Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds
Abstract
:1. Introduction
- (1). The inhibition of lipid oxidation in different systems (oils, solutions of lipids in organic solvents, oil-in-water emulsions, micelles, liposomes, etc.);
- (2). The ability to scavenge free radical species [3].
2. Results and Discussion
2.1. Main Components Present in the Essential Oils
| Components | RI | T. vulgaris | C. aurantium | C. sempervirens | E. globulus | C. limon | F. vulgare |
|---|---|---|---|---|---|---|---|
| α-Pinene | 930 | 49 | |||||
| δ-3-Carene | 1000 | 18 | |||||
| p-Cymene | 1003 | 24 | |||||
| 1,8-Cineole | 1005 | 38 | |||||
| Limonene | 1009 | 32 | 55 | 99 | |||
| Linalool | 1074 | 59 | |||||
| Borneol | 1134 | 16 | |||||
| Linalyl acetate | 1245 | 23 | |||||
| trans-Anethole | 1254 | 75 | |||||
| Thymol | 1275 | 12 | |||||
| Carvacrol | 1286 | 16 |
2.2. Antioxidant Activities of Essential Oils and Standards
2.2.1. Lipid Peroxidation Inhibition by the Thiobarbituric Acid Reactive Species (TBARS) Method
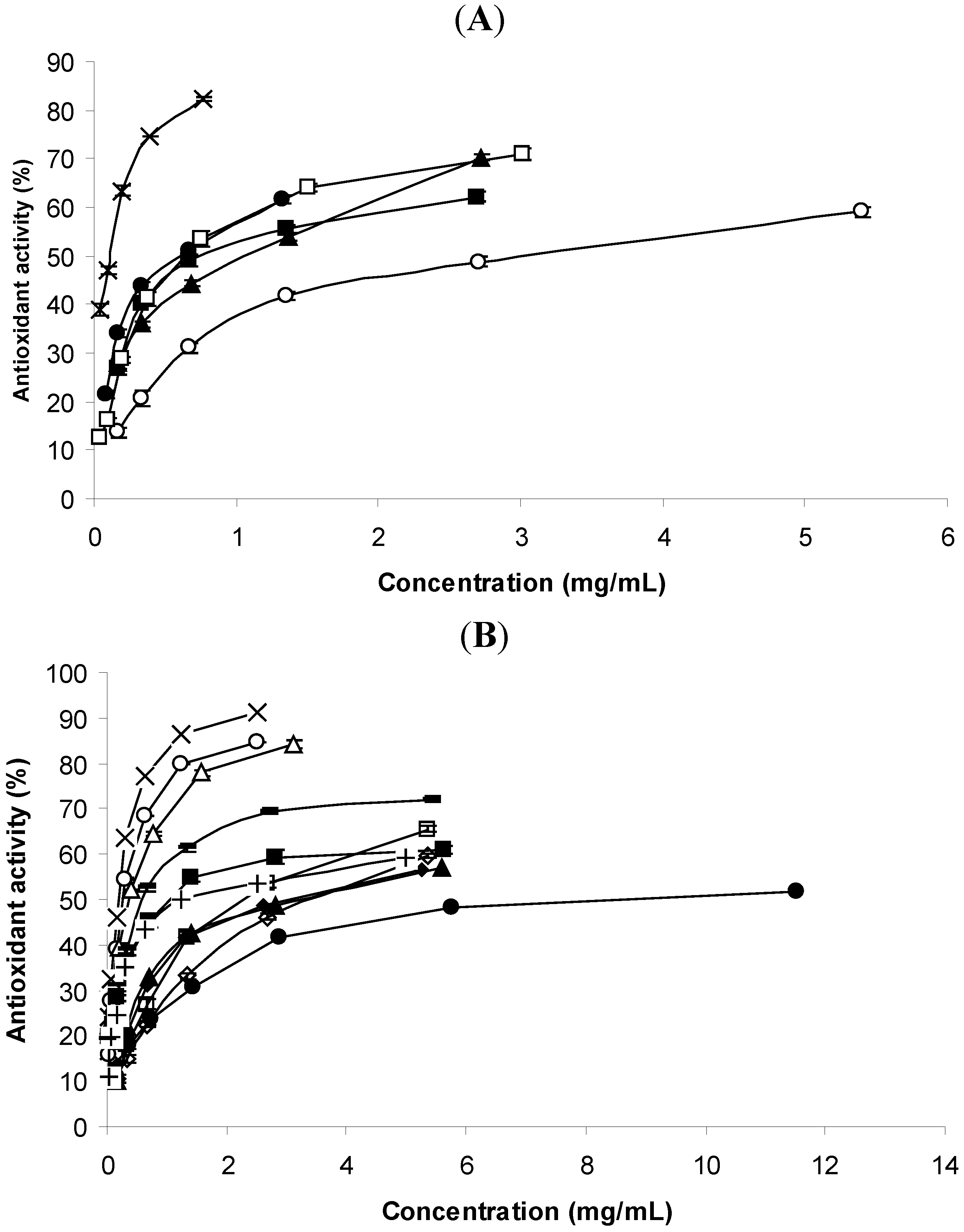
| Plant or standard | TBARS | DPPH | AChE inhibitory activity |
|---|---|---|---|
| IC50 (mg/mL) | IC50 (mg/mL) | IC50 (mg/mL) | |
| T. vulgaris | 0.116 ± 0.125 i | 0.259 ± 0.476 d | 0.2169 ± 0.0115 f |
| C. aurantium | 0.652 ± 0.125 gh | 4.786 ± 0.476 c | nd |
| C. sempervirens | 0.766 ± 0.125 fg | 8.245 ± 0.476 b | 0.2837 ± 0.0115 e |
| C. limon | 3.193 ± 0.125 c | 16.145 ± 0.476 a | 0.8499 ± 0.0115 c |
| E. globulus | 1.109 ± 0.125 f | 0.1298 ± 0.0115 g | |
| F. vulgare | 0.652 ± 0.125 gh | 1.1877 ± 0.0115 b | |
| Thymol | 0.172 ± 0.125 i | 0.051 ± 0.476 d | 0.2124 ± 0.0115 f |
| Carvacrol | 0.267 ± 0.125 hi | 0.052 ± 0.476 d | 0.0917 ± 0.0115 h |
| trans-Anethole | 0.354 ± 0.125 ghi | - | 1.3244 ± 0.0115 a |
| α-Pinene | 3.715 ± 0.125 b | - | nd |
| p-Cymene | 2.313 ± 0.125 d | - | nd |
| Linalool | 1.059 ± 0.125 f | - | nd |
| Linalyl acetate | 3.421 ± 0.125 bc | - | nd |
| 1,8-Cineole | 9.360 ± 0.125 a | - | 0.1082 ± 0.0115 gh |
| Limonene | 3.346 ± 0.125 bc | - | 0.5863 ± 0.0115 d |
| Borneol | 1.689 ± 0.125 e | - | 0.1321 ± 0.0115 g |
| δ-3-Carene | 0.603 ± 0.125 gh | - | 0.0358 ± 0.0115 i |
2.2.2. Ability for Scavenging DPPH Free Radicals
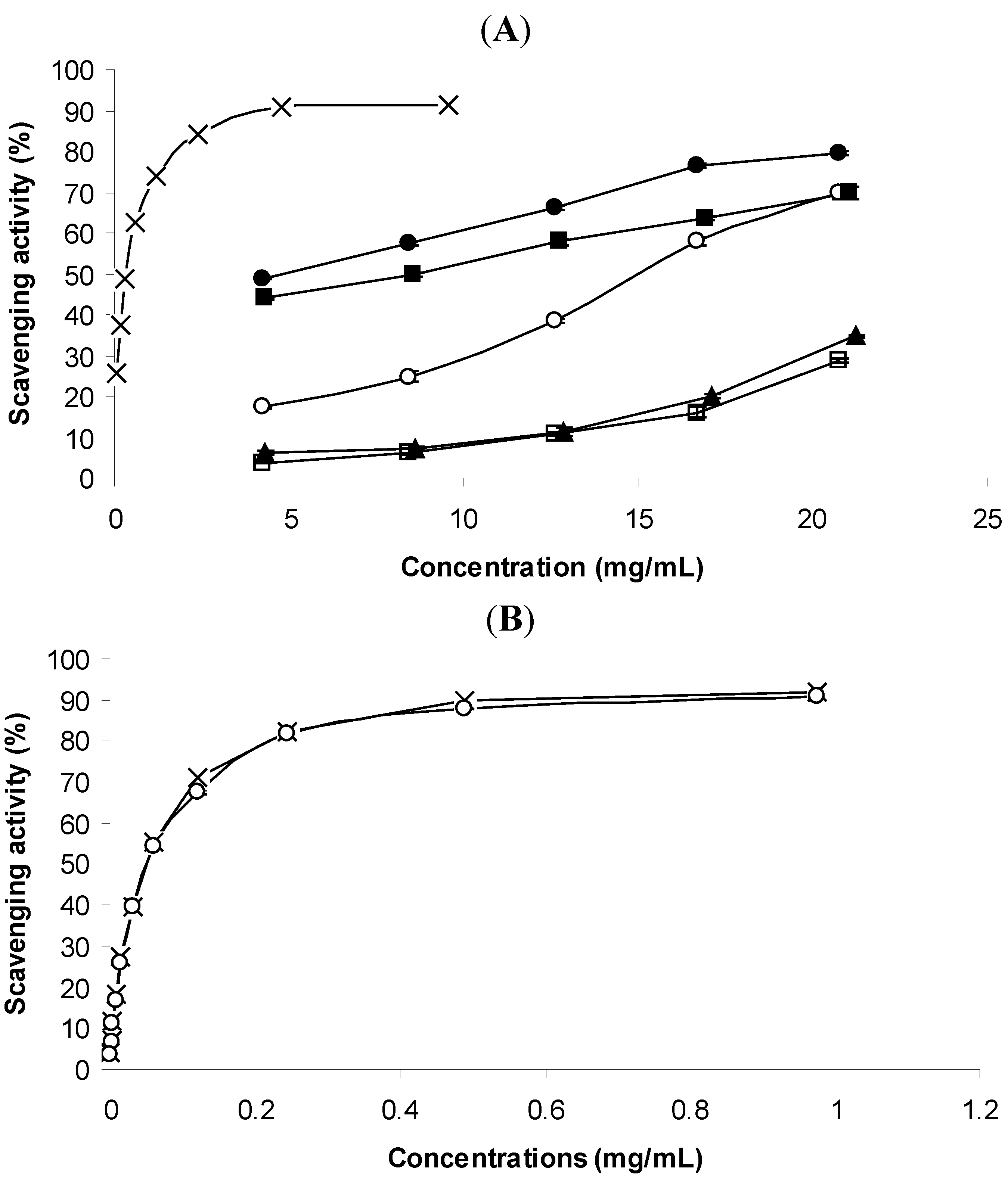
| Plant or standard | DPPH | Reductive potential |
|---|---|---|
| Scavenging activity (%) | Absorbance (770 nm) a | |
| E. globulus | 34.8 ± 0.2 | |
| F. vulgare | 28.8 ± 0.4 | |
| trans-Anethole | 39.8 ± 0.3 | |
| α-Pinene | 40 ± 0.2 | 0.034 ± 0.002 |
| p-Cymene | 36.7 ± 0.3 | 0.090 ± 0.006 |
| Linalool | 38.2 ± 0.2 | 0.052 ± 0.005 |
| Linalyl acetate | 39.1 ± 0.2 | 0.050 ± 0.002 |
| 1,8-Cineole | 36.3 ± 0.3 | 0.068 ± 0.002 |
| Limonene | 38.2 ± 0.5 | |
| α-Borneol | 19.6 ± 1.1 | 0.025 ± 0.002 |
| δ-3-Carene | 21.1 ± 0.5 |
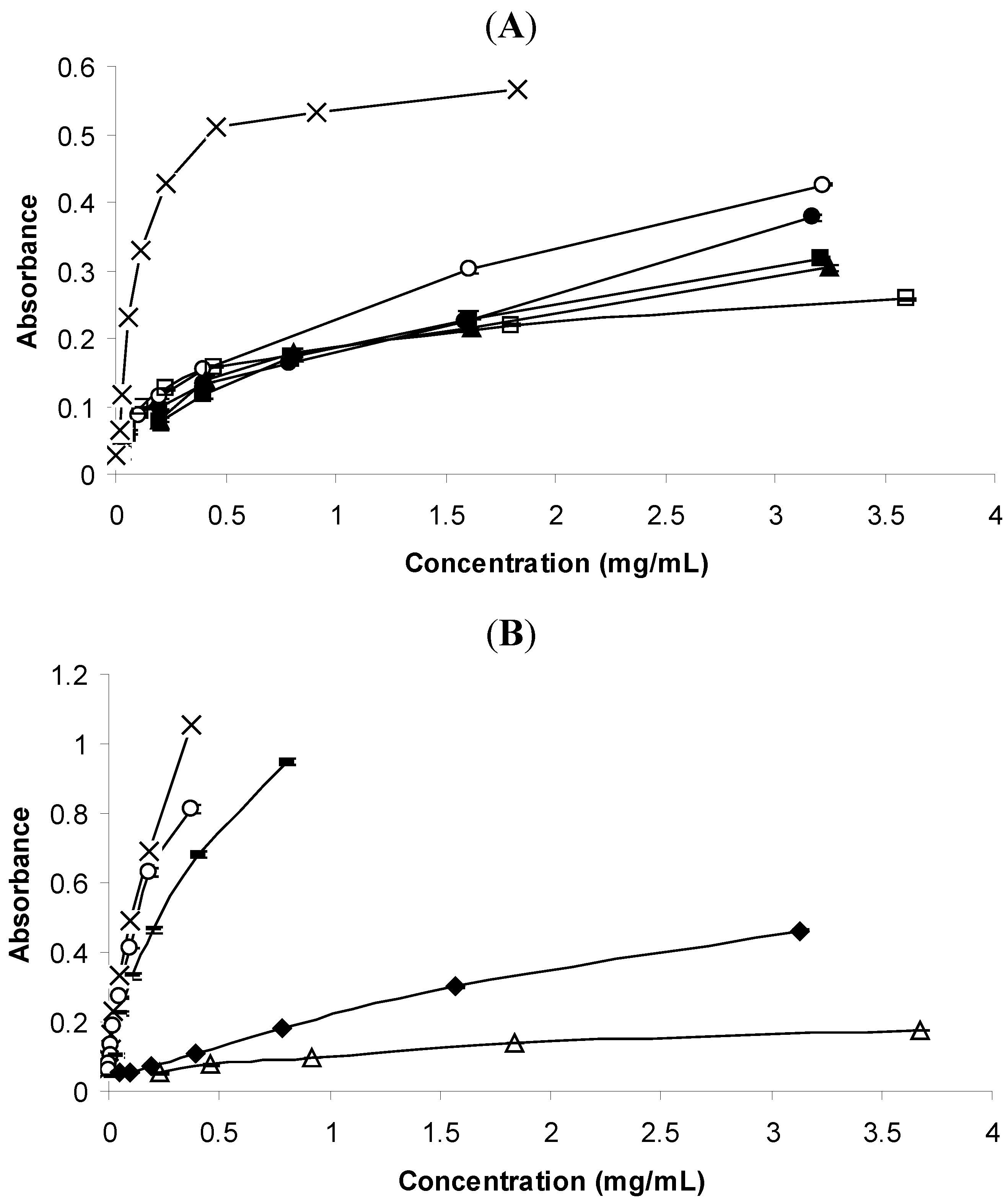
2.2.3. Reductive Potential
2.3. Acetylcholinesterase (AChE) Inhibitory Activity

3. Experimental
3.1. Samples
3.2. Standards and Reagents
3.3. Chemical Analysis of the Essential Oils
3.3.1. Chemical Analysis of the Essential Oils by Gas Chromatography (GC)
3.3.2. Gas Chromatography-Mass Spectrometry (GC-MS)
3.4. Antioxidant Activities
3.4.1. Thiobarbituric Acid Reactive Species (TBARS)
3.4.2. DPPH Free Radical-Scavenging Activity
3.4.3. Reductive Potential
3.5. Acetylcholinesterase (AChE) Inhibitory Activity
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Karadag, A.; Ozcelik, B.; Saner, S. Review of methods to determine antioxidant capacities. Food Anal. Method. 2009, 2, 41–60. [Google Scholar] [CrossRef]
- Miguel, M.G. Antioxidant and anti-inflammatory activities of essential oils: A short review. Molecules 2010, 15, 9252–9286. [Google Scholar] [CrossRef]
- Miguel, M.G. Antioxidant activity of medicinal and aromatic plants. A review. Flavour Fragr. J. 2010, 25, 291–312. [Google Scholar] [CrossRef]
- Fujiwara, M.; Yagi, N.; Miyazawa, M. Acetylcholinesterase inhibitory activity of volatile oil from Peltophorum dasyrachis Kurz ex Bakar (Yellow Batai) and bisabolene-type sesquiterpenoids. J. Agric. Food Chem. 2010, 58, 2824–2829. [Google Scholar] [CrossRef]
- Mantle, D.; Pickering, A.T.; Perry, E.K. Medicinal plant extracts for the treatment of dementia: A review of their pharmacology, efficacy and tolerability. CNS Drugs 2000, 13, 201–213. [Google Scholar] [CrossRef]
- Dohi, S.; Terasaki, M.; Makino, M. Acetylcholinesterase inhibitory activity and chemical composition of commercial essential oils. J. Agric. Food Chem. 2009, 57, 4313–4318. [Google Scholar] [CrossRef]
- Dandlen, S.A.; Miguel, M.G.; Duarte, J.; Faleiro, M.L.; Sousa, M.J.; Lima, A.S.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Acetylcholinesterase inhibition activity of Portuguese Thymus species essential oils. J. Essent. Oil Bear. Pl. 2011, 14, 140–150. [Google Scholar]
- Pultrini, A.M.; Galindo, L.A.; Costa, M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci. 2006, 78, 1720–1725. [Google Scholar] [CrossRef]
- Effects of limonene and essential oil from Citrus aurantium on gastric mucosa: Role of prostaglandins and gastric mucus secretion. Chem.-Biol. Interact. 2009, 180, 499–505. [CrossRef]
- Mabberley, D.J. Citrus (Rutaceae): A review of recent advances in etymology, systematics and medical applications. Blumea 2004, 49, 481–498. [Google Scholar] [CrossRef]
- Pandey, A.; Singh, A.; Garg, P.; Raja, R.B. Biodetection of the mercury level in soil using Citrus limonum plant; a novel method to decrease heavy metal industrial contamination. Ann. Biol. Res. 2011, 2, 189–193. [Google Scholar]
- Sultana, S.; Ali, M.; Ansari, S.H.; Bagri, P. New 4’-substituted flavones from the fruit peels of Citrus limon (L.) Burm.f. J. Asian Nat. Prod. Res. 2008, 10, 1123–1127. [Google Scholar] [CrossRef]
- Prichard, A.J.N. The use of essential oils to treat snoring. Phytother. Res. 2004, 18, 696–699. [Google Scholar] [CrossRef]
- Emami, S.A.; Asili, J.; Rahimizadeh, M.; Fazly-Bazzac, B.S.; Hassanzadeh-Khayyat, M. Chemical and antimicrobial studies of Cupressus sempervirens L. and C. horizentalis Mill. essential oils. Iran. J. Pharm. Sci. 2006, 2, 103–108. [Google Scholar]
- Toroglu, S. In vitro antimicrobial activity and antagonistic effect of essential oils from plant species. J. Environ. Biol. 2007, 28, 551–559. [Google Scholar]
- Tian, L.-W.; Yang, C.-R.; Zhang, Y.-J. Phenolic compounds from the fresh leaves of Eucalyptus maideni. Helv. Chim. Acta 2010, 93, 2194–2202. [Google Scholar] [CrossRef]
- Mulyaningsih, S.; Sporer, F.; Zimmermann, S.; Reichling, J.; Wink, M. Synergistic properties of the terpenoids aromadendrene and 1,8-cineole from the essential oil of Eucalyptus globulus against antibiotic-susceptible and antibiotic-resistant pathogens. Phytomedicine 2010, 17, 1061–1066. [Google Scholar] [CrossRef]
- Miguel, M.G.; Cruz, C.; Faleiro, L.; Simões, M.T.F.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Foeniculum vulgare essential oils: Chemical composition, antioxidant and antimicrobial activities. Nat. Prod. Commun. 2010, 5, 319–328. [Google Scholar]
- Chizzola, R.; Michitsch, H.; Franz, C. Antioxidative properties of Thymus vulgaris leaves: Comparison of different extracts and essential oil chemotypes. J. Agric. Food Chem. 2008, 56, 6897–6904. [Google Scholar]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; MacDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef]
- Laguerre, M.; Lecomte, J.; Villeneuve, P. Evaluation of the ability of antioxidants to counteract lipid oxidation: existing methods, new trends and challenges. Prog. Lipid Res. 2007, 46, 244–282. [Google Scholar] [CrossRef]
- Sacchetti, G.; Maietti, S.; Muzzoli, M.; Scaglianti, M.; Manfredini, S.; Radice, M.; Bruni, R. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem. 2005, 91, 621–632. [Google Scholar] [CrossRef]
- Ruberto, G.; Baratta, M.T. Antioxidant activity of selected essential oil components in two lipid model systems. Food Chem. 2000, 69, 167–174. [Google Scholar] [CrossRef]
- Shahat, A.A.; Ibrahim, A.Y.; Hendawy, S.F.; Omer, E.A.; Hammouda, F.M.; Abdel-Rahman, F.; Saleh, M.A. Chemical composition, antimicrobial and antioxidant activities of essential oils from organically cultivated fennel cultivars. Molecules 2011, 16, 1366–1377. [Google Scholar] [CrossRef]
- Miguel, G.; Simões, M.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G.; Carvalho, L. Composition and antioxidant activities of the essential oils of Thymus caespititius, Thymus camphoratus and Thymus mastichina. Food Chem. 2004, 86, 183–188. [Google Scholar]
- Soares, J.R.; Dinis, T.C.; Cunha, A.P.; Almeida, L.M. Antioxidant activities of some extracts of Thymus zygis. Free Radic. Res. 1997, 26, 469–478. [Google Scholar] [CrossRef]
- Kulisic, T.; Radonic, A.; Milos, M. Antioxidant properties of thyme (Thymus vulgaris L.) and wild thyme (Thymus serpyllum L.) essential oils. Ital. J. Food Sci. 2005, 17, 1–10. [Google Scholar]
- Bozin, B.; Mimica-Dukic, N.; Simin, N.; Anackov, G. Characterization of the volatile composition of essential oils of some Lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. J. Agric. Food Chem. 2006, 54, 1822–1828. [Google Scholar]
- Mastelić, J.; Jerković, I.; Blažević, I.; Poljak-Blaži, M.; Borović, S.; Ivančić-Baće, I.; Smrečki, V.; Zarcović, N.; Brčić-Kostić, K.; Vikić-Topić, D.; Müller, N. Comparative study on the antioxidant and biological activities of carvacrol, thymol, and eugenol derivatives. J. Agric. Food Chem. 2008, 56, 3989–1996. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Frankel, E.N. Chemistry of free radical and singlet oxidation of lipids. Prog. Lipid Res. 1985, 23, 197–221. [Google Scholar] [CrossRef]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 1990, 9, 515–540. [Google Scholar] [CrossRef]
- Bentes, J.; Miguel, M.G.; Monteiro, I.; Costa, M.; Figueiredo, A.C.; Barroso, J.G.; Pedro, L.G. Antioxidant activities of the essential oils and extracts of Portuguese Thymbra capitata and Thymus mastichina. Ital. J. Food Sci. 2009, 21, 183–195. [Google Scholar]
- Rothenberg, G.; Yatziv, Y.; Sasson, Y. Comparative autoxidation of 3-carene and α-pinene: factors governing regioselective hydrogen abstraction reactions. Tetrahedron 1998, 54, 593–598. [Google Scholar] [CrossRef]
- Arbuzov, B.A.; Isaeva, Z.G.; Ibragimova, N.D. Oxidation of 3-carene with oxygen in presence of chromic anhydride. Russ. Chem. Bull. 1961, 11, 599–606. [Google Scholar]
- López, M.D.; Pascual-Villalobos, M.J. Mode of inhibition of acetylcholinesterase by monoterpenoids and implications for pest control. Ind. Crop Prod. 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Kumar, V.; Mal, M.; Houghton, P.J. Acetylcholinesterase inhibitors from plants. Phytomedicine 2007, 14, 289–300. [Google Scholar] [CrossRef]
- Savelev, S.; Okello, E.; Perry, N.S.L.; Wilkins, R.M.; Perry, E.K. Synergistic and antagonistic interactions of anticholinesterase terpenoids in Salvia lavandulaefolia essential oil. Pharmacol. Biochem. Behav. 2003, 75, 661–668. [Google Scholar] [CrossRef]
- Picollo, M.I.; Toloza, A.C.; Cueto, G.M.; Zygadlo, J.; Zerba, E. Anticholinesterase and pediculicidal activities of monoterpenoids. Fitoterapia 2008, 79, 271–278. [Google Scholar] [CrossRef]
- Menichini, F.; Tundis, R.; Loizzo, M.R.; Bonesi, M.; Marreli, M.; Statti, G.A.; Menichini, F.; Conforti, F. Acetylcholinesterase and butyrylcholinesterase inhibition of ethanolic extract and monoterpenes from Pimpinella anisoides V Brig. (Apiaceae). Fitoterapia 2009, 80, 297–300. [Google Scholar] [CrossRef]
- Miyazawa, M.; Yamafuji, C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J. Agric. Food Chem. 2005, 53, 1765–1768. [Google Scholar] [CrossRef]
- Jukic, M.; Politeo, O.; Maksimovic, M.; Milos, M.; Milos, M. In vitro acetylcholinesterase inhibitory properties of thymol, carvacrol and their derivatives thymoquinone and thymohydroquinone. Phytother. Res. 2007, 21, 259–261. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Valentino, A., Jr.; Feathertone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar]
- Hostettmann, K.; Borloz, A.; Urbain, A.; Marston, A. Natural product inhibitors of acetylcholinesterase. Curr. Org. Chem. 2006, 10, 825–847. [Google Scholar] [CrossRef]
- Samples Availability: Samples of the compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Aazza, S.; Lyoussi, B.; Miguel, M.G. Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds. Molecules 2011, 16, 7672-7690. https://doi.org/10.3390/molecules16097672
Aazza S, Lyoussi B, Miguel MG. Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds. Molecules. 2011; 16(9):7672-7690. https://doi.org/10.3390/molecules16097672
Chicago/Turabian StyleAazza, Smail, Badiâ Lyoussi, and Maria G. Miguel. 2011. "Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds" Molecules 16, no. 9: 7672-7690. https://doi.org/10.3390/molecules16097672
APA StyleAazza, S., Lyoussi, B., & Miguel, M. G. (2011). Antioxidant and Antiacetylcholinesterase Activities of Some Commercial Essential Oils and Their Major Compounds. Molecules, 16(9), 7672-7690. https://doi.org/10.3390/molecules16097672





