Synthesis, Characterization, and Antimicrobial Evaluation of Oxadiazole Congeners
Abstract
:1. Introduction
2. Results and Discussion
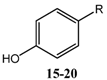
2.1. Synthesis
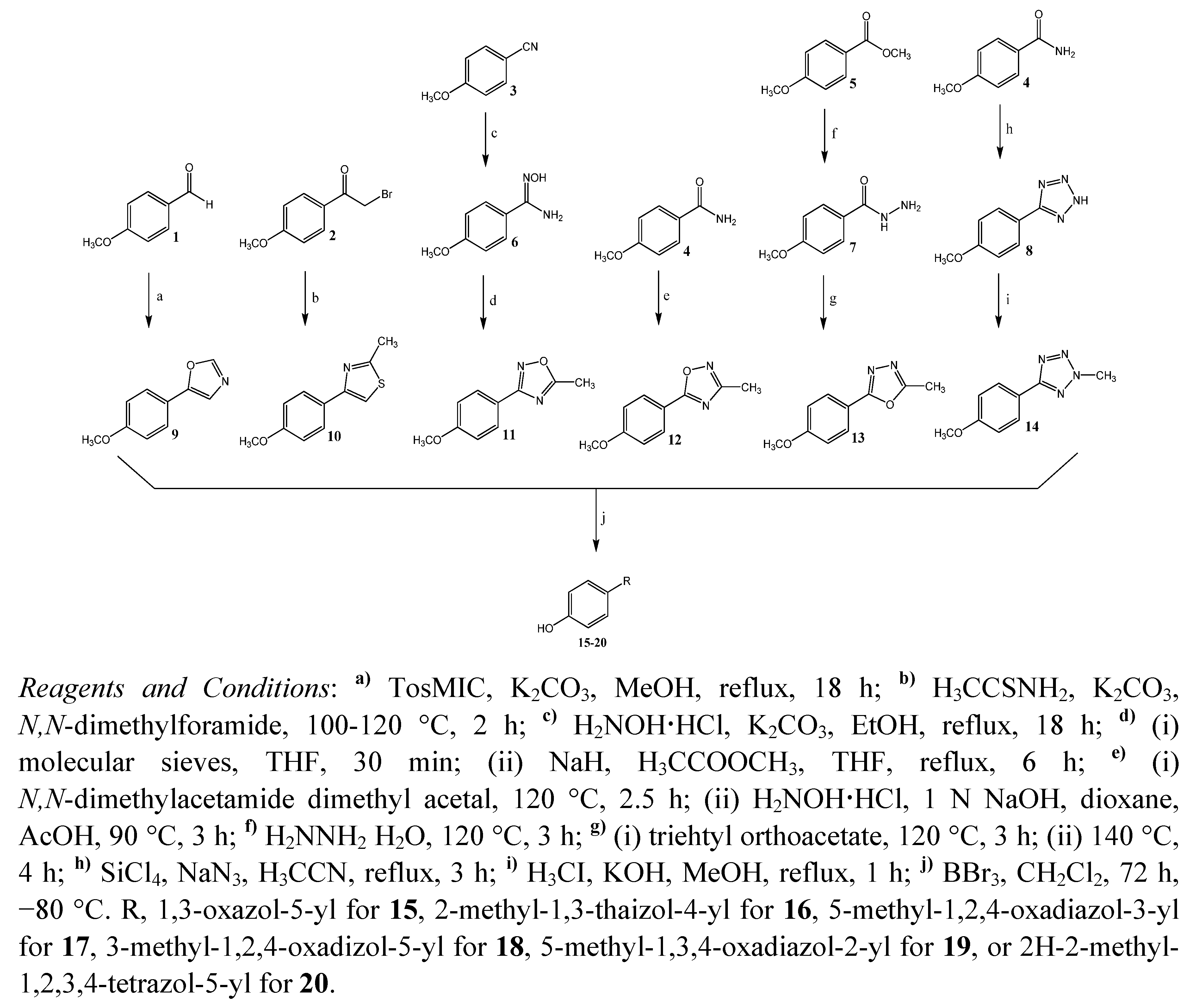
2.2. Biological Activity
| MIC µg/mL | ||||
|---|---|---|---|---|
| Compound | R | S. aureus | E. coli | A. niger |
| 15 |  | 200 | 200 | 200 |
| 16 | 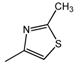 | 150 | 200 | 150 |
| 17 | 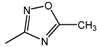 | 25 | 50 | 25 |
| 18 |  | 50 | 25 | 25 |
| 19 | 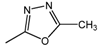 | 25 | 25 | 25 |
| 20 | 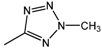 | 200 | 200 | 200 |
| Ofloxacin | 10 | 12.5 | -- | |
| Ketoconazole | -- | -- | 12.5 | |
3. Experimental
3.1. General Procedures
3.2. General Procedure for Ether Cleavage
3.3. Antimicrobial Activity
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Jumat, S.; Nadia, S.; Ayad, H.; Hiba, I.E.Y. Synthesis and antibacterial activity of some new 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives. J. Appl. Sci. Res. 2010, 6, 866–870. [Google Scholar]
- Yang, G.F.; Liu, Z.M.; Qing, X.H. Synthesis of 5,7-dimethyl-2-(5-substituted-1,3,4-oxadiazole-2-yl)-methylenethio-1,2,4-triazolo[1,5-a]pyrimidines as potential fungicides. Chin. Chem. Lett. 2001, 12, 877–880. [Google Scholar]
- Patel, K.; Chandran, J.E.; Shah, R.; Vijaya, J.; Sreenivasa, G.M. Synthesis, characterization and anthelmintic activity (Perituma posthuma) of new oxadiazole incorporated with imidazole and pyrazole. Int. J. Pharm. Bio. Sci. 2010, 1, 1–13. [Google Scholar]
- Dewangan, D.; Pandey, A.; Sivakumar, T.; Rajavel, R.; Dubey, R.D. Synthesis of some novel 2,5- disubstituted 1,3,4-oxadiazole and its analgesic, anti-inflammatory, anti-bacterial and anti-tubercular activity. Int. J. Chem. Tech. Res. 2010, 2, 1397–1412. [Google Scholar]
- Holla, B.S.; Poojary, K.N.; Bhat, K.S.; Ashok, M.; Poojary, B. Synthesis and anticancer activity studies on some 2-choloro-1,4-bis-(5-substituted-1,3,4-oxadiazole-2-ylmethyleneoxy)phenylene derivatives. Indian J. Chem. 2005, 44B, 1669–1673. [Google Scholar]
- El-Sayeda, W.A.; El-Essawyb, F.A.; Alib, O.M.; Nasr, B.S.; Abdalla, M.M.; Abdel-Rahman, A.A.-H. Anti-HIV activity of new substituted 1,3,4-oxadiazole derivatives and their acyclic nucleoside analogues. Z. Naturforsch. 2009, 64C, 773–778. [Google Scholar]
- Cena, C.; Bertinaria, M.; Boschi, D.; Giorgis, M.; Gasco, A. Use of the furoxan (1,2,5-oxadiazole 2-oxide) system in the design of new NO-donor antioxidant hybrids. ARKIVOC 2006, 7, 301–309. [Google Scholar]
- Husain, A.; Ajmal, M. Synthesis of novel 1,3,4-oxadiazole derivatives and their biological properties. Acta Pharm. 2009, 59, 223–233. [Google Scholar] [CrossRef]
- Frank, P.V.; Girish, K.S.; Kalluraya, B. Solvent-free microwave-assisted synthesis of oxadiazoles containing imidazole moiety. J. Chem. Sci. 2007, 119, 41–46. [Google Scholar] [CrossRef]
- Zarghi, A.; Hamedi, S.; Tootooni, F.; Amini, B.; Sharifi, B.; Faizi, M.; Tabatabai, S.A.; Shafiee, A. Synthesis and pharmacological evaluation of new 2-substituted-5-{2-[(2-halobenzyl) thio)phenyl}-1,3,4-oxadiazoles as anticonvulsant agents. Sci. Pharm. 2008, 76, 185–201. [Google Scholar] [CrossRef]
- Muhi-eldeen, Z.; Juma’a, G.; Al-kaissi, E.; Nouri, L. Antimicrobial activity of some new oxadiazole derivatives. J. Chem. 2008, 3, 233–243. [Google Scholar]
- Hussain, S.; Sharma, J.; Amir, M. Synthesis and antimicrobial activities of 1,2,4-triazole and 1,3,4-thiadiazole derivatives of 5-amino-2-hydroxybenzoic acid. E. J.Chem. 2008, 5, 963–968. [Google Scholar] [CrossRef]
- van Leusen, A.M.; Hoogenboom, B.E.; Siderius, H. A novel and efficient synthesis of oxazoles from tosylmethylisocyanide and carbonyl compounds. Tetrahedron Lett. 1972, 23, 2369–2372. [Google Scholar]
- Vermin, G. General Synthesis Methods for Thiazole and Thiazolium Salts; Metzger, J.V., Ed.; Interscience: New York, NY, USA, 1979; Volume 34, pp. 165–335. [Google Scholar]
- Swain, C.J.; Baker, R.; Kneen, C.; Moseley, J.; Saunders, J.; Seward, E.M.; Stevenson, G.; Beer, M.; Stanton, J.; Walting, K. Novel 5-HT3 antagonists. Indole oxadiazoles. J. Med. Chem. 1991, 34, 140–151. [Google Scholar] [CrossRef]
- Lin, Y.I.; Lang, S.A., Jr.; Lovell, M.F.; Perkinson, N.A. New synthesis of 1,2,4-triazoles and 1,2,4-oxadiazoles. J. Org. Chem. 1979, 44, 4160–4164. [Google Scholar] [CrossRef]
- Anisworth, C.J. The condensation of aryl carboxylic acid hydrazides with orthoesters. J. Am. Chem. Soc. 1955, 77, 1148–1152. [Google Scholar] [CrossRef]
- El-Ahl, A.A.S.; Elmorsy, S.S.; Elbeheery, A.H.; Amer, F.A. A novel approach for the synthesis of 5-substituted tetrazole derivatives from primary amides in mild one-step method. Tetrahedron Lett. 1997, 38, 1257–1260. [Google Scholar]
- Begtrup, M.; Larsen, P. Alkylation, acetylation and silylation of azoles. Acta Chem.Scand. 1990, 44, 1050–1057. [Google Scholar] [CrossRef]
- Fraser, R.R.; Haque, K.E. Nuclear magnetic resonance and mass spectral properties of 5-aryltetrazoles. Can.J.Chem. 1968, 46, 2855–2859. [Google Scholar] [CrossRef]
- Mcomie, J.F.W.; Watts, M.L.; West, D.E. Demethylation of aryl methyl ethers by boron tribromide. Tetrahedron 1969, 24, 2289–2292. [Google Scholar]
- Begtrup, M. 13C-NMR spectra of phenyl-substituted azoles: A conformational study. Acta Chem. Scand. 1973, 27, 3101–3110. [Google Scholar] [CrossRef]
- Barry, A.L. The Antimicrobial Susceptibility Test: Principle and Practices; Edited by Illus lea and Febiger: Philadelphia, PA, USA, 1976; p. 180, [Biol. Abstr. 1977, 64, 25183]. [Google Scholar]
- Sample Availability: Samples of the compounds 15-20 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sadek, B.; Fahelelbom, K.M.S. Synthesis, Characterization, and Antimicrobial Evaluation of Oxadiazole Congeners. Molecules 2011, 16, 4339-4347. https://doi.org/10.3390/molecules16064339
Sadek B, Fahelelbom KMS. Synthesis, Characterization, and Antimicrobial Evaluation of Oxadiazole Congeners. Molecules. 2011; 16(6):4339-4347. https://doi.org/10.3390/molecules16064339
Chicago/Turabian StyleSadek, Bassem, and Khairi Mustafa Salem Fahelelbom. 2011. "Synthesis, Characterization, and Antimicrobial Evaluation of Oxadiazole Congeners" Molecules 16, no. 6: 4339-4347. https://doi.org/10.3390/molecules16064339
APA StyleSadek, B., & Fahelelbom, K. M. S. (2011). Synthesis, Characterization, and Antimicrobial Evaluation of Oxadiazole Congeners. Molecules, 16(6), 4339-4347. https://doi.org/10.3390/molecules16064339