Preliminary Phytochemical Screening and In Vitro Anti-Helicobacter pylori Activity of Extracts of the Stem Bark of Bridelia micrantha (Hochst., Baill., Euphorbiaceae)
Abstract
1. Introduction
2. Results and Discussion
2.1. Anti-Helicobacter pylori Activities
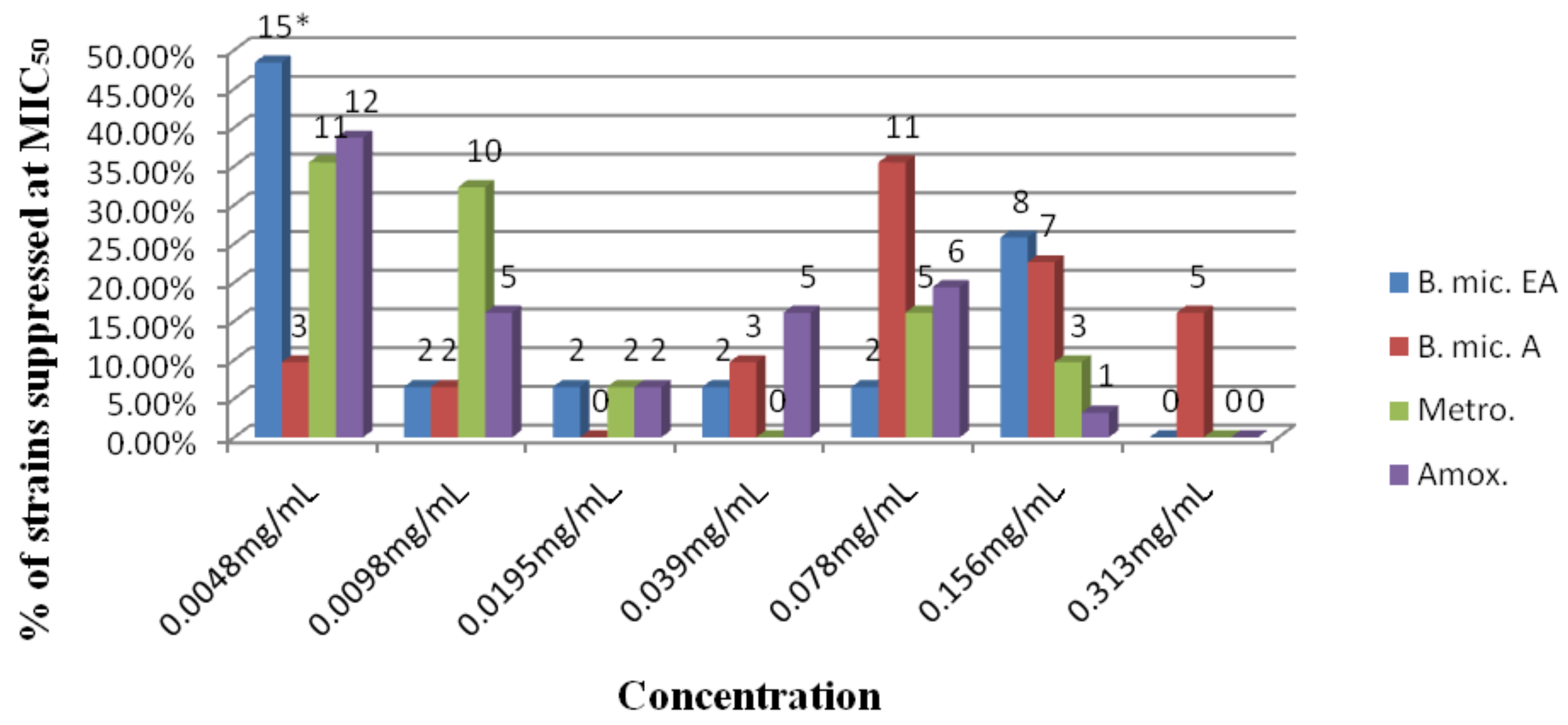
2.2. MIC determination at MIC50 and MIC90
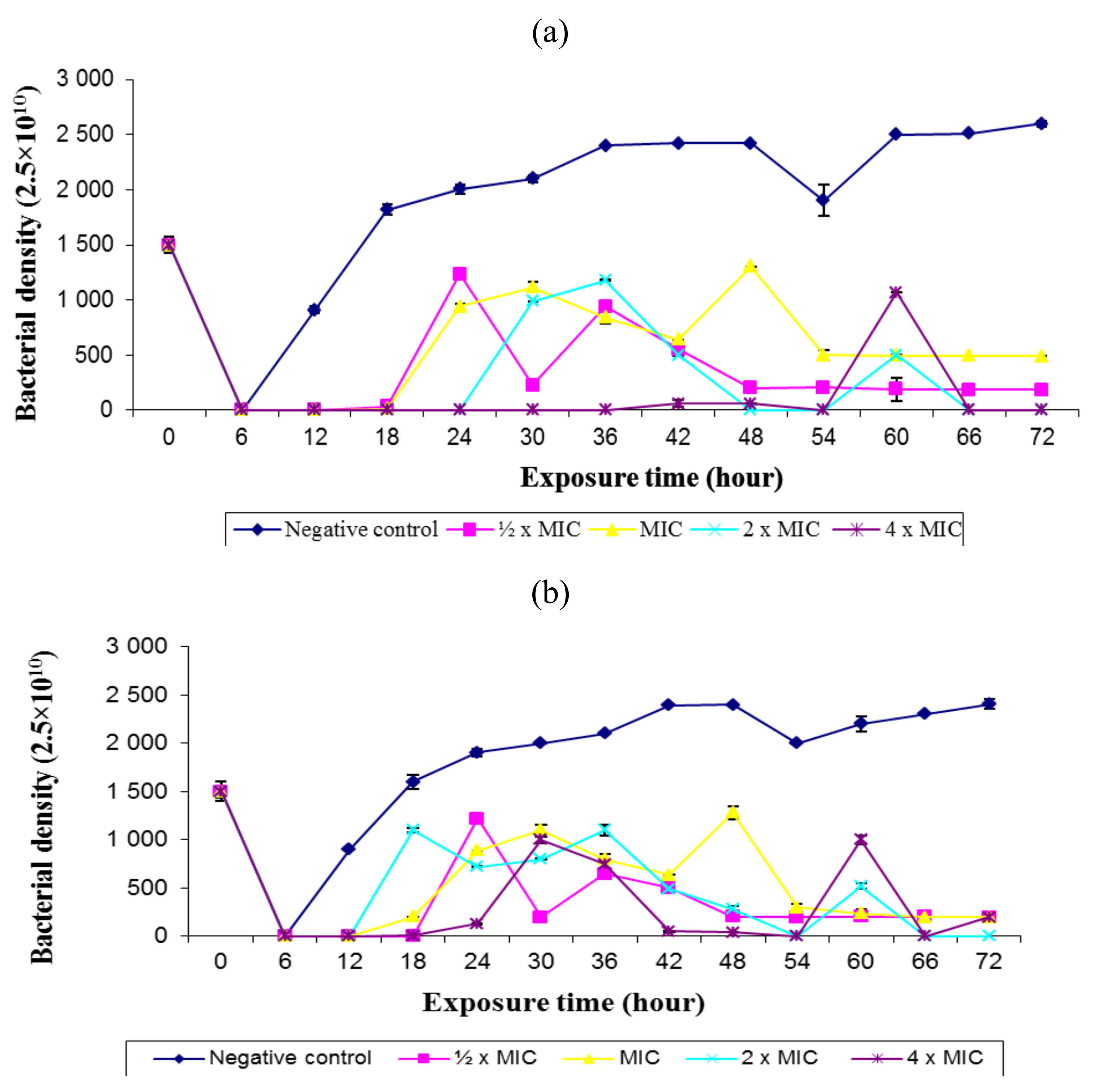
2.3. Rate of Kill
2.4. Phytochemical Compounds
3. Experimental
3.1. Bacterial Strains
3.2. Preparation of Plant Extracts
3.3. Screening of Crude Extracts for Anti-H. pylori Activity
3.4. Determination of Minimum Inhibitory Concentration (MIC50 and MIC90)
3.5. Determination of the Rate of Kill
3.6. Phytochemical Analysis
3.7. Statistical Analysis
4. Conclusions
Acknowledgements
References and Notes
- Konturek, J.W. Discovery by Jaworski of Helicobacter pylori and its pathogenetic role in peptic ulcer, gastritis and gastric cancer. Ann. Clin. Microbiol. Antimicrob. 2004, 10, 3–25. [Google Scholar][Green Version]
- Tanih, N.F.; Clarke, A.M.; Mkwetshana, N.; Green, E.; Ndip, L.M.; Ndip, R.N. Helicobacter pylori infection in Africa: Pathology and microbiological diagnosis. Afr. J. Biotechnol. 2008, 7, 4653–4662. [Google Scholar][Green Version]
- Fritz, E.L.; Slavik, T.; Delport, W.; Olivier, B.; Schalk, W.; Merwel, V. Incidence of Helicobacter felis and the effect of coinfection with Helicobacter pylori on the Gastric Mucosa in the African population. J. Clin. Microbiol. 2006, 44, 1692–1696. [Google Scholar] [CrossRef] [PubMed]
- Ndip, R.N.; Alertia, E.M.T.; Ojongokpoko, J.E.A.; Luma, H.N.; Malongue, A.; Akoachere, J.F.T.K.; Ndip, L.M.; MacMillan, M.; Weaver, L.T. Helicobacter pylori isolates recovered from gastric biopsies of patients with gastro-duodenal pathologies in Cameroon: Current status of antibiogram. Trop. Med. Int. Health. 2008, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Dube, C.; Tanih, N.F.; Clarke, A.M.; Mkwetshana, N.; Green, E.; Ndip, R.N. Helicobacter pylori infection and transmission in Africa: Household hygiene and water sources are plausible factors exacerbating spread. Afr. J. Biotechnol. 2009, 8, 6028–6035. [Google Scholar]
- Stamatisa, G.; Kyriazopoulosb, P.; Golegoub, S.; Basayiannisb, A.; Skaltsasc, S.; Skaltsa, H. In vitro anti-Helicobacter pylori activity of Greek herbal medicines. J. Ethnopharmacol. 2003, 88, 175–179. [Google Scholar] [CrossRef]
- Tanih, N.F.; Okeleye, B.I.; Naidoo, N.; Clarke, A.M.; Mkwetshana, N.; Green, E.; Ndip, L.M.; Ndip, R.N. Marked susceptibility of South African Helicobacter pylori strains to ciprofloxacin and amoxicillin: Clinical implications. S. Afr. Med. J. 2010, 100, 49–52. [Google Scholar] [PubMed]
- Ndip, R.N.; Malange, A.E.; Tarkang, A.E.; Mbullah, S.M.; Luma, H.N.; Malongue, A.; Ndip, L.M.; Nyongbela, K.; Wirmum, C.; Efange, S.M. In vitro anti-Helicobacter pylori activity of extracts of selected medicinal plants from North West Cameroon. J. Ethnopharmacol. 2007, 114, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B.; Pendland, S.L.; Storia, A.; Hamill, F.A.; Fabricant, D.; Dietz, B.M.; Chadwick, L.R. In vitro susceptibility of Helicobacter pylori to botanical extracts used traditionally for the treatment of gastrointestinal disorders. Phytoth. Res. 2005, 19, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Bessong, P.O.; Rojas, L.B.; Obi, L.C.; Tshisikawe, P.M.; Ignore, E.O. Further screening of Venda medicinal plants for activity against HIV type 1 reverse transcriptase and integrase. Afr. J. Biotechnol. 2006, 5, 526–528. [Google Scholar]
- Iwalewa, E.O.; McGaw, L.J.; Naidoo, V.; Eloff, J.N. Inflammation: The foundation of diseases and disorders. A review of phytomedicines of South African origin used to treat pain and inflammatory conditions. Afr. J. Biotechnol. 2007, 25, 2868–2885. [Google Scholar]
- Samie, A.; Obi, C.L.; Bessong, P.O.; Namrita, L. Activity profiles of fourteen selected medicinal plants from rural Venda communities in South Africa against fifteen clinical bacterial species. Afr. J. Biotechnol. 2005, 4, 1443–1451. [Google Scholar]
- Njume, C.; Afolayan, A.J.; Ndip, R.N. An overview of antimicrobial resistance and the future of medicinal plants in the treatment of Helicobacter pylori infections. Afr. J. Pharm. Pharmacol. 2009, 3, 685–699. [Google Scholar]
- Samie, A.; Obi, C.L.; Lall, N.; Meyer, J.J. In-vitro cytotoxicity and antimicrobial activities, against clinical isolates of Campylobacter species and Entamoeba histolytica, of local medicinal plants from the Venda region, in South Africa. Ann. Trop. Med. Parasitol. 2009, 103, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Adeleye, I.A.; Ezekiel, A.O.; Smith, S.; Odusola, O.; Sobande, J. Antibacterial activity of extracts of Alchornea cordifolia (Schum and Thonn) Mull.Arg., Boerhavia diffusa (L) and Bridellia micranthaI (Hoscht) Baill. used in traditional medicine in Nigeria on Helicobacter pylori and four diarrhoeagenic bacterial pathogens. Afr. J. Biotechnol. 2008, 7, 3761–3764. [Google Scholar]
- Theo, A.; Masebe, T.; Suzuki, Y.; Kikuchi, H.; Wada, S.; Obi, L.C.; Bessong, P.O.; Usuzawa, M.; Oshima, Y.; Hattori, T. “Peltophorum Africanum, a traditional South African medicinal plant, contains an anti HIV-1 constituent, betulinic acid”. Tohoku J. Experiment. Med. 2009, 217, 93–99. [Google Scholar] [CrossRef]
- Maoela, M.S.; Arotiba, O.A.; Baker, P.G.L.; Mabusela, W.T.; Jahed, N.; Songa, E.A.; Iwuoha, E.I. Electroanalytical determination of catechin flavonoid in ethyl acetate extracts of Medicinal Plants. Int. J. Electrochem. Sci. 2009, 4, 1497–1510. [Google Scholar]
- Djipa, C.D.; Delméeb, M.; Quetin-Leclercq, J. Antimicrobial activity of bark extracts of Syzygium jambos (L.) Alston (Myrtaceae). J. Ethnopharmacol. 2000, 71, 307–313. [Google Scholar] [CrossRef]
- Asha, S.; Anitha, S.; Anantharajan, R. Antibacterial activity of herbal plant extracts towards the fish pathogens. Int. J. Microbiol. 2008, 4, 1–5. [Google Scholar]
- Evans, W.C. Trease and Evans’ Pharmacognosy, 14th ed.; WB Saunders Company Limited: London, UK, 1996; pp. 176–177. [Google Scholar]
- World Health Organization Expect committee on specification for Pharmaceutical preparation. Quality Assurance of Pharmaceuticals. Thirty-Second Report; WHO: Geneva, Switzerland, 1992; pp. 44–76. [Google Scholar]
- Ali, S.M.; Khan, A.A.; Ahmed, I.; Musaddiq, M.; Ahmed, K.S.; Polasa, H.; Rao, L.V.; Habibullah, C.M.; Sechi, L.A.; Ahmed, N. Antimicrobial activities of Eugenol and Cinnamaldehyde against the human gastric pathogen Helicobacter pylori. Ann. Clin. Microbiol. Antimicrob. 2005, 4, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Gadhi, C.A.; Benharref, A.; Jana, M.; Lozniewski, A. Anti-Helicobacter pylori activity of Aristolochia paucinervis Pomel extracts. J. Ethnopharmacol. 2001, 75, 203–205. [Google Scholar] [CrossRef]
- Akinpelu, D.A.; Aiyegoro, A.O.; Okoh, A.I. Studies on the biocidal and cell membrane disruption potentials of stem bark extracts of Afzelia africana (Smith). Biol. Res. 2009, 42, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Adegboye, M.F.; Akinpelu, D.A.; Okoh, A.I. The bioactive and phytochemical properties of Garcinia kola (Heckel) seed extract on some pathogens. Afr. J. Biotechnol. 2008, 7, 3934–3938. [Google Scholar]
- Hodek, P.; Trefil, P.; Stiborova, M. Flavonoids-Potent and versatile biologically active compounds interacting with cytochrome P450. Chemico-Biol Int. 2002, 139, 1–21. [Google Scholar] [CrossRef]
- Boyanova, L.; Gergova, G.; Nikolov, R.; Derejian, S.; Lazarova, E.; Katsarov, N.; Mitov, I.; Krastev, Z. Activity of Bulgarian propolis against 94 Helicobacter pylori strains in vitro by agar-well diffusion, agar dilution and disc diffusion methods. J. Med. Microbiol. 2005, 54, 481–483. [Google Scholar] [CrossRef] [PubMed]
- Banfi, E.; Scialino, G.; Monti-Bragadin, C. Development of a microdilution method to evaluate Mycobacterium tuberculosis drug susceptibility. J. Antimicrob. Chem. 2003, 52, 796–800. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
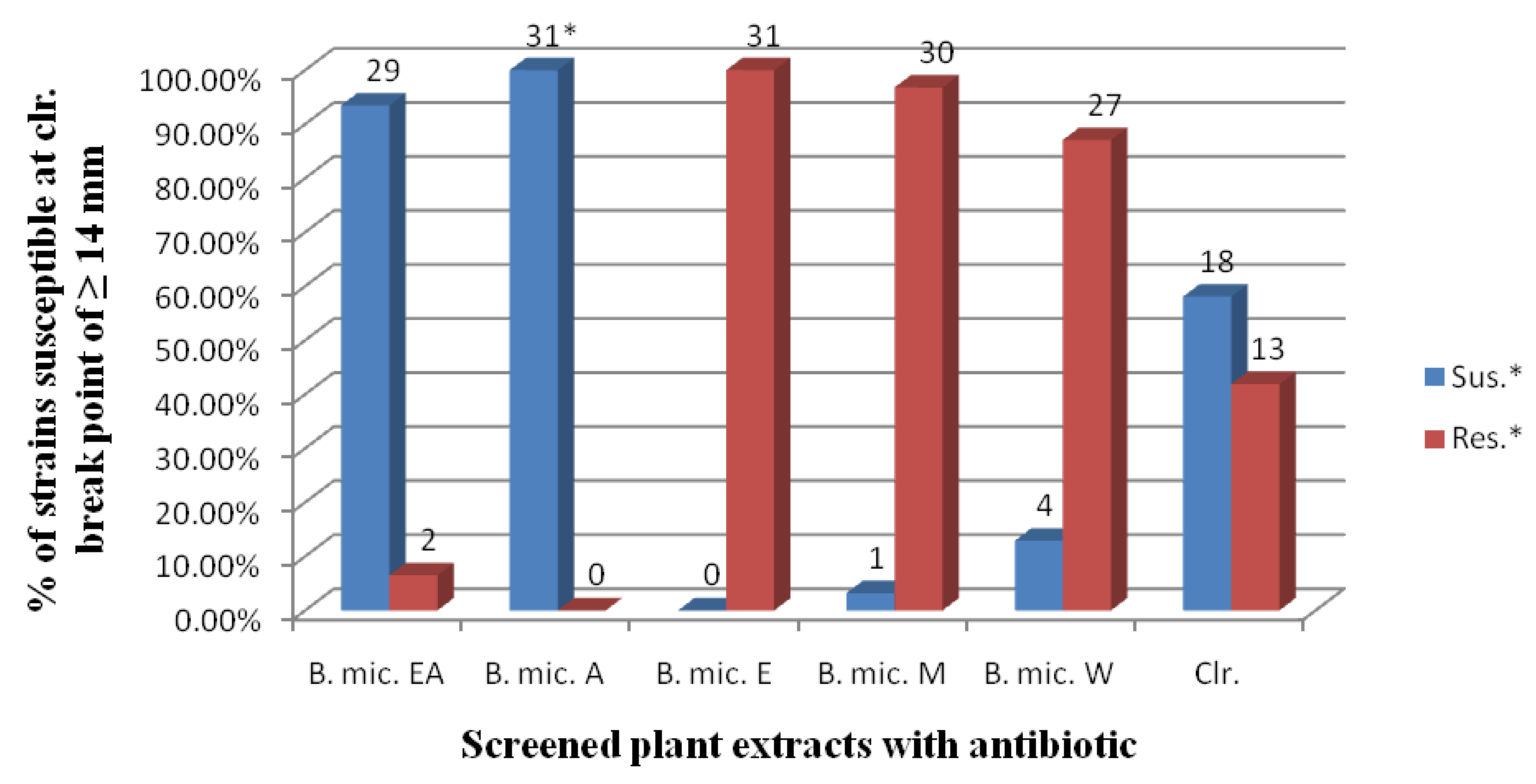
| Zone diameter of inhibition of growth (mm) | ||||||
|---|---|---|---|---|---|---|
| H. pylori strains | B.mic. (EA) | B.mic. (A) | B.mic. (E) | B.mic. (M) | B.mic. (W) | Clr. |
| PE11A | 16 | 22 | 0 | 0 | 8 | 10 |
| PE26A | 17 | 22 | 8 | 8 | 10 | 18 |
| PE93A | 18 | 20 | 0 | 0 | 0 | 28 |
| PE93C | 18 | 20 | 0 | 8 | 9 | 8 |
| PE102C | 16 | 20 | 0 | 0 | 11 | 16 |
| PE115A | 17 | 20 | 0 | 0 | 11 | 10 |
| PE155A | 16 | 20 | 0 | 0 | 9 | 15 |
| PE162A | 12 | 22 | 0 | 8 | 10 | 35 |
| PE219C | 18 | 20 | 0 | 0 | 11 | 12 |
| PE252C | 20 | 20 | 0 | 0 | 11 | 15 |
| PE258C | 15 | 21 | 8 | 8 | 10 | 10 |
| PE265C | 16 | 22 | 0 | 0 | 9 | 13 |
| PE296C | 14 | 21 | 0 | 0 | 9 | 0 |
| PE308C | 19 | 21 | 0 | 0 | 10 | 15 |
| PE369A | 13 | 20 | 0 | 12 | 13 | 20 |
| PE369C | 16 | 20 | 7 | 11 | 12 | 18 |
| PE402A | 14 | 22 | 0 | 11 | 15 | 12 |
| PE406C | 16 | 21 | 7 | 10 | 13 | 27 |
| PE407C | 17 | 23 | 0 | 10 | 12 | 13 |
| PE411C | 15 | 20 | 0 | 0 | 13 | 17 |
| PE430A | 18 | 18 | 10 | 0 | 12 | 21 |
| PE430C | 17 | 19 | 0 | 8 | 15 | 31 |
| PE436A | 14 | 16 | 0 | 0 | 10 | 23 |
| PE436C | 15 | 22 | 8 | 8 | 14 | 20 |
| PE462A | 15 | 21 | 8 | 12 | 13 | 18 |
| PE462C | 16 | 21 | 8 | 8 | 10 | 17 |
| PE466C | 17 | 17 | 0 | 10 | 13 | 25 |
| PE467A | 16 | 20 | 7 | 10 | 14 | 0 |
| PE467C | 16 | 20 | 0 | 12 | 13 | 0 |
| PE469C | 17 | 20 | 7 | 8 | 13 | 8 |
| PE471A | 16 | 21 | 7 | 14 | 9 | 0 |
| Extract/control antibiotic | Mean zone diameter (mm) | Inhibition diameter range |
|---|---|---|
| B.mic. (EA) | 16.13 ± 1.708 | 12–20 mm |
| B.mic. (A) | 20.39 ± 1.476 | 16–23 mm |
| B.mic. (E) | 2.74 ± 3.794 | 0–10 mm |
| B.mic. (M) | 5.68 ± 5.108 | 0–14 mm |
| B.mic. (W) | 11.03 ± 2.811 | 0–15 mm |
| Clr. | 15.32 ± 8.852 | 0–35 mm |
| B.mic. (EA) | B.mic. (A) | Metronidazole | Amoxicillin | |||||
|---|---|---|---|---|---|---|---|---|
| H. pylori strains | MIC50 | MIC90 | MIC50 | MIC90 | MIC50 | MIC90 | MIC50 | MIC90 |
| PE11A | 0.156 | >0.625 * | 0.078 | 0.313 | 0.156 | 0.625 | 0.156 | ND * |
| PE26A | 0.078 | >0.156 | 0.078 | 0.313 | 0.156 | >0.625 | 0.039 | >0.313 |
| PE93A | 0.039 | ND | 0.078 | ND | 0.0098 | ND | 0.039 | ND |
| PE93C | 0.0048 | >0.0048 | 0.078 | >0.313 | 0.0195 | ND | 0.0048 | >0.625 |
| PE102C | 0.156 | >0.625 | 0.078 | 0.313 | 0.078 | 0.156 | 0.039 | 0.156 |
| PE115A | 0.0195 | ND | 0.313 | ND | 0.0098 | ND | 0.0098 | ND |
| PE155A | 0.0048 | >0.0048 | 0.156 | ND | 0.0048 | ND | 0.0048 | 0.313 |
| PE162A | 0.0048 | >0.0048 | 0.078 | >0.625 | 0.0048 | ND | 0.0048 | 0.078 |
| PE219C | 0.0048 | >0.0048 | 0.0048 | >0.156 | 0.0048 | ND | 0.078 | 0.313 |
| PE252C | 0.156 | ND | 0.313 | ND | 0.0098 | ND | 0.0098 | ND |
| PE258C | 0.156 | 2.5 | 0.078 | 0.313 | 0.078 | 0.156 | 0.078 | 0.156 |
| PE265C | 0.0048 | >0.0048 | 0.156 | ND | 0.0048 | ND | 0.0048 | 1.25 |
| PE296C | 0.0048 | 0.0048 | 0.0048 | 0.625 | 0.0098 | 0.625 | 0.0048 | 0.156 |
| PE308C | 0.0048 | >0.0048 | 0.078 | ND | 0.0048 | ND | 0.0048 | >0.625 |
| PE369A | 0.156 | ND | 0.156 | ND | 0.078 | >5 | 0.078 | >2.5 |
| PE369C | 0.0098 | >0.156 | 0.156 | ND | 0.0098 | ND | 0.0098 | ND |
| PE402A | 0.156 | >1.25 | 0.078 | 0.625 | 0.156 | >0.156 | 0.078 | >0.156 |
| PE406C | 0.078 | ND | 0.039 | ND | 0.0098 | >5 | 0.0195 | ND |
| PE407C | 0.156 | 1.25 | 0.078 | 0.313 | 0.078 | >0.156 | 0.039 | 0.625 |
| PE411C | 0.0048 | >0.0048 | 0.313 | ND | 0.0098 | ND | 0.0048 | 0.156 |
| PE430A | 0.0048 | ND | 0.313 | >0.625 | 0.0195 | ND | 0.039 | 0.156 |
| PE430C | 0.156 | >0.156 | 0.156 | ND | 0.078 | >5 | 0.078 | >0.313 |
| PE436A | 0.0048 | >0.156 | 0.0098 | >0.078 | 0.0048 | ND | 0.0048 | ND |
| PE436C | 0.0048 | 0.0048 | 0.0098 | 0.625 | 0.0048 | 0.0098 | 0.0048 | 0.625 |
| PE462A | 0.039 | ND | 0.313 | ND | 0.0098 | ND | 0.0098 | ND |
| PE462C | 0.0195 | ND | 0.039 | ND | 0.0098 | ND | 0.0195 | ND |
| PE466C | 0.0098 | ND | 0.156 | ND | 0.0098 | ND | 0.0098 | ND |
| PE467A | 0.0048 | >0.0098 | 0.039 | >0.078 | 0.0048 | ND | 0.0048 | 0.625 |
| PE467C | 0.0048 | >0.0048 | 0.156 | ND | 0.0048 | ND | 0.078 | 0.313 |
| PE469C | 0.0048 | 0.0048 | 0.0048 | 0.313 | 0.0048 | 1.25 | 0.0048 | 0.313 |
| PE471A | 0.0048 | >0.0048 | 0.078 | >0.313 | 0.0048 | ND | 0.0048 | 0.313 |
| Average | 0.052 | 0.118 | 0.034 | 0.031 | ||||
| Phytochemical | Bridelia micrantha (ethyl acetate extract) |
|---|---|
| Alkaloids | ++ |
| Flavonoids | +++ |
| Steroids | +++ |
| Tannins | +++ |
| Saponins | +++ |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Okeleye, B.I.; Bessong, P.O.; Ndip, R.N. Preliminary Phytochemical Screening and In Vitro Anti-Helicobacter pylori Activity of Extracts of the Stem Bark of Bridelia micrantha (Hochst., Baill., Euphorbiaceae). Molecules 2011, 16, 6193-6205. https://doi.org/10.3390/molecules16086193
Okeleye BI, Bessong PO, Ndip RN. Preliminary Phytochemical Screening and In Vitro Anti-Helicobacter pylori Activity of Extracts of the Stem Bark of Bridelia micrantha (Hochst., Baill., Euphorbiaceae). Molecules. 2011; 16(8):6193-6205. https://doi.org/10.3390/molecules16086193
Chicago/Turabian StyleOkeleye, Benjamin I., Pascal O. Bessong, and Roland N. Ndip. 2011. "Preliminary Phytochemical Screening and In Vitro Anti-Helicobacter pylori Activity of Extracts of the Stem Bark of Bridelia micrantha (Hochst., Baill., Euphorbiaceae)" Molecules 16, no. 8: 6193-6205. https://doi.org/10.3390/molecules16086193
APA StyleOkeleye, B. I., Bessong, P. O., & Ndip, R. N. (2011). Preliminary Phytochemical Screening and In Vitro Anti-Helicobacter pylori Activity of Extracts of the Stem Bark of Bridelia micrantha (Hochst., Baill., Euphorbiaceae). Molecules, 16(8), 6193-6205. https://doi.org/10.3390/molecules16086193




