Effects of Lipoic Acid, Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines
Abstract
:1. Introduction
2. Results and Discussion
2.1. 2-S-lipoyl-caffeic acid synthesis
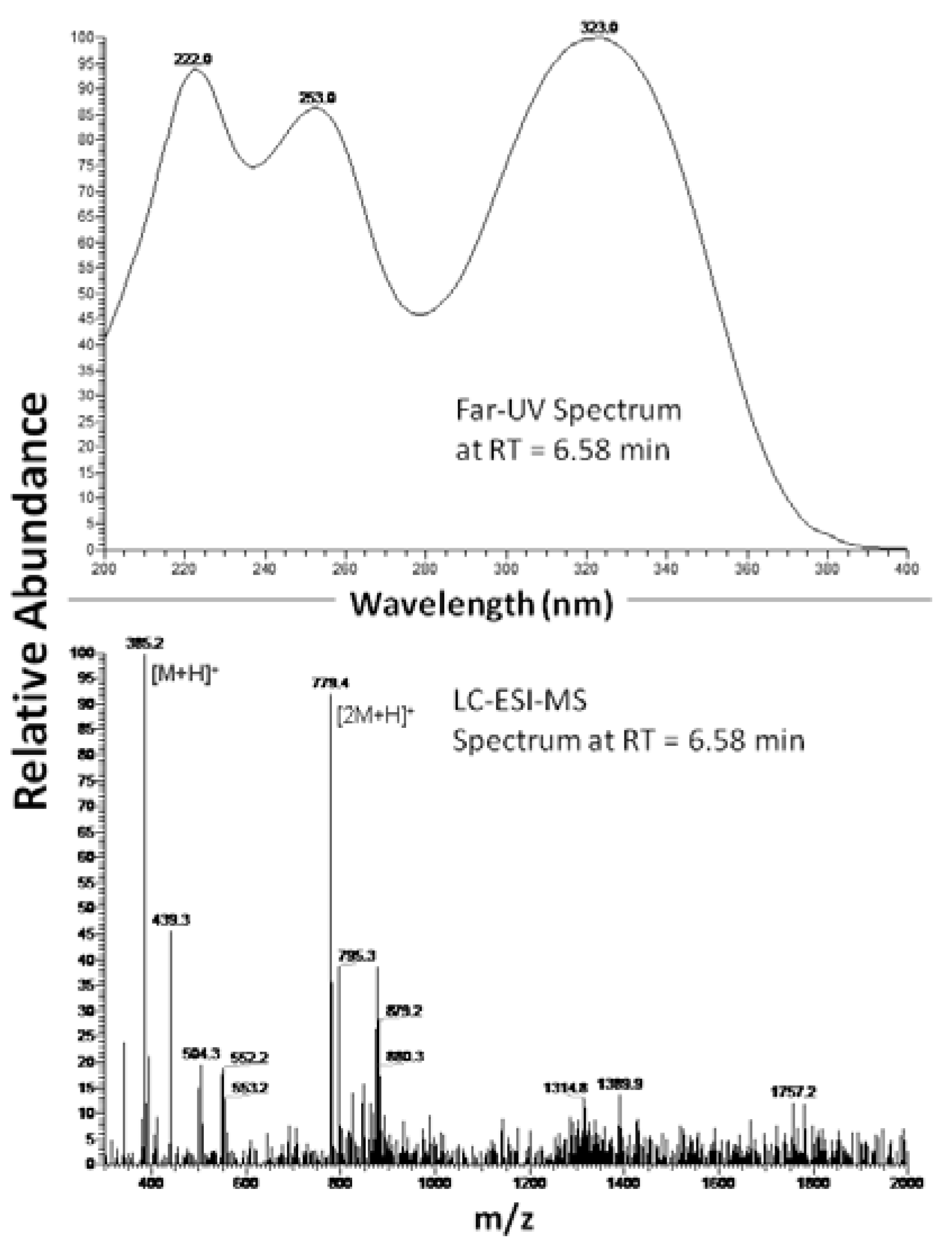
2.2. Colorimetric assay with Sulforhodamine B
2.3. Cytotoxicity assay on HepG2 cells
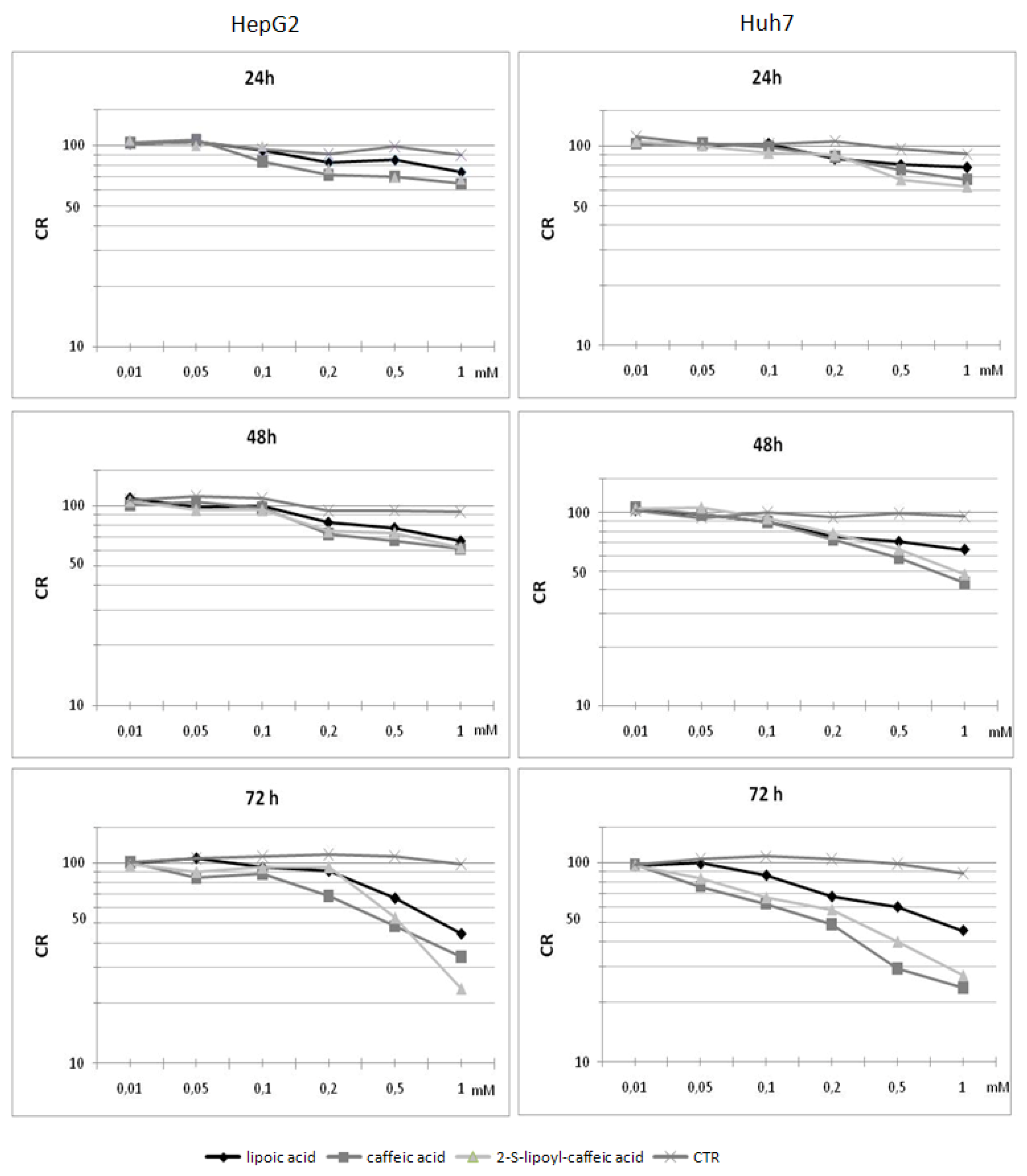
2.4. Cytotoxicity assay on Huh7 cells
2.5. Bio-Plex assay
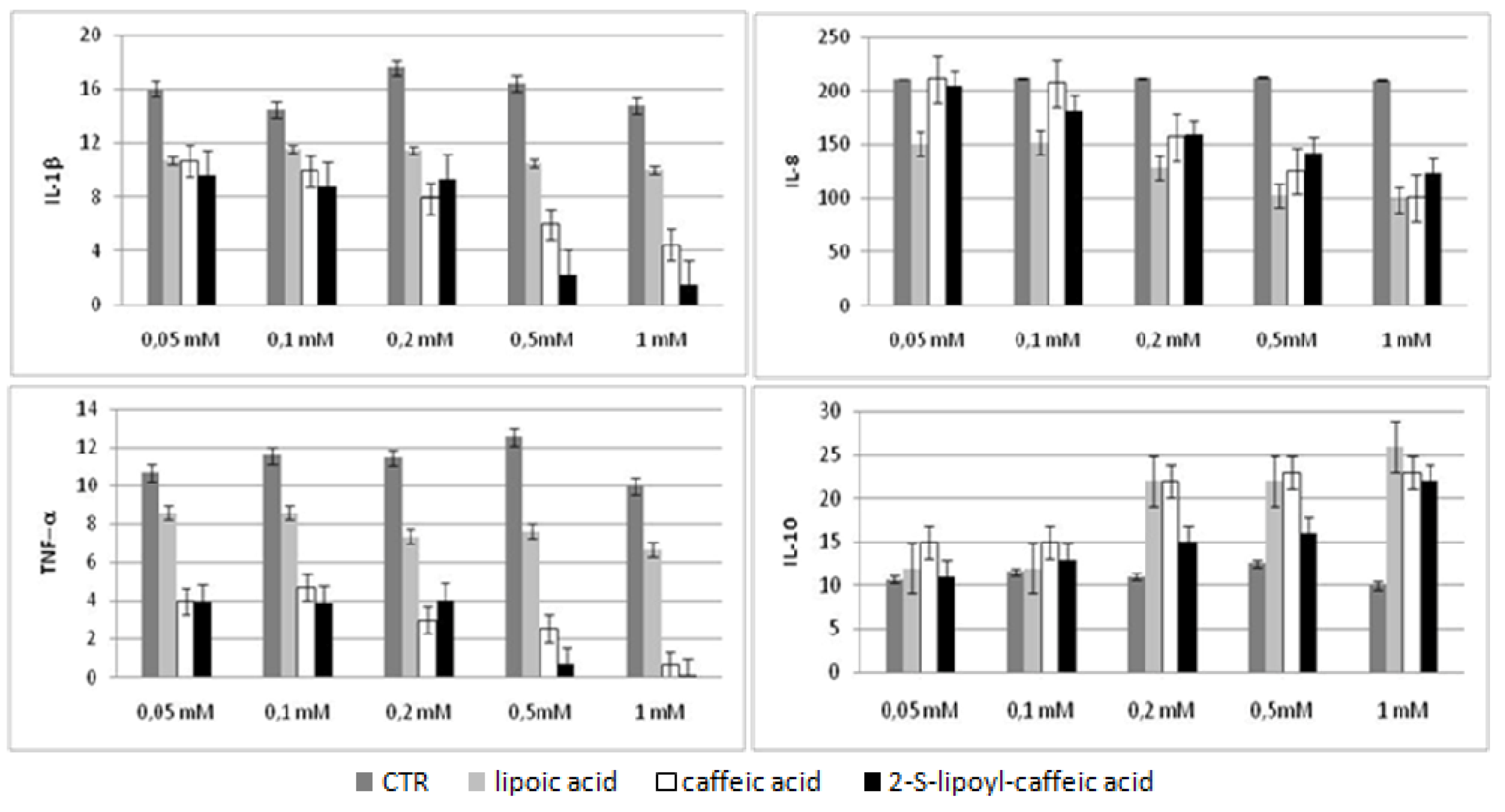
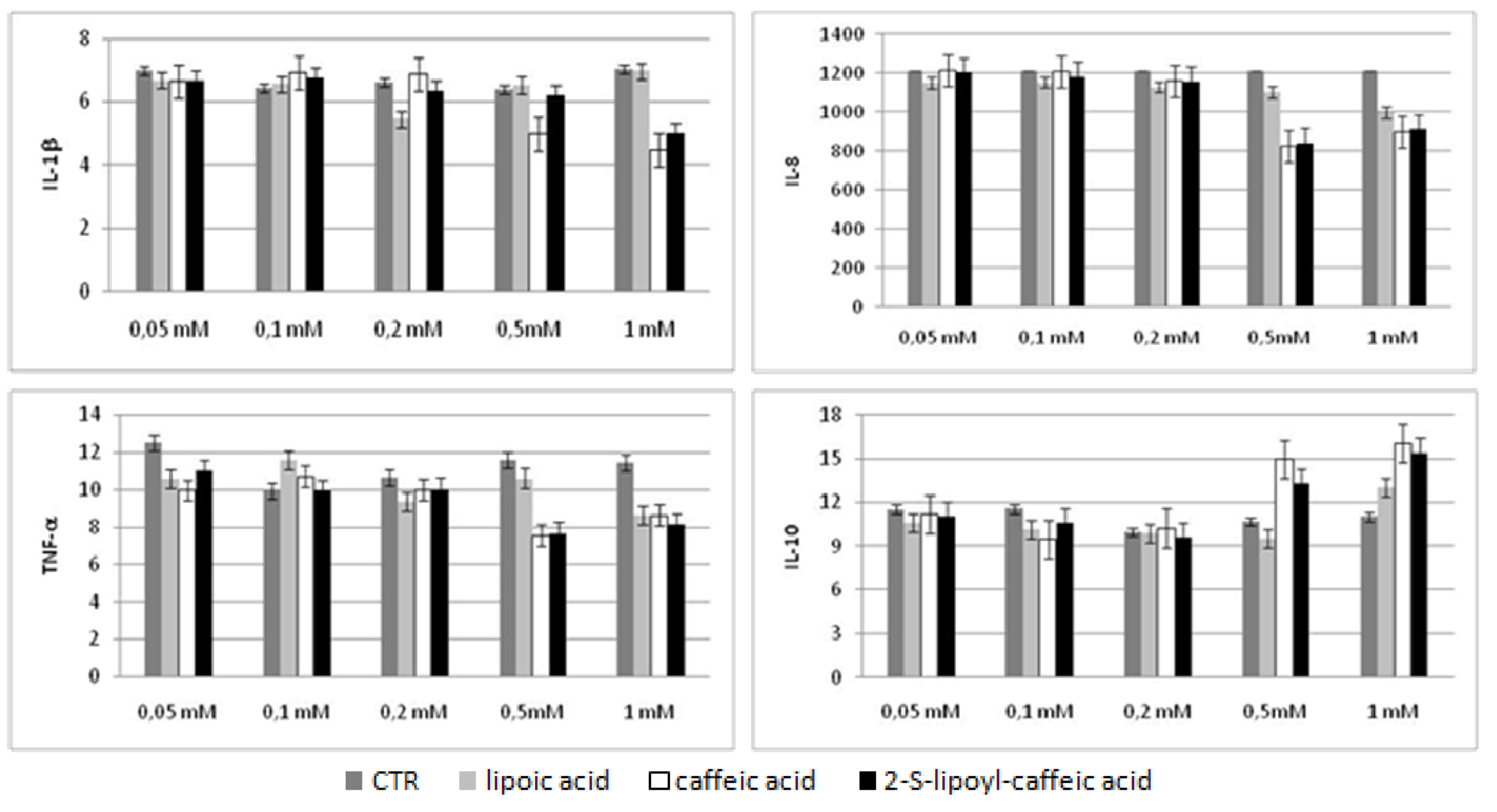
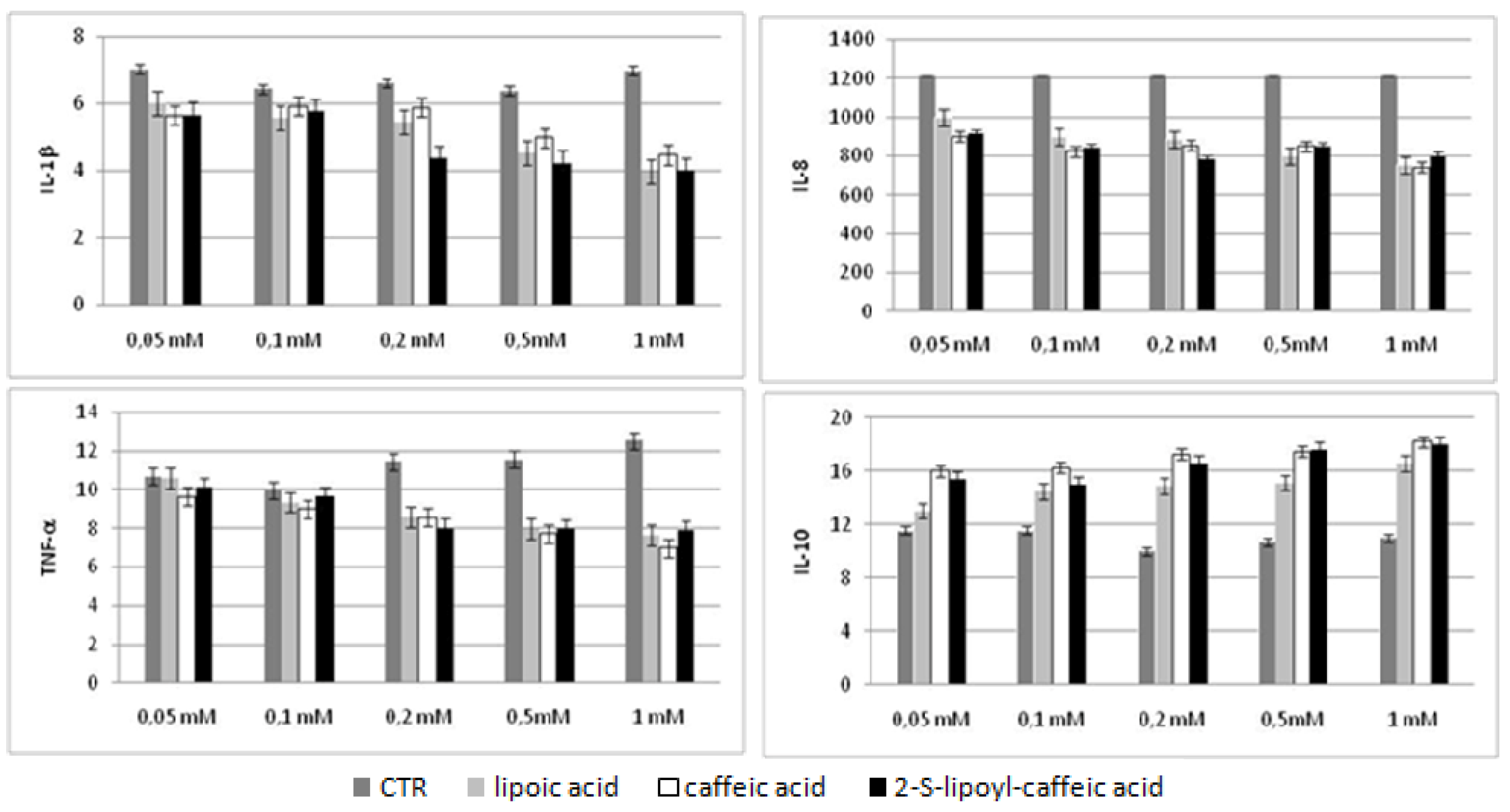
2.6. Discussion
3. Experimental
3.1. General
3.2. Cell culture
3.3. Colorimetric assay with Sulforhodamine B
3.4. Bio-Plex assay
3.5. Statistical analysis
4. Conclusions
Supplementary Materials
Supplementary File 1Conflict of Interest
References
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics, 2002. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar]
- Bosch, F.X.; Ribes, J.; Diaz, M.; Cleries, R. Primary liver cancer: worldwild incidence and trends. Gastroenterology 2004, 127, 5–16. [Google Scholar]
- Edmondson, H.A.; Henderson, B.; Benton, B. Liver-cell adenomas associated with use of oral contraceptives. N. Engl. J. Med. 1976, 294, 470–472. [Google Scholar] [CrossRef]
- McGlynn, K.A.; London, W.T. Epidemiology and natural history of hepatocellular carcinoma. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 3–23. [Google Scholar] [CrossRef]
- Bruix, J.; Sherman, M. Diagnosis of small HCC. Gastroenterology 2005, 129, 1364. [Google Scholar] [CrossRef]
- Stanner, S.A.; Hughes, J.; Kelly, C.N.; Buttriss, J. A review of the epidemiological evidence for the 'antioxidant hypothesis'. Public Health Nutr. 2004, 7, 407–422. [Google Scholar]
- Halliwell, B. Free radicals and antioxidants -quo vadis? Trends Pharmacol. Sci. 2011, 32, 125–130. [Google Scholar] [CrossRef]
- Harman, D. Free-radical theory of aging. Increasing the functional life span. Ann. N.Y. Acad. Sci. 1994, 717, 1–15. [Google Scholar] [CrossRef]
- Simonian, N.A.; Coyle, J.T. Oxidative stress in neurodegenerative diseases. Annu. Rev. Pharmacol.Toxicol. 1996, 36, 83–106. [Google Scholar] [CrossRef]
- Stoner, G.D.; Mukhtar, H. Polyphenols as cancer chemopreventive agents. J. Cell. Biochem. Suppl. 1995, 22, 169–180. [Google Scholar] [CrossRef]
- Reed, L.J.; DeBusk, B.G.; Gunsalus, I.C.; Hornberger, C.S., Jr. Crystalline α-lipoic acid: A catalitic agent associated with pyruvate dehydrogenase. Science 1951, 114, 93–94. [Google Scholar]
- Packer, L.; Witt, E.H.; Tritschler, H.J. alpha-Lipoic acid as a biological antioxidant. Free Radic. Biol. Med. 1995, 19, 227–250. [Google Scholar] [CrossRef]
- Bast, A.; Haenen, G.R. Lipoic acid: a multifunctional antioxidant. Biofactors 2003, 17, 207–13. [Google Scholar] [CrossRef]
- Farah, A.; Monteiro, M.; Donangelo, C.M.; Lafay, S. Chlorogenic acids from green coffee extract are highly bioavailable in humans. J. Nutr. 2008, 138, 2309–2315. [Google Scholar] [CrossRef]
- Sato, Y.; Itagaki, S.; Kurokawa, T.; Ogura, J.; Kobayashi, M.; Hirano, T.; Sugawara, M.; Iseki, K. In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int. J. Pharm. 2011, 17, 403:136–403:138. [Google Scholar]
- Gailit, J. Restoring free sulfhydryl groups in synthetic peptides. Anal. Biochem. 1993, 214, 334–335. [Google Scholar]
- Fulcrand, H.; Cheminat, A.; Brouillard, R.; Cheynier, V. Characterization of compounds obtained by chemical oxidation of caffeic acid in acidic condition. Phytochemistry 1994, 35, 499–505. [Google Scholar]
- Bassil, D.; Makris, D.P.; Kefalas, P. Oxidation of caffeic acid in the presence of L-cysteine: isolation of 2-S-cysteinylcaffeic acid and evaluation of its antioxidant properties. Food Res. Int. 2005, 38, 395–402. [Google Scholar] [CrossRef]
- Cilliers, J.J.L.; Singleton, V.L. Caffeic acid Autoxidation and the effects of Thiols. J. Agric. Food Chem. 1990, 38, 1789–1796. [Google Scholar] [CrossRef]
- Simbula, G.; Columbano, A.; Ledda-Columbano, G.M.; Sanna, L.; Deidda, M.; Diana, A.; Pibiri, M. Increased ROS generation and p53 activation in α-lipoic acid-induced apoptosis of hepatoma cells. Apoptosis 2007, 12, 113–123. [Google Scholar] [CrossRef]
- Chung, T.W.; Moon, S.K.; Chang, Y.C.; Ko, J.H.; Lee, Y.C.; Cho, G.; Kim, S.H.; Kim, J.G.; Kim, C.H. Novel and therapeutic effect of caffeic acid and caffeic acid phenyl ester on hepatocarcinoma cells: Complete regression of hepatoma growth and metastasis by dual mechanism. FASEB J. 2004, 18, 1670–1681. [Google Scholar]
- Won, C.; Lee, C.S.; Lee, J.K.; Kim, T.J.; Lee, K.H.; Yang, Y.M.; Kim, Y.N.; Ye, S.K.; Chung, M.H. CADPE suppresses cyclin D1 expression in hepatocellular carcinoma by blocking IL-6-induced STAT3 activation. Anticancer Res. 2010, 30, 481–488. [Google Scholar]
- Parekh, P.; Rao, K.V. Overexpression of cyclin D1 is associated with elevated levels of MAP kinases, Akt and Pak1 during diethylnitrosamine-induced progressive liver carcinogenesis. Cell. Biol. Int. 2007, 31, 35–43. [Google Scholar] [CrossRef]
- Jabari, S.; Meissnitzer, M.; Quint, K.; Gahr, S.; Wissniowski, T.; Hahn, E.G.; Neureiter, D.; Ocker, M. Cellular plasticity of trans- and dedifferentiation markers in human hepatoma cells in vitro and in vivo. Int. J. Oncol. 2009, 35, 69–80. [Google Scholar]
- Hussain, S.P.; Schwank, J.; Staib, F.; Wang, X.W.; Harris, C.C. TP53 mutations and hepatocellular carcinoma: insights into the etiology and pathogenesis of liver cancer. Oncogene 2007, 26, 2166–2176. [Google Scholar] [CrossRef]
- Capone, F.; Costantini, S.; Guerriero, E.; Calemma, R.; Napolitano, M.; Scala, S.; Izzo, F.; Castello, G. Serum cytokine levels in patients with hepatocellular carcinoma. Eur. Cytokine Netw. 2010, 21, 99–104. [Google Scholar]
- Costantini, S.; Capone, F.; Guerriero, E.; Maio, P.; Colonna, G.; Castello, G. Serum cytokine levels as putative prognostic markers in the progression of chronic HCV hepatitis to cirrhosis. Eur. Cytokine Netw. 2010, 21, 251–256. [Google Scholar]
- Stonāns, I.; Stonāne, E.; Russwurm, S.; Deigner, H.P.; Böhm, K.J.; Wiederhold, M.; Jäger, L.; Reinhart, K. HepG2 human hepatoma cells express multiple cytokine genes. Cytokine 1999, 11, 151–156. [Google Scholar] [CrossRef]
- Shin, E.C.; Choi, Y.H.; Kim, J.S.; Kim, S.J.; Park, J.H. Expression patterns of cytokines and chemokines genes in human hepatoma cells. Yonsei Med. J. 2002, 43, 657–664. [Google Scholar]
- Liu, Z.G.; Han, J. Cellular responses to tumor necrosis factor. Curr. Issues Mol. Biol. 2001, 3, 79–90. [Google Scholar]
- Ataseven, H.; Bahcecioglu, I.H.; Kuzu, N.; Yalniz, M.; Celebi, S.; Erensoy, A.; Ustundag, B. The levels of ghrelin, leptin, TNF-alpha, and IL-6 in liver cirrhosis and hepatocellular carcinoma due to HBV and HDV infection. Mediators Inflamm. 2006, 2006, 78380. [Google Scholar]
- Fabregat, I. Dysregulation of apoptosis in hepatocellular carcinoma cells. World J. Gastroenterol. 2009, 15, 513–520. [Google Scholar] [CrossRef]
- Natarajan, K.; Singh, S.; Burke, T.R., Jr.; Grunberger, D.; Aggarwal, B.B. Caffeic acid phenethyl ester is a potent and specific inhibitor of activation of nuclear transcription factor NF-kappa B. Proc. Natl. Acad. Sci. USA 1996, 93, 9090–9095. [Google Scholar] [CrossRef]
- Zhang, W.J.; Frei, B. Alpha-lipoic acid inhibits TNF-alpha-induced NF-kappaB activation and adhesion molecule expression in human aortic endothelial cells. FASEB J. 2001, 15, 2423–2432. [Google Scholar] [CrossRef]
- Abdel-Latif, M.M.; Windle, H.J.; Homasany, B.S.; Sabra, K.; Kelleher, D. Caffeic acid phenethyl ester modulates Helicobacter pylori-induced nuclear factor-kappa B and activator protein-1 expression in gastric epithelial cells. Br. J. Pharmacol. 2005, 146, 1139–1147. [Google Scholar]
- Jung, W.K.; Choi, I.; Lee, D.Y.; Yea, S.S.; Choi, Y.H.; Kim, M.M.; Park, S.G.; Seo, S.K.; Lee, S.W.; Lee, C.M.; et al. Caffeic acid phenethyl ester protects mice from lethal endotoxin shock and inhibits lipopolysaccharide-induced cyclooxygenase-2 and inducible nitric oxide synthase expression in RAW 264.7 macrophages via the p38/ERK and NF-kappaB pathways. 2008, 40, 2572–2582. [Google Scholar]
- Song, J.J.; Cho, J.G.; Hwang, S.J.; Cho, C.G.; Park, S.W.; Chae, S.W. Inhibitory effect of caffeic acid phenethyl ester (CAPE) on LPS-induced inflammation of human middle ear epithelial cells. Acta Otolaryngol. 2008, 128, 1303–1307. [Google Scholar] [CrossRef]
- Akiba, J.; Yano, H.; Ogasawara, S.; Higaki, K.; Kojiro, M. Expression and function of interleukin-8 in human hepatocellular carcinoma. Int. J. Oncol. 2001, 18, 257–264. [Google Scholar]
- Kubo, F.; Ueno, S.; Hiwatashi, K.; Sakoda, M.; Kawaida, K.; Nuruki, K.; Aikou, T. Interleukin 8 in human hepatocellular carcinoma correlates with cancer cell invasion of vessels but not with tumor angiogenesis. Ann. Surg. Oncol. 2005, 12, 800–807. [Google Scholar] [CrossRef]
- Moon, M.K.; Lee, Y.J.; Kim, J.S.; Kang, D.G.; Lee, H.S. Effect of caffeic acid on tumor necrosis factor-alpha-induced vascular inflammation in human umbilical vein endothelial cells. Biol. Pharm. Bull. 2009, 32, 1371–1377. [Google Scholar]
- Zhao, Z.; Shin, H.S.; Satsu, H.; Totsuka, M.; Shimizu, M. 5-caffeoylquinic acid and caffeic acid down-regulate the oxidative stress- and TNF-alpha-induced secretion of interleukin-8 from Caco-2 cells. J. Agric. Food Chem. 2008, 56, 3863–3868. [Google Scholar]
- Fitzpatrick, L.R.; Wang, J.; Le, T. Caffeic acid phenethyl ester, an inhibitor of nuclear factor-kappaB, attenuates bacterial peptidoglycan polysaccharide-induced colitis in rats. J. Pharmacol. Exp. Ther. 2001, 299, 915–920. [Google Scholar]
- Zhang, T.; Guo, C.J.; Li, Y.; Douglas, S.D.; Qi, X.X.; Song, L.; Ho, W.Z. Interleukin-1beta induces macrophage inflammatory protein-1beta expression in human hepatocytes. Cell. Immunol. 2003, 226, 45–53. [Google Scholar] [CrossRef]
- Santiago-Lomelí, M.; Gómez-Quiroz, L.E.; Ortíz-Ortega, V.M.; Kershenobich, D.; Gutiérrez-Ruiz, M.C. Differential effect of interleukin-10 on hepatocyte apoptosis. Life Sci. 2005, 76, 2569–2579. [Google Scholar] [CrossRef]
- Hsia, C.Y.; Huo, T.I.; Chiang, S.Y.; Lu, M.F.; Sun, C.L.; Wu, J.C.; Lee, P.C.; Chi, C.W.; Lui, W.Y.; Lee, S.D. Evaluation of interleukin-6, interleukin-10 and human hepatocyte growth factor as tumor markers for hepatocellular carcinoma. Eur. J. Surg. Oncol. 2007, 33, 208–212. [Google Scholar]
- Delhem, N.; Cottrez, F.; Carpentier, A.; Miroux, C.; Moralès, O.; François, V.; Groux, H.; Auriault, C.; Pancré, V. Role of the Regulatory T lymphocytes in hepatitis C fibrosis progression. Bull. Cancer 2008, 95, 1029–1038. [Google Scholar]
- Bruzzese, F.; Di Gennaro, E.; Avallone, A.; Pepe, S.; Arra, C.; Caraglia, M.; Tagliaferri, P.; Budillon, A. Synergistic antitumor activity of epidermal growth factor receptor tyrosine kinase inhibitor gefitinib and IFN-alpha in head and neck cancer cells in vitro and in vivo. Clin. Cancer Res. 2006, 12, 617–625. [Google Scholar] [CrossRef]
- Tirosh, O.; Sen, C.K.; Roy, S.; Kobayashi, M.S.; Packer, L. Neuroprotective effects of alpha-lipoic acid and its positively charged amide analogue. Free Radic. Biol. Med. 1999, 26, 1418–1426. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guerriero, E.; Sorice, A.; Capone, F.; Costantini, S.; Palladino, P.; D'ischia, M.; Castello, G. Effects of Lipoic Acid, Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines. Molecules 2011, 16, 6365-6377. https://doi.org/10.3390/molecules16086365
Guerriero E, Sorice A, Capone F, Costantini S, Palladino P, D'ischia M, Castello G. Effects of Lipoic Acid, Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines. Molecules. 2011; 16(8):6365-6377. https://doi.org/10.3390/molecules16086365
Chicago/Turabian StyleGuerriero, Eliana, Angela Sorice, Francesca Capone, Susan Costantini, Pasquale Palladino, Marco D'ischia, and Giuseppe Castello. 2011. "Effects of Lipoic Acid, Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines" Molecules 16, no. 8: 6365-6377. https://doi.org/10.3390/molecules16086365
APA StyleGuerriero, E., Sorice, A., Capone, F., Costantini, S., Palladino, P., D'ischia, M., & Castello, G. (2011). Effects of Lipoic Acid, Caffeic Acid and a Synthesized Lipoyl-Caffeic Conjugate on Human Hepatoma Cell Lines. Molecules, 16(8), 6365-6377. https://doi.org/10.3390/molecules16086365



