Proanthocyanidins from Grape Seeds Modulate the NF-κB Signal Transduction Pathways in Rats with TNBS-Induced Ulcerative Colitis
Abstract
:1. Introduction
2. Results and Discussion
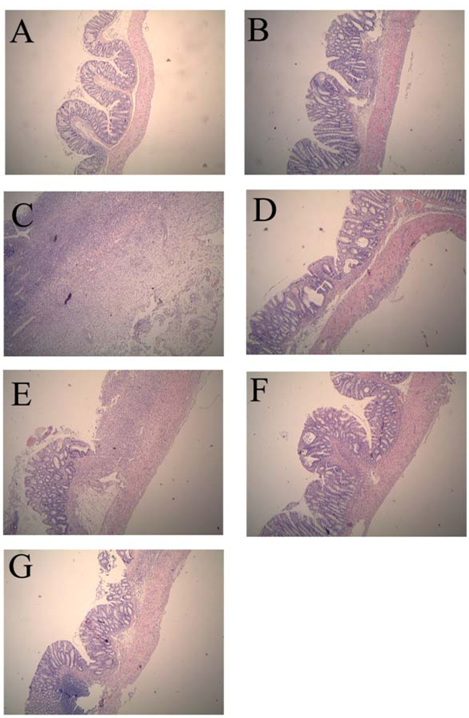
2.1. Colonic Microscopic Damage
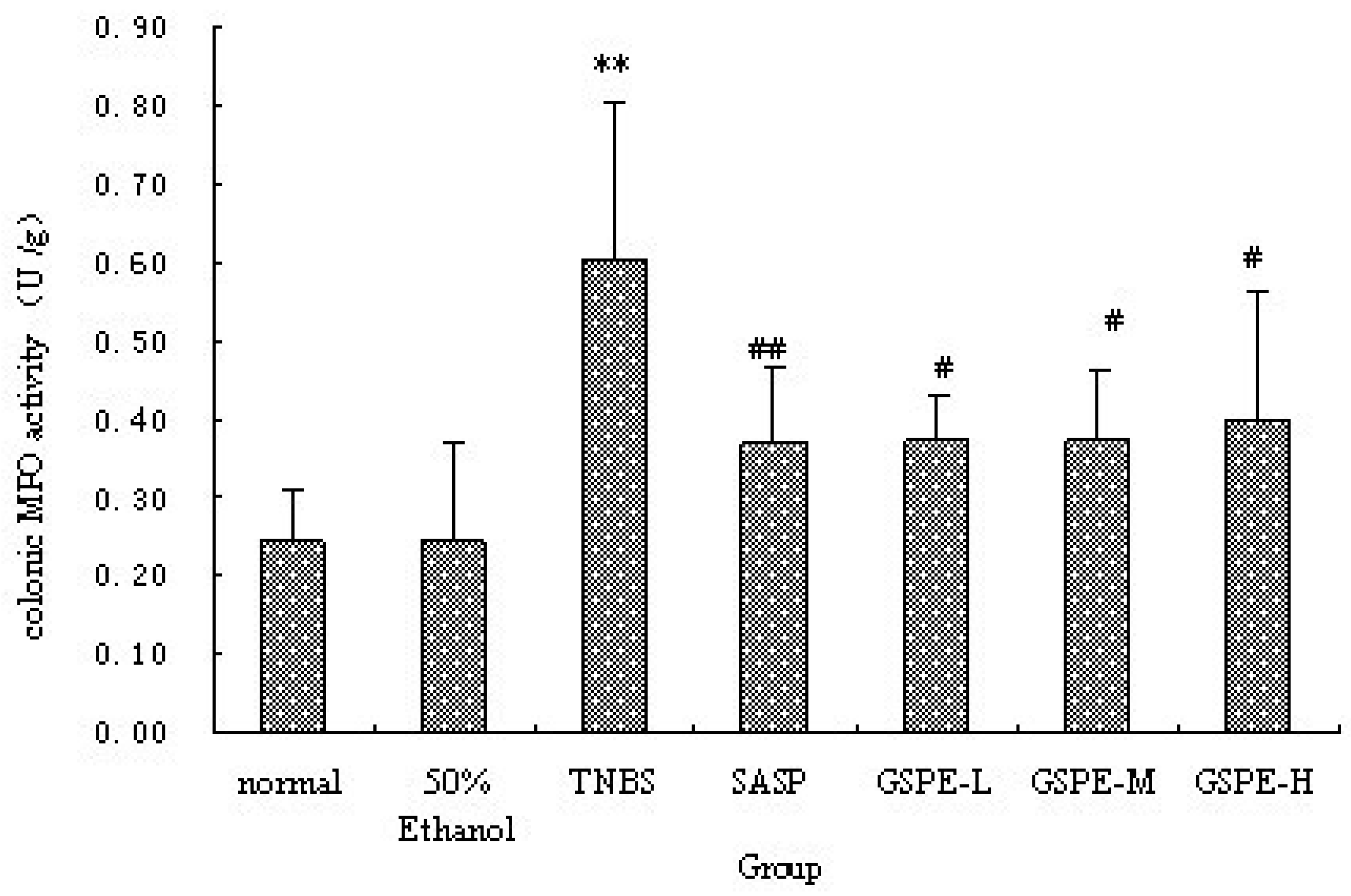
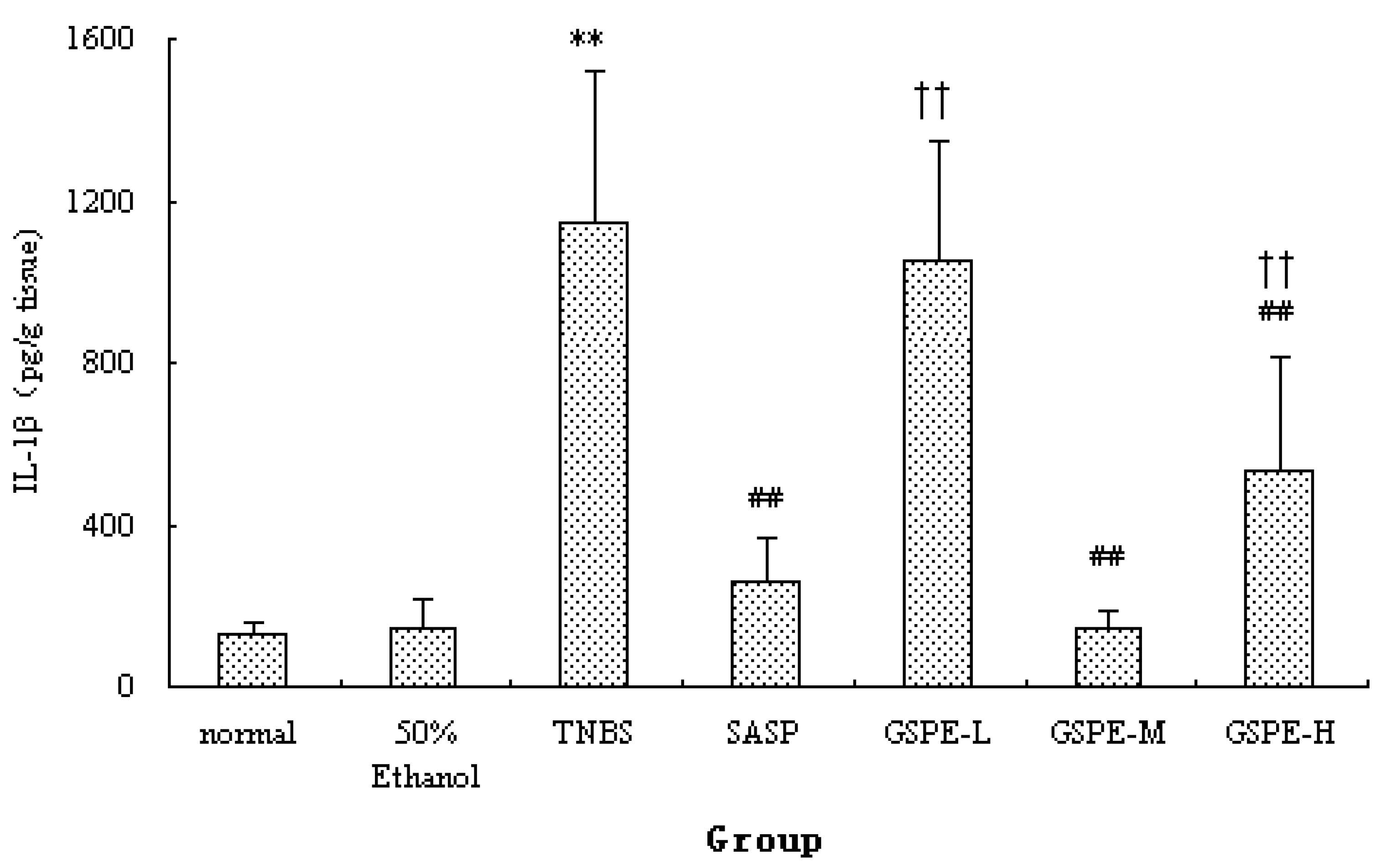
2.2. MPO Activity and IL-1β Levels in Colon
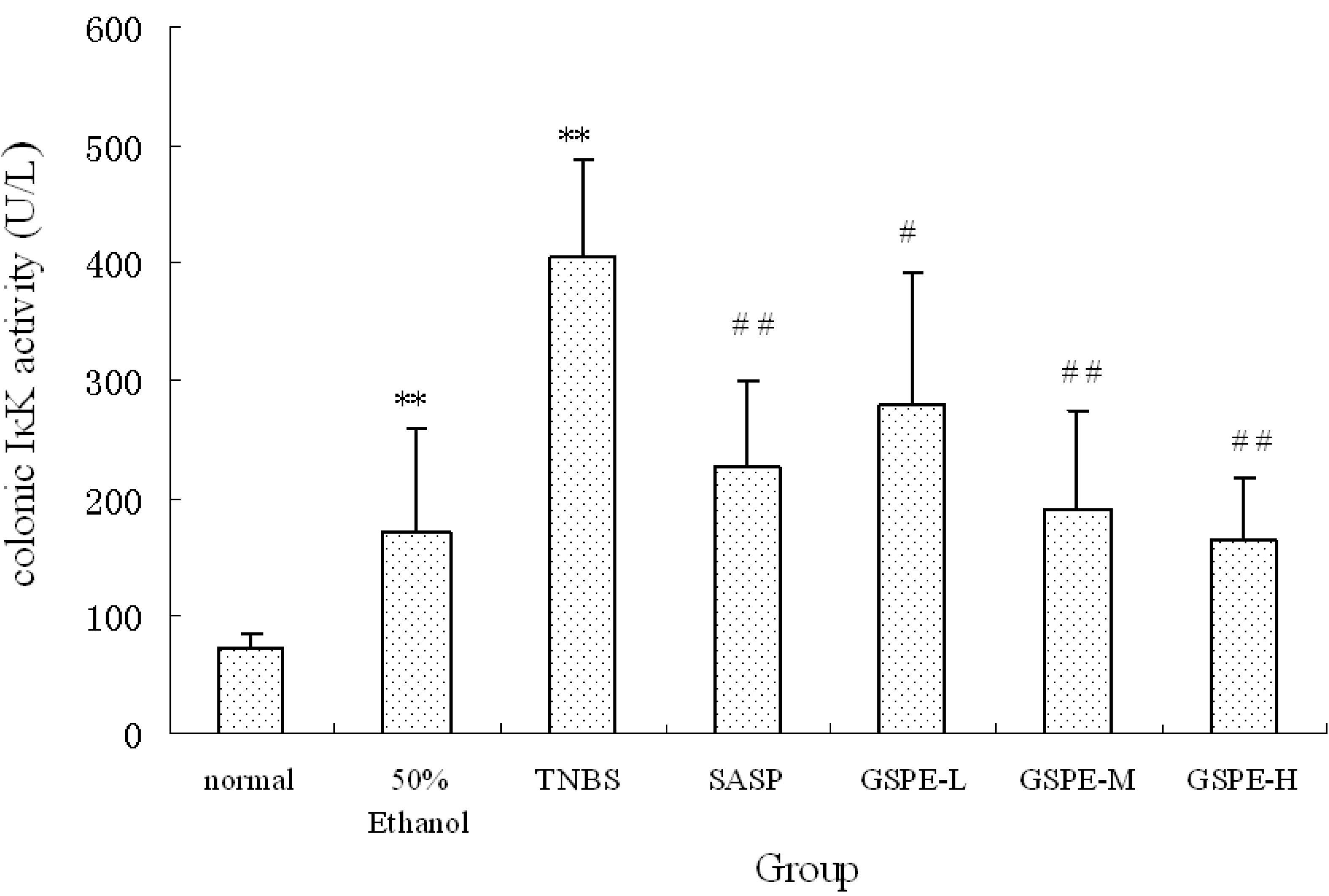
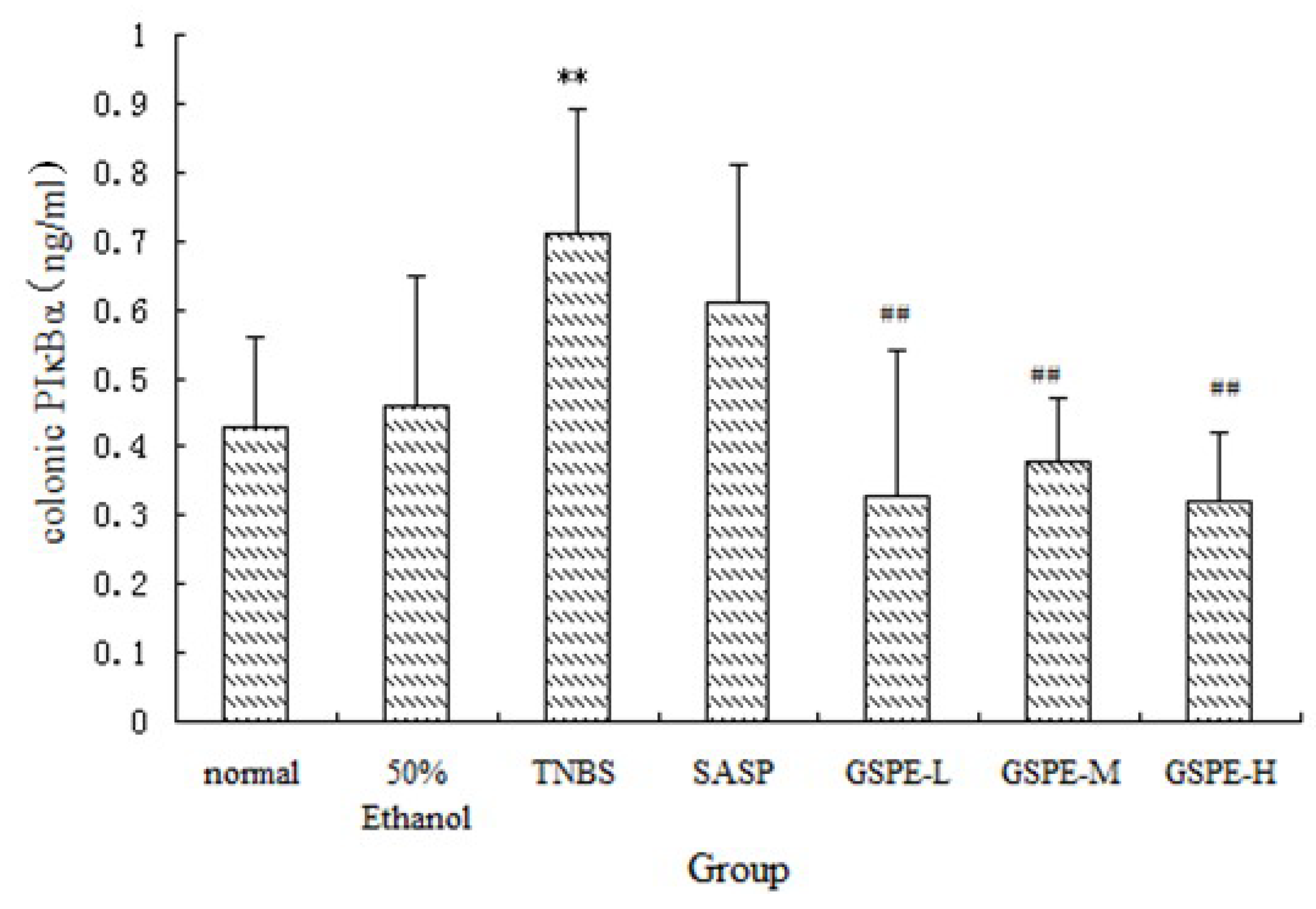
2.3. The Expression of Colonic IκK and PIκBα
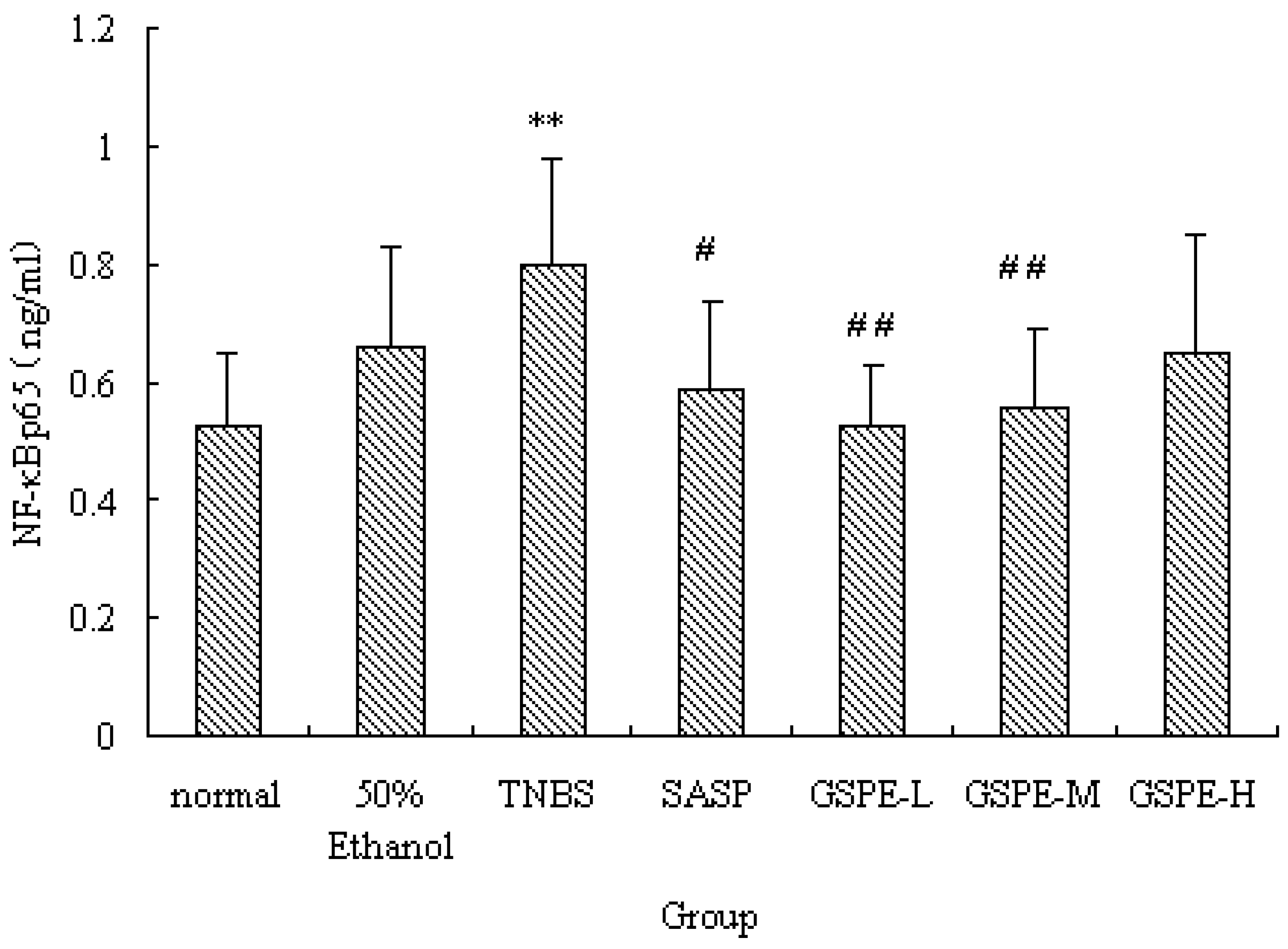
2.4. The Expression of Colonic NF-κB
2.5. Discussion
3. Experimental
3.1. Animals
3.2. Drug and Reagents
3.3. Drug and Agent Preparation
3.4. Induction of Experimental UC and Treatment Protocols
3.5. Induction of UC
3.6. Treatment
3.7. Assessment of Colonic Damage and Acquisition of Specimens
3.8. Preparation of Nuclear and Cytosolic Fractions
3.9. Statistical Analysis
4. Conclusions
Conflicts of interest
Acknowledgements
References and Notes
- Deguchi, Y.; Andoh, A.; Inatomi, O.; Yagi, Y.; Bamba, S.; Araki, Y.; Hata, K.; Tsujikawa, T.; Fujiyama, Y. Curcumin prevents the development of dextran sulfate sodium (DSS)-induced experimental colitis. Dig.Dis.Sci. 2007, 52, 2993–2998. [Google Scholar] [CrossRef]
- Podolsky, D.K. Inflammatory bowel disease. N.Engl.J.Med. 2002, 347, 417–429. [Google Scholar] [CrossRef]
- Liu, C.J.; Jin, J.D.; Lv, T.D.; Wu, Z.Z.; Ha, X.Q. Keratinocyte growth factor gene therapy ameliorates ulcerative colitis in rats. World J. Gastroenterol. 2011, 17, 2632–2640. [Google Scholar] [CrossRef]
- Gálvez-Llompart, M.; Recio, M.C.; García-Domenech, R. Topological virtual screening: A way to find new compounds active in ulcerative colitis by inhibiting NF-κB. Mol Divers. 2011. [Google Scholar] [CrossRef]
- Bayeta, E.; Lau, B. Pycnogenol inhibits generation of inflammatory Mediators in macrophages. Nutr. Res. 2000, 20, 249–259. [Google Scholar] [CrossRef]
- Li, W.G.; Zhang, X.Y.; Wu, Y.J. Anti-inflammatory effect and mechanism of proanthocyanidin from grape seeds. Acta Pharmacol. Sin. 2001, 22, 1117–1120. [Google Scholar]
- Li, X.L.; Cai, Y.Q.; Qin, H.; Wu, Y.J. Therapeutic effect and mechanism of proanthocyanidins from grape seeds in rats with TNBS-induced ulcerative colitis. Can. J. Physiol. Pharmacol. 2008, 86, 841–849. [Google Scholar] [CrossRef]
- Wang, Y.H.; Yang, X.L.; Wang, L.; Cui, M.X.; Cai, Y.Q.; Li, X.L.; Wu, Y.J. Effects of Proanthocyanidins from grape seeds on treatment of recurrent ulcerative colitis in rats. Can. J. Physiol. Pharmacol. 2010, 88, 888–898. [Google Scholar] [CrossRef]
- Michael, K. How NF-kB is activated: The role of the IkB kinase (IKK) complex. Oncogene 1999, 18, 6867–6874. [Google Scholar] [CrossRef]
- Baldwin, A.S., Jr. The NF-kappa B and I kappa B proteins: New discoveries and insights. Annu. Rev. Immunol. 1996, 14, 649–683. [Google Scholar] [CrossRef]
- May, M.J.; Ghosh, S. Signal transduction through NF-kappa B. Immunol. Today 1998, 19, 80–88. [Google Scholar] [CrossRef]
- Carrier, J.C.; Aghdassi, E.; Jeejeebhoy, K.; Allard, J.P. Exacerbation of dextran sulfate sodium-induced colitis by dietary iron supplementation: role of NF-κB. Int. J. Colorectal. Dis. 2006, 21, 381–387. [Google Scholar] [CrossRef]
- Lenardo, M.J.; Baltimore, D. NF-kappa B: A pleiotropic mediator of inducible and tissue-specific gene control. Cell 1989, 58, 227–229. [Google Scholar] [CrossRef]
- Lee, S.J.; Lim, K.T. Chemopreventive effect of plant originated glycoprotein on colitis-mediated colorectal cancer in A/J mice. J. Biomed. Sci. 2008, 15, 111–121. [Google Scholar]
- Churg, A.; Wright, J.L. Airway wall remodeling induced by occupational mineral dusts and air pollutant particles. Chest 2002, 122, 306–309. [Google Scholar] [CrossRef]
- Oldenburg, B.; van Berge Henegouwen, G.P.; Rennick, D.; van Asbeck, B.S.; Koningsberger, J.C. Iron supplementation affects the production of proinflammatory cytokines in IL-10 deficient mice. Eur. J. Clin. Investig. 2000, 30, 505–510. [Google Scholar] [CrossRef]
- Tsukamoto, H.; Lin, M.; Ohata, M.; Giulivi, C.; French, S.W.; Brittenham, G. Iron primes hepatic macrophages for NF-kappa B activation in alcoholic liver injury. Am. J. Physiol. 1999, 277, G1240–G1250. [Google Scholar]
- Morris, G.P.; Beck, P.L.; Herridge, M.S.; Depew, W.T.; Szewczuk, M.R.; Wallace, J.L. Hapten induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology 1989, 96, 7952–8111. [Google Scholar]
- Sample Availability: Samples of the compounds of proanthocyanidins from grape seeds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, X.; Yang, X.; Cai, Y.; Qin, H.; Wang, L.; Wang, Y.; Huang, Y.; Wang, X.; Yan, S.; Wang, L.; et al. Proanthocyanidins from Grape Seeds Modulate the NF-κB Signal Transduction Pathways in Rats with TNBS-Induced Ulcerative Colitis. Molecules 2011, 16, 6721-6731. https://doi.org/10.3390/molecules16086721
Li X, Yang X, Cai Y, Qin H, Wang L, Wang Y, Huang Y, Wang X, Yan S, Wang L, et al. Proanthocyanidins from Grape Seeds Modulate the NF-κB Signal Transduction Pathways in Rats with TNBS-Induced Ulcerative Colitis. Molecules. 2011; 16(8):6721-6731. https://doi.org/10.3390/molecules16086721
Chicago/Turabian StyleLi, Xiaoli, Xiaolai Yang, Yongqing Cai, Hong Qin, Li Wang, Yanhong Wang, Yanhui Huang, Xiaoxia Wang, Shuai Yan, Liping Wang, and et al. 2011. "Proanthocyanidins from Grape Seeds Modulate the NF-κB Signal Transduction Pathways in Rats with TNBS-Induced Ulcerative Colitis" Molecules 16, no. 8: 6721-6731. https://doi.org/10.3390/molecules16086721
APA StyleLi, X., Yang, X., Cai, Y., Qin, H., Wang, L., Wang, Y., Huang, Y., Wang, X., Yan, S., Wang, L., Zhao, X., Li, W., Li, S., Chen, J., & Wu, Y. (2011). Proanthocyanidins from Grape Seeds Modulate the NF-κB Signal Transduction Pathways in Rats with TNBS-Induced Ulcerative Colitis. Molecules, 16(8), 6721-6731. https://doi.org/10.3390/molecules16086721



