Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species
Abstract
:1. Introduction
2. Results and Discussion
2.1. Essential Oil Composition and Variability
3. Experimental
3.1. Plant Material
| Locality in Croatia | Plant species | Habitat Types | Coordinates: Gauss-Krüger (X,Y) | Altidude a.s.l. (m) |
|---|---|---|---|---|
| Elevations between Trogir and Prapatnica | T. polium | Rocky grassland, once an area affected by fire | X = 5598845Y = 4820965 | 277 |
| Marjan, hill above town Split | T. flavum | Dry grasslands as. Querco ilici-Pinetum halepensis Loisel 1971 | X = 5614505Y = 4819218 | 158 |
| Elevations between Trogir and Prapatnica | T. montanum | Rocky grassland, once an area affected by fire | X = 5598845Y = 4820965 | 277 |
| Klis Grlo | T. chamaedrys | Rocky grassland in succession towards under-growth and lower forest as. Carpino orientali-Quercetum virgilianae Trinajstić 1987 | X = 5624263Y = 4826875 | 356 |
3.2. Isolation of Essential Oils
3.3. Gas Chromatography and Mass Spectrometry (GC, GC/MS)
3.4. Data Analysis and Data Evaluation
3.6. Solution of Essential Oil
3.7. Viral Inoculums
3.8. Application to the Local Host Plants
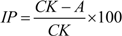
3.9. Statistical Analysis
4. Conclusion
Acknowledgments
- Sample Availability: Samples of the compounds are available from the authors.
References
- Tutin, T.G.; Wood, D. Teucrium. In Flora Europaea; Tutin, T.G., Ed.; Cambridge University Press: Cambridge, UK, 1972; Volume 3. [Google Scholar]
- Nikolić, T. (Ed.) Flora Croatica Database; Department of Botany, Faculty of Science, University of Zagreb: Zagreb, Croatia, 2011. Available online: http://hirc.botanic.hr/fcd (accessed on 25 July 2011).
- Trinajstić, I. Plant Communities of Croatia; Academy of Forestry Sciences: Zagreb, Croatia, 2008; pp. 106–125. [Google Scholar]
- Cavalerio, C.; Salgueiro, L.R.; Antunes, T.; Sevinate-Pinto, I.; Barroso, G. Composition of the essential oil and micromorphology of trichomes of Teucrium salviastrum, an endemic species from Portugal. Flavour Fragr. J. 2002, 17, 287–291. [Google Scholar] [CrossRef]
- Hisham, A.; Pathare, N.; Al-Saidi, S. The Composition and Antimicrobial Activity of the Essential Oil of Teucrium stocksianum subsp. stocksianum Leaf from Oman. Nat. Prod. Comm. 2006, 1, 195–199. [Google Scholar]
- Kazemizadeh, Z.; Basiri, A.; Habibi, Z. Chemical Composition of the Essential Oil of Teucrium hyrcanicum and T. chamaedrys L. subsp. chamaedrys from Iran. Chem. Nat. Comp. 2008, 44, 651–653. [Google Scholar]
- Formisano, C.; Rigano, D.; Senatore, F.; Al-Hillo, M.R.Y.; Piozzi, F.; Rosselli, S. Analysis of Essential Oil from Teucrium maghrebinum Greuter et Burdet Growing Wild in Algeria. Nat. Prod. Comm. 2009, 5, 411–414. [Google Scholar]
- De Martino, L.; Formisano, C.; Mancini, E.; De Feo, V.; Piozzi, F.; Rigano, D.; Senatore, F. Chemical Composition and Phytotoxic Effects of Essential Oils from Four Teucrium Species. Nat. Prod. Comm. 2010, 5, 1969–1976. [Google Scholar]
- Morteza-Semnani, K.; Saeed, M.; Akbarzade, M. Essential oil composition of Teucrium scordium L. Acta Pharm. 2007, 57, 499–504. [Google Scholar] [CrossRef]
- Wink, M. Evolution of secondary metabolites from an ecological and molecular phylogenetic perspective. Phytochemistry 2003, 64, 3–19. [Google Scholar]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007, 3, 408–414. [Google Scholar] [CrossRef]
- Michaelakis, A.; Theotokatos, S.A.; Koliopoulos, G.; Chorianopoulos, N.G. Essential oils of Satureja species: insecticidal effects on Culex pipens larvae (diptera: culicidae). Molecules 2007, 12, 2567–2578. [Google Scholar]
- Bishop, C.D. Antiviral activity of the essential oil of Melaluca alternifolia (Maiden & Betche) cheel (tea tree) against tobacco mosaic virus. J. Essent. Oil Res. 1995, 7, 641–644. [Google Scholar] [CrossRef]
- Juglal, S.; Govinden, R.; Odhav, B. Spice oils for the control of co-occurring mycotoxin-producing fungi. J. Food Prot. 2002, 65, 683–687. [Google Scholar]
- Moon, T.; Wilkinson, J.M.; Cavanagh, H.M.A. Antiparasitic activity of two Lavandula essential oils against Giardia duodenalis, Trichomonas vaginalis and Hexamita inflata. Parasitol. Res. 2006, 99, 722–728. [Google Scholar] [CrossRef]
- Lamiri, A.; Lhaloui, S.; Benjilali, B.; Berrada, M. Insecticidal effects of essential oils against Hessian fly, Mayetiola destructor (Say). Field Crop. Res. 2001, 71, 9–15. [Google Scholar] [CrossRef]
- Othman, B.A.; Shoman, S.A. Antiphytoviral Activity of the Plectranthus tenuiflorus on Some Important Viruses. Int. J. Agri. Biol. 2004, 6, 844–849. [Google Scholar]
- Krcatović, E.; Rusak, G.; Bezić, N.; Krajačić, M. Inhibition of Tobacco Mosaic Virus infection by quercetin and vitexin. Acta Virolog. 2008, 52, 119–124. [Google Scholar]
- Rusak, G.; Krajačić, M.; Pleše, N. Inhibition of tomato bushy stunt virus infection using a quercetagetin flavonoid isolated from Centaurea rupestris L. Antiviral Res. 1997, 36, 125–129. [Google Scholar] [CrossRef]
- Yordanova, A.; Korparov, N.; Stomenova, E.; Starcheva, M. Antiphytoviral activity of 1-morpholinomethyl tetrahydro 2-Pyrimidinone (DDB). Plant Pathol. 1996, 45, 547–551. [Google Scholar]
- Dunkić, V.; Bezić, N.; Vuko, E.; Cukrov, D. Antiphytoviral Activity of Satureja montana L. ssp. variegata (Host) P. W. Ball Essential Oil and Phenol Compounds on CMV and TMV. Molecules 2010, 15, 6713–6721. [Google Scholar] [CrossRef]
- Dunkić, V.; Bezić, N.; Vuko, E. Antiphytoviral Activity of Essential Oil from Endemic Species Teucrium arduini L. Nat. Prod. Comm. 2011, 6, 1385–1388. [Google Scholar]
- Maghtader, M. Chemical Composition of the Essential Oil of Teucrium polium L. From Iran. Amer.-Euroas. J. Agric. Environ. Sci. 2009, 5, 843–856. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bezić, N.; Vuko, E.; Dunkić, V.; Ruščić, M.; Blažević, I.; Burčul, F. Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species. Molecules 2011, 16, 8119-8129. https://doi.org/10.3390/molecules16098119
Bezić N, Vuko E, Dunkić V, Ruščić M, Blažević I, Burčul F. Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species. Molecules. 2011; 16(9):8119-8129. https://doi.org/10.3390/molecules16098119
Chicago/Turabian StyleBezić, Nada, Elma Vuko, Valerija Dunkić, Mirko Ruščić, Ivica Blažević, and Franko Burčul. 2011. "Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species" Molecules 16, no. 9: 8119-8129. https://doi.org/10.3390/molecules16098119
APA StyleBezić, N., Vuko, E., Dunkić, V., Ruščić, M., Blažević, I., & Burčul, F. (2011). Antiphytoviral Activity of Sesquiterpene-Rich Essential Oils from Four Croatian Teucrium Species. Molecules, 16(9), 8119-8129. https://doi.org/10.3390/molecules16098119






