Generation and Detection of Levuglandins and Isolevuglandins In Vitro and In Vivo
Abstract
:1. Introduction
2. Generation of Levuglandins and Isolevuglandins
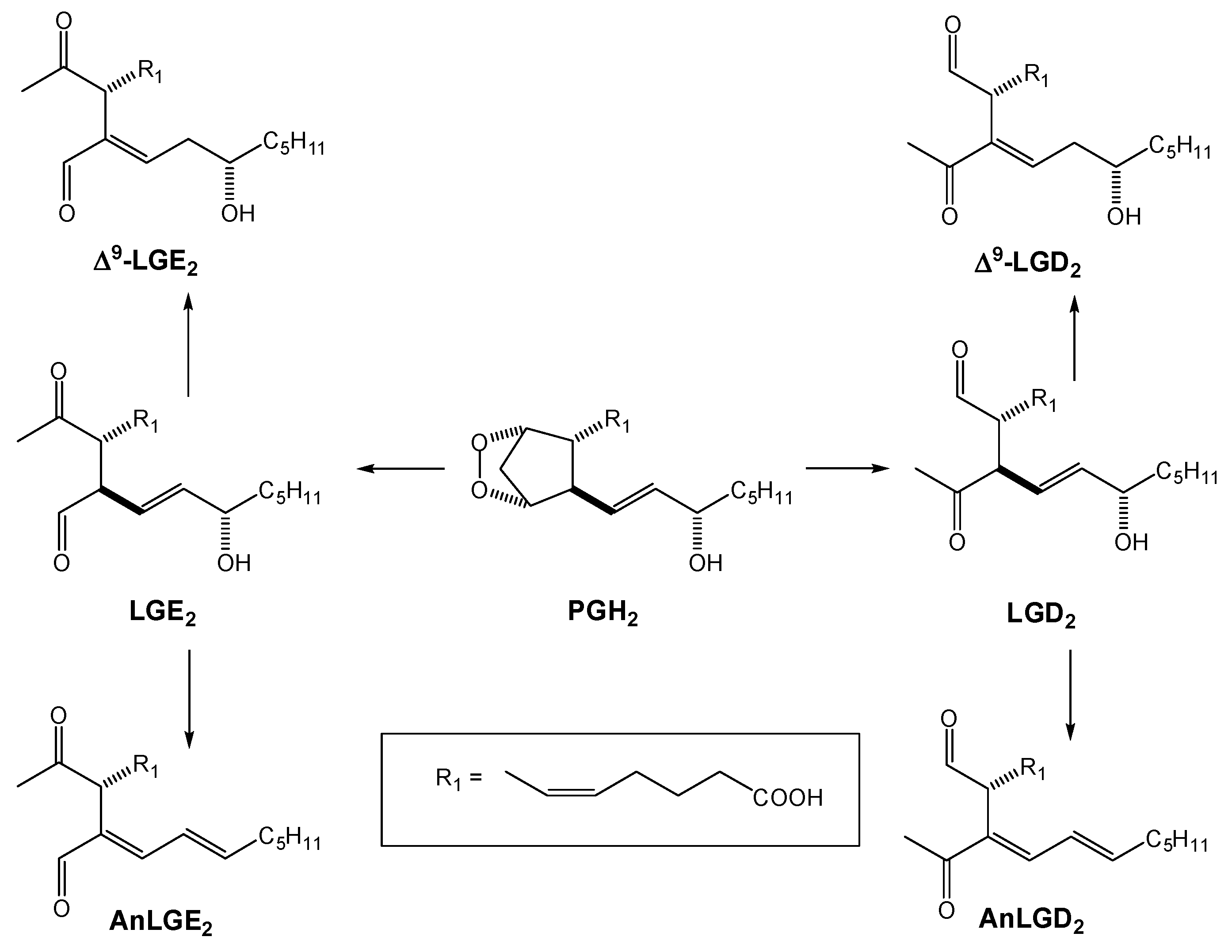
2.1. Enzymatic Pathways
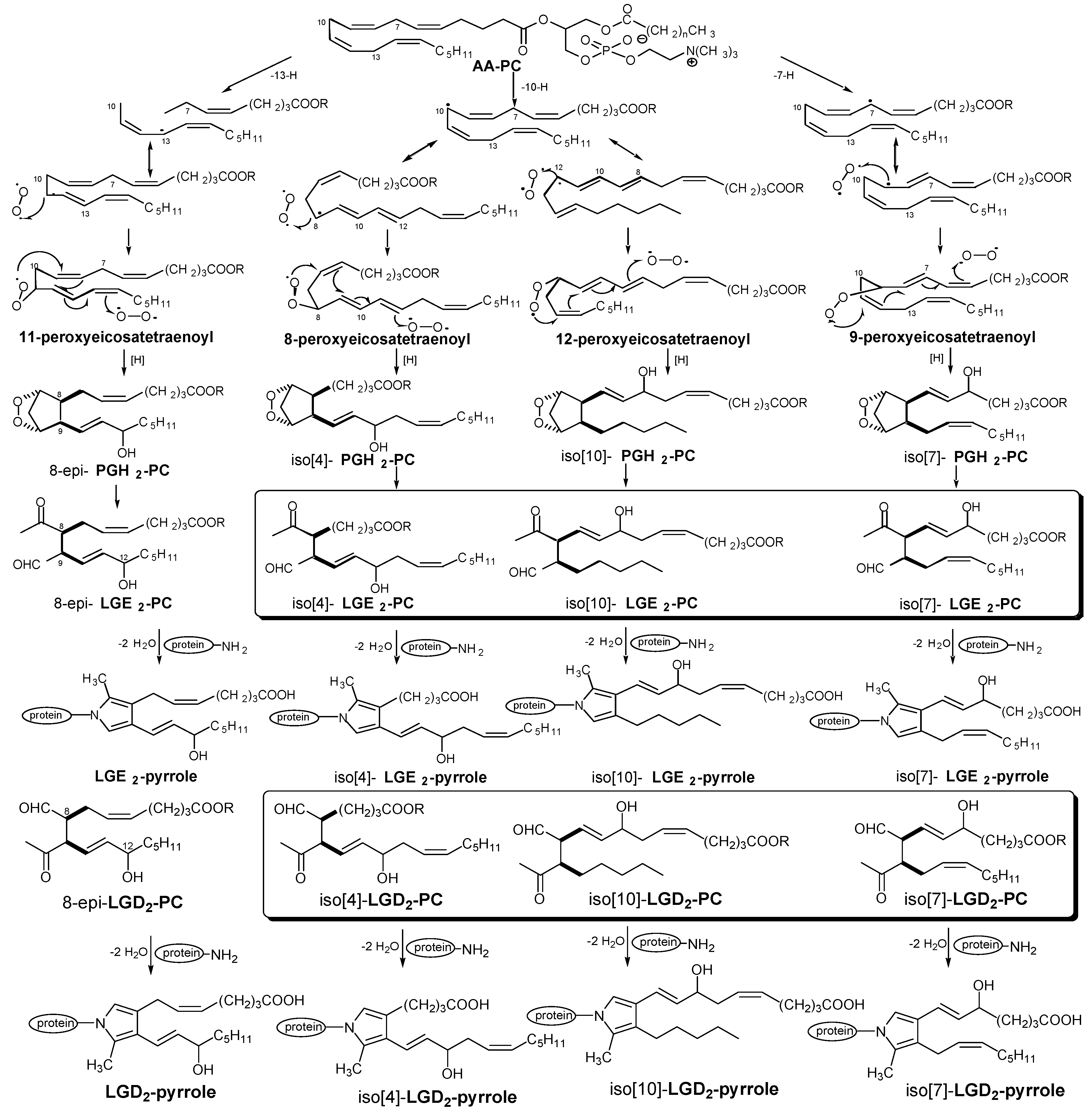
2.2. Free Radical Pathways
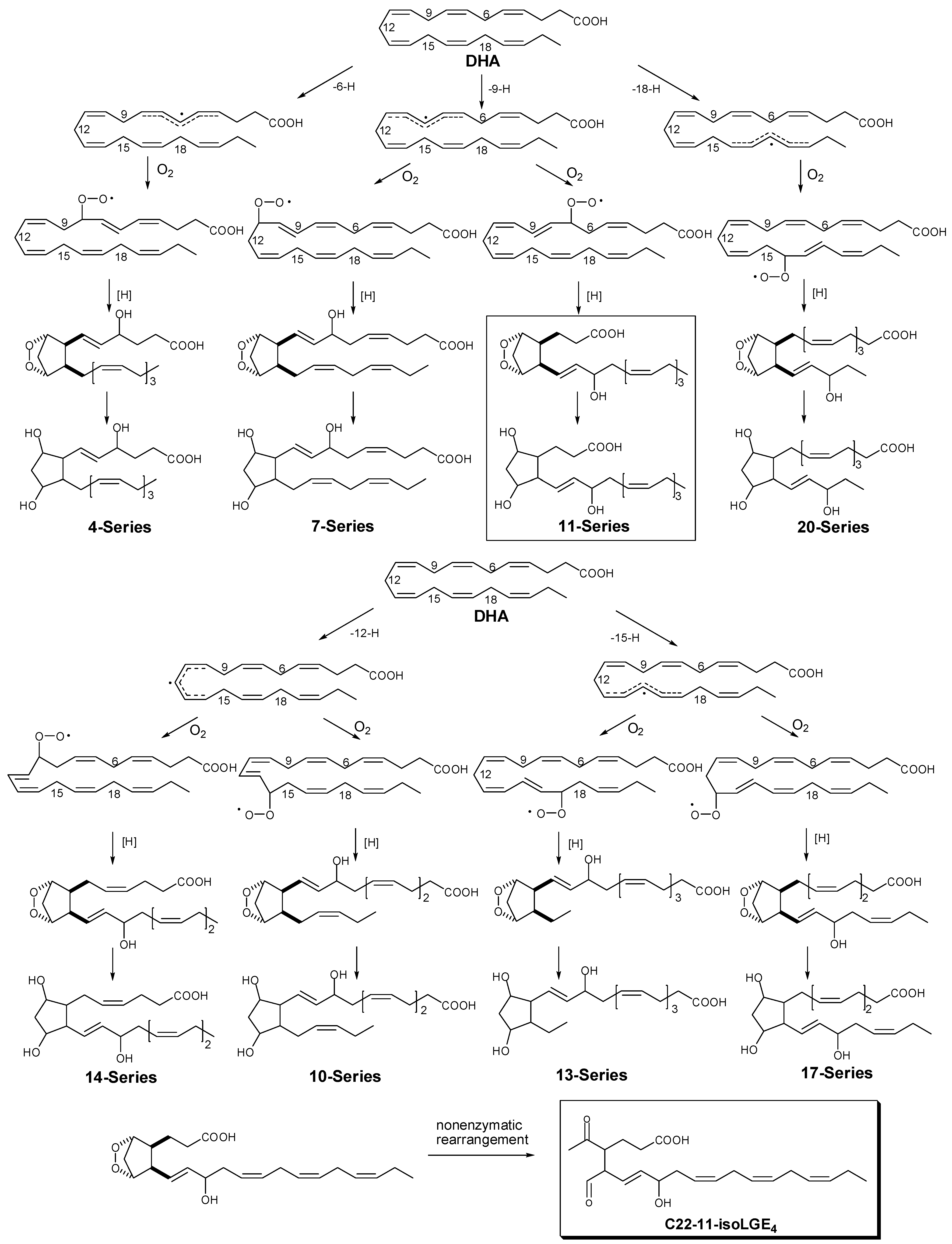
2.3. Free Radical Oxidation of Docosahexaenoic Acid (DHA)
3. Detection of Biological Adducts of Isolevuglandins
3.1. Immunological Detection of Protein Adducts of LGs and IsoLGs
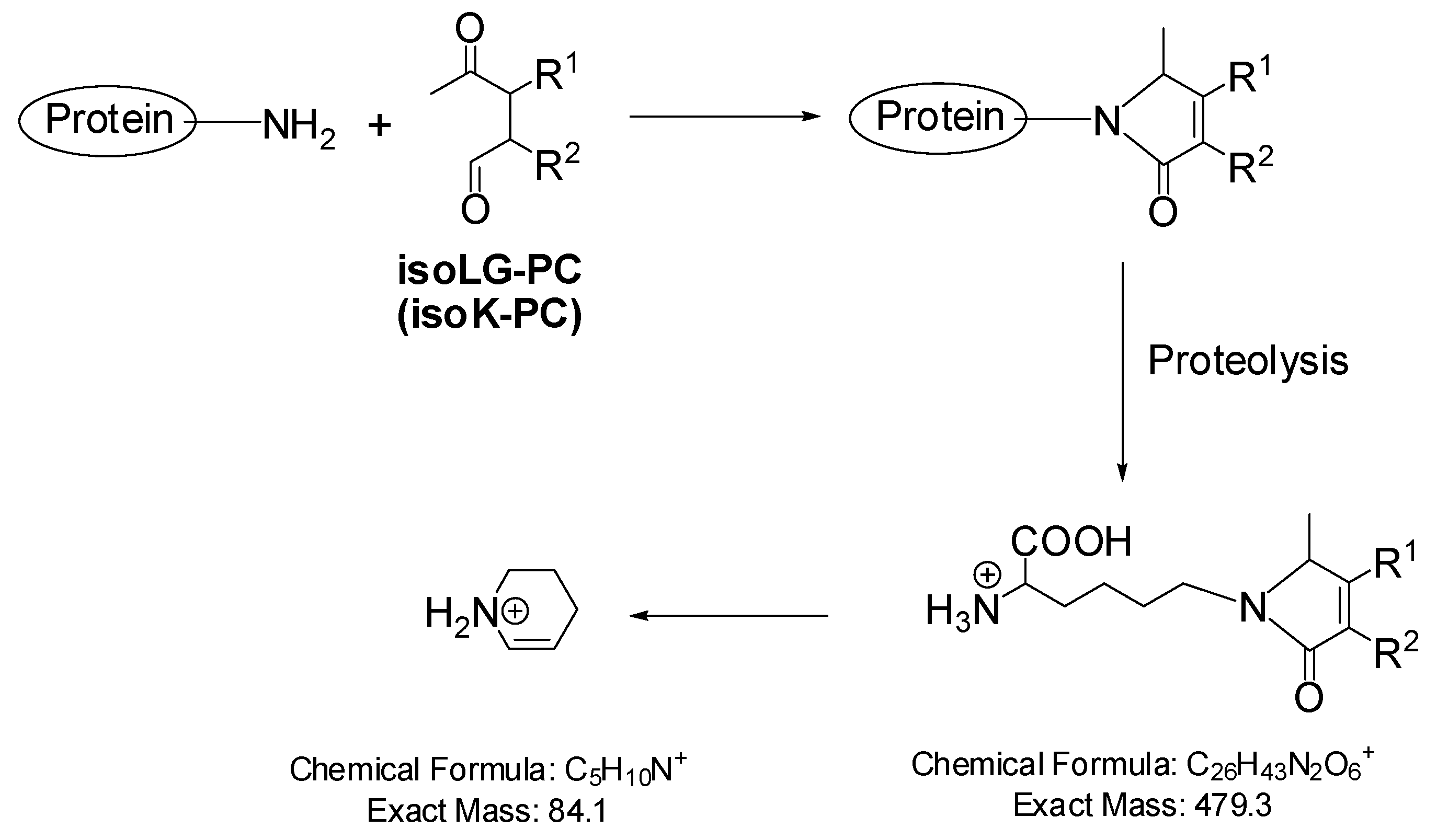
3.2. Mass Spectrometric Detection of Protein Adducts of LGs and IsoLGs
4. Conclusions
References
- Ames, B.N. Dietary carcinogens and anticarcinogens. Oxygen radicals and degenerative diseases. Science 1983, 221, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Montine, T.J.; Neely, M.D.; Quinn, J.F.; Beal, M.F.; Markesbery, W.R.; Roberts, L.J.; Morrow, J.D. Lipid peroxidation in aging brain and Alzheimer’s disease. Free Radic. Biol. Med. 2002, 33, 620–626. [Google Scholar] [CrossRef]
- Smith, M.A.; Perry, G.; Richey, P.L.; Sayre, L.M.; Anderson, V.E.; Beal, M.F.; Kowall, N. Oxidative damage in Alzheimer’s. Nature 1996, 382, 120–121. [Google Scholar] [CrossRef] [PubMed]
- Davies, S.S.; Amarnath, V.; Montine, K.S.; Bernoud-Hubac, N.; Boutaud, O.; Montine, T.J.; Roberts, L.J., 2nd. Effects of reactive gamma-ketoaldehydes formed by the isoprostane pathway (isoketals) and cyclooxygenase pathway (levuglandins) on proteasome function. FASEB J. 2002, 16, 715–717. [Google Scholar] [CrossRef] [PubMed]
- Amarnath, V.; Valentine, W.M.; Amarnath, K.; Eng, M.A.; Graham, D.G. The mechanism of nucleophilic substitution of alkylpyrroles in the presence of oxygen. Chem. Res. Toxicol. 1994, 7, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.J., 2nd; Salomon, R.G.; Morrow, J.D.; Brame, C.J. New developments in the isoprostane pathway: Identification of novel highly reactive gamma-ketoaldehydes (isolevuglandins) and characterization of their protein adducts. FASEB J. 1999, 13, 1157–1168. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Subbanagounder, G.; Singh, U.; O’Neil, J.; Hoff, H.F. Oxidation of low-density lipoproteins produces levuglandin-protein adducts. Chem. Res. Toxicol. 1997, 10, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Sha, W.; Brame, C.; Kaur, K.; Subbanagounder, G.; O’Neil, J.; Hoff, H.F.; Roberts, L.J., 2nd. Protein adducts of iso[4]levuglandin E2, a product of the isoprostane pathway, in oxidized low density lipoprotein. J. Biol. Chem. 1999, 274, 20271–20280. [Google Scholar] [CrossRef] [PubMed]
- Poliakov, E.; Meer, S.G.; Roy, S.C.; Mesaros, C.; Salomon, R.G. Iso[7]LGD2-protein adducts are abundant in vivo and free radical-induced oxidation of an arachidonyl phospholipid generates this D series isolevuglandin in vitro. Chem. Res. Toxicol. 2004, 17, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Subbanagounder, G.; O’Neil, J.; Kaur, K.; Smith, M.A.; Hoff, H.F.; Perry, G.; Monnier, V.M. Levuglandin E2-protein adducts in human plasma and vasculature. Chem. Res. Toxicol. 1997, 10, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Batyreva, E.; Kaur, K.; Sprecher, D.L.; Schreiber, M.J.; Crabb, J.W.; Penn, M.S.; DiCorletoe, A.M.; Hazen, S.L.; Podrez, E.A. Isolevuglandin-protein adducts in humans: Products of free radical-induced lipid oxidation through the isoprostane pathway. Biochim. Biophys. Acta 2000, 1485, 225–235. [Google Scholar] [CrossRef]
- Morrow, J.D.; Harris, T.M.; Roberts, L.J., 2nd. Noncyclooxygenase oxidative formation of a series of novel prostaglandins: Analytical ramifications for measurement of eicosanoids. Anal. Biochem. 1990, 184, 1–10. [Google Scholar] [CrossRef]
- Morrow, J.D.; Hill, K.E.; Burk, R.F.; Nammour, T.M.; Badr, K.F.; Roberts, L.J., 2nd. A series of prostaglandin F2-like compounds are produced in vivo in humans by a non-cyclooxygenase, free radical-catalyzed mechanism. Proc. Natl. Acad. Sci. USA 1990, 87, 9383–9387. [Google Scholar] [CrossRef] [PubMed]
- Morrow, J.D.; Awad, J.A.; Boss, H.J.; Blair, I.A.; Roberts, L.J., 2nd. Non-cyclooxygenase-derived prostanoids (F2-isoprostanes) are formed in situ on phospholipids. Proc. Natl. Acad. Sci. USA 1992, 89, 10721–10725. [Google Scholar] [CrossRef] [PubMed]
- Awad, J.A.; Morrow, J.D.; Takahashi, K.; Roberts, L.J., 2nd. Identification of non-cyclooxygenase-derived prostanoid (F2-isoprostane) metabolites in human urine and plasma. J. Biol. Chem. 1993, 268, 4161–4169. [Google Scholar] [PubMed]
- Poliakov, E.; Brennan, M.L.; Macpherson, J.; Zhang, R.; Sha, W.; Narine, L.; Salomon, R.G.; Hazen, S.L. Isolevuglandins, a novel class of isoprostenoid derivatives, function as integrated sensors of oxidant stress and are generated by myeloperoxidase in vivo. FASEB J. 2003, 17, 2209–2220. [Google Scholar] [CrossRef] [PubMed]
- Hamberg, M.; Samuelsson, B. Detection and isolation of an endoperoxide intermediate in prostaglandin biosynthesis. Proc. Natl. Acad. Sci. USA 1973, 70, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Ramwell, P.W.; Leovey, E.M.; Sintetos, A.L. Regulation of the arachidonic acid cascade. Biol. Reprod. 1977, 16, 70–87. [Google Scholar] [CrossRef] [PubMed]
- Nugteren, D.H.; Hazelhof, E. Isolation and properties of intermediates in prostaglandin biosynthesis. Biochim. Biophys. Acta 1973, 326, 448–461. [Google Scholar] [CrossRef]
- Samuelsson, B. From studies of biochemical mechanism to novel biological mediators: Prostaglandin endoperoxides, thromboxanes, and leukotrienes. Nobel Lecture, 8 December 1982. Angew. Chem. Int. Ed. Engl. 1983, 22, 805–815. [Google Scholar] [CrossRef]
- Hamberg, M.; Svensson, J.; Wakabayashi, T.; Samuelsson, B. Isolation and structure of two prostaglandin endoperoxides that cause platelet aggregation. Proc. Natl. Acad. Sci. USA 1974, 71, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Bhagwat, S.S.; Hamann, P.R.; Still, W.C.; Bunting, S.; Fitzpatrick, F.A. Synthesis and structure of the platelet aggregation factor thromboxane A2. Nature 1985, 315, 511–513. [Google Scholar] [CrossRef] [PubMed]
- Bunting, S.; Gryglewski, R.; Moncada, S.; Vane, J.R. Arterial walls generate from prostaglandin endoperoxides a substance (prostaglandin X) which relaxes strips of mesenteric and coeliac ateries and inhibits platelet aggregation. Prostaglandins 1976, 12, 897–913. [Google Scholar] [CrossRef]
- Gryglewski, R.J.; Bunting, S.; Moncada, S.; Flower, R.J.; Vane, J.R. Arterial walls are protected against deposition of platelet thrombi by a substance (prostaglandin X) which they make from prostaglandin endoperoxides. Prostaglandins 1976, 12, 685–713. [Google Scholar] [CrossRef]
- Hamberg, M.; Svensson, J.; Samuelsson, B. Thromboxanes: A new group of biologically active compounds derived from prostaglandin endoperoxides. Proc. Natl. Acad. Sci. USA 1975, 72, 2994–2998. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Gryglewski, R.; Bunting, S.; Vane, J.R. An enzyme isolated from arteries transforms prostaglandin endoperoxides to an unstable substance that inhibits platelet aggregation. Nature 1976, 263, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, N.; Bunting, S.; Salmon, J.; Moncada, S.; Vane, J.R.; Johnson, R.A.; Morton, D.R.; Kinner, J.H.; Gorman, R.R.; McGuire, J.C.; et al. The chemical structure of prostaglandin X (prostacyclin). Prostaglandins 1976, 12, 915–928. [Google Scholar] [PubMed]
- Coughlin, D.J.; Salomon, R.G. Synthesis and thermal reactivity of some 2,3-dioxabicyclo[2.2.1]heptane models of prostaglandin endoperoxides. J. Am. Chem. Soc. 1977, 99, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Salomon, M.F. 2,3-Dioxabicyclo[2.2.1]heptane-strained bicyclic peroxide nucleus of prostaglandin endoperoxides. J. Am. Chem. Soc. 1977, 99, 3501–3503. [Google Scholar] [CrossRef] [PubMed]
- Zagorski, M.G.; Salomon, R.G. Prostaglandin endoperoxides. 11. Mechanism of amine-catalyzed fragmentation of 2,3-dioxabicyclo[2.2.1]heptane. J. Am. Chem. Soc. 1980, 102, 2501–2503. [Google Scholar] [CrossRef]
- Zagorski, M.G.; Salomon, R.G. Prostaglandin endoperoxides. 12. Carboxylate catalysis and the effects of proton donors on the decomposition of 2,3-dioxabicyclo[2.2.1]heptane. J. Am. Chem. Soc. 1982, 104, 3498–3503. [Google Scholar] [CrossRef]
- Iyer, R.S.; Miller, D.B.; Salomon, R.G. Prostaglandin endoperoxides. 26. Decomposition of levuglandin-E2-dehydration and allylic rearrangement products. J. Org. Chem. 1990, 55, 3175–3180. [Google Scholar] [CrossRef]
- Salomon, R.G.; Miller, D.B.; Raychaudhuri, S.R.; Avasthi, K.; Lal, K.; Levison, B.S. Prostaglandin endoperoxides. 15. Asymmetric total synthesis of levuglandin E2. J. Am. Chem. Soc. 1984, 106, 8296–8298. [Google Scholar] [CrossRef]
- Nugteren, D.H.; Vonkeman, H.; Vandorp, D.A. Non-enzymic conversion of all-cis 8,11,14-eicosatrienoic acid into prostaglandin E1. Recueil des Travaux Chimiques des Pays-Bas 1967, 86, 1237–1245. [Google Scholar] [CrossRef]
- Morrow, J.D.; Minton, T.A.; Mukundan, C.R.; Campbell, M.D.; Zackert, W.E.; Daniel, V.C.; Badr, K.F.; Blair, I.A.; Roberts, L.J. Free radical-induced generation of isoprostanes in-vivo—Evidence for the formation of D-ring and E-ring isoprostanes. J. Biol. Chem. 1994, 269, 4317–4326. [Google Scholar] [PubMed]
- Morrow, J.D.; Awad, J.A.; Wu, A.P.; Zackert, W.E.; Daniel, V.C.; Roberts, L.J. Nonenzymatic free radical-catalyzed generation of thromboxane-like compounds (isothromboxanes) in vivo. J. Biol. Chem. 1996, 271, 23185–23190. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G. Prostaglandin endoperoxide reaction-mechanisms and the discovery of levuglandins. Acc. Chem. Res. 1985, 18, 294–301. [Google Scholar] [CrossRef]
- Hobbs, H.H.; Russell, D.W.; Brown, M.S.; Goldstein, J.L. The LDL receptor locus in familial hypercholesterolemia: Mutational analysis of a membrane protein. Annu. Rev. Genet. 1990, 24, 133–170. [Google Scholar] [CrossRef] [PubMed]
- Bernoud-Hubac, N.; Davies, S.S.; Boutaud, O.; Montine, T.J.; Roberts, L.J., 2nd. Formation of highly reactive gamma-ketoaldehydes (neuroketals) as products of the neuroprostane pathway. J. Biol. Chem. 2001, 276, 30964–30970. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.L.; Brennan, M.L.; Shen, Z.Z.; MacPherson, J.C.; Schmitt, D.; Molenda, C.E.; Hazen, S.L. Myeloperoxidase functions as a major enzymatic catalyst for initiation of lipid peroxidation at sites of inflammation. J. Biol. Chem. 2002, 277, 46116–46122. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, S.J.; Clark, R.A. The Neutrophil: Function and Clinical Disorders; North Holland Biomedical Press: Amsterdam, The Netherlands, 1978. [Google Scholar]
- Daugherty, A.; Dunn, J.L.; Rateri, D.L.; Heinecke, J.W. Myeloperoxidase, a catalyst for lipoprotein oxidation, is expressed in human atherosclerotic lesions. J. Clin. Invest. 1994, 94, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Hazell, L.J.; Arnold, L.; Flowers, D.; Waeg, G.; Malle, E.; Stocker, R. Presence of hypochlorite-modified proteins in human atherosclerotic lesions. J. Clin. Invest. 1996, 97, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Hazen, S.L.; Heinecke, J.W. 3-Chlorotyrosine, a specific marker of myeloperoxidase-catalyzed oxidation, is markedly elevated in low density lipoprotein isolated from human atherosclerotic intima. J. Clin. Invest. 1997, 99, 2075–2081. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.E.; Schultz, J. Studies on the chlorinating activity of myeloperoxidase. J. Biol. Chem. 1976, 251, 1371–1374. [Google Scholar] [PubMed]
- Heinecke, J.W.; Li, W.; Daehnke, H.L., 3rd; Goldstein, J.A. Dityrosine, a specific marker of oxidation, is synthesized by the myeloperoxidase-hydrogen peroxide system of human neutrophils and macrophages. J. Biol. Chem. 1993, 268, 4069–4077. [Google Scholar] [PubMed]
- Brennan, M.L.; Wu, W.; Fu, X.; Shen, Z.; Song, W.; Frost, H.; Vadseth, C.; Narine, L.; Lenkiewicz, E.; Borchers, M.T.; et al. A tale of two controversies: defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J. Biol. Chem. 2002, 277, 17415–17427. [Google Scholar] [CrossRef] [PubMed]
- van der Vliet, A.; Eiserich, J.P.; Halliwell, B.; Cross, C.E. Formation of reactive nitrogen species during peroxidase-catalyzed oxidation of nitrite. A potential additional mechanism of nitric oxide-dependent toxicity. J. Biol. Chem. 1997, 272, 7617–7625. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; McCall, M.R.; Frei, B. Oxidation of LDL by myeloperoxidase and reactive nitrogen species: reaction pathways and antioxidant protection. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Podrez, E.A.; Abu-Soud, H.M.; Hazen, S.L. Myeloperoxidase-generated oxidants and atherosclerosis. Free Radic. Biol. Med. 2000, 28, 1717–1725. [Google Scholar] [CrossRef]
- Anderson, G.J.; Connor, W.E.; Corliss, J.D. Docosahexaenoic acid is the preferred dietary N-3 fatty-acid for the development of the brain and retina. Pediatr. Res. 1990, 27, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Connor, W.E.; Neuringer, M.; Lin, D.S. Dietary effects on brain fatty acid composition: The reversibility of n-3 fatty acid deficiency and turnover of docosahexaenoic acid in the brain, erythrocytes, and plasma of rhesus monkeys. J. Lipid Res. 1990, 31, 237–247. [Google Scholar] [PubMed]
- Connor, W.E.; Neuringer, M.; Reisbick, S. Essential fatty-acids—The importance of N-3 fatty-acids in the retina and brain. Nutr. Rev. 1992, 50, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.E.; Rapp, L.M.; Wiegand, R.D. Lipid-peroxidation and retinal degeneration. Curr. Eye Res. 1984, 3, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Reactive oxygen species and the central-nervous-system. J. Neurochem. 1992, 59, 1609–1623. [Google Scholar] [CrossRef] [PubMed]
- Simonian, N.A.; Coyle, J.T. Oxidative stress in neurodegenerative diseases. Ann. Rev. Pharm. Toxicol. 1996, 36, 83–106. [Google Scholar] [CrossRef] [PubMed]
- Knight, J.A. Reactive oxygen species and the neurodegenerative disorders. Ann. Clin. Lab. Sci. 1997, 27, 11–25. [Google Scholar] [PubMed]
- Markesbery, W.R. Oxidative stress hypothesis in Alzheimer’s disease. Free Radic. Biol. Med. 1997, 23, 134–147. [Google Scholar] [CrossRef]
- Roberts, L.J.; Montine, T.J.; Markesbery, W.R.; Tapper, A.R.; Hardy, P.; Chemtob, S.; Dettbarn, W.D.; Morrow, J.D. Formation of isoprostane-like compounds (neuroprostanes) in vivo from docosahexaenoic acid. J. Biol. Chem. 1998, 273, 13605–13612. [Google Scholar] [CrossRef] [PubMed]
- Nourooz-Zadeh, J.; Liu, E.H.C.; Anggard, E.E.; Halliwell, B. F-4-isoprostanes: A novel class of prostanoids formed during peroxidation of docosahexaenoic acid (DHA). Biochem. Biophys. Res. Commun. 1998, 242, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Salomon, R.G.; Miller, D.B.; Zagorski, M.G.; Coughlin, D.J. Solvent induced fragmentation of prostaglandin endoperoxides. New aldehyde products from PGH2 and a novel intramolecular 1,2-hydride shift during endoperoxide fragmentation in aqueous solution. J. Am. Chem. Soc. 1984, 106, 6049–6060. [Google Scholar] [CrossRef]
- Salomon, R.G.; Jirousek, M.R.; Ghosh, S.; Sharma, R.B. Prostaglandin endoperoxides 21. Covalent binding of levuglandin E2 with proteins. Prostaglandins 1987, 34, 643–656. [Google Scholar] [CrossRef]
- Brame, C.J.; Salomon, R.G.; Morrow, J.D.; Roberts, L.J., 2nd. Identification of extremely reactive gamma-ketoaldehydes (isolevuglandins) as products of the isoprostane pathway and characterization of their lysyl protein adducts. J. Biol. Chem. 1999, 274, 13139–13146. [Google Scholar] [CrossRef] [PubMed]
- Boutaud, O.; Brame, C.J.; Salomon, R.G.; Roberts, L.J., 2nd; Oates, J.A. Characterization of the lysyl adducts formed from prostaglandin H2 via the levuglandin pathway. Biochemistry 1999, 38, 9389–9396. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.S.; Kobierski, M.E.; Salomon, R.G. Generation of pyrroles in the reaction of levuglandin E2 with proteins. J. Org. Chem. 1994, 59, 6038–6043. [Google Scholar] [CrossRef]
- Bernoud-Hubac, N.; Fay, L.B.; Armarnath, V.; Guichardant, M.; Bacot, S.; Davies, S.S.; Roberts, L.J., 2nd; Lagarde, M. Covalent binding of isoketals to ethanolamine phospholipids. Free Radic. Biol. Med. 2004, 37, 1604–1611. [Google Scholar] [CrossRef] [PubMed]
- Difranco, E.; Subbanagounder, G.; Kim, S.; Murthi, K.; Taneda, S.; Monnier, V.M.; Salomon, R.G. Formation and stability of pyrrole adducts in the reaction of levuglandin E(2) with proteins. Chem. Res. Toxicol. 1995, 8, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Kobierski, M.E.; Kim, S.C.; Murthi, K.K.; Iyer, R.S.; Salomon, R.G. Synthesis of a pyrazole isostere of pyrroles formed by the reaction of the epsilon-amino groups of protein lysyl residues with levuglandin E(2). J. Org. Chem. 1994, 59, 6044–6050. [Google Scholar] [CrossRef]
- Davies, S.S.; Amarnath, V.; Roberts, L.J., 2nd. Isoketals: Highly reactive gamma-ketoaldehydes formed from the H2-isoprostane pathway. Chem. Phys. Lipids 2004, 128, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, M.; Fenn, J.B. Electrospray ion-source—Another variation on the free-jet theme. J. Phys. Chem. 1984, 88, 4451–4459. [Google Scholar] [CrossRef]
- Tanaka, K.; Waki, H.; Ido, Y.; Akita, S.; Yoshida, Y.; Yoshida, T.; Matsuo, T. Protein and polymer analyses up to m/z 100 000 by laser ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom 1988, 2, 151–153. [Google Scholar] [CrossRef]
- Karas, M.; Hillenkamp, F. Laser desorption ionization of proteins with molecular masses exceeding 10000 Daltons. Anal. Chem. 1988, 60, 2299–2301. [Google Scholar] [CrossRef] [PubMed]
- Domingues, M.R.M.; Reis, A.; Domingues, P. Mass spectrometry analysis of oxidized phospholipids. Chem. Phys. Lipids 2008, 156, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Li, W. Oxidative modification of ethanolamine phospholipids by isolevuglandins: Detection by LC-MS/MS in vitro and in vivo. Ph.D. Thesis, Case Western Reserve University, Cleveland, OH, USA, 2009. [Google Scholar]
- Brame, C.J.; Boutaud, O.; Davies, S.S.; Yang, T.; Oates, J.A.; Roden, D.; Roberts, L.J., 2nd. Modification of proteins by isoketal-containing oxidized phospholipids. J. Biol. Chem. 2004, 279, 13447–13451. [Google Scholar] [CrossRef] [PubMed]
- Boutaud, O.; Li, J.; Zagol, I.; Shipp, E.A.; Davies, S.S.; Roberts, L.J., 2nd; Oates, J.A. Levuglandinyl adducts of proteins are formed via a prostaglandin H2 synthase-dependent pathway after platelet activation. J. Biol. Chem. 2003, 278, 16926–16928. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Laird, J.M.; Lu, L.; Roychowdhury, S.; Nagy, L.E.; Zhou, R.; Crabb, J.W.; Salomon, R.G. Isolevuglandins covalently modify phosphatidylethanolamines in vivo: Detection and quantitative analysis of hydroxylactam adducts. Free Radic. Biol. Med. 2009, 47, 1539–1552. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, S.; McMullen, M.R.; Pritchard, M.T.; Li, W.; Salomon, R.G.; Nagy, L.E. Formation of gamma-ketoaldehyde-protein adducts during ethanol-induced liver injury in mice. Free Radic. Biol. Med. 2009, 47, 1526–1538. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, M.; Li, W.; Li, T. Generation and Detection of Levuglandins and Isolevuglandins In Vitro and In Vivo. Molecules 2011, 16, 5333-5348. https://doi.org/10.3390/molecules16075333
Zhang M, Li W, Li T. Generation and Detection of Levuglandins and Isolevuglandins In Vitro and In Vivo. Molecules. 2011; 16(7):5333-5348. https://doi.org/10.3390/molecules16075333
Chicago/Turabian StyleZhang, Ming, Wei Li, and Tao Li. 2011. "Generation and Detection of Levuglandins and Isolevuglandins In Vitro and In Vivo" Molecules 16, no. 7: 5333-5348. https://doi.org/10.3390/molecules16075333
APA StyleZhang, M., Li, W., & Li, T. (2011). Generation and Detection of Levuglandins and Isolevuglandins In Vitro and In Vivo. Molecules, 16(7), 5333-5348. https://doi.org/10.3390/molecules16075333



