Synthesis of Some Oxadiazole Derivatives as New Anticandidal Agents
Abstract
:1. Introduction
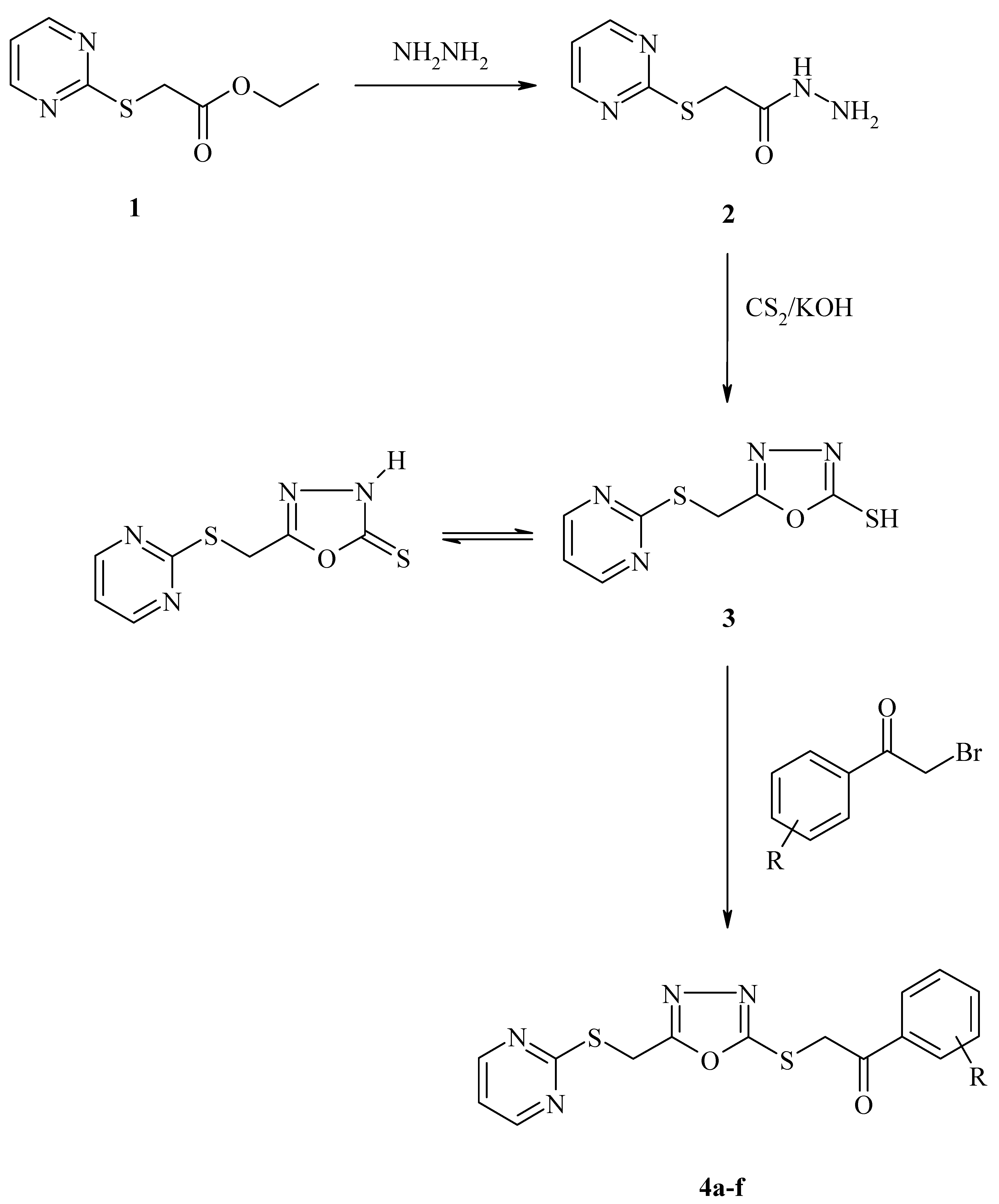
2. Results and Discussion
| Compound | R | Yield (%) | M.p. (°C) | Molecular formula | Molecular weight |
|---|---|---|---|---|---|
| 4a | H | 75 | 112-114 | C15H12N4O2S2 | 344 |
| 4b | p-Cl | 85 | 106-108 | C15H11ClN4O2S2 | 378,5 |
| 4c | p-NO2 | 95 | 145-149 | C15H11N5O4S2 | 389 |
| 4d | m-NO2 | 92 | 79-80 | C15H11N5O4S2 | 389 |
| 4e | m-Cl | 84 | 89-90 | C15H11ClN4O2S2 | 378,5 |
| 4f | 2,4-Cl | 88 | 75-77 | C15H10Cl2N4O2S2 | 413 |
| 4a | 4b | 4c | 4d | 4e | 4f | Ref. | |
|---|---|---|---|---|---|---|---|
| A | 0.015 | 0.015 | 0.06 | 0.125 | 0.015 | 0.03 | 0.007 |
| B | 0.03 | 0.03 | 0.03 | 0.06 | 0.03 | 0.06 | 0.007 |
| C | 0.03 | 0.125 | 0.06 | 0.06 | 0.125 | 0.06 | 0.003 |
| D | 0.015 | 0.015 | 0.015 | 0.06 | 0.06 | 0.06 | 0.001 |
| E | 0.007 | 0.03 | 0.015 | 0.125 | 0.03 | 0.015 | 0.001 |
| F | 0.06 | 0.06 | 0.03 | 0.06 | 0.06 | 0.06 | 0.001 |
| G | 0.06 | 0.06 | 0.06 | 0.125 | 0.06 | 0.06 | 0.001 |
| H | 0.06 | 0.03 | 0.03 | 0.06 | 0.03 | 0.015 | 0.001 |
3. Experimental
3.1. General
3.2. General Procedure for Synthesis of the Compounds
3.3. Microbiology
3.3.1. Anticandidal Evaluation
3.3.2. Microbroth Dilution Assay
4. Conclusions
Conflict of Interest
- Samples Availability: Samples of the compounds 4a–f are available from the author.
References
- Ruan, S.Y.; Hsueh, P.R. Invasive Candidiasis: An Overview from Taiwan. J. Formos. Med. Assoc. 2009, 108, 443–451. [Google Scholar] [CrossRef]
- Canuto, M.M.; Rodero, F.G. Antifungal drug resistance to azoles and polyenes. Lancet Infect. Dis. 2002, 2, 550–563. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Louis, B.R. Antifungal Agents: Mode of Action, Mechanisms of Resistance and Correlation of These Mechanisms with Bacterial Resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of Invasive Candidiasis: A Persistent Public Health Problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef]
- Sheehan, D.J.; Hitchcock, C.A.; Sibley, C.M. Current and emerging azole antifungal agents. Clin. Microbiol. Rev. 1999, 12, 40–79. [Google Scholar]
- Jiang, L.; Tan, Y.; Zhu, X.; Wang, Z.; Zuo, Y.; Chen, Q.; Xi, Z.; Yang, G. Design, Synthesis, and 3D-QSAR Analysis of Novel 1,3,4-Oxadiazol-2(3H)-ones as Protoporphyrinogen Oxidase Inhibitors. J. Agric. Food Chem. 2010, 58, 2643–2651. [Google Scholar]
- Mayekar, A.N.; Yathirajan, H.S.; Narayana, B.; Sarojini, B.K.; Suchetha Kumari, N. Synthesis and Antimicrobial Studies on New Substituted 1,3,4-Oxadiazole Derivatives Bearing 6-Bromonaphthalene Moiety. Int. J. Chem. 2010, 2, 38–54. [Google Scholar]
- Farshori, N.N.; Banday, M.R.; Ahmad, A.; Khan, A.U.; Rauf, A. Synthesis, characterization, and in vitro antimicrobial activities of 5-alkenyl/hydroxyalkenyl-2-phenylamine-1,3,4-oxadiazoles and thiadiazoles. Bioorg. Med. Chem. Lett. 2010, 20, 1933–1938. [Google Scholar] [CrossRef]
- Chikhalia, K.H.; Vashi, D.B.; Patel, M.J. Synthesis of a novel class of some 1,3,4-oxadiazole derivatives as antimicrobial agents. J. Enzym. Inhib. Med. Chem. 2009, 24, 617–622. [Google Scholar] [CrossRef]
- Karabasanagouda, T.; Adhikari, A.V.; Shetty, N.S. Synthesis and Antimicrobial Activities of Some novel 1,3,4-Oxadiazoles Carrying Alkylthio and Alkylsulphonylphenoxy Moieties. Phosphor. Sulfur Silicon 2007, 182, 2925–2941. [Google Scholar] [CrossRef]
- Bakht, M.A.; Shahar Yar, M.; Abdel-Hamid, S.G.; Al Qasoumi, S.I.; Samad, A. Molecular properties prediction, synthesis and antimicrobial activity of some newer oxadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5862–5869. [Google Scholar] [CrossRef]
- Bala, S.; Kamboj, S.; Kumar, A. Heterocyclic 1,3,4-oxadiazole compounds with diverse biological activities: A comprehensive review. J. Pharm. Res. 2010, 3, 2993–2997. [Google Scholar]
- Padmavathi, V.; Sudhakar Reddy, G.; Padmaja, A.; Kondaiah, P.; Shazia, A. Synthesis, antimicrobial and cytotoxic activities of 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazoles. Eur. J. Med. Chem. 2009, 44, 2106–2112. [Google Scholar]
- Dolman, S.J.; Gosselin, F.; O’Shea, P.D.; Davies, I.W. Superior Reactivity of Thiosemicarbazides in the Synthesis of 2-Amino-1,3,4-oxadiazoles. J. Org. Chem. 2006, 71, 9548–9551. [Google Scholar] [CrossRef]
- Horning, D.E.; Muchowski, J.M. Five-membered Heterocyclic Thiones. Part I. 1,3,4-Oxadiazole-2-thione. Can. J. Chem. 1972, 50, 3079–3082. [Google Scholar] [CrossRef]
- Mekuskiene, G.; Burbuliene, M.M.; Jakubkiene, V.; Udrenaite, E.; Gaidelis, P.; Vainilavicius, P. 5-(4,6-diphenyl-2-pyrimidinyl)-1,3,4-oxadiazole-2-thione with some C-electrophiles and N nucleophiles. Chem. Heterocycl. Comp. 2003, 39, 1364–1368. [Google Scholar]
- Kalluraya, B.; Lingappa, B.; Nooji, S.R.; Kumari, N.S. Regioselective reaction: synthesis of novel Mannich bases derived from 5-(4,6-disubstituted-2-thiomethyl pyrimidyl)-1,3,4-oxadiazole-2-thiones and their biological properties. Org. Chem. Indian J. 2006, 2, 158–164. [Google Scholar]
- Liu, T.T.; Lee, R.E.B.; Barker, K.S.; Lee, R.E.; Wei, L.; Homayouni, R.; Rogers, P.D. Genome-wide expression profiling of the response to azole, polyene, echinocandin, and pyrimidine antifungal agents in Candida albicans. Antimicrob. Agents Ch. 2005, 49, 2226–2236. [Google Scholar] [CrossRef]
- Shadomy, S.; Kirchoff, C.B.; Ingroff, A.E. In vitro activity of 5-fluorocytosine against Candida and Torulopsis species. Antimicrob. Agents Ch. 1973, 3, 9–14. [Google Scholar] [CrossRef]
- Gülerman, N.N.; Doğan, H.N.; Rollas, S.; Johansson, C.; Çelik, C. Synthesis and structure elucidation of some new thioether derivatives of 1,2,4-triazoline-3-thiones and their antimicrobial activities. Farmaco 2001, 56, 953–958. [Google Scholar] [CrossRef]
- Dhayanithi, V.; Syed, S.S.; Kumaran, K.; Sankar, K.R.J.; Ragavan, R.V.; Goud, P.S.K.; Kumari, N.S.; Pati, H.N. Synthesis of selected 5-thio-substituted tetrazole derivatives and evaluation of their antibacterial and antifungal activities. J. Serb. Chem. Soc. 2011, 76, 165–175. [Google Scholar] [CrossRef]
- Beggs, W.H. Influence of growth phase on the susceptibility of Candida albicans to butoconazole, oxiconazole, and sulconazole. J. Antimicrob. Chemoth. 1985, 16, 397–399. [Google Scholar] [CrossRef]
- Koneman, E.W.; Allen, S.D.; Janda, W.M.; Schreckenberger, P.C.; Winn, W.C. Color Atlas and Textbook of Diagnostic Microbiology, 5th ed; Lippincott-Raven: Philadelphia, PA, USA, 1997; pp. 785–856. [Google Scholar]
- Özdemir, A.; Turan-Zitouni, G.; Kaplancıklı, Z.A.; İşcan, G.; Khan, S.; Demirci, F. Synthesis and the selective antifungal activity of 5,6,7,8-tetrahydroimidazo[1,2-a]pyridine derivatives. Eur. J. Med. Chem. 2010, 45, 2080–2084. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kaplancikli, Z.A. Synthesis of Some Oxadiazole Derivatives as New Anticandidal Agents. Molecules 2011, 16, 7662-7671. https://doi.org/10.3390/molecules16097662
Kaplancikli ZA. Synthesis of Some Oxadiazole Derivatives as New Anticandidal Agents. Molecules. 2011; 16(9):7662-7671. https://doi.org/10.3390/molecules16097662
Chicago/Turabian StyleKaplancikli, Zafer Asim. 2011. "Synthesis of Some Oxadiazole Derivatives as New Anticandidal Agents" Molecules 16, no. 9: 7662-7671. https://doi.org/10.3390/molecules16097662
APA StyleKaplancikli, Z. A. (2011). Synthesis of Some Oxadiazole Derivatives as New Anticandidal Agents. Molecules, 16(9), 7662-7671. https://doi.org/10.3390/molecules16097662





