Screening of Natural Organic Volatiles from Prunus mahaleb L. Honey: Coumarin and Vomifoliol as Nonspecific Biomarkers
Abstract
:1. Introduction
2. Results and Discussion
2.1. Organic Volatiles Isolated by HS-SPME
| No. | Compound | RI | Min. | Max. | Av. | SD. |
|---|---|---|---|---|---|---|
| 1 | Dimethyl sulfide | <900 | 0.0 | 0.9 | 0.43 | 0.45 |
| 2 | 2-Methylbutanal | <900 | 0.0 | 0.9 | 0.37 | 0.47 |
| 3 | 2,5-Dimethylfuran | <900 | 0.0 | 2.9 | 1.30 | 1.47 |
| 4 | 3-Methylpentan-3-one | <900 | 2.0 | 2.4 | 2.20 | 0.20 |
| 5 | Octane | <900 | 0.0 | 1.7 | 0.83 | 0.85 |
| 6 | Furfural | <900 | 0.0 | 0.4 | 0.23 | 0.21 |
| 7 | 3-Methylpentan-1-ol | <900 | 0.0 | 0.4 | 0.20 | 0.20 |
| 8 | 3-Methylpentanoic acid | 941 | 1.0 | 1.3 | 1.13 | 0.15 |
| 9 | Benzaldehyde | 965 | 2.0 | 4.5 | 3.37 | 1.27 |
| 10 | Phenylacetaldehyde | 1048 | 0.5 | 2.8 | 1.43 | 1.21 |
| 11 | trans-Linalool oxide (furan type) | 1076 | 2.0 | 3.1 | 2.53 | 0.55 |
| 12 | cis-Linalool oxide (furan type) | 1091 | 0.6 | 1.0 | 0.80 | 0.20 |
| 13 | Linalool | 1101 | 4.0 | 6.6 | 5.13 | 1.33 |
| 14 | Nonanal | 1102 | 0.0 | 0.8 | 0.37 | 0.40 |
| 15 | Hotrienol | 1106 | 0.5 | 1.9 | 1.10 | 0.72 |
| 16 | α-Isophorone | 1124 | 2.1 | 6.5 | 4.10 | 2.23 |
| 17 | Lilac aldehyde (isomer I) | 1151 | 1.0 | 4.9 | 3.30 | 2.04 |
| 18 | 4-Ketoisophorone | 1152 | 5.0 | 7.4 | 6.17 | 1.20 |
| 19 | Lilac aldehyde (isomer II) | 1159 | 6.1 | 11.7 | 9.00 | 2.81 |
| 20 | Lilac aldehyde (isomer III) | 1174 | 2.2 | 4.9 | 3.37 | 1.39 |
| 21 | 3,5,5-Trimethylcyclohexan-1,4-dione | 1177 | 0.0 | 1.8 | 0.93 | 0.90 |
| 22 | Benzoic acid | 1181 | 0.2 | 1.4 | 0.87 | 0.61 |
| 23 | Safranal | 1206 | 0.0 | 1.0 | 0.40 | 0.53 |
| 24 | Lilac alcohol (isomer I) | 1210 | 0.1 | 1.1 | 0.47 | 0.55 |
| 25 | Lilac alcohol (isomer II) | 1220 | 0.0 | 0.6 | 0.27 | 0.31 |
| 26 | Lilac alcohol (isomer III) | 1222 | 0.1 | 0.6 | 0.30 | 0.26 |
| 27 | Car-2-en-4-one* | 1228 | 0.0 | 1.2 | 0.67 | 0.61 |
| 28 | 2-Hydroxy-3,5,5-trimethyl-cyclohex-2-en-1,4-dione(2-Hydroxy-4-ketoisophorone) | 1245 | 0.0 | 0.6 | 0.30 | 0.30 |
| 29 | 5-Hydroxymethylfurfural | 1254 | 0.7 | 1.8 | 1.17 | 0.57 |
| 30 | 4-Methoxybenzaldehyde (4-Anisaldehyde) | 1264 | 1.4 | 10.9 | 4.93 | 5.20 |
| 31 | 1-Methoxy-4-propylbenzene | 1307 | 0.5 | 5.3 | 2.60 | 2.46 |
| 32 | 3,4,5-Trimethylphenol** | 1336 | 1.1 | 3.6 | 2.40 | 1.25 |
| 33 | Coumarin | 1444 | 0.9 | 1.8 | 1.23 | 0.49 |
| 34 | Hexadecanoic acid | 1981 | 0.5 | 3.0 | 1.50 | 1.32 |
2.2. Organic Volatiles Isolated by USE
| No. | Compound | RI | Area percentages (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Solvent A | Solvent B | |||||||||||
| Min. | Max. | Av. | SD. | Min. | Max. | Av. | SD. | |||||
| 1 | 3-Methylbutanoic acid | < 900 | - | - | - | - | 0.0 | 0.1 | 0.07 | 0.06 | ||
| 2 | 1,3-Dimethylbenzene** | < 900 | - | - | - | - | 0.0 | 0.1 | 0.07 | 0.06 | ||
| 3 | Nonane | 900 | 0.1 | 0.8 | 0.33 | 0.32 | - | - | - | - | ||
| 4 | 3-Methylpentanoic acid | 941 | 0.0 | 1.1 | 0.35 | 0.51 | 0.0 | 0.2 | 0.10 | 0.10 | ||
| 5 | Benzaldehyde | 965 | 0.0 | 0.3 | 0.15 | 0.13 | 0.0 | 0.2 | 0.13 | 0.12 | ||
| 6 | Benzyl alcohol | 1037 | 0.0 | 0.2 | 0.08 | 0.10 | 0.0 | 0.1 | 0.07 | 0.06 | ||
| 7 | Pantoic lactone | 1044 | - | - | - | - | 0.0 | 0.1 | 0.07 | 0.06 | ||
| 8 | Phenylacetaldehyde | 1048 | 0.0 | 0.4 | 0.18 | 0.17 | 0.0 | 0.1 | 0.05 | 0.06 | ||
| 9 | 4,5-Dimethyl-2-formylfuran | 1078 | 0.1 | 0.5 | 0.25 | 0.19 | 0.1 | 0.6 | 0.30 | 0.22 | ||
| 10 | Methyl 2-furoate | 1084 | 0.1 | 0.3 | 0.18 | 0.10 | 0.2 | 0.7 | 0.45 | 0.24 | ||
| 11 | 4-Ketoisophorone | 1152 | 0.2 | 1.9 | 0.98 | 0.85 | 0.1 | 0.6 | 0.40 | 0.25 | ||
| 12 | Benzoic acid | 1181 | 2.5 | 4.9 | 3.65 | 1.10 | 0.3 | 0.8 | 0.58 | 0.21 | ||
| 13 | (E)-3,7-dimethylocta-1,5-diene-3,7-diol(Terpenediol I) | 1191 | 0.1 | 0.2 | 0.13 | 0.05 | 0.0 | 0.2 | 0.10 | 0.10 | ||
| 14 | 2,3-dihydrobenzofuran (coumaran) | 1245 | 0.0 | 0.7 | 0.25 | 0.33 | - | - | - | - | ||
| 15 | 5-Hydroxymethylfurfural | 1251 | 1.0 | 2.6 | 2.13 | 0.75 | 1.0 | 5.0 | 3.33 | 1.70 | ||
| 16 | 4-Methoxybenzaldehyde (4-Anisaldehyde) | 1262 | 1.0 | 1.9 | 1.53 | 0.38 | 0.1 | 0.3 | 0.23 | 0.10 | ||
| 17 | Phenylacetic acid | 1283 | 0.8 | 1.4 | 1.10 | 0.26 | 0.1 | 0.2 | 0.15 | 0.06 | ||
| 18 | 4-Methoxybenzyl alcohol | 1292 | 0.0 | 0.3 | 0.15 | 0.13 | - | - | - | - | ||
| 19 | 1-Methoxy-4-propylbenzene | 1307 | 0.0 | 1.6 | 0.88 | 0.80 | 0.0 | 0.4 | 0.20 | 0.16 | ||
| 20 | 4-Hydroxy-3,5,5-trimethyl-cyclohexan-1-one-2-ene (4-Hydroxy-α-isophorone) | 1320 | 0.3 | 0.6 | 0.45 | 0.13 | 0.0 | 0.1 | 0.07 | 0.06 | ||
| 21 | 3,4,5-Trimethylphenol** | 1333 | 0.5 | 0.8 | 0.65 | 0.13 | 0.5 | 1.5 | 0.80 | 0.48 | ||
| 22 | 4-Methoxy-2-phenylethanol | 1377 | 0.3 | 0.5 | 0.43 | 0.10 | 0.0 | 0.1 | 0.05 | 0.06 | ||
| 23 | 4-Hydroxybenzaldehyde | 1393 | 0.0 | 0.3 | 0.13 | 0.25 | 0.0 | 0.1 | 0.05 | 0.06 | ||
| 24 | Vanillin | 1412 | 0.0 | 0.5 | 0.25 | 0.24 | 0.0 | 0.2 | 0.10 | 0.08 | ||
| 25 | Coumarin | 1444 | 1.1 | 2.4 | 1.83 | 0.56 | 0.3 | 0.4 | 0.35 | 0.06 | ||
| 26 | 4-Methoxybenzoic acid | 1452 | 0.4 | 4.7 | 3.35 | 1.99 | 0.2 | 1.8 | 1.03 | 0.66 | ||
| 27 | Methyl 4-hydroxybenzoate | 1482 | 0.0 | 0.5 | 0.23 | 0.22 | 0.0 | 0.2 | 0.10 | 0.10 | ||
| 28 | 3,4-Dimethoxybenzaldehyde (methyl vanillin) | 1492 | 0.0 | 0.9 | 0.38 | 0.41 | - | - | - | - | ||
| 29 | Acetylvanillone | 1498 | - | - | - | - | 0.0 | 0.2 | 0.10 | 0.08 | ||
| 30 | 4-Methyl-2,6-bis(1,1-dimethylethyl)-phenol | 1522 | 0.7 | 1.3 | 0.98 | 0.32 | - | - | - | - | ||
| 31 | Dibenzyl | 1526 | 0.0 | 0.4 | 0.18 | 0.17 | - | - | - | - | ||
| 32 | Methyl 4-hydroxy-3-methoxy benzoate | 1530 | 0.9 | 2.9 | 1.65 | 0.87 | 0.1 | 0.5 | 0.28 | 0.17 | ||
| 33 | Methyl 4-methoxybenzoate | 1546 | 2.0 | 3.3 | 2.50 | 0.53 | 0.3 | 1.0 | 0.50 | 0.36 | ||
| 34 | Methyl 2,5-dihydroxybenzoate | 1551 | 0.0 | 1.0 | 0.45 | 0.42 | 0.0 | 0.3 | 0.13 | 0.15 | ||
| 35 | 4-Hydroxybenzoic acid | 1571 | 0.4 | 3.7 | 2.08 | 1.88 | - | - | - | - | ||
| 36 | 1,4-Dimethylindanyl acetate | 1661 | 1.8 | 2.7 | 2.15 | 0.55 | 0.7 | 1.6 | 1.03 | 0.39 | ||
| 37 | 3,4-Dimethoxybenzoic acid | 1672 | - | - | - | - | 0.3 | 1.0 | 0.78 | 0.32 | ||
| 38 | 3-Ethyl-2-(but-3-enyl)-cyclohex-2-en-1-one* | 1705 | 0.0 | 1.1 | 0.70 | 0.50 | 0.0 | 1.5 | 0.68 | 0.67 | ||
| 39 | Methyl syringate | 1788 | 4.5 | 6.8 | 6.03 | 1.05 | 3.8 | 4.2 | 4.00 | 0.16 | ||
| 40 | Vomofoliol | 1820 | 10.7 | 13.8 | 12.15 | 1.28 | 21.6 | 24.2 | 22.95 | 1.06 | ||
| 41 | Hexadecan-1-ol | 1892 | 0.0 | 0.5 | 0.30 | 0.22 | 0.5 | 1.4 | 0.95 | 0.47 | ||
| 42 | Dibuthyl phtalate | 1975 | 0.3 | 0.4 | 0.35 | 0.06 | 0.0 | 1.0 | 0.68 | 0.46 | ||
| 43 | Hexadecanoic acid | 1988 | 1.0 | 1.6 | 1.38 | 0.26 | 0.3 | 1.1 | 0.78 | 0.34 | ||
| 44 | Pinocembrin | 2039 | 0.0 | 0.5 | 0.20 | 0.25 | - | - | - | - | ||
| 45 | (Z)-Octadec-9-en-1-ol | 2073 | 1.5 | 5.5 | 3.25 | 1.66 | 1.0 | 2.7 | 1.35 | 0.90 | ||
| 46 | Heneicosane | 2100 | 0.0 | 2.9 | 1.78 | 1.25 | 0.0 | 0.2 | 0.10 | 0.10 | ||
| 47 | (Z)-Octadec-9-enoic acid | 2181 | 1.2 | 3.2 | 2.15 | 0.82 | 0.5 | 0.8 | 0.68 | 0.15 | ||
| 48 | Octadecanoic acid | 2209 | 0.0 | 0.6 | 0.25 | 0.30 | 0.0 | 0.2 | 0.10 | 0.10 | ||
| 49 | Tricosane | 2300 | 0.5 | 1.8 | 0.83 | 0.66 | 0.1 | 0.2 | 0.15 | 0.06 | ||
2.3. Potential Chemical Biomarkers of P. mahaleb Honey
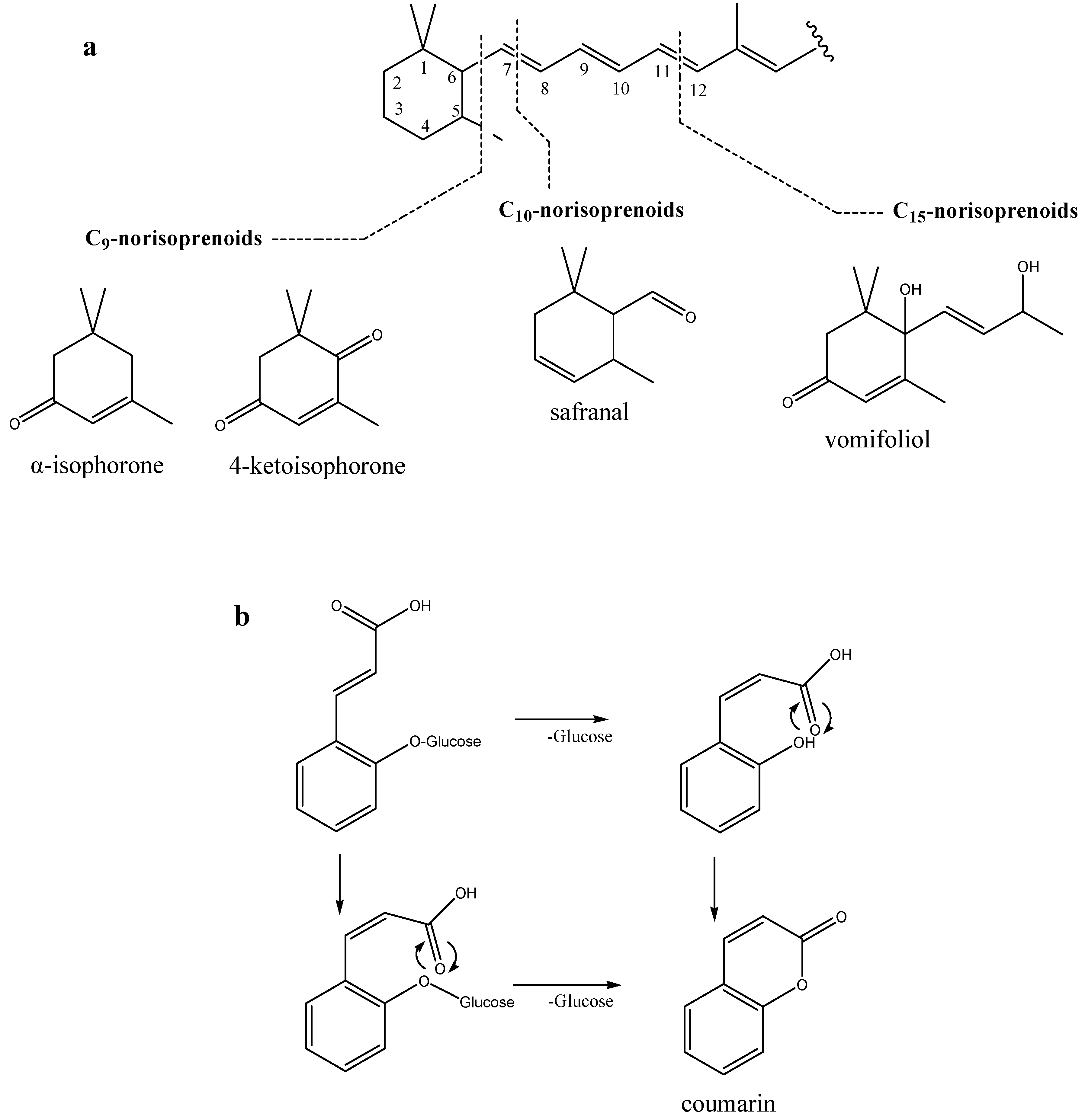
3. Experimental
3.1. Honey Samples
3.2. Headspace Solid-Phase Microextraction (HS-SPME)
3.3. Ultrasonic Solvent Extraction (USE)
3.4. Gas Chromatography and Mass Spectrometry (GC, GC-MS)
3.5. Data Analysis and Data Evaluation
4. Conclusions
Acknowledgements
References
- Zima, D. Contribution to the knowledge of honey rich plants in Croatia. Agronomski Glasnik 2007, 2, 147–160. [Google Scholar]
- Alsaid, M.S.; Hifnawy, M.S. Dihydrocoumarin and certain other coumarins from Prunus mahaleb seeds. J. Nat. Prod. (Lloydia) 1986, 49, 721. [Google Scholar] [CrossRef]
- Favre-Bonvin, J.; Massias, M.; Mentzer, C.; Massicot, J. Sites de biosynthèse des coumarines chez Prunus mahaleb. Ètude par greffage et administration d’un précurseur marquè. Phytochemistry 1968, 7, 1555–1560. [Google Scholar] [CrossRef]
- Moreno, M.A.; Montanes, L.; Tabuenca, M.C.; Cambra, R. The performance of adara as a cherry rootstock. Sci. Hort. 1996, 65, 85–91. [Google Scholar] [CrossRef]
- Aydin, C.; Ögüt, H.; Konak, M. Some physical properties of Turkish Mahaleb. Biosyst. Eng. 2002, 82, 231–234. [Google Scholar] [CrossRef]
- Mastelić, J.; Jerković, I.; Mesić, M. Volatile constituents from flowers, leaves, bark and wood of Prunus mahaleb L. Flavour Fragr. J. 2006, 21, 306–313. [Google Scholar] [CrossRef]
- Persano Oddo, L.; Piana, L.; Bogdanov, S.; Bentabol, A.; Gotsiou, P.; Kerkvliet, J.; Martin, P.; Morlot, M.; Ortiz Valbuena, A.; Ruoff, K.; von der Ohe, K. Botanical species giving unifloral honey in Europe. Apidologie 2004, 35, S82–S93. [Google Scholar] [CrossRef]
- Cuevas-Glory, L.F.; Pino, J.A.; Santiago, L.S.; Sauri-Duch, E. A review of volatile analytical methods for determining the botanical origin of honey. Food Chem. 2007, 103, 1032–1043. [Google Scholar] [CrossRef]
- de la Fuente, E.; Valencia-Barrera, R.M.; Martïnez-Castro, I.; Sanz, J. Occurrence of 2-hydroxy-5-methyl-3-hexanone and 3-hydroxy-5-methyl-2-hexanone as indicators of botanic origin in eucalyptus honeys. Food Chem. 2007, 103, 1176–1180. [Google Scholar] [CrossRef]
- Guyot, C.; Bouseta, A.; Scheirman, V.; Collin, S. Floral origin markers of chestnut and lime treehoneys. J. Agric. Food Chem. 1998, 46, 625–633. [Google Scholar] [CrossRef]
- Guyot, C.; Scheirman, V.; Collin, S. Floral origin markers of heather honeys: Calluna vulgaris and Erica arborea. Food Chem. 1999, 64, 3–11. [Google Scholar] [CrossRef]
- Piasenzotto, L.; Gracco, L.; Conte, L. Solid phase microextraction (SPME) applied to honey quality control. J. Sci. Food Agric. 2003, 83, 1037–1044. [Google Scholar] [CrossRef]
- Peña, R.M.; Barciela, J.; Herrero, C.; García-Martín, S. Solid-phase microextraction gas chromatography-mass spectrometry determination of monoterpenes in honey. J. Sep. Sci. 2004, 27, 1540–1544. [Google Scholar] [CrossRef]
- Pontes, M.; Marques, J.C.; Câmara, J.S. Screening of volatile composition from Portuguese multifloral honeys using headspace solid-phase microextraction - gas chromatography - quadrupole mass spectrometry. Talanta 2007, 74, 91–103. [Google Scholar] [CrossRef]
- Soria, A.C.; Sanz, J.; Martínez-Castro, I. SPME followed by GC-MS: A powerful techniques for qualitative analysis of honey volatiles. Eur. Food Res. Technol. 2009, 228, 579–590. [Google Scholar] [CrossRef]
- Jerković, I.; Marijanović, Z.; Tuberoso, C.I.G.; Bubalo, D.; Kezić, N. Molecular diversity of volatile compounds in rare willow (Salix spp.) honeydew honey: Identification of chemical biomarkers. Mol. Divers. 2010, 14, 237–248. [Google Scholar] [CrossRef]
- Jerković, I.; Hegić, G.; Marijanović, Z.; Bubalo, D. Organic extractives from Mentha spp. honey and the bee-stomach: Methyl syringate, vomifoliol, terpenediol I, hotrienol and other compounds. Molecules 2010, 15, 2911–2924. [Google Scholar] [CrossRef]
- Jerković, I.; Marijanović, Z. Screening of volatile composition of Lavandula hybrida Reverchon II honey using headspace solid phase microextraction and ultrasonic solvent extraction. Chem. Biodivers. 2009, 6, 421–430. [Google Scholar] [CrossRef]
- Guyot-Declerck, C.; Renson, S.; Bouseta, A.; Collin, S. Floral quality and discrimination of Lavandula stoechas, Lavandula angustifolia, and Lavandula angustifolia × latifolia honeys. Food Chem. 2002, 79, 453–459. [Google Scholar] [CrossRef]
- Jerković, I.; Tuberoso, C.I.G.; Marijanović, Z.; Jelić, M.; Kasum, A. Headspace, volatile and semi-volatile patterns of Paliurus spina-christi unifloral honey as markers of botanical origin. Food Chem. 2009, 112, 239–245. [Google Scholar] [CrossRef]
- Wilkins, A.L.; Lu, Y.; Tan, S.T. Extractives from New Zealand honeys. 4. Linalool derivatives and other substances from nodding thistle (Carduus nutans) honey. J. Agric. Food Chem. 1993, 41, 873–878. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Daferera, D.; Tarantalis, P.A.; Polissiou, M.; Harizanis, P.C. Ultrasound-assisted extraction of volatile compounds from citrus flowers and citrus honey. Food Chem. 2003, 82, 575–582. [Google Scholar] [CrossRef]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of melissopalynology. Bee World 1978, 59, 139–153. [Google Scholar]
- El-Sayed, A.M. The Pherobase: Database of Insect Pheromones and Semiochemicals. Available online: http://www.pherobase.com/ (accessed on 12 February 2011).
- Sample Availability: Contact the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jerković, I.; Marijanović, Z.; Staver, M.M. Screening of Natural Organic Volatiles from Prunus mahaleb L. Honey: Coumarin and Vomifoliol as Nonspecific Biomarkers. Molecules 2011, 16, 2507-2518. https://doi.org/10.3390/molecules16032507
Jerković I, Marijanović Z, Staver MM. Screening of Natural Organic Volatiles from Prunus mahaleb L. Honey: Coumarin and Vomifoliol as Nonspecific Biomarkers. Molecules. 2011; 16(3):2507-2518. https://doi.org/10.3390/molecules16032507
Chicago/Turabian StyleJerković, Igor, Zvonimir Marijanović, and Mladenka Malenica Staver. 2011. "Screening of Natural Organic Volatiles from Prunus mahaleb L. Honey: Coumarin and Vomifoliol as Nonspecific Biomarkers" Molecules 16, no. 3: 2507-2518. https://doi.org/10.3390/molecules16032507
APA StyleJerković, I., Marijanović, Z., & Staver, M. M. (2011). Screening of Natural Organic Volatiles from Prunus mahaleb L. Honey: Coumarin and Vomifoliol as Nonspecific Biomarkers. Molecules, 16(3), 2507-2518. https://doi.org/10.3390/molecules16032507






