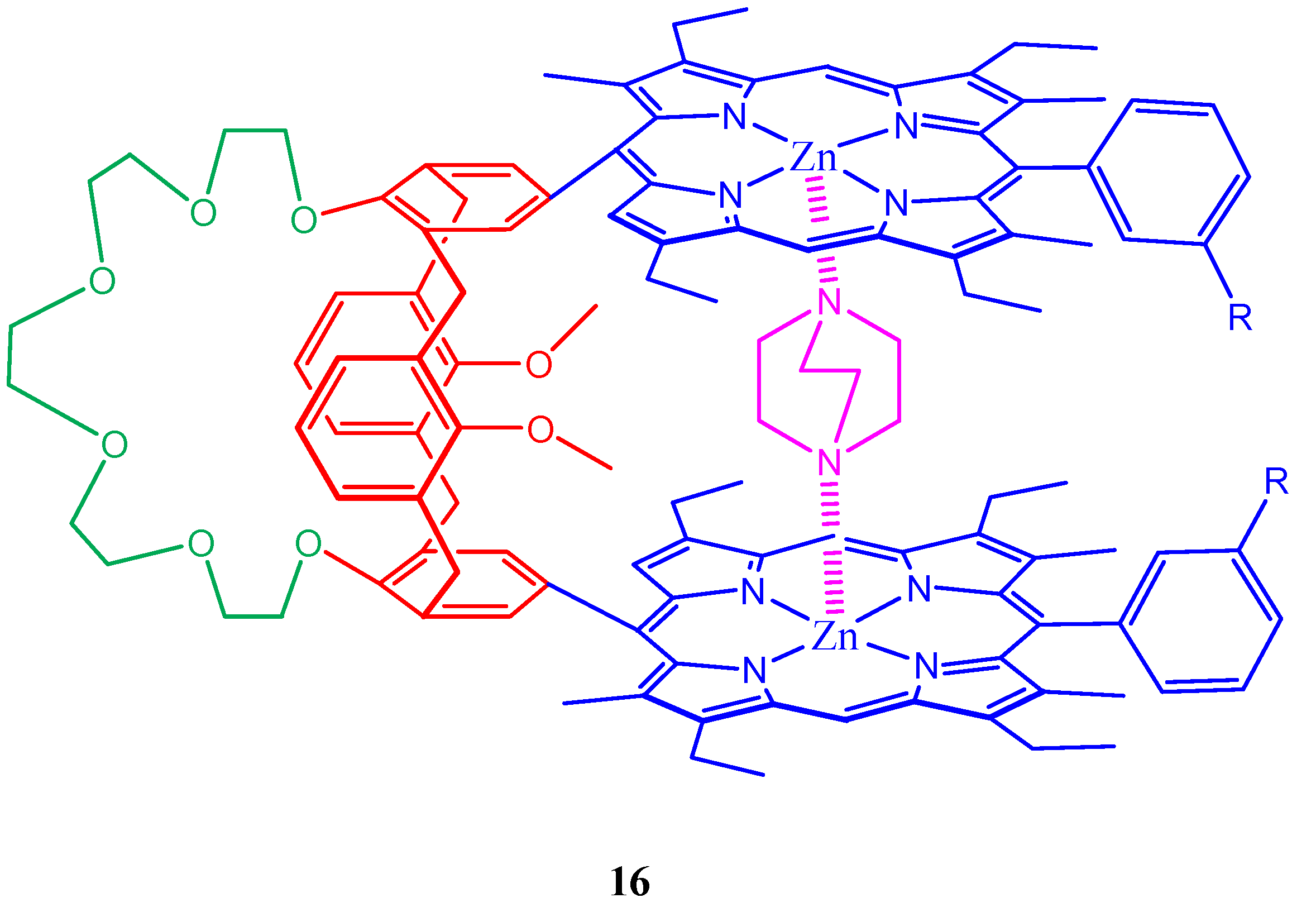
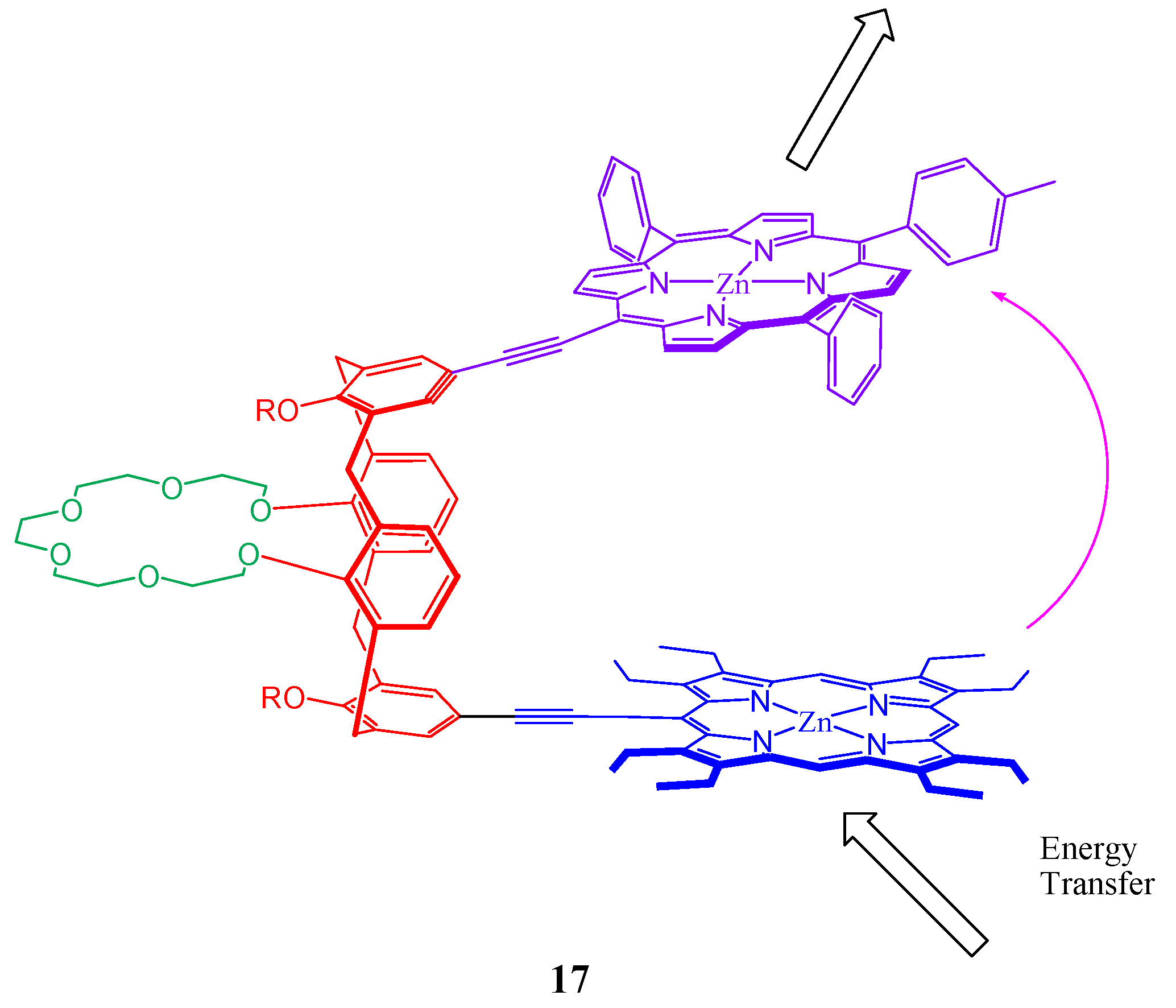
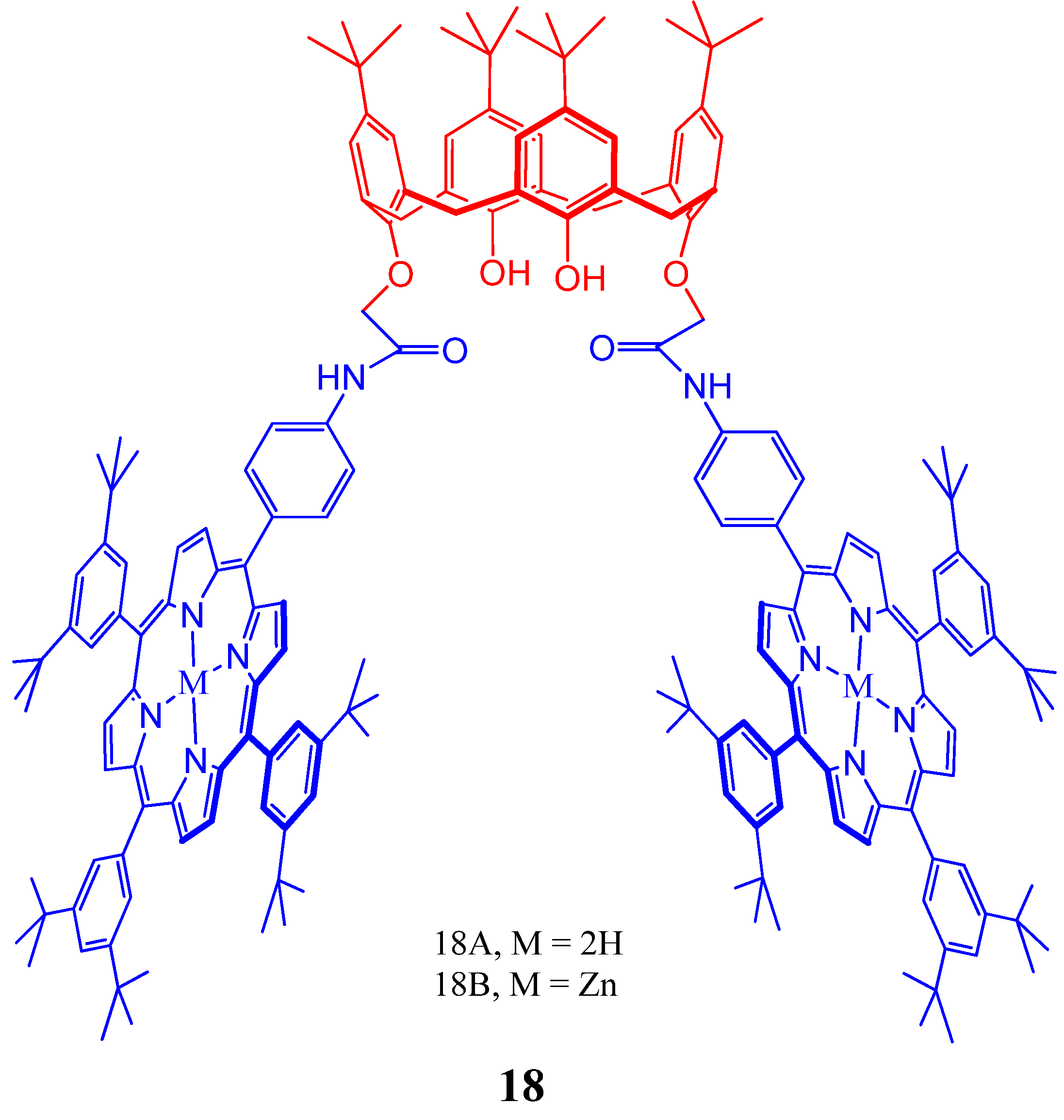
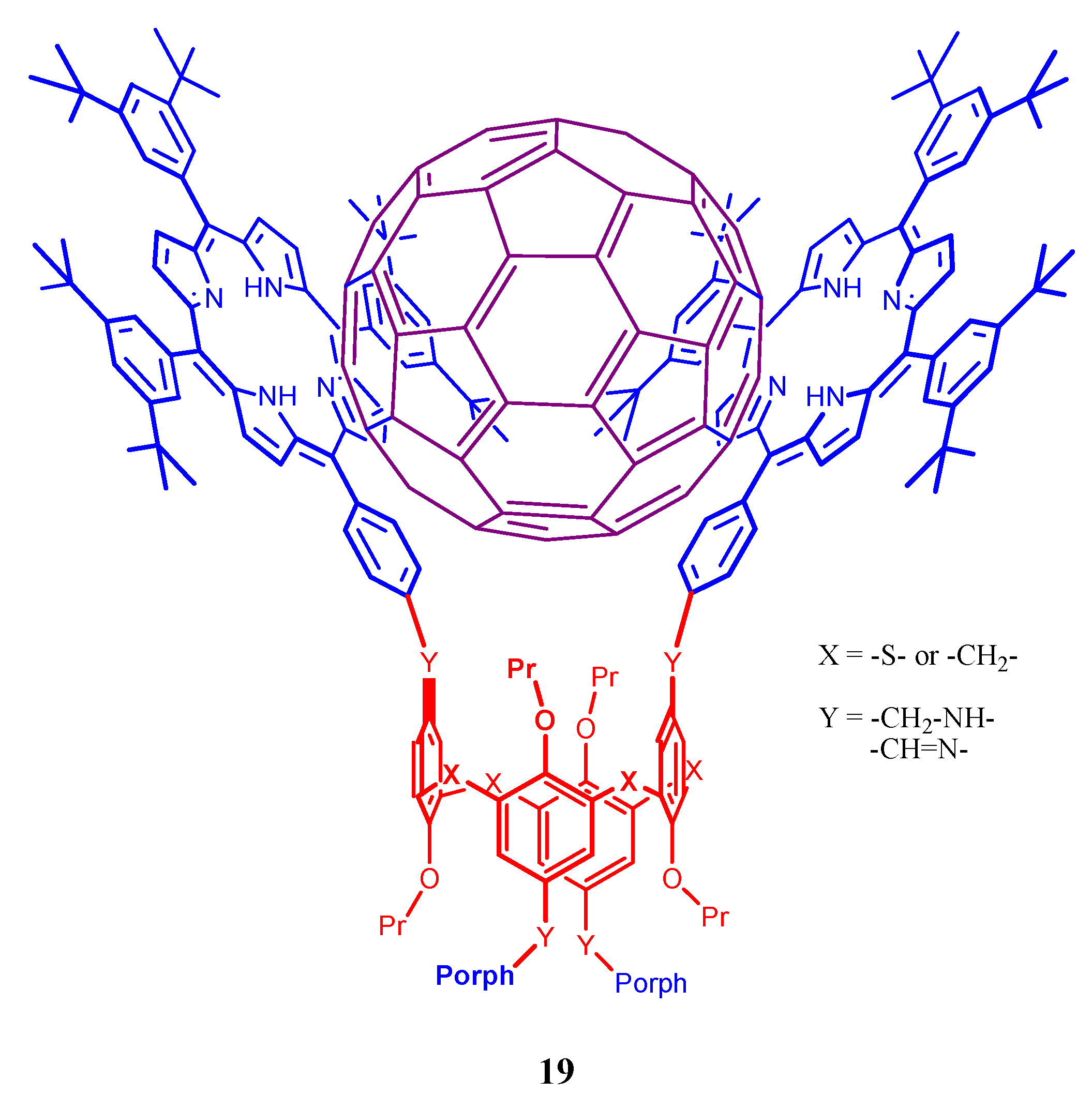
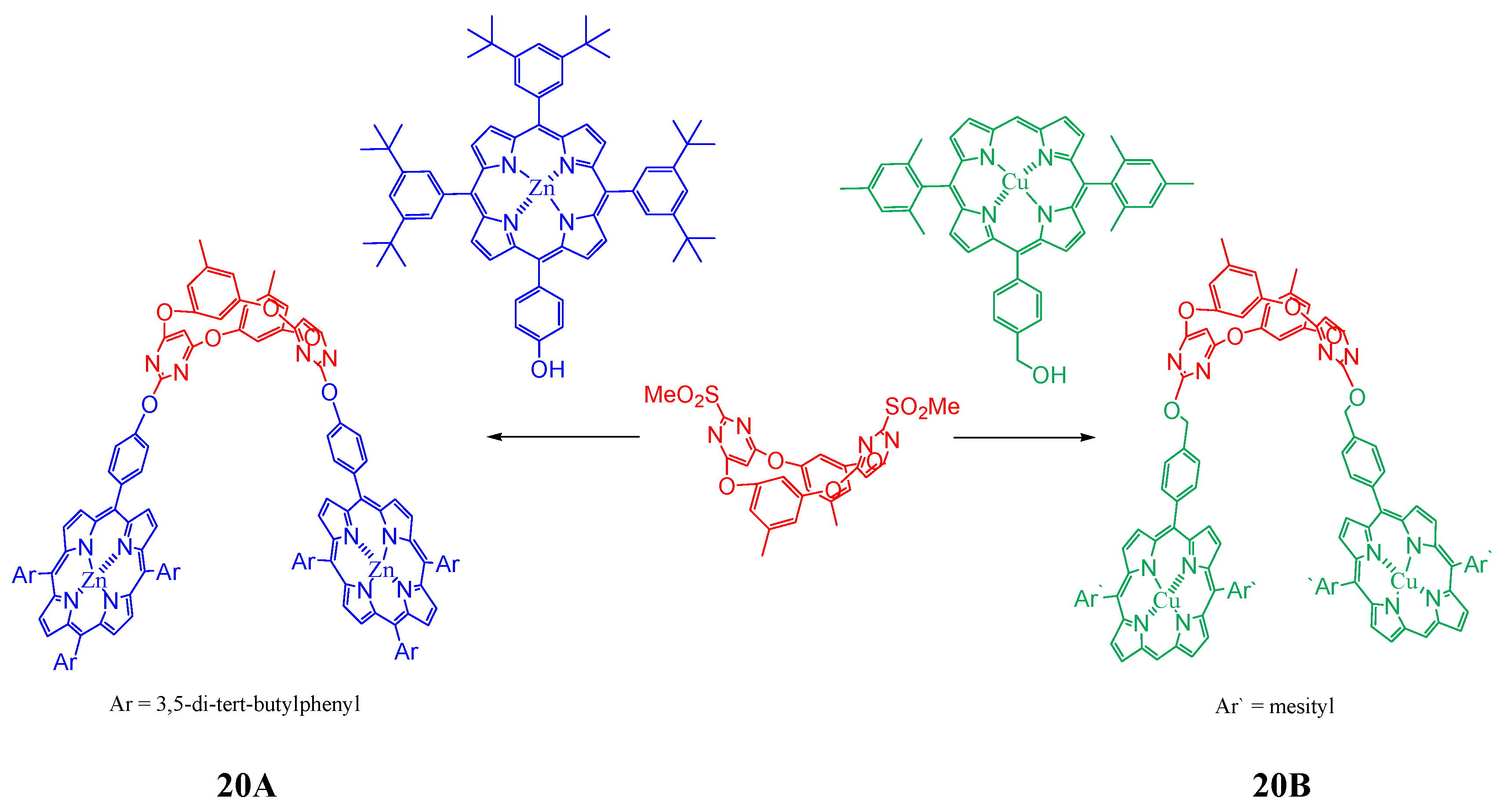
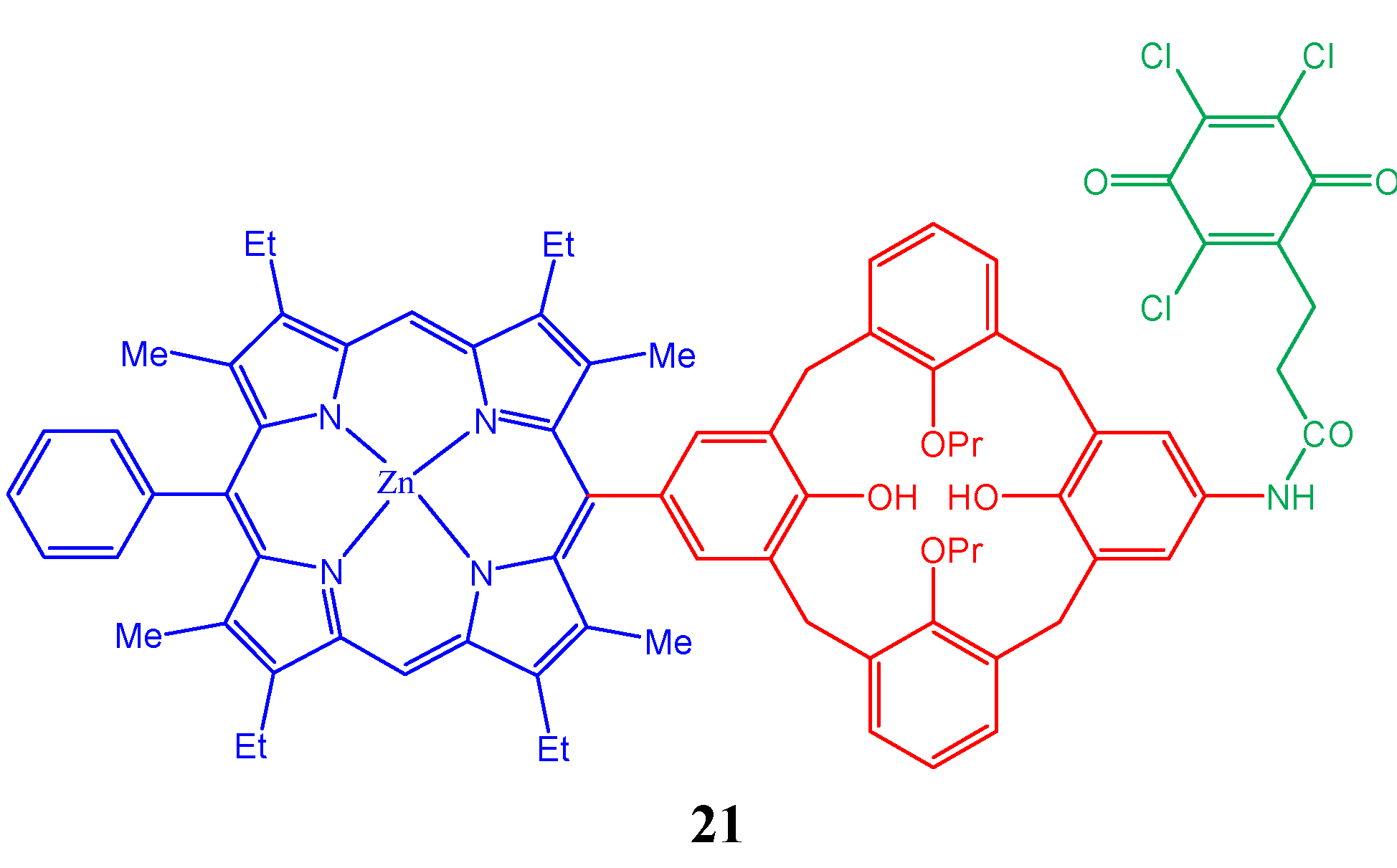
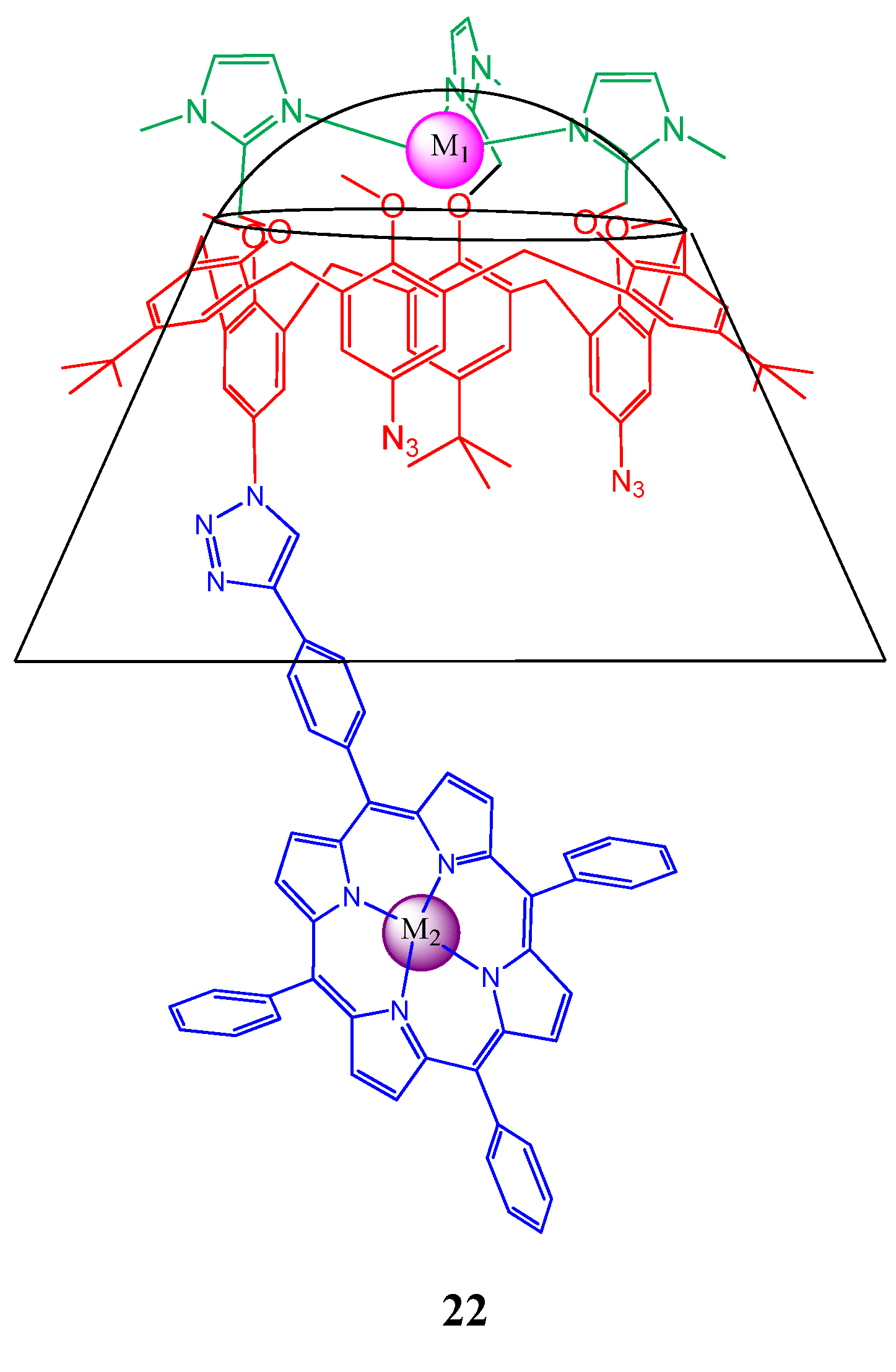
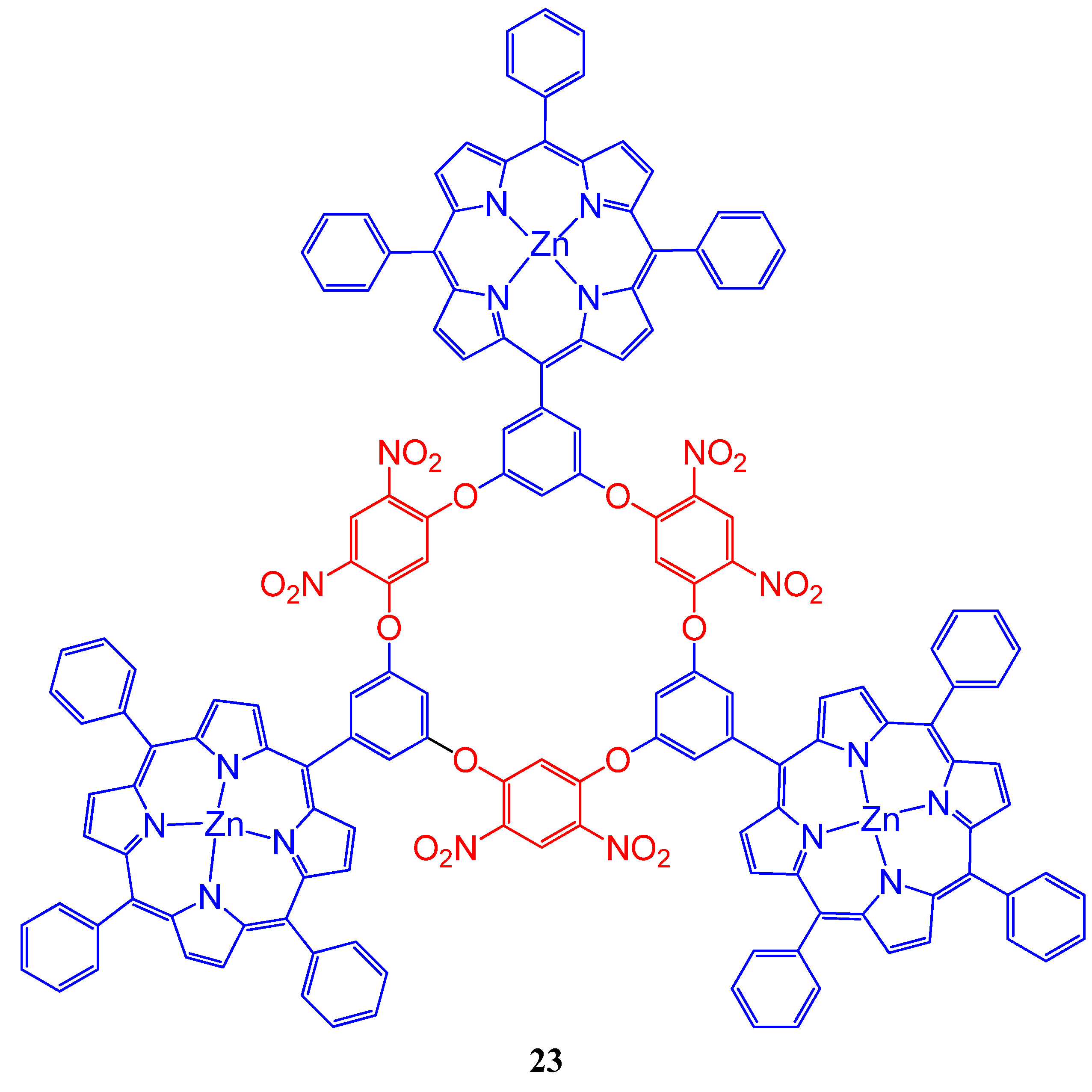
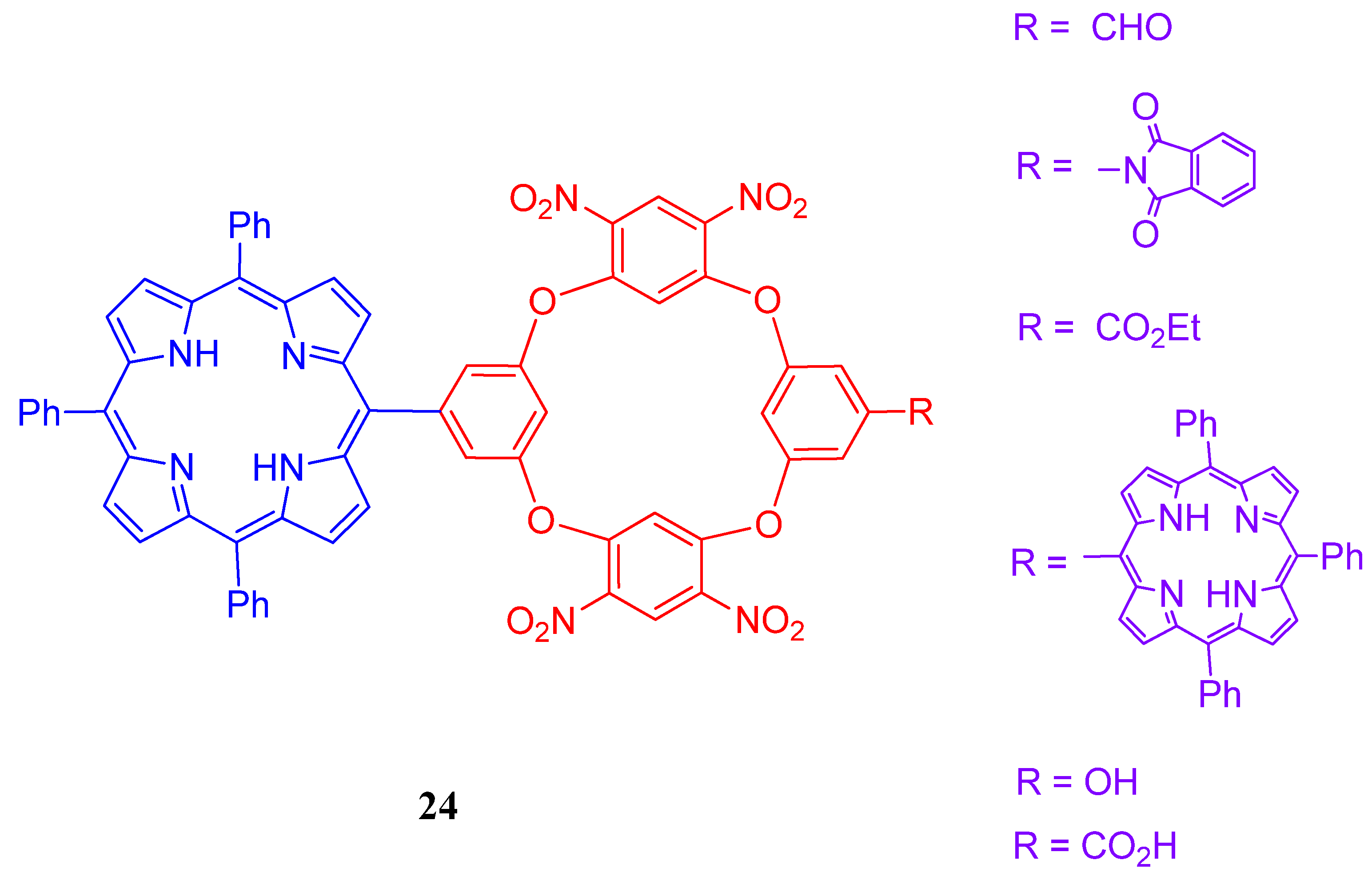

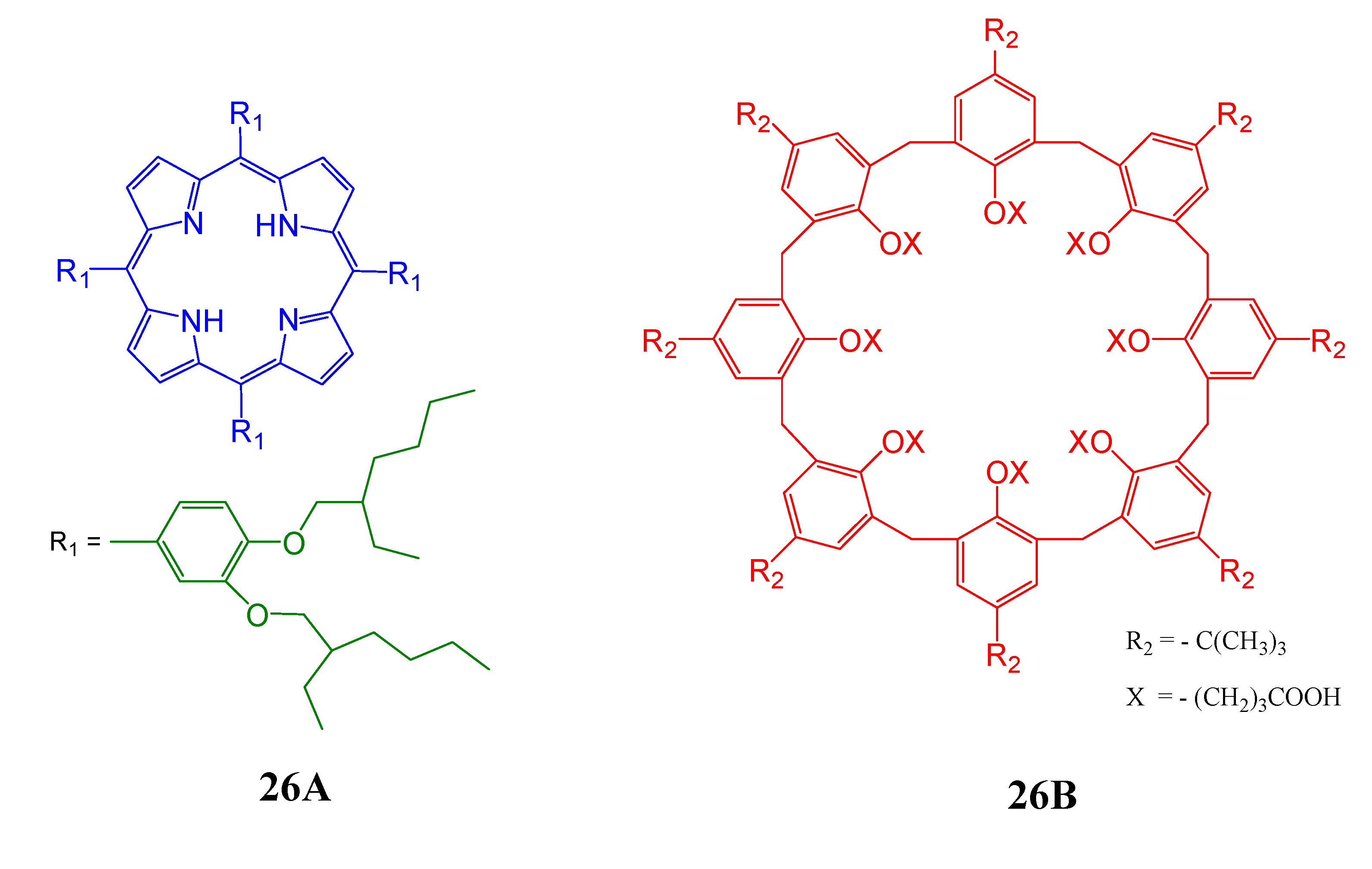
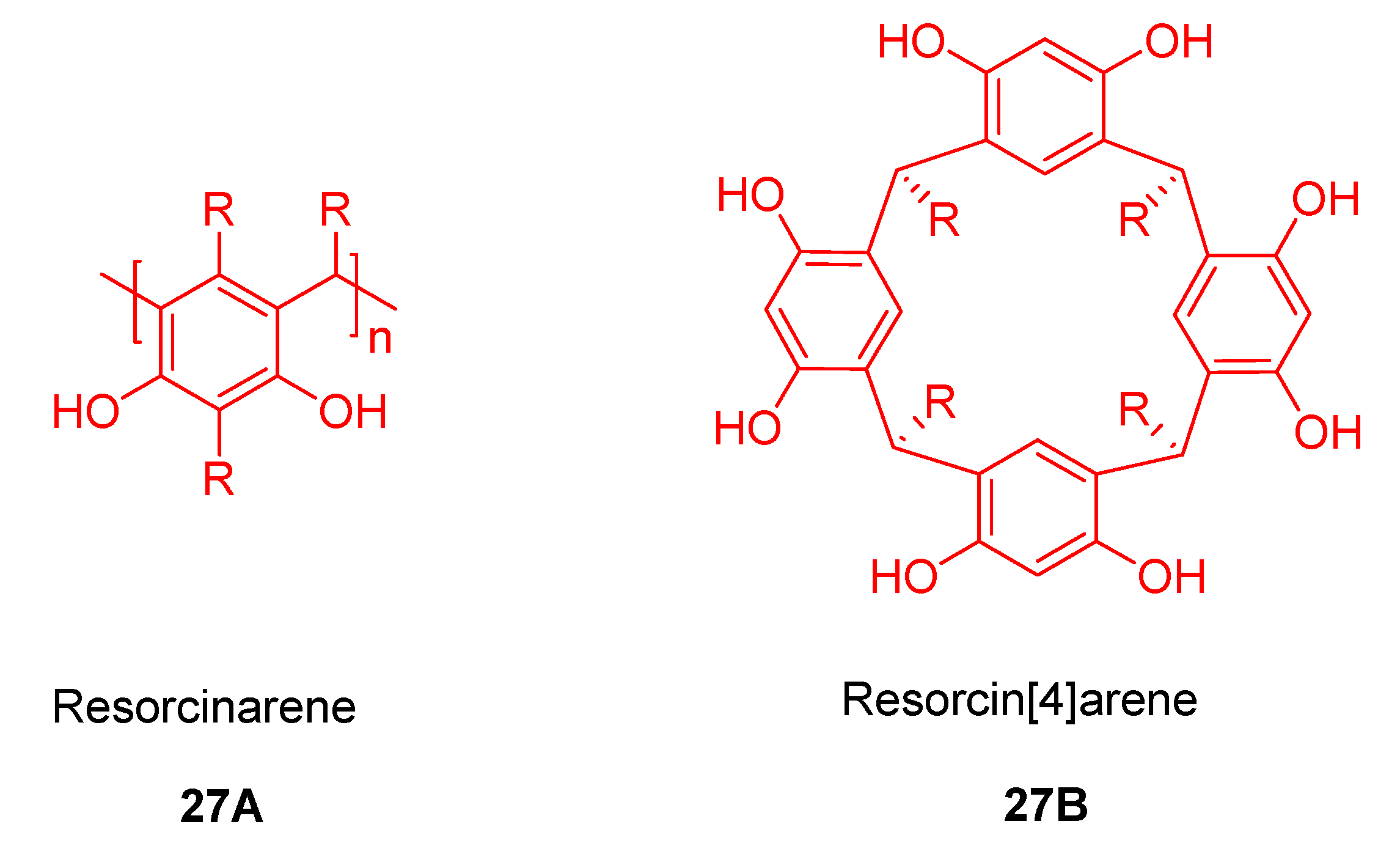
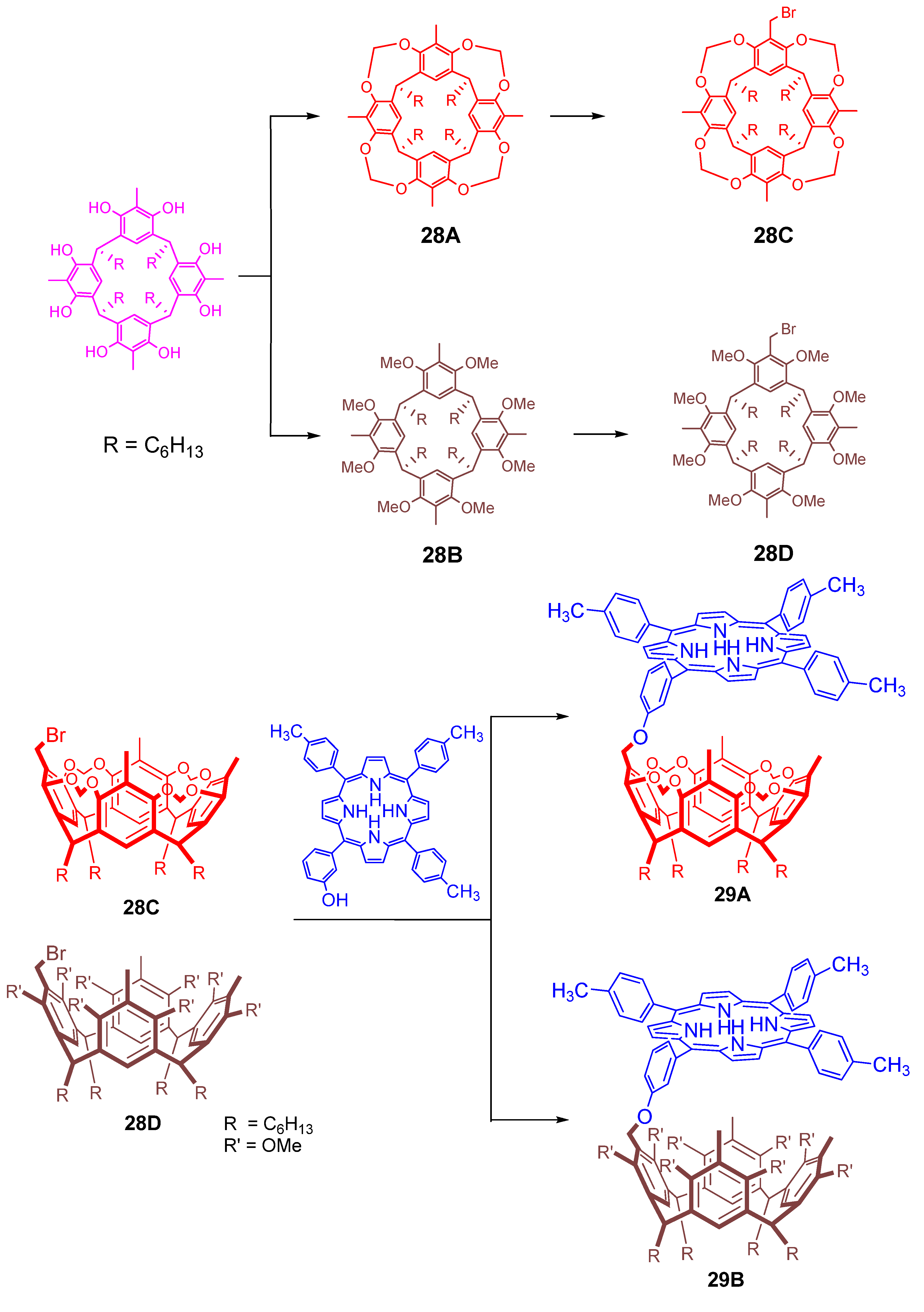
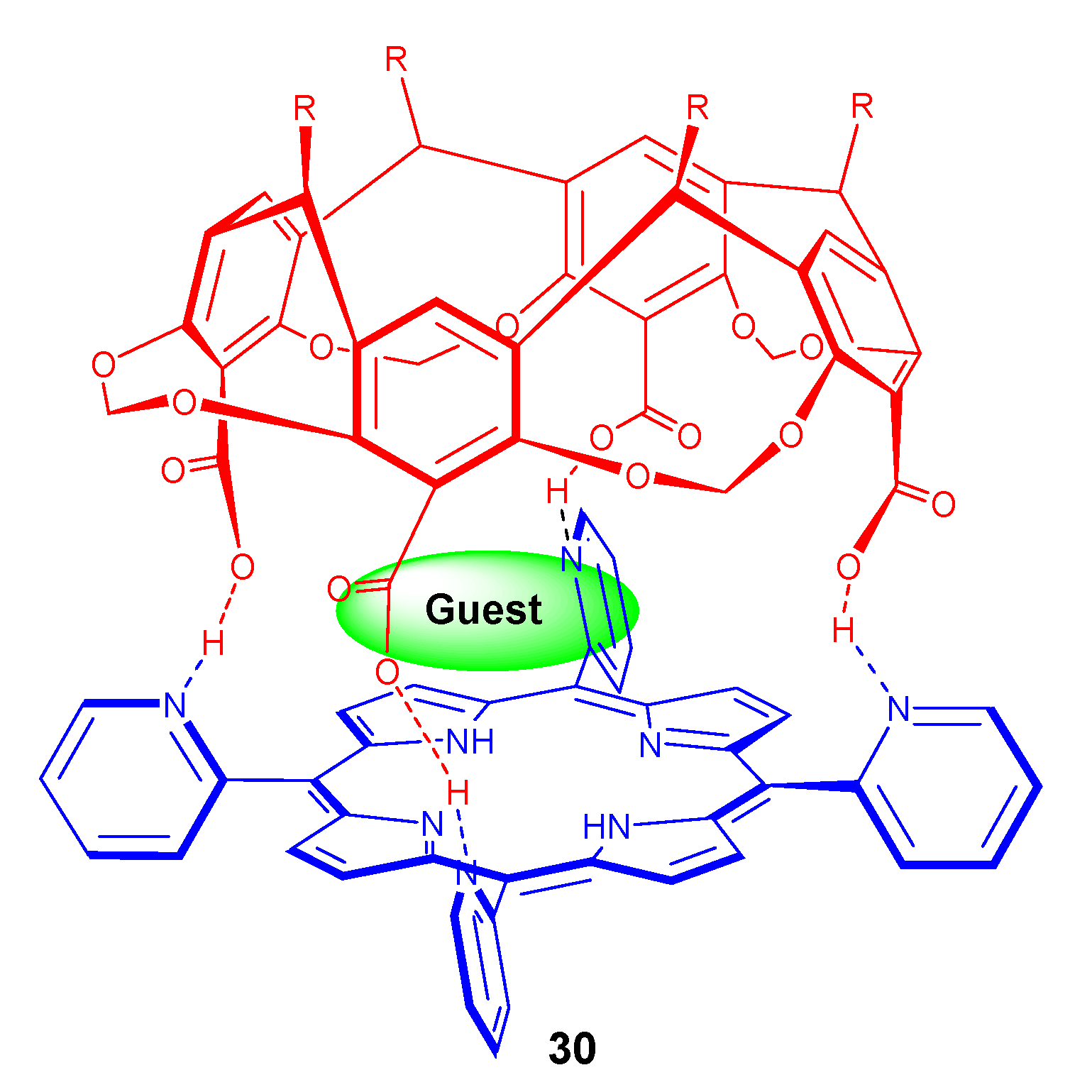
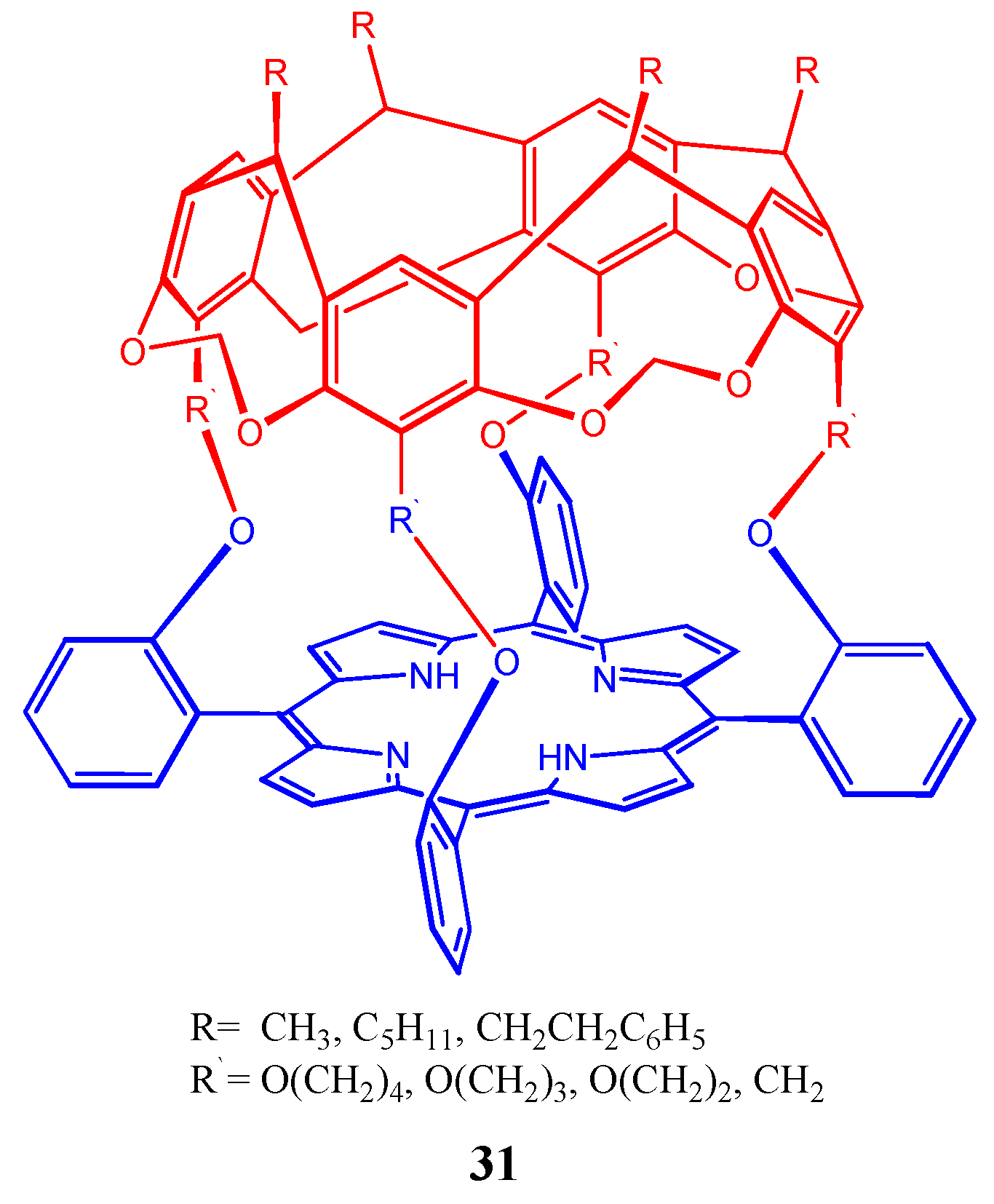
4. Resorcinarenes-Porphyrin Assemblies
5. Conclusions
Acknowledgments
References
- Voet, D.; Voet, J.D. Biochemistry, 3rd ed; Wiley: New York, NY, USA, 2004; pp. 1–1606. [Google Scholar]
- Kadish, K.M., Smith. Biochemistry and Binding: Activation of Small Molecules. In The Porphyrin Handbook; Academic Press: San Diego, CA, USA, 2000; Volume 4, pp. 1–341. [Google Scholar]
- Kadish, K.M., Smith. Bioinorganic and Bioorganic Chemistry. In The Porphyrin Handbook; Academic Press: San Diego, CA, USA, 2003; Volume 11, pp. 1–277. [Google Scholar]
- Milanesio, M.E.; Alvarez, M.G.; Durantini, E.N. Methoxyphenylporphyrin derivatives as phototherapeutic agents. Curr. Bioact. Compd. 2010, 6, 97–105. [Google Scholar] [CrossRef]
- De Visser, S.P.; Valentine, J.S.; Nam, W. A biomimetic ferric hydroperoxoporphyrin intermediate. Angew. Chem. Int. Ed. Engl. 2010, 49, 2099–2101. [Google Scholar] [CrossRef]
- Rocha Gonsalves, A.M.A.; Serra, A.C.; Pineiro, M. The small stones of Coimbra in the huge tetrapyrrolic chemistry building. J. Porphyr. Phthalocya. 2009, 13, 429–445. [Google Scholar] [CrossRef]
- Maeda, C.; Kamada, T.; Aratani, N.; Osuka, A. Chiral self-discriminative self-assembling of meso-meso linked diporphyrins. Coord. Chem. Rev. 2007, 251, 2743–2752. [Google Scholar] [CrossRef]
- Hori, T.; Nakamura, Y.; Aratani, N.; Osuka, A. Exploration of electronically interactive cyclic porphyrin arrays. J. Organomet. Chem. 2007, 692, 148–155. [Google Scholar]
- Savitsky, A.; Möbius, K. Photochemical reactions and photoinduced electron-transfer processes in liquids, frozen solutions, and proteins as studied by multifrequency time-resolved EPR spectroscopy. Helv. Chim. Acta 2006, 89, 2544–2589. [Google Scholar] [CrossRef]
- Balaban, T.S. Tailoring porphyrins and chlorins for self-assembly in biomimetic artificial antenna systems. Acc. Chem. Res. 2005, 38, 612–623. [Google Scholar] [CrossRef]
- Carofiglio, T.; Lubian, E.; Varotto, A. Synthesis, heterogenization and sensing properties of melamine-bridged bis-porphyrin dimers. J. Porphyr. Phthalocya. 2010, 14, 701–707. [Google Scholar] [CrossRef]
- Samaroo, D.; Vinodu, M.; Chen, X.; Drain, C.M. meso-Tetra(pentafluorophenyl)porphyrin as an efficient platform for combinatorial synthesis and the selection of new photodynamic therapeutics using a cancer cell line. J. Comb. Chem. 2007, 9, 998–1011. [Google Scholar] [CrossRef]
- Tsuda, A. Design of porphyrin nanoclusters toward discovery of novel properties and functions. Bull. Chem. Soc. Jpn. 2009, 82, 11–28. [Google Scholar] [CrossRef]
- Goldberg, I. Crystal engineering of nanoporous architectures and chiral porphyrin assemblies. CrystEngComm 2008, 10, 637–645. [Google Scholar] [CrossRef]
- Shinoda, S. Nanoscale substrate recognition by porphyrin dendrimers with patched structures. J. Incl. Phenom. Macro. 2007, 59, 1–9. [Google Scholar] [CrossRef]
- Boyd, P.D.W.; Reed, C.A. Fullerene-porphyrin constructs. Acc. Chem. Res. 2005, 38, 235–242. [Google Scholar] [CrossRef]
- Jurow, M.; Schuckman, A.E.; Batteas, J.D.; Drain, C.M. Porphyrins as molecular electronic components of functional devices. Coord. Chem. Rev. 2010, 254, 2297–2310. [Google Scholar] [CrossRef]
- Beletskaya, I.; Tyurin, V.S.; Tsivadze, A.Y.; Gulard, R.; Stern, C. Supramolecular chemistry of metalloporphyrins. Chem. Rev. 2009, 109, 1659–1713. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Liu, Y. Molecular selective binding and nano-fabrication of cucurbituril/cyclodextrin pairs. Isr. J. Chem. 2011, 51, 515–524. [Google Scholar] [CrossRef]
- Endo, T. Vital significance of addition of “shape similarity” between solutes to shape complementarity for more precise molecular recognition in aqueous binary solvents. Chem. Rec. 2011, 11, 146–157. [Google Scholar] [CrossRef]
- Kraus, T. Modified cyclodextrins with pendant cationic and anionic moieties as hosts for highly stable inclusion complexes and molecular recognition. Curr. Org. Chem. 2011, 15, 802–814. [Google Scholar] [CrossRef]
- De Rossi, R.H.; Silva, O.F.; Vico, R.V.; Gonzalez, C.J. Molecular organization and recognition properties of amphiphiliccyclodextrins. Pure Appl. Chem. 2009, 81, 755–765. [Google Scholar] [CrossRef]
- Sallas, F.; Darcy, R. Amphiphiliccyclodextrins—Advances in synthesis and supramolecular chemistry. Eur. J. Org. Chem. 2008, 6, 957–969. [Google Scholar] [CrossRef]
- Korendovych, I.V.; Roesner, R.A.; Rybak-Akimova, E.V. Molecular recognition of neutral and charged guests using metallomacrocyclic hosts. Adv. Inorg. Chem. 2007, 59, 109–173. [Google Scholar]
- Wang, M. Nitrogen and oxygen bridged calixaromatics: Synthesis, structure, functionalization, and molecular recognition. Acc. Chem. Res. 2012, 45, 182–195. [Google Scholar] [CrossRef]
- Joseph, R.; Rao, C.P. Ion and molecular recognition by lower rim 1,3-di-conjugates of calix[4]arene as receptors. Chem. Rev. 2011, 111, 4658–4702. [Google Scholar] [CrossRef]
- Evtugyn, G.A.; Stoikova, E.E.; Shamagsumova, R.V. Molecular receptors and electrochemical sensors based on functionalized calixarenes. Russ. Chem. Rev. 2010, 79, 1071–1097. [Google Scholar]
- Mokhtari, B.; Pourabdollah, K.; Dalali, N. Molecule and ion recognition of nano-baskets of calixarenes since 2005. J. Coord. Chem. 2011, 64, 743–794. [Google Scholar] [CrossRef]
- Sansone, F.; Baldini, L.; Casnati, A.; Ungaro, R. Calixarenes: From biomimetic receptors to multivalent ligands for biomolecular recognition. New J. Chem. 2010, 34, 2715–2728. [Google Scholar] [CrossRef]
- Thatiparti, T.R.; Shoffstall, A.J.; von Recum, H.A. Cyclodextrin-based device coatings for affinity-based release of antibiotics. Biomaterials 2010, 31, 2335–2347. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y. Cooperative binding and multiple recognition by bridged bis(β-cyclodextrin)s with functional linkers. Acc. Chem. Res. 2006, 39, 681–691. [Google Scholar] [CrossRef]
- Easton, C.J.; Lincoln, S.F. Chiral discrimination by modified cyclodextrins. Chem. Soc. Rev. 1996, 25, 163–170. [Google Scholar]
- Murakami, Y.; Kikuchi, J.; Hisaeda, Y.; Hayashida, O. Artificial enzymes. Chem. Rev. 1996, 96, 721–758. [Google Scholar] [CrossRef]
- Walker, B.D.; Joshi, G.; Davis, A.P. Progress in biomimetic carbohydrate recognition. Cell. Mol. Life Sci. 2009, 66, 3177–3191. [Google Scholar] [CrossRef]
- Puglisi, A.; Purrello, R.; Rizzarelli, E.; Sortino, S.; Vecchio, G. Spectroscopic and self-association behavior of a porphyrin-β-cyclodextrin conjugate. New J. Chem. 2007, 31, 1499–1506. [Google Scholar] [CrossRef]
- Jayawickramarajah, J.; Tagore, D.M.; Tsou, L.K.; Hamilton, A.D. Allosteric control of self-assembly: Modulating the formation of guanine quadruplexes through orthogonal aromatic interactions. Angew. Chem. Int. Ed. Engl. 2007, 46, 7583–7586. [Google Scholar] [CrossRef]
- Hosokawa, K.; Miura, Y.; Kiba, T.; Kakuchi, T.; Sato, S. Fluorescence resonance energy transfer in host-guest inclusion complexes of cyclodextrin-porphyrin composite in aqueous solution. Chem. Lett. 2008, 37, 60–61. [Google Scholar] [CrossRef]
- Fathalla, M.; Li, S.; Diebold, U.; Alb, A.; Jayawickramarajah, J. Water-soluble nano-rods self-assembled via pristine C60 and porphyrin moieties. Chem. Commun. 2009, 2009, 4209–4211. [Google Scholar]
- Kralova, J.; Kejik, Z.; Briza, T.; Pouckova, P.; Kral, A.; Martasek, P.; Kral, V. Porphyrin-cyclodextrinconjugates as a nanosystem for versatile drug delivery and multimodal cancer therapy. J. Med. Chem. 2010, 53, 128–138. [Google Scholar] [CrossRef]
- Kejik, Z.; Briza, T.; Kralova, J.; Pouckova, P.; Kral, A.; Martasek, P.; Kral, V. Coordination conjugates of therapeutic proteins with drug carriers: A new approach for versatile advanced drug delivery. Bioorg. Med. Chem. Lett. 2011, 21, 5514–5520. [Google Scholar]
- Guo, Y.; Zhang, P.; Chao, J.; Shuang, S.; Dong, C. Study on the supramolecular system of 5-(p-hydroxyphenyl)-10,15,20-tris-(4-chlorophenyl)porphyrin with cyclodextrins and its analytical characteristics. Spectrochim. Acta A 2008, 71A, 946–950. [Google Scholar]
- Zhang, L.; Li, X.; Kang, S.-Z.; Mu, J. Complex of porphyrins and cyclodextrin-modified multi-walled carbon nanotubes: Preparation and its properties. Supramol. Chem. 2008, 20, 601–604. [Google Scholar] [CrossRef]
- Fathalla, M.; Neuberger, A.; Li, S.-C.; Schmehl, R.; Diebold, U.; Jayawickramarajah, J. Straightforward self-assembly of porphyrin nanowires in water: Harnessing adamantane/β-cyclodextrin interactions. J. Am. Chem. Soc. 2010, 132, 9966–9967. [Google Scholar]
- Li, X.-X.; Wang, J.-W.; Guo, Y.-J.; Kong, L.-H.; Pan, J.-H. Study on inclusion complexes of meso-tetrakis(2-thienyl)porphyrin and Cu-meso-tetrakis(2-thienyl)porphyrin with cyclodextrins by spectroscopy method. J. Incl. Phenom. Macro. 2007, 58, 307–315. [Google Scholar] [CrossRef]
- Arimura, T.; Tsuchiya, Y.; Tashiro, M.; Tachiya, M. Photocurrent generators derived from non-covalently assembled cyclodextrinnano-system. J. Photopolym. Sci. Tech. 2007, 20, 533–538. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Chao, J.-B.; Pan, J.-H. Study on the interaction of 5-pyridine-10,15,20-tris-(p-chlorophenyl)porphyrin with cyclodextrins and DNA by spectroscopy. Spectrochim. Acta A 2007, 68A, 231–236. [Google Scholar]
- Kiba, T.; Suzuki, H.; Hosokawa, K.; Kobayashi, H.; Baba, S.; Kakuchi, T.; Sato, S. Supramolecular J-aggregate assembly of a covalently linked zinc Porphyrin-β-cyclodextrin conjugate in a water/ethanol binary mixture. J. Phys. Chem. B. 2009, 113, 11560–11563. [Google Scholar]
- Yang, Y.; Zhang, Y.-M.; Chen, Y.; Zhao, D.; Chen, J.-T.; Liu, Y. Construction of a grapheme oxide based noncovalent multiple nanosupramolecular assembly as a scaffold for drug delivery. Chem. Eur. J. 2012, 18, 4208–4215. [Google Scholar]
- Gu, Z.-Y.; Guo, D.-S.; Sun, M.; Liu, Y. Effective enlargement of fluorescence resonance energy transfer of poly-porphyrinmediated by β-cyclodextrindimers. J. Org. Chem. 2010, 75, 3600–3607. [Google Scholar]
- Ermilov, E.A.; Menting, R.; Lau, J.T.F.; Leng, X.; Roeder, B.; Ng, D.K.P. Switching the photoinduced processes in host-guest complexes of β-cyclodextrin-substituted silicon(iv) phthalocyanines and a tetrasulfonatedporphyrin. Phys. Chem. Chem. Phys. 2011, 13, 17633–17641. [Google Scholar]
- Xu, H.; Ermilov, E.A.; Roeder, B.; Ng, D.K.P. Formation and energy transfer property of a subphthalocyanine-porphyrin complex held by host-guest interactions. Phys. Chem. Chem. Phys. 2010, 12, 7366–7370. [Google Scholar]
- Khavasi, H.R.; Sasan, K.; Safari, N. Inclusion complex of iron meso-tetrakis(p-sulfonatophenyl)porphyrin and 2-hydroxypropyl-β-cyclodextrin as a functional model of cytochrome P-450; study on a supramolecular formation and its application in aqueous oxidation of styrene. J. Porphyr. Phthalocya. 2007, 11, 874–882. [Google Scholar] [CrossRef]
- Liu, Y.; Ke, C.-F.; Zhang, H.-Y.; Cui, J.; Ding, F. Complexation-induced transition of nanorod to network aggregates: Alternate porphyrin and cyclodextrinarrays. J. Am. Chem. Soc. 2008, 130, 600–605. [Google Scholar]
- Liang, P.; Zhang, H.-Y.; Yu, Z.-L.; Liu, Y. Solvent-controlled photoinduced electron transfer between porphyrin and carbon nanotubes. J. Org. Chem. 2008, 73, 2163–2168. [Google Scholar]
- Qiu, W.-G.; Li, Z.-F.; Bai, G.-M.; Meng, S.-N.; Dai, H.-X.; He, H. Study on the inclusion behavior between meso-tetrakis[4-(3-pyridinium-propoxy)phenyl]porphyrintetrakis-bromide and β-cyclodextrinderivatives in aqueous solution. Spectrochim. Acta A 2007, 66A, 1189–1193. [Google Scholar]
- Guo, Y.-J.; Guo, L.; Pan, J.-H. Study on the supramolecular system of tetrakis(2-hydroxy-5-sulfonatophenyl)porphyrin with cyclodextrins by spectroscopy. Phys. Chem. Liq. 2007, 45, 261–269. [Google Scholar] [CrossRef]
- Kano, K.; Ishida, Y. Supramolecular complex of cytochrome c with a polyanionic β-cyclodextrin. Angew. Chem. Int. Ed. Engl. 2007, 46, 727–730. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Yamano, A.; Shiraki, T.; Sada, K.; Shinkai, S. Single-crystal structure of porphyrin bicapped with trimethyl-β-cyclodextrins: A novel dye-oriented material. Chem. Lett. 2011, 40, 99–101. [Google Scholar] [CrossRef]
- Callari, F.L.; Mazzaglia, A.; Scolaro, L.M.; Valli, L.; Sortino, S. Biocompatible nanoparticles of amphiphilic cyclodextrins entangling porphyrins as suitable vessels for light-induced energy and electron transfer. J. Mater. Chem. 2008, 18, 802–805. [Google Scholar] [CrossRef]
- Yu, M.; Chen, Y.; Zhang, N.; Liu, Y. Construction and transmembrane dissociation behavior of supramolecular assembly of quinolinocyclodextrin with porphyrin. Org. Biomol. Chem. 2010, 8, 4148–4154. [Google Scholar] [CrossRef]
- Wang, Y.; Cohen, B.; Jicsinszky, L.; Douhal, A. Femtosecond to second studies of a water-soluble porphyrin derivative in chemical and biological nanocavities. Langmuir 2012, 28, 4363–4372. [Google Scholar] [CrossRef]
- Mazzaglia, A.; Valerio, A.; Micali, N.; Villari, V.; Quaglia, F.; Castriciano, M.A.; Scolaro, L.M.; Giuffre, M.; Siracusano, G.; Sciortino, M.T. Effective cell uptake of nanoassemblies of a fluorescent amphiphilic cyclodextrin and an anionic porphyrin. Chem. Commun. 2011, 47, 9140–9142. [Google Scholar]
- Ferro, S.; Jori, G.; Sortino, S.; Stancanelli, R.; Nikolov, P.; Tognon, G.; Ricchelli, F.; Mazzaglia, A. Inclusion of 5-[4-(1-Dodecanoylpyridinium)]-10,15,20-triphenylporphine in supramolecular aggregates of cationic amphiphilic cyclodextrins: Physicochemical characterization of the complexes and strengthening of the antimicrobial photosensitizing activity. Biomacromolecules 2009, 10, 2592–2600. [Google Scholar] [CrossRef]
- Mora, S.J.; Cormick, M.P.; Milanesio, M.E.; Durantini, E.N. The photodynamic activity of a novel porphyrin derivative bearing a fluconazole structure in different media and against Candida albicans. Dyes Pigm. 2010, 87, 234–240. [Google Scholar] [CrossRef]
- Zhang, Y.-M.; Chen, Y.; Zhuang, R.-J.; Liu, Y. Supramolecular architecture of tetrathiafulvalene-bridged bis(β-cyclodextrin) with porphyrin and its electron transfer behaviors. Photochem. Photobiol. Sci. 2011, 10, 1393–1398. [Google Scholar] [CrossRef]
- Zhang, Y.-M.; Chen, Y.; Yang, Y.; Liu, P.; Liu, Y. Supramolecular architectures by fullerene-bridged bis(permethyl-β-cyclodextrin)s with porphyrins. Chem. Eur. J. 2009, 15, 11333–11340. [Google Scholar] [CrossRef]
- Leng, X.; Choi, C.-F.; Lo, P.-C.; Ng, D.K.P. Assembling a mixed phthalocyanine-porphyrin array in aqueous media through host-guest interactions. Org. Lett. 2007, 9, 231–234. [Google Scholar]
- Valli, L.; Giancane, G.; Mazzaglia, A.; Scolaro, L.M.; Conoci, S.; Sortino, S. Photoresponsive multilayer films by assembling cationic amphiphilic cyclodextrins and anionic porphyrins at the air/water interface. J. Mater. Chem. 2007, 17, 1660–1663. [Google Scholar] [CrossRef]
- Wang, K.-R; Guo, D.-S.; Jiang, B.-P.; Liu, Y. Excitonic coupling interactions in the self-assembly of perylene-bridged bis(β-cyclodextrin)s and porphyrin. Chem. Commun. 2012, 48, 3644–3646. [Google Scholar] [CrossRef]
- Long, L.; Jin, J.Y.; Zhang, Y.; Yang, R.; Wang, K. Interactions between protein and porphyrin-containing cyclodextrin supramolecular system: A fluorescent sensing approach for albumin. Analyst 2008, 133, 1201–1208. [Google Scholar] [CrossRef]
- Yu, S.; Yin, Y.; Zhu, J.; Huang, X.; Luo, Q.; Xu, J.; Shen, J.; Liu, J. A modulatory bifunctional artificial enzyme with both SOD and GPx activities based on a smart star-shaped pseudo-block copolymer. Soft Matter 2010, 6, 5342–5350. [Google Scholar] [CrossRef]
- Yu, S.; Huang, X.; Miao, L.; Zhu, J.; Yin, Y.; Luo, Q.; Xu, J.; Shen, J.; Liu, J. A supramolecular bifunctional artificial enzyme with superoxide dismutase and glutathione peroxidase activities. Bioorg. Chem. 2010, 38, 159–164. [Google Scholar] [CrossRef]
- Oliveri, V.; Puglisi, A.; Vecchio, G. New conjugates of β-cyclodextrin with manganese(III) salophen and porphyrin complexes as antioxidant systems. Dalton Trans. 2011, 40, 2913–2919. [Google Scholar]
- Watanabe, K.; Kano, K. Time-dependent enzyme activity dominated by dissociation of J-aggregates bound to protein surface. Bioconjug. Chem. 2010, 21, 2332–2338. [Google Scholar] [CrossRef]
- Kano, K.; Ishida, Y. Regulation of α-chymotrypsin catalysis by ferric porphyrins and cyclodextrins. Chem. Asian J. 2008, 3, 678–686. [Google Scholar] [CrossRef]
- Kitagishi, H.; Tamaki, M.; Ueda, T.; Hirota, S.; Ohta, T.; Naruta, Y.; Kano, K. Oxoferryl porphyrin/hydrogen peroxide system whose behavior is equivalent to hydroperoxoferric porphyrin. J. Am. Chem. Soc. 2010, 132, 16730–16732. [Google Scholar] [CrossRef]
- Kano, K. Porphyrin-cyclodextrin supramolecular complexes as myoglobin model in water. Colloid Polym. Sci. 2008, 286, 79–84. [Google Scholar] [CrossRef]
- Kano, K.; Ochi, T.; Okunaka, S.; Ota, Y.; Karasugi, K.; Ueda, T.; Kitagishi, H. Preparation and function of poly(acrylic acid)s modified by supramolecular complex composed of porphinatoiron and a cyclodextrin dimer that bind diatomic molecules (O2 and CO) in aqueous solution. Chem. Asian J. 2011, 6, 2946–2955. [Google Scholar] [CrossRef]
- Kano, K.; Kitagishi, H. Hemo CD as an artificial oxygen carrier: Oxygen binding and autoxidation. Artif. Organs 2009, 33, 177–182. [Google Scholar] [CrossRef]
- Watanabe, K.; Kitagishi, H.; Kano, K. Supramolecular ferric porphyrins as cyanide receptors in aqueous solution. ACS Med. Chem. Lett. 2011, 2, 943–947. [Google Scholar] [CrossRef]
- Kano, K.; Kitagishi, H.; Dagallier, C.; Kodera, M.; Matsuo, T.; Hayashi, T.; Hisaeda, Y.; Hirota, S. Iron porphyrin-cyclodextrin supramolecular complex as a functional model of myoglobin in aqueous solution. Inorg. Chem. 2006, 45, 4448–4460. [Google Scholar]
- Baldini, L.; Casnati, A.; Sansone, F.; Ungaro, R. Calixarene-based multivalent ligands. Chem. Soc. Rev. 2007, 36, 254–266. [Google Scholar] [CrossRef]
- Hosseini, A.; Taylor, S.; Accorsi, G.; Armaroli, N.; Reed, C.A.; Boyd, P.D. Calix[4]arene-linked bisporphyrin hosts for fullerenes: Binding strength, solvation effects, and porphyrin-fullerene charge transfer bands. J. Am. Chem. Soc. 2006, 128, 15903–15913. [Google Scholar]
- Pognon, G.; Boudon, C.; Schenk, K.J.; Bonin, M.; Bach, B.; Weiss, J. Electrochemically triggered open and closed pacman bis-metalloporphyrins. J. Am. Chem. Soc. 2006, 128, 3488–3489. [Google Scholar]
- Ivanova, Y.B.; Churakhina, Y.I.; Mamardashvili, N.Z. Synthesis and basic properties of bisporphyrinocalix[4]arene. Russ. J. Gen. Chem 2008, 78, 673–677. [Google Scholar] [CrossRef]
- Mamardashvili, G.M.; Zvezdina, S.V.; Mamardashvili, N.Z. Bisporphyrin-calix[4]arene heterotopic receptors of multifunctional substrates. Russ. J. Gen. Chem. 2011, 81, 594–601. [Google Scholar] [CrossRef]
- Arimura, T.; Nishioka, T.; Suga, Y.; Kumamoto, S.; Tsuchiya, Y.; Yamaguchi, T.; Tachiya, M. 13C-NMR longitudinal relaxation time studies of a molecular tweezers derived from a calixarene-porphyrin conjugate. J. Oleo Sci. 2007, 56, 155–158. [Google Scholar] [CrossRef]
- Pognon, G.; Wytko, J.A.; Weiss, J. Unsymmetrical calix[4]arene bisporphyrin pacman. Org. Lett. 2007, 9, 785–788. [Google Scholar] [CrossRef]
- Ivanova, Y.B.; Kumeev, R.S.; Mamardashvili, N.Z. pH-Dependent conformational changes in bisporphyrincalix[4]arene. Russ. J. Gen. Chem. 2008, 78, 485–492. [Google Scholar] [CrossRef]
- Zvezdina, S.V.; Churakhina, Y.I.; Mamardashvili, N.Z. Synthesis and design of supramolecular systems on the basis of tetrapyrrole macrocycles. Russ. J. Org. Chem. 2007, 43, 1864–1869. [Google Scholar] [CrossRef]
- D’Urso, A.; Nicotra, P.F.; Centonze, G.; Fragala, M.E.; Gattuso, G.; Notti, A.; Pappalardo, A.; Pappalardo, S.; Parisi, M.F.; Purrello, R. Induction of chirality in porphyrin-(bis)calixarene assemblies: A mixed covalent-non-covalent vs. a fully non-covalent approach. Chem. Commun 2012, 48, 4046–4048. [Google Scholar]
- Mamardashvili, N.Z.; Koifman, O.I. Synthesis of calix[4]arene bis-porphyrin on the basis of biladiene-a,c dihydrobromide. Macrohetrocycles 2009, 2, 30–32. [Google Scholar]
- Mamardashvili, G.M.; Shinkar’, I.A.; Mamardashvili, N.Z.; Koifman, O.I. Calix[4]arene-porphyrin molecular receptors for selective binding of ethylenediamines. Russ. J. Coord. Chem. 2007, 33, 774–778. [Google Scholar]
- Kas, M.; Lang, K.; Stibor, I.; Lhotak, P. Novel fullerene receptors based on calixarene-porphyrin conjugates. Tetrahedron Lett. 2007, 48, 477–481. [Google Scholar] [CrossRef]
- Grimm, B.; Schornbaum, J.; Cardona, C.M.; van Paauwe, J.D.; Boyd, P.D.W.; Guldi, D.M. Enhanced binding strengths of acyclic porphyrin hosts with endohedral metallofullerenes. Chem. Sci. 2011, 2, 1530–1537. [Google Scholar] [CrossRef]
- Kundrat, O.; Kas, M.; Tkadlecova, M.; Lang, K.; Cvacka, J.; Stibor, I.; Lhotak, P. Thiacalix[4]arene-porphyrin conjugates with high selectivity towards fullerene C70. TetrahedronLett. 2007, 48, 6620–6623. [Google Scholar]
- Rossom, W.V.; Kundrat, O.; Ngo, T.H.; Lhotak, P.; Dehaen, W.; Maes, W. n oxacalix[2]arene[2]pyrimidine-bis(Zn-porphyrin) tweezer as a selective receptor towards fullerene C-70. Tetrahedron Lett. 2010, 51, 2423–2426. [Google Scholar] [CrossRef]
- Sun, D.; Tham, F.S.; Reed, C.A.; Chaker, L.; Burgess, M.; Boyd, P.D.W. Porphyrin-fullerene host-guest chemistry. J. Am. Chem. Soc. 2000, 122, 10704–10705. [Google Scholar]
- Guldi, D.M. Fullerene-porphyrin architectures; photosynthetic antenna and reaction center models. Chem. Soc. Rev. 2002, 31, 22–36. [Google Scholar] [CrossRef]
- Arimura, T.; Ide, S.; Suga, Y.; Tachiya, M. Syntheses and characterizations of a new calixarene-porphyrin conjugate as electron transfer system. J. Oleo Sci. 2007, 56, 149–153. [Google Scholar] [CrossRef]
- Snurnikova, O.V.; Lukyanenko, A.P.; Alekseeva, E.A.; Korovin, Y.V.; Rusakova, N.V. Synthesis and spectral-luminescent properties of polynuclear lanthanide complexes with functionalized calix[4]arenes. Macroheterocycles 2011, 4, 93–96. [Google Scholar] [CrossRef]
- Tsuge, A.; Kunimune, T.; Ikeda, Y.; Moriguchi, T.; Araki, K. Binding properties of calixarene-based cofacial bisporphyrins. Chem. Lett. 2010, 39, 1155–1157. [Google Scholar] [CrossRef]
- Monnereau, C.; Rebilly, J.-N.; Reinaud, O. Synthesis and first studies of the host-guest and substrate recognition properties of a porphyrin-tethered calix[6]arene ditopic ligand. Eur. J. Org. Chem. 2011, 2011, 166–175. [Google Scholar] [CrossRef]
- Holler, M.; Schmitt, M.; Nierengarten, J.-F. Synthesis and conformational analysis of porphyrin derivatives substituted with calix[4]arene subunits. J. Porphyr. Phthalocya. 2011, 15, 1183–1188. [Google Scholar] [CrossRef]
- Wu, L.; Jiao, L.; Lu, Q.; Hao, E.; Zhou, Y. Spectrofluorometric studies on the interaction between oxacalix[6]arene-locked trizinc(II)porphyrins and crystal violet. Spectrochim. Acta A 2009, 73, 353–357. [Google Scholar] [CrossRef]
- Jiao, L.; Hao, E.; Fronczek, F.R.; Smith, K.M.; Vicente, M.G.H. Syntheses and properties of functionalized oxacalix[4]arene porphyrins. Tetrahedron 2007, 63, 4011–4017. [Google Scholar] [CrossRef]
- Iwamoto, H.; Nishi, S.; Haino, T. Highly shape-selective guest encapsulation in the precisely defined cavity of a calix[4]arene-capped metalloporphyrin. Chem. Commun. 2011, 47, 12670–12672. [Google Scholar] [CrossRef]
- Giancane, G.; Valli, L. State of art in porphyrin Langmuir-Blodgett films as chemical sensors. Adv. Colloid Interface Sci. 2012, 171-172, 17–35. [Google Scholar] [CrossRef]
- Brittle, S.A.; Richardson, T.H.; Varley, L.; Hunter, C.A. Amine-sensing properties of a covalently linked calix[4]arene-porphyrin (“calixporph”) multilayered. J. Porphyr. Phthalocya. 2010, 14, 1027–1033. [Google Scholar] [CrossRef]
- De Miguel, G.; Perez-Morales, M.; Martin-Romero, M.T.; Munoz, E.; Richardson, T.H.; Camacho, L. J-aggregation of a water-soluble tetracationic porphyrin in mixed LB films with a calix[8]arene carboxylic acid derivative. Langmuir 2007, 23, 3794–3801. [Google Scholar] [CrossRef]
- Roales, J.; Pedrosa, J.M.; Castillero, P.; Cano, M.; Richardson, T.H. Optimization of mixed Langmuir-Blodgett films of a water insoluble porphyrin in a calixarene matrix for optical gas sensing. Thin Solid Films 2011, 519, 2025–2030. [Google Scholar] [CrossRef]
- De Miguel, G.; Martin-Romero, M.T.; Pedrosa, J.M.; Munoz, E.; Perez-Morales, M.; Richardson, T.H.; Camacho, L. Improvement of optical gas sensing using LB films containing a water insoluble porphyrin organized in a calixarene matrix. J. Mater. Chem. 2007, 17, 2914–2920. [Google Scholar] [CrossRef]
- Brittle, S.A.; Richardson, T.H.; Hutchinson, J.; Hunter, C.A. Comparing zinc and manganese porphyrin LB films as amine vapor sensing materials. Colloids Surf. A 2008, 321, 29–33. [Google Scholar] [CrossRef]
- De Miguel, G.; Martin-Romero, M.T.; Pedrosa, J.M.; Munoz, E.; Perez-Morales, M.; Richardson, T.H.; Camacho, L. Dis-aggregation of an insoluble porphyrin in a calixarene matrix: Characterization of aggregate modes by extended dipole model. Phys. Chem. Chem. Phys. 2008, 10, 1569–1576. [Google Scholar]
- D’Urso, A.; Cristaldi, D.A.; Fragala, M.E.; Gattuso, G.; Pappalardo, A.; Villari, V.; Micali, N.; Pappalardo, S.; Parisi, M.F.; Purrello, R. Sequence, stoichiometry, and dimensionality control in porphyrin/bis-calix[4]arene self-assemblies in aqueous solution. Chem. Eur. J. 2010, 16, 10439–10446. [Google Scholar] [CrossRef]
- Tabbi, G.; di Mauro, G.; Purrello, R.; Bonomo, R.P. Electrochemical characterization of ordered arrays of metallo-porphyrins in aqueous solution. Dalton Trans. 2011, 40, 4223–4229. [Google Scholar] [CrossRef]
- De Zorzi, R.; Guidolin, N.; Randaccio, L.; Purrello, R.; Geremia, S. Nanoporous crystals of calixarene/porphyrin supramolecular complex functionalized by diffusion and coordination of metal ions. J. Am. Chem. Soc. 2009, 131, 2487–2489. [Google Scholar] [CrossRef]
- De Zorzi, R.; Guidolin, N.; Randaccio, L.; Geremia, S. A bifunctionalized porous material containing discrete assemblies of copper-porphyrins and calixarenes metalated by ion diffusion. CrystEngComm 2010, 12, 4056–4058. [Google Scholar] [CrossRef]
- Mamardashvili, G.M.; Chizhova, N.V.; Mamardashvili, N.Z. Synthesis of calix[4]arene-bis(tin(IV)porphyrins) and supramolecular complexes on their basis. Russ. J. Inorg. Chem. 2012, 57, 390–397. [Google Scholar]
- Guo, D.-S.; Chen, K.; Zhang, H.-Q.; Liu, Y. Nano-supramolecular assemblies constructed from water-soluble bis(calix[5]arenes) with porphyrins and their photoinduced electron transfer properties. Chem. Asian J. 2009, 4, 436–445. [Google Scholar] [CrossRef]
- D’Urso, A.; Fragala, M. E.; Purrello, R. From self-assembly to noncovalent synthesis of programmable porphyrins’ arrays in aqueous solution. Chem. Commun. 2012, 48, 8165–8176. [Google Scholar] [CrossRef]
- Kubat, P.; Sebera, J.; Zalis, S.; Langmaier, J.; Fuciman, M.; Polivka, T.; Lang, K. Charge transfer in porphyrin-calixarene complexes: Ultrafast kinetics, cyclic voltammetry, and DFT calculations. Phys. Chem. Chem. Phys. 2011, 13, 6947–6954. [Google Scholar]
- Kubat, P.; Lang, K.; Lhotak, P.; Janda, P.; Sykora, J.; Matejicek, P.; Hof, M.; Prochazka, K.; Zelinger, Z. Porphyrin/calixarene self-assemblies in aqueous solution. J. Photochem. Photobiol. A Chem. 2008, 198, 18–25. [Google Scholar] [CrossRef]
- Schroeder, T.; Sahu, S.N.; Mattay, J. Molecular capsules derived from resorcin[4]arenes by metal-coordination. Top. Curr. Chem. 2012, 319, 99–124. [Google Scholar]
- Beyeh, N.K.; Rissanen, K. Dimeric resorcin[4]arene capsules in the solid state. Isr. J. Chem. 2011, 51, 769–780. [Google Scholar]
- Pinalli, R.; Boccini, F.; Dalcanale, E. Cavitand-based coordination cages: Achievements and current challenges. Isr. J. Chem. 2011, 51, 781–797. [Google Scholar]
- Jain, V.K.; Kanaiya, P.H. Chemistry of calix[4]resorcinarenes. Russ. Chem. Rev. 2011, 80, 75–102. [Google Scholar] [CrossRef]
- Dalgarno, S.J.; Power, N.P.; Atwood, J.L. Metallo-supramolecular capsules. Coord. Chem. Rev. 2008, 252, 825–841. [Google Scholar] [CrossRef]
- Al-Azemi, T.F.; Vinodh, M. Synthesis of porphyrin conjugates based on conformationally rigid and flexible resorcin[4]arene frameworks. Tetrahedron 2011, 67, 2585–2590. [Google Scholar] [CrossRef]
- Wu, R.; Al-Azemi, T.F.; Bisht, K.S. Spatially directional multiarm poly(ε-caprolactone) based on resorcin[4]arene cavitand core. Chem. Commun. 2009, 2009, 1822–1824. [Google Scholar]
- Nakazawa, J.; Mizuki, M.; Shimazaki, Y.; Tani, F.; Naruta, Y. Encapsulation of small molecules by a cavitand porphyrin self-assembled via quadruple hydrogen bonds. Org. Lett. 2006, 8, 4275–4278. [Google Scholar] [CrossRef]
- Nakazawa, J.; Sakae, Y.; Aida, M.; Naruta, Y. Kinetic investigations of the process of encapsulation of small hydrocarbons into a cavitand-porphyrin. J. Org. Chem. 2007, 72, 9448–9455. [Google Scholar] [CrossRef]
- McKay, M.G.; Cwele, T.; Friedrich, H.B.; Maguire, G.E.M. Microwave-assisted synthesis of a new series of resorcin[4]arene cavitand-capped porphyrin capsules. Org. Biomol. Chem. 2009, 7, 3958–3968. [Google Scholar] [CrossRef]
- Stefanelli, M.; Monti, D.; Van Axel Castelli, V.; Ercolani, G.; Venanzi, M.; Pomarico, G.; Paolesse, R. Chiral supramolecular capsule by ligand promoted self-assembly of resorcinarene-Zn porphyrin conjugate. J. Porphyr. Phthalocya. 2008, 12, 1279–1288. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vinodh, M.; Alipour, F.H.; Mohamod, A.A.; Al-Azemi, T.F. Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications. Molecules 2012, 17, 11763-11799. https://doi.org/10.3390/molecules171011763
Vinodh M, Alipour FH, Mohamod AA, Al-Azemi TF. Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications. Molecules. 2012; 17(10):11763-11799. https://doi.org/10.3390/molecules171011763
Chicago/Turabian StyleVinodh, Mickey, Fatemeh H. Alipour, Abdirahman A. Mohamod, and Talal F. Al-Azemi. 2012. "Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications" Molecules 17, no. 10: 11763-11799. https://doi.org/10.3390/molecules171011763
APA StyleVinodh, M., Alipour, F. H., Mohamod, A. A., & Al-Azemi, T. F. (2012). Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications. Molecules, 17(10), 11763-11799. https://doi.org/10.3390/molecules171011763



